Gait Alteration in Individual with Limb Loss: The Role of Inertial Sensors
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Extraction
2.2. Methodological Assessment
3. Results
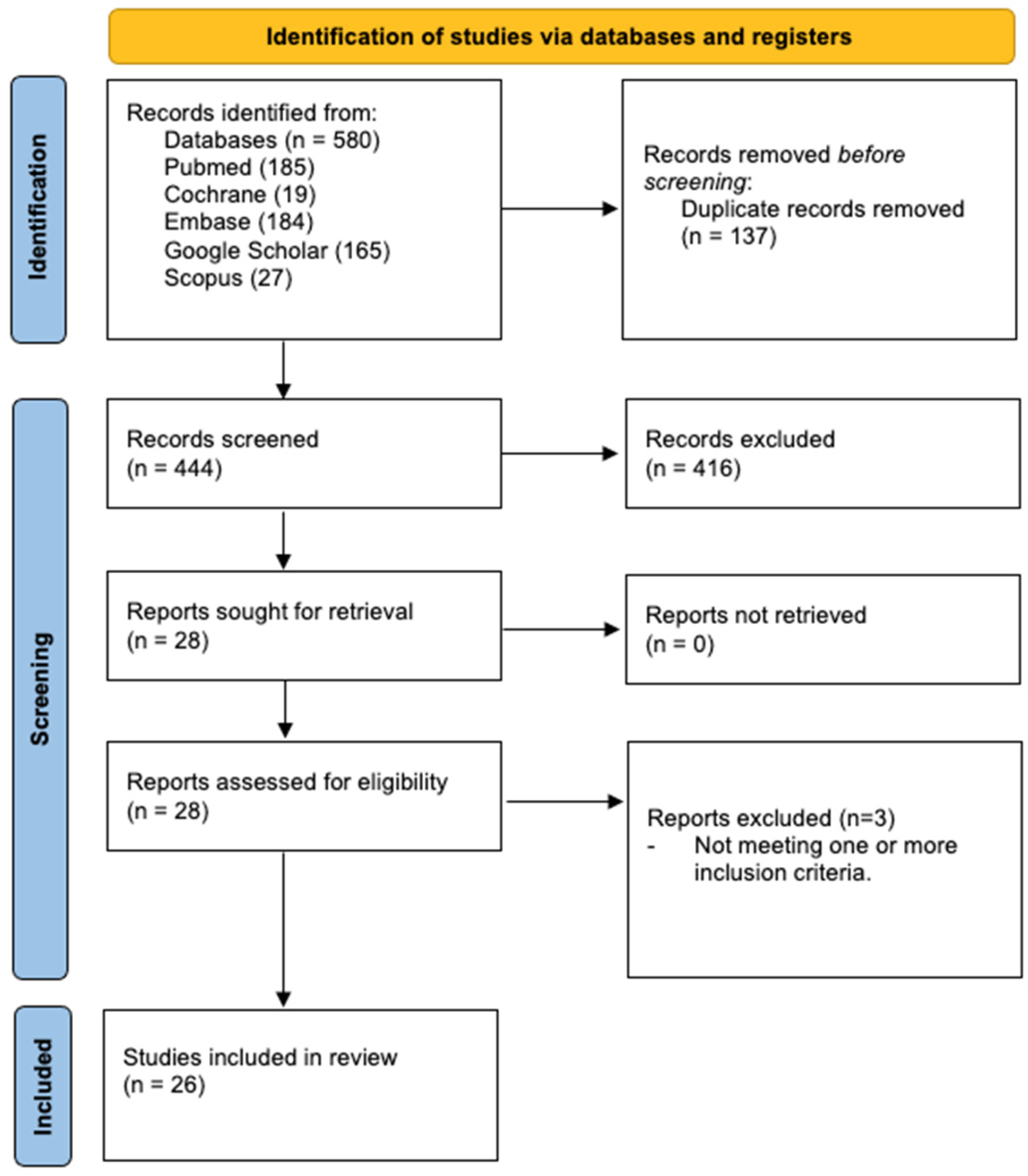
3.1. Study Selection
3.2. Main Characteristics of the Studies
3.3. Methodological Quality
4. Discussion
4.1. Gait Analysis
4.1.1. Joint Angle Calculation
4.1.2. Detection of the Onset of Gait
4.1.3. Acceleration and Walking Speed
4.1.4. Step Length
4.1.5. Variability and Stability, Symmetries and Regularity
4.2. Fall Risk Classification
4.3. Prosthesis Development
4.4. Energy Expenditure and Metabolism
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Penn-Barwell, J.G. Outcomes in lower limb amputation following trauma: A systematic review and meta-analysis. Injury 2011, 42, 1474–1479. [Google Scholar] [CrossRef] [PubMed]
- Varma, P.; Stineman, M.G.; Dillingham, T.R. Physical Medicine and Rehabilitation Clinics of North America Epidemiology of Limb Loss. Phys. Med. Rehabil. Clin. N. Am. 2014, 25, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Edwards, D.S.; Phillip, R.D.; Bosanquet, N.; Bull, A.M.J.; Clasper, J.C. What Is the Magnitude and Long-term Economic Cost of Care of the British Military Afghanistan Amputee Cohort? Clin. Orthop. Relat. Res. 2015, 473, 2848–2855. [Google Scholar] [CrossRef] [PubMed]
- Ma, V.Y.; Chan, L.; Carruthers, K.J. Incidence, prevalence, costs, and impact on disability of common conditions requiring rehabilitation in the United States: Stroke, spinal cord injury, traumatic brain injury, multiple sclerosis, osteoarthritis, rheumatoid arthritis, limb loss, and back pain. Arch. Phys. Med. Rehabil. 2014, 95, 986–995.e1. [Google Scholar] [CrossRef] [PubMed]
- Traballesi, M.; Brunelli, S.; Pulcini, M.; Pitidis, A.; Taggi, F. Trattamento Riabilitativo del Paziente Geriatrico Vasculopatico Amputato di Coscia: Sperimentazione di un Protocollo. Available online: https://www.iss.it/documents/20126/955767/0346.1109238140.pdf/60022757-fe48-872b-d086-cd948b33b309?t=1575578559152 (accessed on 15 November 2022).
- Webster, J.B.; Hakimi, K.N.; Williams, R.M.; Turner, A.P.; Norvell, D.C.; Czerniecki, J.M. Prosthetic fitting, use, and satisfaction following lower-limb amputation: A prospective study. J. Rehabil. Res. Dev. 2012, 49, 1493–1504. [Google Scholar] [CrossRef]
- Geertzen, J.H.B.; Bosmans, J.C.; van der Schans, C.P.; Dijkstra, P.U. Claimed walking distance of lower limb amputees. Disabil. Rehabil. 2005, 27, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.M.; Kalbaugh, C.A.; Blackhurst, D.W.; Hamontree, S.E.; Cull, D.L.; Messich, H.S.; Robertson, R.T.; Langan, E.M.; York, J.W.; Carsten, C.G.; et al. Preoperative clinical factors predict postoperative functional outcomes after major lower limb amputation: An analysis of 553 consecutive patients. J. Vasc. Surg. 2005, 42, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Latlief, G.; Elnitsky, C.; Hart-Hughes, S.; Phillips, S.L.; Adams-Koss, L.; Kent, R.; Highsmith, M.J. Patient safety in the rehabilitation of the adult with an amputation. Phys. Med. Rehabil. Clin. N. Am. 2012, 23, 377–392. [Google Scholar] [CrossRef] [PubMed]
- Pierrie, S.N.; Gaston, R.G.; Loeffler, B.J. Current Concepts in Upper-Extremity Amputation. J. Hand Surg. Am. 2018, 43, 657–667. [Google Scholar] [CrossRef]
- Ülger, Ö.; Yıldırım Şahan, T.; Çelik, S.E. A systematic literature review of physiotherapy and rehabilitation approaches to lower-limb amputation. Physiother. Theory Pract. 2018, 34, 821–834. [Google Scholar] [CrossRef]
- Fang, Y.; Morse, L.R.; Nguyen, N.; Tsantes, N.G.; Troy, K.L. Anthropometric and biomechanical characteristics of body segments in persons with spinal cord injury. J. Biomech. 2017, 55, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Hebert, J.S.; Wolfe, D.L.; Miller, W.C.; Deathe, A.B.; Devlin, M.; Pallaveshi, L. Outcome measures in amputation rehabilitation: ICF body functions. Disabil. Rehabil. 2009, 31, 1541–1554. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-J.; Bose, N.H. Six-Minute Walk Test in Persons with Transtibial Amputation. Arch. Phys. Med. Rehabil. 2008, 89, 2354–2359. [Google Scholar] [CrossRef] [PubMed]
- Iosa, M.; Picerno, P.; Paolucci, S.; Morone, G. Wearable inertial sensors for human movement analysis. Expert Rev. Med. Dev. 2016, 13, 641–659. [Google Scholar] [CrossRef] [PubMed]
- Saunders, J.B.; Inman, V.T.; Eberhart, H.D. The major determinants in normal and pathological gait. J. Bone Jt. Surg. Am. 1953, 35, 543–558. [Google Scholar] [CrossRef]
- Demeco, A.; Marotta, N.; Moggio, L.; Pino, I.; Marinaro, C.; Barletta, M.; Petraroli, A.; Palumbo, A.; Ammendolia, A. Quantitative analysis of movements in facial nerve palsy with surface electromyography and kinematic analysis. J. Electromyogr. Kinesiol. 2021, 56, 102485. [Google Scholar] [CrossRef]
- Bijalwan, V.; Semwal, V.B.; Mandal, T.K. Fusion of multi-sensor-based biomechanical gait analysis using vision and wearable sensor. IEEE Sens. J. 2021, 21, 14213–14220. [Google Scholar] [CrossRef]
- Carozzo, S.; Vatrano, M.; Coschignano, F.; Battaglia, R.; Calabrò, R.S.; Pignolo, L.; Contrada, M.; Tonin, P.; Cerasa, A.; Demeco, A. Efficacy of Visual Feedback Training for Motor Recovery in Post-Operative Subjects with Knee Replacement: A Randomized Controlled Trial. J. Clin. Med. 2022, 11, 7355. [Google Scholar] [CrossRef]
- Hatamzadeh, M.; Busé, L.; Chorin, F.; Alliez, P.; Favreau, J.-D.; Zory, R. A kinematic-geometric model based on ankles’ depth trajectory in frontal plane for gait analysis using a single RGB-D camera. J. Biomech. 2022, 145, 111358. [Google Scholar] [CrossRef]
- Petraglia, F.; Scarcella, L.; Pedrazzi, G.; Brancato, L.; Puers, R.; Costantino, C. Inertial sensors versus standard systems in gait analysis: A systematic review and meta-analysis. Eur. J. Phys. Rehabil. Med. 2019, 55, 265–280. [Google Scholar] [CrossRef]
- O’Keeffe, K.; Argent, R.; Bourke, A.; Shabani, S.; Praestgaard, J.; Muaremi, A.; Goulding, C.; Brom, M.; Scotti, C.; Walsh, L.C.; et al. Test-retest reliability of wireless inertial-sensor derived measurements of knee joint kinematics. In Proceedings of the 2022 44th Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Glasgow, UK, 11–15 July 2022; Volume 2022, pp. 4218–4221. [Google Scholar] [CrossRef]
- Di Raimondo, G.; Vanwanseele, B.; van der Have, A.; Emmerzaal, J.; Willems, M.; Killen, B.A.; Jonkers, I. Inertial Sensor-to-Segment Calibration for Accurate 3D Joint Angle Calculation for Use in OpenSim. Sensors 2022, 22, 3259. [Google Scholar] [CrossRef] [PubMed]
- Kiernan, D.; Hawkins, D.A.; Manoukian, M.A.C.; McKallip, M.; Oelsner, L.; Caskey, C.F.; Coolbaugh, C.L. Accelerometer-based prediction of running injury in National Collegiate Athletic Association track athletes. J. Biomech. 2018, 73, 201–209. [Google Scholar] [CrossRef]
- Lee, S.; Kim, K.; Kim, Y.H.; Lee, S. Motion anlaysis in lower extremity joints during Ski carving turns using wearble inertial sensors and plantar pressure sensors. In Proceedings of the 2017 IEEE International Conference on Systems, Man, and Cybernetics (SMC), Banff, AB, Canada, 5–8 October 2017; pp. 695–698. [Google Scholar]
- Fall, C.L.; Quevillon, F.; Blouin, M.; Latour, S.; Campeau-Lecours, A.; Gosselin, C.; Gosselin, B. A Multimodal Adaptive Wireless Control Interface for People with Upper-Body Disabilities. IEEE Trans. Biomed. Circuits Syst. 2018, 12, 564–575. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, L.; Tigra, W.; Navarro, B.; Guiraud, D.; Fattal, C.; Bó, A.; Fachin-Martins, E.; Leynaert, V.; Gélis, A.; Azevedo-Coste, C. Assisted Grasping in Individuals with Tetraplegia: Improving Control through Residual Muscle Contraction and Movement. Sensors 2019, 19, 4532. [Google Scholar] [CrossRef] [PubMed]
- Bo, F.; Yerebakan, M.; Dai, Y.; Wang, W.; Li, J.; Hu, B.; Gao, S. IMU-Based Monitoring for Assistive Diagnosis and Management of IoHT: A Review. Healthcare 2022, 10, 1210. [Google Scholar] [CrossRef]
- Shuvra, S.B.; Deprettere, F.E.; Leupers, R.; Takala, J. (Eds.) Bhattacharyya, Handbook of Signal Processing Systems, 2nd ed.; Springer: New York, NY, USA, 2013. [Google Scholar]
- Andrenacci, I.; Boccaccini, R.; Bolzoni, A.; Colavolpe, G.; Costantino, C.; Federico, M.; Ugolini, A.; Vannucci, A. A Comparative Evaluation of Inertial Sensors for Gait and Jump Analysis. Sensors 2021, 21, 5990. [Google Scholar] [CrossRef] [PubMed]
- Milosevic, B.; Leardini, A.; Farella, E. Kinect and wearable inertial sensors for motor rehabilitation programs at home: State of the art and an experimental comparison. Biomed. Eng. Online 2020, 19, 25. [Google Scholar] [CrossRef]
- Demeco, A.; de Sire, A.; Marotta, N.; Spanò, R.; Lippi, L.; Palumbo, A.; Iona, T.; Gramigna, V.; Palermi, S.; Leigheb, M.; et al. Match Analysis, Physical Training, Risk of Injury and Rehabilitation in Padel: Overview of the Literature. Int. J. Environ. Res. Public Health 2022, 19, 4153. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. STROBE Initiative the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef]
- Beausoleil, S.; Miramand, L.; Turcot, K. Evolution of gait parameters in individuals with a lower-limb amputation during a six-minute walk test. Gait Posture 2019, 72, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Maqbool, H.F.; Husman, M.A.B.; Awad, M.I.; Abouhossein, A.; Iqbal, N.; Dehghani-Sanij, A.A. A Real-Time Gait Event Detection for Lower Limb Prosthesis Control and Evaluation. IEEE Trans. Neural Syst. Rehabil. Eng. 2017, 25, 1500–1509. [Google Scholar] [CrossRef] [PubMed]
- Seel, T.; Raisch, J.; Schauer, T. IMU-based joint angle measurement for gait analysis. Sensors 2014, 14, 6891–6909. [Google Scholar] [CrossRef] [PubMed]
- Wentink, E.C.; Schut, V.G.H.; Prinsen, E.C.; Rietman, J.S.; Veltink, P.H. Detection of the onset of gait initiation using kinematic sensors and EMG in transfemoral amputees. Gait Posture 2014, 39, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Simonetti, E.; Bergamini, E.; Vannozzi, G.; Bascou, J.; Pillet, H. Estimation of 3D Body Center of Mass Acceleration and Instantaneous Velocity from a Wearable Inertial Sensor Network in Transfemoral Amputee Gait: A Case Study. Sensors 2021, 21, 3129. [Google Scholar] [CrossRef]
- Paradisi, F.; Di Stanislao, E.; Summa, A.; Brunelli, S.; Traballesi, M.; Vannozzi, G. Upper body accelerations during level walking in transtibial amputees. Prosthet. Orthot. Int. 2019, 43, 204–212. [Google Scholar] [CrossRef]
- Dauriac, B.; Bonnet, X.; Pillet, H.; Lavaste, F. Estimation of the walking speed of individuals with transfemoral amputation from a single prosthetic shank-mounted IMU. Proc. Inst. Mech. Eng. H J. Eng. Med. 2019, 233, 931–937. [Google Scholar] [CrossRef]
- Major, M.J.; Raghavan, P.; Gard, S. Assessing a low-cost accelerometer-based technique to estimate spatial gait parameters of lower-limb prosthesis users. Prosthet. Orthot. Int. 2016, 40, 643–648. [Google Scholar] [CrossRef]
- Howcroft, J.; Lemaire, E.D.; Kofman, J.; Kendell, C. Understanding dynamic stability from pelvis accelerometer data and the relationship to balance and mobility in transtibial amputees. Gait Posture 2015, 41, 808–812. [Google Scholar] [CrossRef]
- Lamoth, C.J.C.; Ainsworth, E.; Polomski, W.; Houdijk, H. Variability and stability analysis of walking of transfemoral amputees. Med. Eng. Phys. 2010, 32, 1009–1014. [Google Scholar] [CrossRef]
- Tura, A.; Raggi, M.; Rocchi, L.; Cutti, A.G.; Chiari, L. Gait symmetry and regularity in transfemoral amputees assessed by trunk accelerations. J. Neuroeng. Rehabil. 2010, 7, 4. [Google Scholar] [CrossRef]
- Clemens, S.; Kim, K.J.; Gailey, R.; Kirk-Sanchez, N.; Kristal, A.; Gaunaurd, I. Inertial sensor-based measures of gait symmetry and repeatability in people with unilateral lower limb amputation. Clin. Biomech. 2020, 72, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Daines, K.J.F.; Baddour, N.; Burger, H.; Bavec, A.; Lemaire, E.D. Fall risk classification for people with lower extremity amputations using random forests and smartphone sensor features from a 6-minute walk test. PLoS ONE 2021, 16, e0247574. [Google Scholar] [CrossRef] [PubMed]
- Shawen, N.; Lonini, L.; Mummidisetty, C.K.; Shparii, I.; Albert, M.V.; Kording, K.; Jayaraman, A. Fall Detection in Individuals with Lower Limb Amputations Using Mobile Phones: Machine Learning Enhances Robustness for Real-World Applications. JMIR mHealth uHealth 2017, 5, e151. [Google Scholar] [CrossRef] [PubMed]
- Hordacre, B.; Barr, C.; Crotty, M. Community activity and participation are reduced in transtibial amputee fallers: A wearable technology study. BMJ Innov. 2015, 1, 10. [Google Scholar] [CrossRef]
- Kapti, A.O.; Muhurcu, G. Wearable acceleration sensor application in unilateral trans-tibial amputation prostheses. Biocybern. Biomed. Eng. 2014, 34, 53–62. [Google Scholar] [CrossRef]
- Chang, M.; Kim, K.; Jeon, D. Research on Terrain Identification of the Smart Prosthetic Ankle by Fuzzy Logic. IEEE Trans. Neural Syst. Rehabil. Eng. 2019, 27, 1801–1809. [Google Scholar] [CrossRef] [PubMed]
- Su, B.-Y.; Wang, J.; Liu, S.-Q.; Sheng, M.; Jiang, J.; Xiang, K. A CNN-Based Method for Intent Recognition Using Inertial Measurement Units and Intelligent Lower Limb Prosthesis. IEEE Trans. Neural Syst. Rehabil. Eng. 2019, 27, 1032–1042. [Google Scholar] [CrossRef]
- Keri, M.-I.; Shehata, A.W.; Marasco, P.D.; Hebert, J.S.; Vette, A.H. A Cost-Effective Inertial Measurement System for Tracking Movement and Triggering Kinesthetic Feedback in Lower-Limb Prosthesis Users. Sensors 2021, 21, 1844. [Google Scholar] [CrossRef]
- Krasoulis, A.; Vijayakumar, S.; Nazarpour, K. Multi-Grip Classification-Based Prosthesis Control with Two EMG-IMU Sensors. IEEE Trans. Neural Syst. Rehabil. Eng. 2020, 28, 508–518. [Google Scholar] [CrossRef]
- Sharba, G.K.; Wali, M.K.; Ai-Timemy, A.H. Real-time classification of shoulder girdle motions for multifunctional prosthetic hand control: A preliminary study. Int. J. Artif. Organs 2019, 42, 508–515. [Google Scholar] [CrossRef]
- Ladlow, P.; Nightingale, T.E.; McGuigan, M.P.; Bennett, A.N.; Phillip, R.D.; Bilzon, J.L.J. Predicting ambulatory energy expenditure in lower limb amputees using multi-sensor methods. PLoS ONE 2019, 14, e0209249. [Google Scholar] [CrossRef] [PubMed]
- Ladlow, P.; Nightingale, T.E.; McGuigan, M.P.; Bennett, A.N.; Phillip, R.; Bilzon, J.L.J. Impact of anatomical placement of an accelerometer on prediction of physical activity energy expenditure in lower-limb amputees. PLoS ONE 2017, 12, e0185731. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.D.; Guerra, G. Quantifying Step Count and Oxygen Consumption with Portable Technology during the 2-Min Walk Test in People with Lower Limb Amputation. Sensors 2021, 21, 2080. [Google Scholar] [CrossRef]
- Desveaux, L.; Goldstein, R.S.; Mathur, S.; Hassan, A.; Devlin, M.; Pauley, T.; Brooks, D. Physical Activity in Adults with Diabetes Following Prosthetic Rehabilitation. Can. J. Diabetes 2016, 40, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Wensman, J.; Colabianchi, N.; Gates, D.H. The influence of powered prostheses on user perspectives, metabolics, and activity: A randomized crossover trial. J. Neuroeng. Rehabil. 2021, 18, 49. [Google Scholar] [CrossRef]
- van Velzen, J.M.; van Bennekom, C.A.; Polomski, W.; Slootman, J.R.; van der Woude, L.H.; Houdijk, H. Physical capacity and walking ability after lower limb amputation: A systematic review. Clin. Rehabil. 2006, 20, 999–1016. [Google Scholar] [CrossRef]
- Quigley, M.; Dillon, M.P. Quality of life in persons with partial foot or transtibial amputation: A systematic review. Prosthet. Orthot. Int. 2016, 40, 18–30. [Google Scholar] [CrossRef]
- Esquenazi, A. Gait analysis in lower-limb amputation and prosthetic rehabilitation. Phys. Med. Rehabil. Clin. N. Am. 2014, 25, 153–167. [Google Scholar] [CrossRef]
- Wasser, J.G.; Vincent, K.R.; Herman, D.C.; Vincent, H.K. Potential lower extremity amputation-induced mechanisms of chronic low back pain: Role for focused resistance exercise. Disabil. Rehabil. 2020, 42, 3713–3721. [Google Scholar] [CrossRef]
- Simonetti, E.; Villa, C.; Bascou, J.; Vannozzi, G.; Bergamini, E.; Pillet, H. Gait event detection using inertial measurement units in people with transfemoral amputation: A comparative study. Med. Biol. Eng. Comput. 2020, 58, 461–470. [Google Scholar] [CrossRef]
- Zhang, F.; Dou, Z.; Nunnery, M.; Huang, H. Real-time implementation of an intent recognition system for artificial legs. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; Volume 2011, pp. 2997–3000. [Google Scholar] [CrossRef]
- Varrecchia, T.; Serrao, M.; Rinaldi, M.; Ranavolo, A.; Conforto, S.; De Marchis, C.; Simonetti, A.; Poni, I.; Castellano, S.; Silvetti, A.; et al. Common and specific gait patterns in people with varying anatomical levels of lower limb amputation and different prosthetic components. Hum. Mov. Sci. 2019, 66, 9–21. [Google Scholar] [CrossRef]
- Isakov, E.; Keren, O.; Benjuya, N. Trans-tibial amputee gait: Time-distance parameters and EMG activity. Prosthet. Orthot. Int. 2000, 24, 216–220. [Google Scholar] [CrossRef]
- Uchytil, J.; Jandacka, D.; Zahradnik, D.; Farana, R.; Janura, M. Temporal-spatial parameters of gait in transfemoral amputees: Comparison of bionic and mechanically passive knee joints. Prosthet. Orthot. Int. 2014, 38, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Lythgo, N.; Marmaras, B.; Connor, H. Physical function, gait, and dynamic balance of transfemoral amputees using two mechanical passive prosthetic knee devices. Arch. Phys. Med. Rehabil. 2010, 91, 1565–1570. [Google Scholar] [CrossRef]
- Hak, L.; van Dieën, J.H.; van der Wurff, P.; Prins, M.R.; Mert, A.; Beek, P.J.; Houdijk, H. Walking in an Unstable Environment: Strategies Used by Transtibial Amputees to Prevent Falling during Gait. Arch. Phys. Med. Rehabil. 2013, 94, 2186–2193. [Google Scholar] [CrossRef] [PubMed]
- Kendell, C.; Lemaire, E.D.; Dudek, N.L.; Kofman, J. Indicators of dynamic stability in transtibial prosthesis users. Gait Posture 2010, 31, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Silverman, A.K.; Neptune, R.R. Muscle and prosthesis contributions to amputee walking mechanics: A modeling study. J. Biomech. 2012, 45, 2271–2278. [Google Scholar] [CrossRef] [PubMed]
- Harandi, V.J.; Ackland, D.C.; Haddara, R.; Lizama, L.E.C.; Graf, M.; Galea, M.P.; Lee, P.V.S. Gait compensatory mechanisms in unilateral transfemoral amputees. Med. Eng. Phys. 2020, 77, 95–106. [Google Scholar] [CrossRef]
- Hoskin, A.F. Fatal falls: Trends and characteristics. Stat. Bull. 1998, 79, 10–15. [Google Scholar]
- Vu, K.; Payne, M.W.C.; Hunter, S.W.; Viana, R. Risk Factors for Falls in Individuals with Lower Extremity Amputations during the Pre-Prosthetic Phase: A Retrospective Cohort Study. PM&R 2019, 11, 828–833. [Google Scholar] [CrossRef]
- Steinberg, N.; Gottlieb, A.; Siev-Ner, I.; Plotnik, M. Fall incidence and associated risk factors among people with a lower limb amputation during various stages of recovery—A systematic review. Disabil. Rehabil. 2019, 41, 1778–1787. [Google Scholar] [CrossRef]
- Hausdorff, J.M. Gait variability: Methods, modeling and meaning. J. Neuroeng. Rehabil. 2005, 2, 19. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.B.; Miller, M.J.; Murray, A.M.; Fields, T.T.; So, N.F.; Christiansen, C.L. Falls After Dysvascular Transtibial Amputation: A Secondary Analysis of Falling Characteristics and Reduced Physical Performance. PM&R 2021, 13, 19–29. [Google Scholar] [CrossRef]
- Subramaniam, S.; Faisal, A.I.; Deen, M.J. Wearable Sensor Systems for Fall Risk Assessment: A Review. Front. Digit. Health 2022, 4, e921506. [Google Scholar] [CrossRef] [PubMed]
- Aziz, O.; Klenk, J.; Schwickert, L.; Chiari, L.; Becker, C.; Park, E.J.; Mori, G.; Robinovitch, S.N. Validation of accuracy of SVM-based fall detection system using real-world fall and non-fall datasets. PLoS ONE 2017, 12, e0180318. [Google Scholar] [CrossRef] [PubMed]
- Scheurer, S.; Koch, J.; Kucera, M.; Bryn, H.; Bärtschi, M.; Meerstetter, T.; Nef, T.; Urwyler, P. Optimization and Technical Validation of the AIDE-MOI Fall Detection Algorithm in a Real-Life Setting with Older Adults. Sensors 2019, 19, 1357. [Google Scholar] [CrossRef] [PubMed]
- Flynn, L.; Geeroms, J.; Jimenez-Fabian, R.; Heins, S.; Vanderborght, B.; Munih, M.; Molino Lova, R.; Vitiello, N.; Lefeber, D. The Challenges and Achievements of Experimental Implementation of an Active Transfemoral Prosthesis Based on Biological Quasi-Stiffness: The CYBERLEGs Beta-Prosthesis. Front. Neurorobot. 2018, 12, 80. [Google Scholar] [CrossRef]
- Shultz, A.H.; Lawson, B.E.; Goldfarb, M. Variable Cadence Walking and Ground Adaptive Standing with a Powered Ankle Prosthesis. IEEE Trans. Neural Syst. Rehabil. Eng. 2016, 24, 495–505. [Google Scholar] [CrossRef]
- Nakamura, B.H.; Hahn, M.E. Myoelectric Activation Pattern Changes in the Involved Limb of Individuals with Transtibial Amputation during Locomotor State Transitions. Arch. Phys. Med. Rehabil. 2017, 98, 1180–1186. [Google Scholar] [CrossRef]
- Waters, R.L.; Mulroy, S. The energy expenditure of normal and pathologic gait. Gait Posture 1999, 9, 207–231. [Google Scholar] [CrossRef]
- Robins, K.; Stankorb, S.M.; Salgueiro, M. Energy expenditure in acute posttraumatic amputation: Comparison of four methods for assessment. Nutr. Clin. Pract. 2013, 28, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Brage, S.; Brage, N.; Ekelund, U.; Luan, J.; Franks, P.W.; Froberg, K.; Wareham, N.J. Effect of combined movement and heart rate monitor placement on physical activity estimates during treadmill locomotion and free-living. Eur. J. Appl. Physiol. 2006, 96, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Göktepe, A.S.; Cakir, B.; Yilmaz, B.; Yazicioglu, K. Energy expenditure of walking with prostheses: Comparison of three amputation levels. Prosthet. Orthot. Int. 2010, 34, 31–36. [Google Scholar] [CrossRef] [PubMed]
| Article | Study Design | Participants | Aim | Procedure | Outcome |
|---|---|---|---|---|---|
| Beausoleil S. et al., 2019 [34] | Case Study | 15 LLA Patients | To assess kinematic gait parameters during 6MWT and clinical applicability of IMU. | Post-Rehab assessment of gait, during 6MWT, with IMU (3D Acc + 3D Gyro) on both feet. | High stance and cadence variability on both limbs. High and variable minimal Toe clearance on AL. Gait kinematic parameters variability are correlated with future falls. Relevant IMU applicability in clinical context. |
| Maqbool H.F. et al., 2017 [35] | Controlled Clinical Trial | 8 Healthy Control, 1 TFA Patients, 1 TTA Patient | To evaluate the reliability of RT Gait event detection algorithm in both flat and inclined surfaces for TFA patients. | Patients walked for 10 min at self-selected walking speed over flat surfaces and walk up and down a ramp (5° inclination) with a 6-DOF IMU (Acc + Gyro) fixed on the shank and insole with footswitches. The algorithm was written in MAT-lab. | 100% detection accuracy for Initial Contact and Toe Off with different prostheses. Reliable algorithm for gait event detection. |
| Seel T. et al., 2014 [36] | Case Study | 1 TFA Patients | To asses Joint Angle using IMUs and validate it’s measure with a 3D optoelectronic movement detection system (MDS). | IMUs (Xsense MTw) mounted on proximal and distal legs as well as foots. Body marker for 3D MDS (Vicon V612) mounted on both legs. Participant is requested to walk 10 m at self-selected speed. Data are gathered and confronted with 3D MDS | Joint angle calculation using accelerometer and gyroscope showed high precision and correlation with 3D MDS (1° RMSE for prosthetic leg and 3° for contralateral). |
| Wentinik E.C. et al., 2014 [37] | Case Study | 3 TFA, 3 TKA | To detect onset of gait with IMUs. | Footswitches positioned in the heel center and under the first metatarsal head. Two inertial sensors (Xsens, Acc + Gyro) placed on anterior side of the proximal and distal leg. sEMG electrodes placed on residual muscles of the amputated leg. Each patient is asked to walk. | IMUs demonstrated reliable in detecting onset of gait in both healthy (Gyro) and prosthetic limb (Acc). |
| Simonetti E. et al., 2021 [38] | Case Study | 1 TFA | To use a framework of MIMUs to evaluate BCoM acceleration and instantaneous velocity. Validate the measure versus a 3D MDS. | Full body marker set + 7 MIMUs on feet, shanks, thighs and trunk. The participant is requested to walk at self-selected speed through an 8 m path with 3 force platform in the middle. | Moderate to Strong correlation between MIMUs and Force Platform for SCoMs and BCoM acceleration and velocity. |
| Paradisi F. et al., 2019 [39] | Case-Control Study | 20 TTA,20 Healthy Control | To investigate upper body acceleration and how these propagate from pelvis to head. | 3 MIMUs located at head, sternum, and lumbo-pelvic segment. Participants were asked to walk thorough a 10 m pathway at self-selected speed. | Amputees have a larger coefficient of attenuation of acceleration from pelvis to sternum, greater medio-lateral and head acceleration. Attenuation coefficient may be a useful index for mobility assessment in LLA. |
| Dauriac B. et al., 2019 [40] | Case study | 9 TFA | To evaluate the walking speed by estimating COM speed during gait cycle using a single IMU integrated in a microprocessor-controlled knee ankle prosthesis. | Several sped and slop conditions were tested at treadmill | This method estimates the walking speed with a 9% of RMSE in patients walking on a treadmill with 0° slope. The RMSE slightly increased when the slope is taken to 5% (but still acceptable). |
| Major M.J. et al., 2016 [41] | Controlled Trial | 20 LLP (8 TFA, 9 TTA, 2 TT/TTA, 1 TT/TFA), 5 Healthy Control | To asses step length (SL) in patients with LLP. | A three-axis accelerometer was fixed at lumbar level in 20 LLP. The patients were asked to walk in a 20-m pathway from a standing position to a complete stop. | SL was correlated positively with previous literature study. Method was validated only on Healthy subject but not for LLP users. |
| Howcroft J. et al., 2014 [42] | Case study | 11 TTA | To investigate if accelerometer derivate measures can differentiate between dynamic states and how those data correlate with clinical measures scores. | Community Balance and Mobility scales, Balance Berg scales, Prosthesis Evaluation Questionnaire were administered to the participants. An inertial sensor was affixed to the pelvis and then the participants walked in two scenarios: a 10-metre path on level ground (LG) and an 8-metre path covered by foam mattresses (uneven ground—UG). | Statistically significant differences were found between LG and UG walking in TTA participants. Stride time, vertical and AP acceleration FFT first quartile and ML Harmonic ratio were greater in UG than LG. Vertical acceleration and cadence were greater in LG than UG. ML acceleration range, AP acceleration standard deviation and stride time were correlated with change in clinical outcome measures scales. |
| Lamoth C.J. et al., 2010 [43] | Controlled Trial | 8 TFA, 8 Healthy Control | To asses variability and stability of gait in LLA patients and healthy subjects. | All participants were equipped with a tri-axial accelerometer and walked for 6 min in various context: (i) indoor walking, (ii) indoor walking with cognitive dual tasks, (iii) outside walking (even terrain) in a square circuit (260 m long), (iv) outside walking (uneven terrain) in a square circuit (260 m long). | There was significant statistically differences in trunk acceleration (variability on ML acceleration) and walking speed (LLA patients are slower than healthy subject) in amputees’ group. Those two parameters are directly correlated with stability of the gait. |
| Tura A. et al., 2010 [44] | Controlled Trial | 10 TFA, 10 Healthy Control | To evaluate a method for assessing gait regularity and symmetry of LLP users using a single accelerometer. | All participants were equipped with a single tri-axial accelerometer mounted at thoracic level and foot insoles. Patients are asked to walk a straight path 70 m long at natural, lower and faster speed. Step and stride regularity and duration were used to determine symmetry and regularity of the gait. | Step and stride regularity and step and stride duration are good index of regularity and symmetry of gait. A single accelerometer is capable to determine these parameters with good sensibility and specificity. |
| Clemens S. et al., 2020 [45] | Cohort Study | 65 TTA, 63 TFA | To evaluate test-retest reliability of IMU based measures of segmental symmetry between lower limbs and differences between TTA and TFA in segmental symmetry score (SSS) and segmental repeatability score (SRS). | Participants wore knee sleeves equipped with 4 IMUs. They were asked to undergo a 10MWT on a Zeno Electronic Walkway system. Using sagittal angular velocities of thigh and shank SSS and SRS where calculated. | Good test-retest reliability, can differentiate between healthy and AL. Cannot differentiate between TFA and TTA. |
| Daines K.J.F. et al., 2021 [46] | Cohort Study | 89 LLA (4 BiTTA, 1 TT/TFA, 63 TTA, 18 TFA, 2 TKA, 1 TAA) | To evaluate if the use of a random forest classificator is able to classify risk of fall in LLA. | An android smartphone was placed in posterior pelvis. All patients performed a 6MWT in a 20 m pathway. Data were collected in a custom-made application installed on to the smartphone. | Random forest classificator applied to data collected with a smartphone showed a good specificity (near 95%), good accuracy (81.3%) in classifying risk of fall in LLA patients. |
| Shawen N. et al., 2017 [47] | Controlled Trial | 7 TFA, 10 Healthy Controls | To develop a classifier that integrates data from healthy participants to detect falls in individual with LLA. | All participants carried a Galaxy S4 Smartphone (Acc + Gyro) in natural position (pocket, hand, waist) during activities of daily living. 3 LLA participants took the phone with them for three days for quantifying false alarms. | Using a machine learning approach, data recorded from smartphones regarding angular and linear accelerations of healthy subjects can be used to classify falls risk in LLA subjects more specifically than a threshold approach (2 false alarms vs. 122). |
| Hordacre B. et al., 2015 [48] | Cohort Study | 47 TFA | To asses activity and participation at home and various settings both for fallers and non- fallers LLA. | All participants were equipped with a stepwatch 3 activity monitor sensor and a GPS linked to the prosthesis. The community activity was defined as counting steps outside the house in various settings, and the home activity as counting steps inside the house. Participation was defined as an event in which participants had to leave home. | A statistically significant difference was demonstrated between LLA fallers versus non-fallers participants for commercial activity, recreational activity and total community activity. In addition, a statistically significant difference was found in recreational and total community participation. |
| Kapti et al., 2013 [49] | Case study | 1 Healthy Subject | To investigate the use of accelerometric data recorded from TTA for trajectory control of an experimental active ankle joint prosthesis | Two acceleration sensors were used to register AP, ML and Vertical acceleration on the sound leg. | Data acquired from registration from the sound leg of a TTA may be used for controlling the trajectory of an LLP with active ankle joint users. |
| Chang M. et al., 2019 [50] | Case Study | 4 TTA | To use a fuzzy logic system for terrain detection and automatic prosthetic ankle angle correction. | All participants wore a prosthesis with smart ankle system (equipped with an IMU sensor and a load cell for GRF detection) and walked on five different terrain condition (flat, upslope, downslope, upstairs and downstair) for at least 20 steps. | This fuzzy logic system had a 97.5% accuracy in terrain detection. |
| Su B.Y. et al., 2019 [51] | Case Study | 1 TFA, 10 Healthy Participants | To evaluate a new method for training and intent recognition system using Convolutional Neural Network (CNN) algorithm. | Three IMUs were positioned on thigh, shank and ankle of the healthy leg. All participants were instructed to walk at a comfortable speed and walked among different motion states as well as steady state. | CNN can be used effectively for intent recognition with a system of 3 IMUs, and potentially to control a powered prosthesis for allowing natural transition trough motion states. |
| Keri MI et al., 2021 [52] | Case Study | 1 TFA | To develop a low cost IMU based vibratory feedback system and use it to trigger prosthesis motion illusion (kinesthetics illusion -KI). | Vibratory feedback system (VFS) was composed with: an Arduino microcontroller, two 3-DOF Gyroscope, a lithium battery, a vibratory actuator. The accuracy of the VFS is quantified using an MDS and commercial IMU. Vibratory actuator was fixed on thigh and IMUs to a Robotic arm. | Participant in this study experienced KI for 16 degrees in knee flexion. Illusion of motion may improve gait parameters and reduce risk of falling. |
| Krasoulis A. et al., 2019 [53] | Controlled Trial | 12 Healthy controls, 2 TRA | To develop a multi-grip classification system for prosthesis control in TRP users. | For HC 16 EMG-IMU sensors were placed in two 8 Sensor row on the forearm. For TRA 12 and 13 sensor were placed on the stump. Participants were asked to execute different grasp for calibration (power grasp, lateral grasp, tripod grasp, index pointer and hand opening). Consequentially they were asked to pick an object that stimulate a specific grasp. | Authors developed a multi-grip classification system using only two EMG-IMU sensors that can be used for real time prosthesis control during grip tasks. |
| Sharba G.K. et al., 2019 [54] | Controlled Trial | 4 Healthy Control, 1 HlULA | To develop a RT shoulder girdle movement classifier to help high level ULA to control a prosthetic hand. | EMG and 3DOF Acc. were fixed on shoulder girdle of all participants. A set of five motions were chosen for classification: (i) elevation, (ii) depression, (iii) protraction, (iv) retraction and rest. The above classification was the used to control elbow, wrist and fingers of a 3D printed prosthesis. | Results showed a 92.8% accuracy in classifying shoulder girdle movement of the ULA participants. Classification was used to control a 3D printed UL prosthesis. |
| Ladlow P. et al., 2019 [55] | Controlled Trial | 19 LLA (9 ULLA and 10 BiLLA), 9 Healthy Control | To asses validity of an algorithm combining data from accelerometer and HR (GT3X+ + Polar T31) monitor to assess energy expenditure (EE) during Physical Activity versus Actiheart Monitor (AHR) | All participants wore a Metamax 3B mask for Indirect calorimetry and were equipped with an AHR and a Polar T31 HR monitor. An Actigraph GTX3+ (3-DOF Acc) mounted on the waist near the shortest residual limb. All participants, then, are asked to walk on a treadmill at 5 progressive velocities and two slope (2% and 5%). Physiological Cost Index is then calculated (ΔHR/Walking speed). | The use of integrated Acc. data and HR data provided the most valid estimation of EE in ambulatorial setting for both amputation group. Level amputation impacts on accuracy of predicting EE. |
| Ladlow P. et al., 2017 [56] | Controlled trial | 10 ULLA,10 BiLLA, 10 Healthy Control | To assess the impact of anatomical positioning of GT3X+ activity monitor in LLA participants and to develop algorithm on predicting EE. | All participants wore a Metamax 3B mask for Indirect calorimetry and were equipped with a GT3X+ activity monitor on either side of the waist above the hip and at L2 level. Participants were asked to walk on a treadmill at 5 progressive speeds. Moreover, all participants performed a sitting-based arm crank ergometry. | The anatomical positioning of accelerometers impacts the ability to predict EE in LLA. The positioning that better correlates with EE is on the amputated side of the waist, just above the hip. |
| Smith J.D. et al., 2021 [57] | Cross-sectional Study | 23 TTA 9 TFA 3 BiTTA | To determine step count and step count accuracy with different activity monitor and O2 consumption during a 2MWT. | All participants were equipped with an Actigraph GT9X+ and a Garmin Vivofit ® 3 both on wrist and ankle of the non-dominant side. A modus Stepwatch 4 is placed on the non-dominant ankle in addition to the above sensors. All Participants are fitted with a Polar HR sensor and a Cosmed 5 portable metabolic analyzer. After three minutes sitting, participants performed a 2MWT as fast and safetly possible. | There were no differences in distance walked, VO2, HR and RPE between different amputation level. Step count and cadence were greater in TTA vs. TFA. Stepwatch on the ankle and Vivofit on the wrist provided the most accurate step count. |
| Desveaux L. et al., 2016 [58] | Cross-sectional | 15 TTA | To asses if TTA patients with diabetes meet recommended level of physical activity and daily steps count. To investigate if physical functioning measures are correlated with objective measures of physical activity. | Participants were provided with a Stepwatch activity monitor (SAM) fixed around the ankle of intact limb. Participants were asked to wear SAM for 9 consecutive days. Physical activity was measured by steps count and number of minutes engaging activity involving >90 steps/min. Each participants underwent a 2MWT and performed an L test. Activities-specific Balance Confidence Scale (ABC) and WHO QoL-Brief Questionnaire were administered. | Despite improvement in functional mobility (L test) over 6-month follow-up, step count were below 6500/day and participants spent <150 min/week for vigorous physical activity (>90 steps/min). These results indicate the needs of post-rehabilitation intervention to promote active lifestyles. |
| Kim J. et al., 2021 [59] | Randomized Cross-over Trial | 10 TTA | To quantifying metabolic cost, step count, walking, perception of mobility and quality of life between powered and non-powered prostheses users. | Participants were randomly assigned to perform testing with a powered prosthesis or with an unpowered prosthesis. All participants were equipped with two ActiGraph GT9X Link (one mounted on the prosthetic foot and one mounted on the prosthetic pylon) and a GPS enabled system on their phone active for two weeks. At the end of the two weeks, data were collected and participants underwent a metabolic measurement with Kosmed K4b2. | Authors did not find any differences in metabolic cost between powered prosthesis. |
| Articles | Criteria for the Quality Scoring | Score | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
| Beausoleil S. et al., 2019 [34] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 |
| Maqbool H.F. et al., 2017 [35] | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 7 |
| Seel T. et al., 2014 [36] | 1 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 1 | 0 | 5 |
| Wentink E.C. et al., 2014 [37] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 |
| Simonetti E. et al., 2021 [38] | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 9 |
| Paradisi F. et al., 2019 [39] | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 9 |
| Dauriac B. et al., 2019 [40] | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 9 |
| Major M.J. et al., 2016 [41] | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 9 |
| Howcroft J. et al., 2014 [42] | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 7 |
| Lamoth C.J. et al., 2010 [43] | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 7 |
| Tura A. et al., 2010 [44] | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 7 |
| Clemens S. et al., 2020 [45] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 9 |
| Daines K.J.F. et al., 2021 [14] | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 9 |
| Shawen N. et al., 2017 [47] | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 9 |
| Hordacre B. et al., 2015 [48] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 |
| Kapti A.O. et al., 2013 [49] | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 6 |
| Chang M. et al., 2019 [50] | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 5 |
| Su B.Y. et al., 2019 [51] | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 7 |
| Keri MI et al., 2021 [52] | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 8 |
| Krasoulis A. et al., 2019 [53] | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 7 |
| Sharba G.K. et al., 2019 [54] | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 7 |
| Ladlow P. et al., 2019 [55] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 |
| Ladlow P. et al., 2017 [56] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 |
| Smith J.D. et al., 2021 [57] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 |
| Desveaux L. et al., 2016 [58] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 9 |
| Kim J. et al., 2021 [59] | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demeco, A.; Frizziero, A.; Nuresi, C.; Buccino, G.; Pisani, F.; Martini, C.; Foresti, R.; Costantino, C. Gait Alteration in Individual with Limb Loss: The Role of Inertial Sensors. Sensors 2023, 23, 1880. https://doi.org/10.3390/s23041880
Demeco A, Frizziero A, Nuresi C, Buccino G, Pisani F, Martini C, Foresti R, Costantino C. Gait Alteration in Individual with Limb Loss: The Role of Inertial Sensors. Sensors. 2023; 23(4):1880. https://doi.org/10.3390/s23041880
Chicago/Turabian StyleDemeco, Andrea, Antonio Frizziero, Christian Nuresi, Giovanni Buccino, Francesco Pisani, Chiara Martini, Ruben Foresti, and Cosimo Costantino. 2023. "Gait Alteration in Individual with Limb Loss: The Role of Inertial Sensors" Sensors 23, no. 4: 1880. https://doi.org/10.3390/s23041880
APA StyleDemeco, A., Frizziero, A., Nuresi, C., Buccino, G., Pisani, F., Martini, C., Foresti, R., & Costantino, C. (2023). Gait Alteration in Individual with Limb Loss: The Role of Inertial Sensors. Sensors, 23(4), 1880. https://doi.org/10.3390/s23041880










