AI-Enabled Advanced Development for Assessing Low Circulating Blood Volume for Emergency Medical Care: Comparison of Compensatory Reserve Machine-Learning Algorithms
Abstract
:1. Introduction
2. Methods
2.1. Subject Volunteers
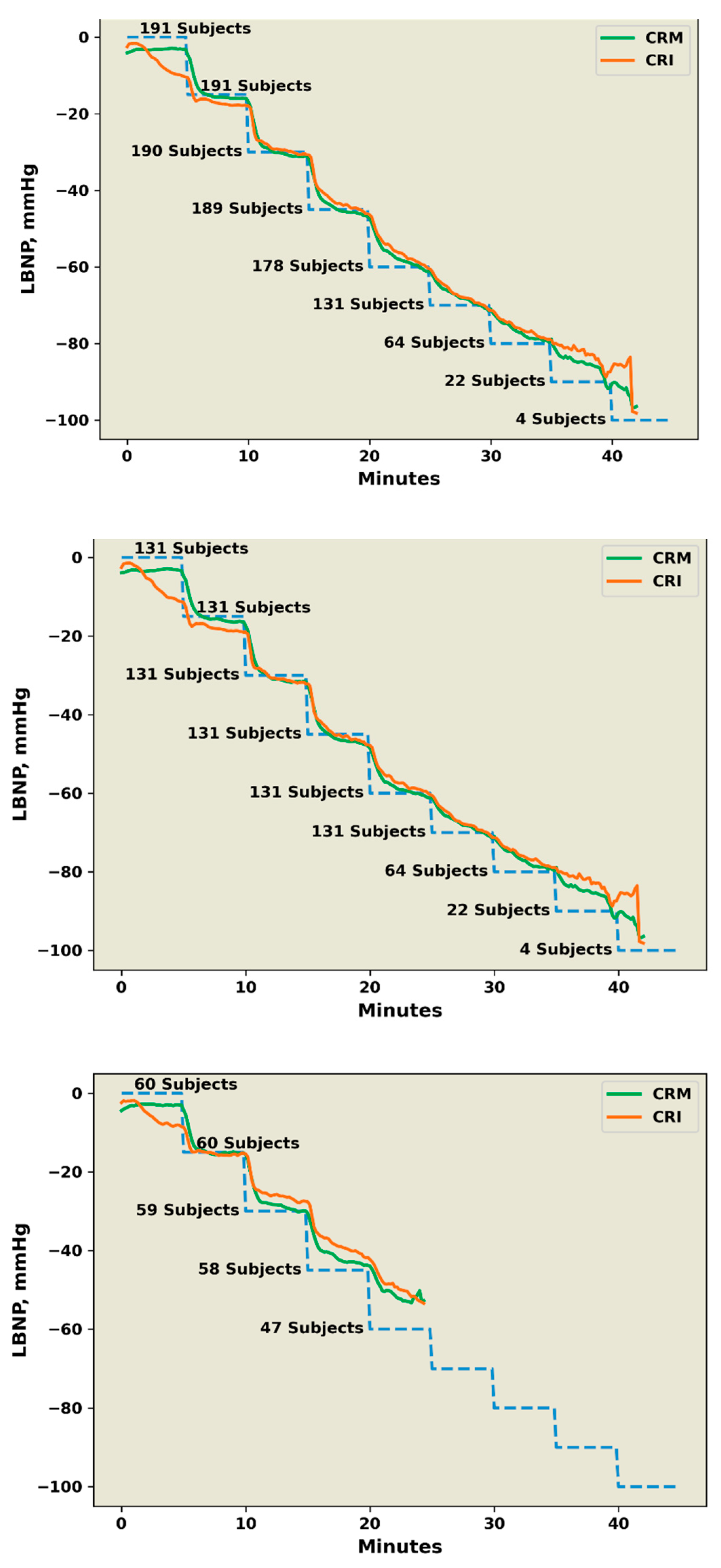
2.2. Experimental Protocol
2.3. Measuring the Compensatory Reserve
2.3.1. Compensatory Reserve Index (CRI)
2.3.2. Compensatory Reserve Metric (CRM)
2.4. High versus Low Tolerance Classification
2.5. Statistical Analysis
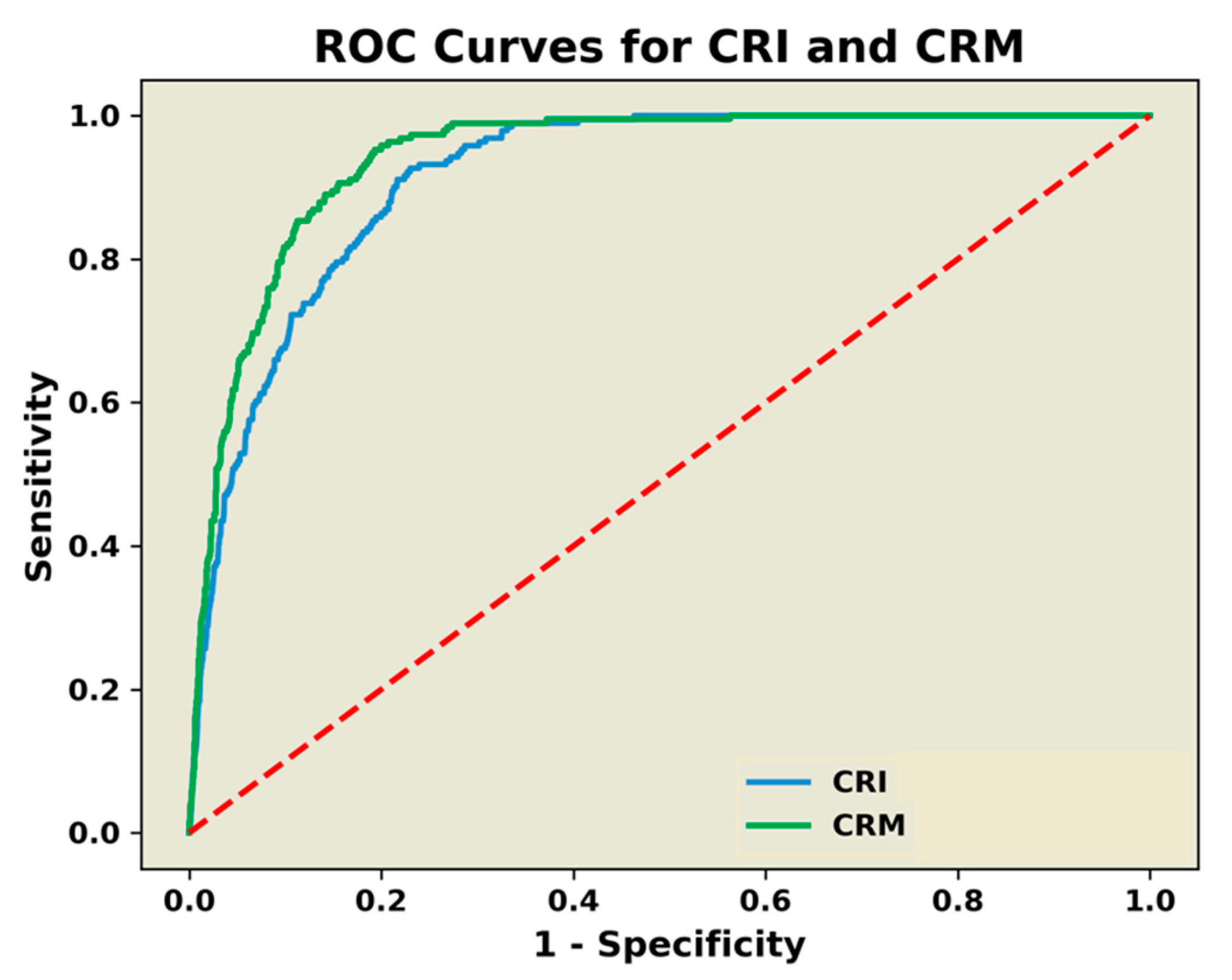
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Convertino, V.A.; Koons, N.J.; Suresh, M. The physiology of human hemorrhage and compensation. Comp. Physiol. 2020, 11, 1531–1574. [Google Scholar]
- Convertino, V.A.; Schauer, S.G.; Weitzel, E.K.; Cardin, S.; Stackle, M.E.; Talley, M.J.; Sawka, M.N.; Inan, O.T. Wearable sensors integrated with compensatory reserve monitoring in critically injured trauma patients. Sensors 2020, 20, 6413. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A.; Koons, N.J. The compensatory reserve: Potential for accurate individualized goal-directed whole blood resuscitation. Transfusion 2020, 60, S150–S157. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A.; Grudic, G.; Mulligan, J.; Moulton, S. Estimation of individual-specific progression to impending cardiovascular instability using arterial waveforms. J. Appl. Physiol. 2013, 115, 1196–1202. [Google Scholar] [CrossRef]
- Convertino, V.A.; Wirt, M.D.; Glenn, J.F.; Lein, B.C. The compensatory reserve for early and accurate prediction of hemodynamic compromise: A review of the underlying physiology. Shock 2016, 45, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A.; Schiller, A.M. Measuring the compensatory reserve to identify shock. J. Trauma Acute Care Surg. 2017, 82, S57–S65. [Google Scholar] [CrossRef]
- Moulton, S.L.; Mulligan, J.; Grudic, G.Z.; Convertino, V.A. Running on empty? The compensatory reserve index. J. Trauma Acute Care Surg. 2013, 75, 1053–1059. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Convertino, V.A.; Cardin, S.; Batchelder, P.; Grudic, G.Z.; Mulligan, J.; Moulton, S.L.; MacLeod, D. A novel measurement for accurate assessment of clinical status in patients with significant blood loss: The compensatory reserve. Shock 2015, 44, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Howard, J.T.; Janak, J.C.; Hinojosa-Laborde, C.; Convertino, V.A. Specificity of compensatory reserve and tissue oxygenation as early predictors of tolerance to progressive reductions in central blood volume. Shock 2016, 46, 68–73. [Google Scholar] [CrossRef]
- Janak, J.C.; Howard, J.T.; Goei, K.A.; Weber, R.; Muniz, G.W.; Hinojosa-Laborde, C.; Convertino, V.A. Predictors of the onset of hemodynamic decompensation during progressive central hypovolemia: Comparison of the peripheral perfusion index, pulse pressure variability, and compensatory reserve index. Shock 2015, 44, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Schiller, A.M.; Howard, J.T.; Lye, K.R.; Magby, C.G.; Convertino, V.A. Comparisons of traditional metabolic markers and compensatory reserve as early predictors of tolerance to central hypovolemia in humans. Shock 2018, 50, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.L.; Mulligan, J.; Grudic, G.Z.; Convertino, V.A.; Moulton, S.L. Detection of low-volume blood loss: The compensatory reserve index versus traditional vital signs. J. Trauma Acute Care Surg. 2014, 77, 892–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schlotman, T.E.; Suresh, M.; Koons, N.J.; Howard, J.T.; Convertino, V.A. Predictors of hemodynamic decompensation in progressive hypovolemia: Compensatory reserve versus heart rate variability. J. Trauma Acute Care Surg. 2020, 89, S161–S168. [Google Scholar] [CrossRef]
- Convertino, V.A.; Wampler, M.R.; Johnson, M.; Alarhayem, A.; Le, T.D.; Nicholson, S.; Myers, J.G.; Chung, K.K.; Struck, K.R.; Cuenca, C.; et al. Validating clinical threshold values for a dashboard view of the compensatory reserve measurement for hemorrhage detection. J. Trauma Acute Care Surg. 2020, 89, S169–S174. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Alarhayem, A.; Convertino, V.; Carter, R., 3rd; Chung, K.; Stewart, R.; Myers, J.; Dent, D.; Liao, L.; Cestero, R.; et al. Compensatory reserve index: Performance of a novel monitoring technology to identify the bleeding trauma patient. Shock 2018, 49, 295–300. [Google Scholar] [CrossRef]
- Nadler, R.; Convertino, V.A.; Gendler, S.; Lending, G.; Lipsky, A.M.; Cardin, S.; Lowenthal, A.; Glassberg, E. The value of non-invasive mesurement of the compensatory reserve index in monitoring and triage of patients experiencing minimal blood loss. Shock 2014, 42, 93–98. [Google Scholar] [CrossRef]
- Benov, A.; Yaslowitz, O.; Hakim, T.; Amir-Keret, R.; Nadler, R.; Brand, A.; Glassberg, E.; Yitzhak, A.; Convertino, V.A.; Paran, H. The effect of blood transfusion on compensatory reserve: A prospective clinical trial. J. Trauma Acute Care Surg. 2017, 83, S71–S76. [Google Scholar] [CrossRef] [PubMed]
- Benov, A.; Brand, A.; Rozenblat, T.; Antebi, B.; Ben-Ari, A.; Amir-Keret, R.; Nadler, R.; Chen, J.; Chung, K.K.; Convertino, V.A.; et al. Evaluation of sepsis using compensatory reserve measurement: A prospective clinical trial. J. Trauma Acute Care Surg. 2020, 89, S153–S160. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A.; Johnson, M.C.; Alarhayem, A.; Nicholson, S.E.; Chung, K.K.; DeRosa, M.; Eastridge, B.J. Compensatory reserve detects subclinical phases of shock with more expeditious prediction for need of life-saving interventions compared to vital signs and arterial lactate. Transfusion 2021, 61, S167–S173. [Google Scholar] [CrossRef] [PubMed]
- Schauer, S.G.; April, M.D.; Arana, A.A.; Maddry, J.K.; Escandon, M.A.; Linscomb, C.D.; Rodriguez, D.C.; Convertino, V.A. Efficacy of the compensatory reserve measurement in an emergency department trauma population. Transfusion 2021, 61, S174–S182. [Google Scholar] [CrossRef]
- Schiller, A.M.; Howard, J.T.; Convertino, V.A. The physiology of blood loss and shock: New insights from a human laboratory model of hemorrhage. Exp. Biol. Med. 2017, 242, 874–883. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hinojosa-Laborde, C.; Shade, R.E.; Muniz, G.W.; Bauer, C.; Goei, K.A.; Pidcoke, H.F.; Chung, K.K.; Cap, A.P.; Convertino, V.A. Validation of lower body negative pressure as an experimental model of hemorrhage. J. Appl. Physiol. 2014, 116, 406–415. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A.; Rickards, C.A.; Ryan, K.L. Autonomic mechanisms associated with heart rate and vasoconstrictor reserves. Clin. Auton. Res. 2012, 22, 123–130. [Google Scholar] [CrossRef]
- Engelke, K.A.; Doerr, D.F.; Crandall, C.G.; Convertino, V.A. Application of acute maximal exercise to protect orthostatic tolerance after simulated microgravity. Am. J. Physiol. 1996, 271, R837–R847. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A. G-Factor as a tool in basic research: Mechanisms of orthostatic tolerance. J. Gravit. Physiol. 1999, 6, 73–76. [Google Scholar]
- Schlotman, T.E.; Akers, K.S.; Nessen, S.C.; Convertino, V.A. Differentiating compensatory mechanisms associated with low tolerance to central hypovolemia in women. Am. J. Physiol. Heart Circ. Physiol. 2019, 316, H609–H616. [Google Scholar] [CrossRef]
- Convertino, V.A. Blood pressure measurement for accurate assessment of patient status in emergency medical settings. Aviat. Space Environ. Med. 2012, 83, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A.; Moulton, S.L.; Grudic, G.Z.; Rickards, C.A.; Hinojosa-Laborde, C.; Ryan, K.L. Use of advanced machine-learning techniques for non-invasive monitoring of hemorrhage. J. Trauma 2011, 71, S25–S32. [Google Scholar]
- Hinojosa-Laborde, C.; Howard, J.T.; Mulligan, J.; Grudic, G.Z.; Convertino, V.A. Comparison of comensatory reserve during lower-body negative pressure and hemorrhage in nonhuman primates. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 310, R1154–R1159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goswami, N.; Blaber, A.; Hinghofer-Szalkay, H.; Convertino, V.A. Lower body negative pressure: Physiological effects, applications and implementations. Physiol. Rev. 2019, 99, 807–851. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.D.; van Helmond, N.; Curry, T.B.; van Buskirk, C.M.; Convertino, V.A.; Joyner, M.J. Reductions in central venous pressure by lower body negative pressure or blood loss elicit similar hemodynamic responses. J. Appl. Physiol. 2014, 117, 131–141. [Google Scholar] [CrossRef] [Green Version]
- Techentin, R.W.; Felton, C.L.; Schlotman, T.E.; Gilbert, B.K.; Joyner, M.J.; Curry, T.B.; Convertino, V.A.; Holmes, D.R., III; Haider, C.R. 1D Convolutional neural networks for estimation of compensatory reserve from blood pressure waveforms. In Proceedings of the 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; Volume 2019, pp. 2169–2173. [Google Scholar]
- Greiner, M.; Pfeiffer, D.; Smith, R.D. Principles and practical application of the receiver-operating characteristic analysis for diagnostic tests. Prev. Vet. Med. 2000, 45, 23–41. [Google Scholar] [CrossRef]
- Stewart, C.L.; Mulligan, J.; Grudic, G.Z.; Talley, M.E.; Jurkovich, G.J.; Moulton, S.L. The Compensatory reserve index following injury: Results of a prospective clinical trial. Shock 2016, 46, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Chew, M.S.; Aneman, A. Haemodynamic monitoring using arterial waveform analysis. Curr. Opin. Crit. Care 2013, 19, 234–241. [Google Scholar] [CrossRef]
- Hatib, F.; Jian, Z.; Buddi, S.; Lee, C.; Settels, J.; Sibert, K.; Rinehart, J.; Cannesson, M. Machine-learning algorithm to predict hypotension based on high-fidelity arterial pressure waveform analysis. Anesthesiology 2018, 129, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Thiele, R.H.; Durieux, M.E. Arterial waveform analysis for the anesthesiologist: Past, present, and future concepts. Anesth. Analg. 2011, 113, 766–776. [Google Scholar] [CrossRef] [PubMed]
- Reisner, A.; Shaltis, P.A.; McCombie, D.; Asada, H.H. Utility of the photoplethysmogram in circulatory monitoring. Anesthesiology 2008, 108, 950–958. [Google Scholar] [CrossRef] [Green Version]
- Mackenzie, C.F.; Wang, Y.; Hu, P.F.; Chen, S.-Y.; Chen, H.H.; Hagegeorge, G.; Stansbury, L.G.; Shackelford, S.; The ONPOINT Study Group. Automated prediction of early blood transfusion and mortality in trauma patients. J. Trauma Acute Care Surg. 2014, 76, 1379–1385. [Google Scholar] [CrossRef]


| CRM, % | CRI, % | p Value | ||
|---|---|---|---|---|
| N | R2 | R2 | ||
| All subjects | 191 | 0.958 | 0.978 | 0.344 |
| HT subjects | 131 | 0.965 | 0.980 | 0.232 |
| LT subjects | 60 | 0.999 | 0.991 | <0.0001 |
| References | N | Clinical Condition | CRM | SBP | HR | PP | SI | Lac |
|---|---|---|---|---|---|---|---|---|
| Nadler et al. [16] | 230 | Blood Donation | 0.84 | 0.60 | 0.73 | 0.51 | 0.64 | - |
| Stewart et al. [12] | 122 | Blood Donation | 0.90 | 0.84 | 0.55 | - | - | - |
| Mackenzie et al. [39] | 556 | Trauma Hemorrhage | 0.78–0.89 | - | 0.56–0.62 | - | - | - |
| Stewart et al. [34] | 44 | Trauma Hemorrhage | 0.97 | 0.81 | 0.64 | - | 0.74 | 0.73 |
| Benov et al. [17] | 31 | GI Bleeding | 0.79 | 0.62 | 0.60 | 0.36 | - | - |
| Johnson et al. [15] | 89 | Trauma Hemorrhage | 0.83 | 0.62 | - | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Convertino, V.A.; Techentin, R.W.; Poole, R.J.; Dacy, A.C.; Carlson, A.N.; Cardin, S.; Haider, C.R.; Holmes III, D.R.; Wiggins, C.C.; Joyner, M.J.; et al. AI-Enabled Advanced Development for Assessing Low Circulating Blood Volume for Emergency Medical Care: Comparison of Compensatory Reserve Machine-Learning Algorithms. Sensors 2022, 22, 2642. https://doi.org/10.3390/s22072642
Convertino VA, Techentin RW, Poole RJ, Dacy AC, Carlson AN, Cardin S, Haider CR, Holmes III DR, Wiggins CC, Joyner MJ, et al. AI-Enabled Advanced Development for Assessing Low Circulating Blood Volume for Emergency Medical Care: Comparison of Compensatory Reserve Machine-Learning Algorithms. Sensors. 2022; 22(7):2642. https://doi.org/10.3390/s22072642
Chicago/Turabian StyleConvertino, Victor A., Robert W. Techentin, Ruth J. Poole, Ashley C. Dacy, Ashli N. Carlson, Sylvain Cardin, Clifton R. Haider, David R. Holmes III, Chad C. Wiggins, Michael J. Joyner, and et al. 2022. "AI-Enabled Advanced Development for Assessing Low Circulating Blood Volume for Emergency Medical Care: Comparison of Compensatory Reserve Machine-Learning Algorithms" Sensors 22, no. 7: 2642. https://doi.org/10.3390/s22072642
APA StyleConvertino, V. A., Techentin, R. W., Poole, R. J., Dacy, A. C., Carlson, A. N., Cardin, S., Haider, C. R., Holmes III, D. R., Wiggins, C. C., Joyner, M. J., Curry, T. B., & Inan, O. T. (2022). AI-Enabled Advanced Development for Assessing Low Circulating Blood Volume for Emergency Medical Care: Comparison of Compensatory Reserve Machine-Learning Algorithms. Sensors, 22(7), 2642. https://doi.org/10.3390/s22072642







