Indicators and Instruments to Assess Components of Disability in Community-Dwelling Older Adults: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Information Sources and Search Strategy
2.3. Selection and Data Collection Process
2.4. Assessment of Methodologic Quality
3. Results
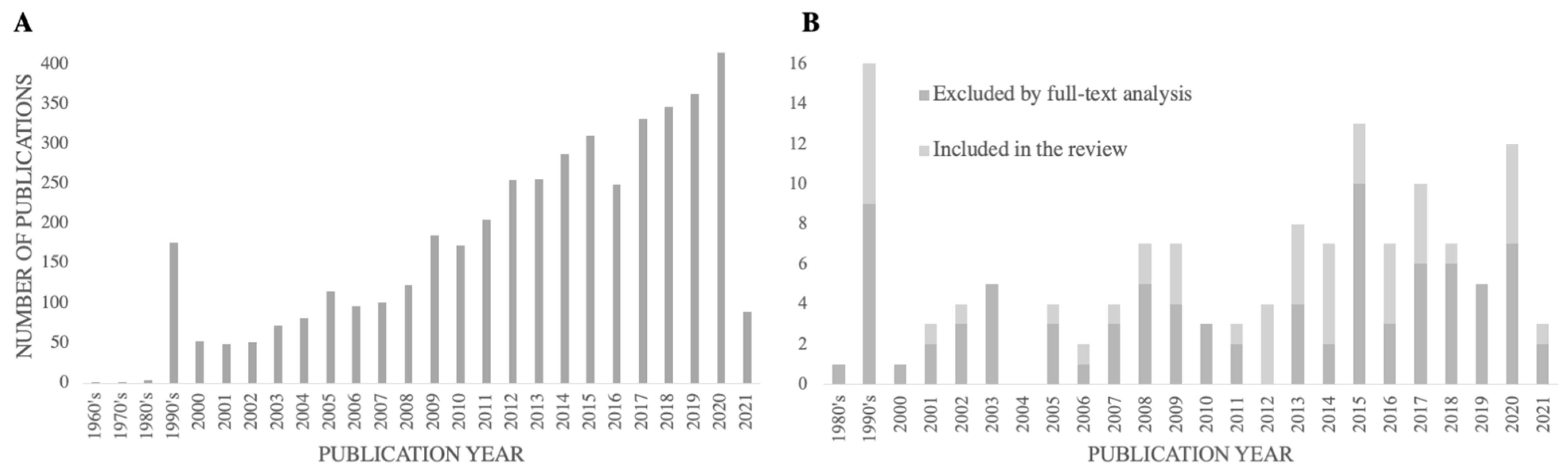
3.1. Literature Search and Study Identification, Screening and Selection
3.2. Methodological Quality Assessment
3.3. Content Analysis
3.3.1. Indicators Expressing a Statistical Association with Age
3.3.2. Indicators with an Inconclusive Statistical Association with Age
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Béjot, Y.; Yaffe, K. Ageing Population: A Neurological Challenge. Neuroepidemiology 2019, 52, 76–77. [Google Scholar] [CrossRef] [PubMed]
- Eurostat. Ageing Europe—Looking at the Lives of Older People in the EU—Statistics Explained; European Union: Brussels, Belgium, 2019. [CrossRef]
- WHO. World Report on Ageing and Health; WHO: Geneva, Switzerland, 2015.
- Chang, A.; Skirbekk, V.; Tyrovolas, S.; Kassebaum, N.; Dieleman, J. Measuring Population Ageing: An Analysis of the Global Burden of Disease Study 2017. Lancet Public Health 2019, 4, e159–e167. [Google Scholar] [CrossRef]
- National Center for Health Statistics. Older Americans 2016: Key Indicators of Well-Being. Federal Interagency Forum on Aging-Related Statistics; National Center for Health Statistics: Hyattsville, MD, USA, 2016.
- Grube, M.M.; Möhler, R.; Fuchs, J.; Gaertner, B.; Scheidt-Nave, C. Indicator-based Public Health Monitoring in Old Age in OECD Member Countries: A Scoping Review. BMC Public Health 2019, 19, 1068. [Google Scholar] [CrossRef]
- Kilpeläinen, K.; Tuomi-Nikula, A.; Thelen, J.; Gissler, M.; Sihvonen, A.; Kramers, P.; Aromaa, A. Health Indicators in Europe: Availability and Data Needs. Eur. J. Public Health 2012, 22, 716–721. [Google Scholar] [CrossRef]
- Khan, S.S.; Singer, B.D.; Vaughan, D.E. Molecular and physiological manifestations and measurement of aging in humans. Aging Cell 2017, 16, 624–633. [Google Scholar] [CrossRef] [PubMed]
- Manini, T. Development of physical disability in older adults. Curr. Aging Sci. 2011, 4, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Ebrahim, S. Disability in older people: A mass problem requiring mass solutions. Lancet 1999, 353, 1990–1992. [Google Scholar] [CrossRef]
- Nagi, S.Z. Some conceptual issues in disability and rehabilitation. In Sociology and Rehabilitation; Sussman, M.B., Ed.; American Sociological Association: Washington, DC, USA, 1965; pp. 100–113. [Google Scholar]
- Verbrugge, L.M.; Jette, A.M. The disablement process. Soc. Sci. Med. 1994, 38, 1–14. [Google Scholar] [CrossRef]
- WHO. International Classification of Functioning, Disability and Health: ICF; WHO: Geneva, Switzerland, 2001.
- Stuck, A.E.; Walthert, J.M.; Nikolaus, T.; Bula, C.J.; Hohmann, C.; Beck, J.C. Risk factors for functional status decline in community-living elderly people: A systematic literature review. Soc. Sci. Med. 1999, 48, 445–469. [Google Scholar] [CrossRef]
- Tas, U.; Verhagen, A.P.; Bierma-Zeinstra, S.M.A.; Odding, E.; Koes, B.W. Prognostic factors of disability in older people: A systematic review. Br. J. Gen. Pract. J. R. Coll. Gen. Pract. 2007, 57, 319–323. [Google Scholar]
- Freedman, V.A.; Martin, L.G.; Schoeni, R.F. Recent trends in disability and functioning among older adults in the United States: A systematic review. JAMA 2002, 288, 3137–3146. [Google Scholar] [CrossRef] [PubMed]
- Amilon, A.; Hansen, K.M.; Kjær, A.A.; Steffensen, T. Estimating disability prevalence and disability-related inequalities: Does the choice of measure matter? Soc. Sci. Med. 2021, 272, 113740. [Google Scholar] [CrossRef]
- Chung, J.; Demiris, G.; Thompson, H.J. Instruments to assess mobility limitation in community-dwelling older adults: A systematic review. J. Aging Phys. Act. 2015, 23, 298–313. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Ding, X.; Dong, B. The measurement of disability in the elderly: A systematic review of self-reported questionnaires. J. Am. Med. Dir. Assoc. 2014, 15, 150.e1–150.e9. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Sanderson, S.; Tatt, I.D.; Higgins, J.P.T. Tools for assessing quality and susceptibility to bias in observational studies in epidemiology: A systematic review and annotated bibliography. Int. J. Epidemiol. 2007, 36, 666–676. [Google Scholar] [CrossRef]
- Krishnamurthi, R.V.; Ikeda, T.; Feigin, V.L. Global, Regional and Country-Specific Burden of Ischaemic Stroke, Intracerebral Haemorrhage and Subarachnoid Haemorrhage: A Systematic Analysis of the Global Burden of Disease Study 2017. Neuroepidemiology 2020, 54, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Avan, A.; Hachinski, V. Stroke and dementia, leading causes of neurological disability and death, potential for prevention. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2021, 17, 1072–1076. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Sun, Q.; Li, J.; Zhang, W.; He, Y.; Zhou, Y. Disability Status and Its Influencing Factors Among Stroke Patients in Northeast China: A 3-Year Follow-Up Study. Neuropsychiatr. Dis. Treat. 2021, 17, 2567–2573. [Google Scholar] [CrossRef] [PubMed]
- Adamson, J.; Beswick, A.; Ebrahim, S. Is stroke the most common cause of disability? J. Stroke Cerebrovasc. Dis. Off. J. Natl. Stroke Assoc. 2004, 13, 171–177. [Google Scholar] [CrossRef]
- Sousa, R.M.; Ferri, C.P.; Acosta, D.; Albanese, E.; Guerra, M.; Huang, Y.; Jacob, K.S.; Jotheeswaran, A.T.; Rodriguez, J.J.L.; Pichardo, G.R.; et al. Contribution of chronic diseases to disability in elderly people in countries with low and middle incomes: A 10/66 Dementia Research Group population-based survey. Lancet 2009, 374, 1821–1830. [Google Scholar] [CrossRef]
- Carvalho-Pinto, B.P.B.; Faria, C.D.C.M. Health, function and disability in stroke patients in the community. Braz. J. Phys. Ther. 2016, 20, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Losilla, J.-M.; Oliveras, I.; Marin-Garcia, J.A.; Vives, J. Three risk of bias tools lead to opposite conclusions in observational research synthesis. J. Clin. Epidemiol. 2018, 101, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Mueller, M.; D’Addario, M.; Egger, M.; Cevallos, M.; Dekkers, O.; Mugglin, C.; Scott, P. Methods to systematically review and meta-analyse observational studies: A systematic scoping review of recommendations. BMC Med. Res. Methodol. 2018, 18, 44. [Google Scholar] [CrossRef]
- Rollo, S.; Antsygina, O.; Tremblaya, M.S. The whole day matters: Understanding 24-hour movement guideline adherence and relationships with health indicators across the lifespan. J. Sport Health Sci. 2020, 9, 493–510. [Google Scholar] [CrossRef]
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Community Health 1998, 52, 377–384. [Google Scholar] [CrossRef]
- Hooper, P.; Jutai, J.W.; Strong, G.; Russell-Minda, E. Age-related macular degeneration and low-vision rehabilitation: A systematic review. Can. J. Ophthalmol. 2008, 43, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Adachi, D.; Nishiguchi, S.; Fukutani, N.; Kayama, H.; Tanigawa, T.; Yukutake, T.; Hotta, T.; Tashiro, Y.; Morino, S.; Yamada, M.; et al. Factors associating with shuttle walking test results in community-dwelling elderly people. Aging Clin. Exp. Res. 2015, 27, 829–834. [Google Scholar] [CrossRef]
- Alcock, L.; O’Brien, T.D.; Vanicek, N. Age-related changes in physical functioning: Correlates between objective and self-reported outcomes. Physiotherapy 2015, 101, 204–213. [Google Scholar] [CrossRef]
- Amarasena, N.; Keuskamp, D.; Balasubramanian, M.; Brennan, D.S. Health behaviours and quality of life in independently living South Australians aged 75 years or older. Aust. Dent. J. 2018, 63, 156–162. [Google Scholar] [CrossRef]
- Andrade, H.B.; Costa, S.M.; Pirôpo, U.S.; Schettino, L.; Casotti, C.A.; Pereira, R. Lower limb strength, but not sensorial integration, explains the age-associated postural control impairment. Muscles Ligaments Tendons J. 2018, 7, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Araújo, L.; Ribeiro, Ó. Incapacity paradox of very-old people. Acta Med. Port. 2011, 24 (Suppl. 2), 189–196. [Google Scholar] [PubMed]
- Arroyo, P.; Lera, L.; Sánchez, H.; Bunout, D.; Santos, J.L.; Albala, C. Anthropometry, body composition and functional limitations in the elderly. Rev. Med. Chile 2007, 135, 846–854. [Google Scholar] [CrossRef] [PubMed]
- Barberger-Gateau, P.; Chaslerie, A.; Dartigues, J.F.; Commenges, D.; Gagnon, M.; Salamon, R. Health measures correlates in a French elderly community population: The PAQUID study. J. Gerontol. 1992, 47, S88–S97. [Google Scholar] [CrossRef]
- Chang, E.-S.; Dong, X. A battery of tests for assessing cognitive function in U.S. Chinese older adults--findings from the PINE Study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 69 (Suppl. 2), S23–S30. [Google Scholar] [CrossRef]
- Chao, J.; Li, Y.; Xu, H.; Yu, Q.; Wang, Y.; Liu, P. Health status and associated factors among the community-dwelling elderly in China. Arch. Gerontol. Geriatr. 2013, 56, 199–204. [Google Scholar] [CrossRef]
- Chen, K.-M.; Lin, M.-H.; Wang, Y.-C.; Huang, H.-T.; Li, C.-H. A model-based survey of physical health in community-dwelling older adults. J. Nurs. Res. 2012, 20, 239–248. [Google Scholar] [CrossRef]
- Choudhary, R. Age and Gender- Related Test Performance in Community Dwelling Elderly Population: Six-Minute Step Test and Four Square Step Test. Indian J. Physiother. Occup. Ther. 2020, 14, 202–206. [Google Scholar]
- Chung, P.-K.; Zhao, Y.; Liu, J.-D.; Quach, B. Functional fitness norms for community-dwelling older adults in Hong Kong. Arch. Gerontol. Geriatr. 2016, 65, 54–62. [Google Scholar] [CrossRef]
- Confortin, S.C.; Barbosa, A.R. Factors Associated With Muscle Strength Among Rural Community-Dwelling Older Women in Southern Brazil. J. Geriatr. Phys. Ther. 2015, 38, 162–168. [Google Scholar] [CrossRef]
- Desrosiers, J.; Robichaud, L.; Demers, L.; Gélinas, I.; Noreau, L.; Durand, D. Comparison and correlates of participation in older adults without disabilities. Arch. Gerontol. Geriatr. 2009, 49, 397–403. [Google Scholar] [CrossRef]
- Dodge, H.H.; Kita, Y.; Takechi, H.; Hayakawa, T.; Ganguli, M.; Ueshima, H. Healthy cognitive aging and leisure activities among the oldest old in Japan: Takashima study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2008, 63, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Chang, E.-S.; Simon, M.A. Physical Function Assessment in a Community-Dwelling Population of U.S. Chinese Older Adults. J. Gerontol. Ser. A 2014, 69, S31–S38. [Google Scholar] [CrossRef] [PubMed]
- Ekström, H.; Schmidt, S.M.; Iwarsson, S. Home and health among different sub-groups of the ageing population: A comparison of two cohorts living in ordinary housing in Sweden. BMC Geriatr. 2016, 16, 90. [Google Scholar] [CrossRef] [PubMed]
- Fastame, M.C.; Penna, M.P.; Hitchcott, P.K. Psychological markers of longevity in Sardinian centenarians: The impact of developmental factors and social desirability. Aging Clin. Exp. Res. 2020, 32, 107–114. [Google Scholar] [CrossRef]
- Furuna, T.; Nagasaki, H.; Nishizawa, S.; Sugiura, M.; Okuzumi, H.; Ito, H.; Kinugasa, T.; Hashizume, K.; Maruyama, H. Longitudinal change in the physical performance of older adults in the community. J. Jpn. Phys. Ther. Assoc. 1998, 1, 1–5. [Google Scholar] [CrossRef]
- Ghinescu, M.; Olaroiu, M.; Dijk, J.P.v.; Olteanu, T.; Heuvel, W.J.A.v.d. Health status of independently living older adults in Romania. Geriatr. Gerontol. Int. 2014, 14, 926–933. [Google Scholar] [CrossRef]
- Grassi, L.; Caruso, R.; Ronch, C.D.; Härter, M.; Schulz, H.; Volkert, J.; Dehoust, M.; Sehner, S.; Suling, A.; Wegscheider, K.; et al. Quality of life, level of functioning, and its relationship with mental and physical disorders in the elderly: Results from the MentDis_ICF65+ study. Health Qual. Life Outcomes 2020, 18, 61. [Google Scholar] [CrossRef] [PubMed]
- Hara, K.; Tohara, H.; Namiki, C.; Yamaguchi, K.; Chantaramanee, A.; Kobayashi, K.; Saito, T.; Nakagawa, K.; Okumura, T.; Yoshimi, K.; et al. Relationship between displacement of the masseter muscle during biting and masseter muscle quality and bite force in healthy elderly persons. J. Oral Rehabil. 2020, 47, 441–448. [Google Scholar] [CrossRef]
- Hayashi, K.; Sato, J.; Fujiwara, N.; Kajita, M.; Fukuharu, M.; Hu, X.; Kuriki, K.; Hoshino, H.; Kato, R.; Tokudome, S.; et al. Associations between physical strength, cerebral function and mental health in independent-living elderly Japanese women. Environ. Health Prev. Med. 2002, 7, 123–128. [Google Scholar] [CrossRef][Green Version]
- Hershman, D.L.; Simonoff, P.A.; Frishman, W.H.; Paston, F.; Aronson, M.K. Drug utilization in the old-old and how it relates to self-perceived health and all-cause mortality—Results from the bronx aging study. J. Am. Geriatr. Soc. 1995, 43, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Ignasiak, Z.; Sebastjan, A.; Sławińska, T.; Skrzek, A.; Czarny, W.; Król, P.; Rzepko, M.; Duda-Biernacka, B.; Marchewka, A.; Filar-Mierzwa, K.; et al. Functional fitness normative values for elderly polish population. BMC Geriatr. 2020, 20, 384. [Google Scholar] [CrossRef]
- Incel, N.A.; Sezgin, M.; As, I.; Bolgen, O.; Sahin, C.G. The geriatric hand: Correlation of hand-muscle function and activity restriction in elderly. Int. J. Rehabil. Res. 2009, 32, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Jansen, C.W.S.; Niebuhr, B.R.; Coussirat, D.J.; Hawthorne, D.; Moreno, L.; Phillip, M. Hand force of men and women over 65 years of age as measured by maximum pinch and grip force. J. Aging Phys. Act. 2008, 16, 24–41. [Google Scholar] [CrossRef] [PubMed]
- Makizako, H.; Shimada, H.; Doi, T.; Tsutsumimoto, K.; Lee, S.; Lee, S.C.; Harada, K.; Hotta, R.; Nakakubo, S.; Bae, S.; et al. Age-dependent changes in physical performance and body composition in community-dwelling Japanese older adults. J. Cachexia Sarcopenia Muscle 2017, 8, 607–614. [Google Scholar] [CrossRef] [PubMed]
- Marques, E.A.; Baptista, F.; Santos, R.; Vale, S.; Santos, D.A.; Silva, A.M.; Mota, J.; Sardinha, L.B. Normative functional fitness standards and trends of Portuguese older adults: Cross-cultural comparisons. J. Aging Phys. Act. 2014, 22, 126–137. [Google Scholar] [CrossRef]
- Moreira, P.L.; Correa, C.R.; Corrente, J.E.; Martin, L.C.; Boas, P.J.F.V.; Ferreira, A.L.A. Anthropometric, functional capacity, and oxidative stress changes in Brazilian community-living elderly subjects. A longitudinal study. Arch. Gerontol. Geriatr. 2016, 66, 140–146. [Google Scholar] [CrossRef]
- Nakagawa, H.B.; Ferraresi, J.R.; Prata, M.G.; Scheicher, M.E. Postural balance and functional independence of elderly people according to gender and age: Cross-sectional study. Sao Paulo Med. J. 2017, 135, 260–265. [Google Scholar] [CrossRef]
- Nakamura, M.; Tazaki, F.; Nomura, K.; Takano, T.; Hashimoto, M.; Hashizume, H.; Kamei, I. Cognitive impairment associated with locomotive syndrome in community-dwelling elderly women in Japan. Clin. Interv. Aging 2017, 12, 1451–1457. [Google Scholar] [CrossRef]
- Neri, A.L.; Ongaratto, L.L.; Yassuda, M.S. Mini-Mental State Examination sentence writing among community-dwelling elderly adults in Brazil: Text fluency and grammar complexity. Int. Psychogeriatr. 2012, 24, 1732–1737. [Google Scholar] [CrossRef]
- Papadakis, M.A.; Grady, D.; Tierney, M.J.; Black, D.; Wells, L.; Grunfeld, C. Insulin-like growth factor 1 and functional status in healthy older men. J. Am. Geriatr. Soc. 1995, 43, 1350–1355. [Google Scholar] [CrossRef]
- Pinheiro, P.A.; Passos, T.D.E.-R.O.; Coqueiro, R.d.S.; Fernandes, M.H.; Barbosa, A.R. Motor performance of the elderly in northeast Brazil: Differences with age and sex. Rev. Esc. Enferm. USP 2013, 47, 128–136. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pisciottano, M.V.C.; Pinto, S.S.; Szejnfeld, V.L.; Castro, C.H.M. The relationship between lean mass, muscle strength and physical ability in independent healthy elderly women from the community. J. Nutr. Health Aging 2014, 18, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Poon, L.W.; Martin, P.; Clayton, G.M.; Messner, S.; Noble, C.A.; Johnson, M.A. The influences of cognitive resources on adaptation and old age. Int. J. Aging Hum. Dev. 1992, 34, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Prata, M.G.; Scheicher, M.E. Correlation between balance and the level of functional independence among elderly people. Sao Paulo Med. J. 2012, 130, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Romero-Ortuno, R.; Cogan, L.; Cunningham, C.U.; Kenny, R.A. Do older pedestrians have enough time to cross roads in Dublin? A critique of the Traffic Management Guidelines based on clinical research findings. Age Ageing 2009, 39, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Salvà, A.; Bolibar, I.; Lucas, R.; Rojano-Luque, X. Use of the POMA in our environment to evaluate balance in gait in a population of community-dwelling elderly individuals. Rev. Esp. Geriatr. Gerontol. 2005, 40, 36–44. [Google Scholar] [CrossRef]
- Sarvestan, J.; Kovacikova, Z.; Linduska, P.; Gonosova, Z.; Svoboda, Z. Contribution of lower limb muscle strength to walking, postural sway and functional performance in elderly women. Isokinet. Exerc. Sci. 2021, 29, 49–57. [Google Scholar] [CrossRef]
- Sauvel, C.; Barberger-Gateau, P.; Dequae, L.; Letenneur, L.; Dartigues, J.F. Factors associated with one-year change in functional status of elderly community dwellers. Rev. Epidemiol. Sante Publique 1994, 42, 13–23. [Google Scholar]
- Sherman, S.E.; Reuben, D. Measures of functional status in community-dwelling elders. J. Gen. Intern. Med. 1998, 13, 817–823. [Google Scholar] [CrossRef][Green Version]
- Smee, D.J.; Anson, J.M.; Waddington, G.S.; Berry, H.L. Association between Physical Functionality and Falls Risk in Community-Living Older Adults. Curr. Gerontol. Geriatr. Res. 2012, 2012, 864516. [Google Scholar] [CrossRef] [PubMed]
- Tomita, A.; Burns, J.K. Depression, disability and functional status among community-dwelling older adults in South Africa: Evidence from the first South African National Income Dynamics Study. Int. J. Geriatr. Psychiatry 2013, 28, 1270–1279. [Google Scholar] [CrossRef]
- Tomsone, S.; Horstmann, V.; Oswald, F.; Iwarsson, S. Aspects of housing and perceived health among ADL independent and ADL dependent groups of older people in three national samples. Aging Clin. Exp. Res. 2013, 25, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.D.; Lim, A.S.; Leurgans, S.E.; Bennett, D.A.; Buchman, A.S.; Barnes, L.L. Self-Reported Sleep in Older African Americans and White Americans. Ethn. Dis. 2016, 26, 521–528. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Uttl, B.; Graf, P.; Miller, J.; Tuokko, H. Pro- and retrospective memory in late adulthood. Conscious. Cogn. 2001, 10, 451–472. [Google Scholar] [CrossRef] [PubMed]
- Zunzunegui, M.V.; Nunez, O.; Durban, M.; Yébenes, M.J.G.d.; Otero, A. Decreasing prevalence of disability in activities of daily living, functional limitations and poor self-rated health: A 6-year follow-up study in Spain. Aging Clin. Exp. Res. 2006, 18, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Cieza, A.; Fayed, N.; Bickenbach, J.; Prodinger, B. Refinements of the ICF Linking Rules to strengthen their potential for establishing comparability of health information. Disabil. Rehabil. 2019, 41, 574–583. [Google Scholar] [CrossRef]
- Stucki, G.; Cieza, A. The International Classification of Functioning, Disability and Health (ICF) Core Sets for rheumatoid arthritis: A way to specify functioning. Ann. Rheum. Dis. 2004, 63 (Suppl. 2), ii40–ii45. [Google Scholar] [CrossRef]
- Naughton, N.; Algar, L. Linking commonly used hand therapy outcome measures to individual areas of the International Classification of Functioning: A systematic review. J. Hand Ther. Off. J. Am. Soc. Hand Ther. 2019, 32, 243–261. [Google Scholar] [CrossRef]
- Yang, M.; Luo, L.; Hao, Q.; Dong, B. Content comparison of self-reported disability measures for the elderly according to the international classification of functioning, disability and health. Disabil. Rehabil. 2014, 36, 884–893. [Google Scholar] [CrossRef]
- Chandrasekaran, R.; Katthula, V.; Moustakas, E. Too old for technology? Use of wearable healthcare devices by older adults and their willingness to share health data with providers. Health Inform. J. 2021, 27, 14604582211058073. [Google Scholar] [CrossRef] [PubMed]
- Mlinac, M.E.; Feng, M.C. Assessment of Activities of Daily Living, Self-Care, and Independence. Arch. Clin. Neuropsychol. Off. J. Natl. Acad. Neuropsychol. 2016, 31, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Devi, J. The scales of functional assessment of Activities of Daily Living in geriatrics. Age Ageing 2018, 47, 500–502. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Moreno, F.M.; Bermudez-Edo, M.; Rodríguez-García, E.; Pérez-Mármol, J.M.; Garrido, J.L.; Rodríguez-Fórtiz, M.J. A machine learning approach for semi-automatic assessment of IADL dependence in older adults with wearable sensors. Int. J. Med. Inform. 2022, 157, 104625. [Google Scholar] [CrossRef] [PubMed]
- Camp, N.; Lewis, M.; Hunter, K.; Johnston, J.; Zecca, M.; Di Nuovo, A.; Magistro, D. Technology Used to Recognize Activities of Daily Living in Community-Dwelling Older Adults. Int. J. Environ. Res. Public Health 2020, 18, 163. [Google Scholar] [CrossRef]
- Gallagher, J.E.; Wilkie, A.A.; Cordner, A.; Hudgens, E.E.; Ghio, A.J.; Birch, R.J.; Wade, T.J. Factors associated with self-reported health: Implications for screening level community-based health and environmental studies. BMC Public Health 2016, 16, 640. [Google Scholar] [CrossRef]
- Wuorela, M.; Lavonius, S.; Salminen, M.; Vahlberg, T.; Viitanen, M.; Viikari, L. Self-rated health and objective health status as predictors of all-cause mortality among older people: A prospective study with a 5-, 10-, and 27-year follow-up. BMC Geriatr. 2020, 20, 120. [Google Scholar] [CrossRef]
- Lorem, G.; Cook, S.; Leon, D.A.; Emaus, N.; Schirmer, H. Self-reported health as a predictor of mortality: A cohort study of its relation to other health measurements and observation time. Sci. Rep. 2020, 10, 4886. [Google Scholar] [CrossRef]
- Syddall, H.E.; Martin, H.J.; Harwood, R.H.; Cooper, C.; Sayer, A.A. The SF-36: A simple, effective measure of mobility-disability for epidemiological studies. J. Nutr. Health Aging 2009, 13, 57–62. [Google Scholar] [CrossRef]
- Walters, S.J.; Munro, J.F.; Brazier, J.E. Using the SF-36 with older adults: A cross-sectional community-based survey. Age Ageing 2001, 30, 337–343. [Google Scholar] [CrossRef]
- Parker, S.G.; Bechinger-English, D.; Jagger, C.; Spiers, N.; Lindesay, J. Factors affecting completion of the SF-36 in older people. Age Ageing 2006, 35, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Kostanjsek, N. Use of The International Classification of Functioning, Disability and Health (ICF) as a conceptual framework and common language for disability statistics and health information systems. BMC Public Health 2011, 11 (Suppl. 4), S3. [Google Scholar] [CrossRef] [PubMed]
- Tomandl, J.; Heinmüller, S.; Selb, M.; Graessel, E.; Freiberger, E.; Kühlein, T.; Hueber, S.; Book, S.; Gotthardt, S. Laying the foundation for a Core Set of the International Classification of Functioning, Disability and Health for community-dwelling older adults in primary care: Relevant categories of their functioning from the research perspective, a scoping review. BMJ Open 2021, 11, e037333. [Google Scholar] [CrossRef]
- Spoorenberg, S.L.; Reijneveld, S.A.; Middel, B.; Uittenbroek, R.J.; Kremer, H.P.; Wynia, K. The Geriatric ICF Core Set reflecting health-related problems in community-living older adults aged 75 years and older without dementia: Development and validation. Disabil. Rehabil. 2015, 37, 2337–2343. [Google Scholar] [CrossRef]
- Book, S.; Ulbrecht, G.; Tomandl, J.; Kuehlein, T.; Gotthardt, S.; Freiberger, E.; Graessel, E. Laying the foundation for an International Classification of Functioning, Disability and Health Core Set for community-dwelling elderly adults in primary care: The clinical perspective identified in a cross-sectional study. BMJ Open 2020, 10, e038434. [Google Scholar] [CrossRef]
- Vasconcelos, K.S.d.S.; Bastone, A.d.C.; Vieira, R.A.; Andrade, A.C.d.S.; Dias, R.C.; Dias, J.M.D.; Perracini, M.R.; Guerra, R.O.; Moreira, B.d.S. Examining the factors associated with functional capacity of community-dwelling older adults using the ICF framework: A cross-sectional study from the Frailty in Brazilian Older Adults Study (FIBRA). Physiother. Theory Pract. 2022, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Rejeski, W.J.; Ip, E.H.; Marsh, A.P.; Miller, M.E.; Farmer, D.F. Measuring disability in older adults: The International Classification System of Functioning, Disability and Health (ICF) framework. Geriatr. Gerontol. Int. 2008, 8, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yu, J.; Shen, W.; Zhang, Y.; Xiang, Y.; Zhang, X.; Lin, Z.; Yan, T. A mobile app implementing the international classification of functioning, disability and health rehabilitation set. BMC Med. Inform. Decis. Mak. 2020, 20, 12. [Google Scholar] [CrossRef]
- Leonardi, M.; Lee, H.; Kostanjsek, N.; Fornari, A.; Raggi, A.; Martinuzzi, A.; Yáñez, M.; Almborg, A.H.; Fresk, M.; Besstrashnova, Y.; et al. 20 Years of ICF-International Classification of Functioning, Disability and Health: Uses and Applications around the World. Int. J. Environ. Res. Public Health 2022, 19, 11321. [Google Scholar] [CrossRef]
- Elsman, E.B.M.; Butcher, N.J.; Mokkink, L.B.; Terwee, C.B.; Tricco, A.; Gagnier, J.J.; Aiyegbusi, O.L.; Barnett, C.; Smith, M.; Moher, D.; et al. Study protocol for developing, piloting and disseminating the PRISMA-COSMIN guideline: A new reporting guideline for systematic reviews of outcome measurement instruments. Syst. Rev. 2022, 11, 121. [Google Scholar] [CrossRef]

| Study Identification | Modified Downs and Black Items and Score | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | Score | |
| Adachi et al., 2015 [33] | + | + | + | + | + | + | + | + | + | UD | − | + | + | + | + | + | + | − | − | 15 |
| Alcock et al., 2015 [34] | + | + | − | + | − | + | + | + | + | − | − | + | + | + | + | − | + | + | − | 13 |
| Amarasena et al., 2018 [35] | + | + | + | + | − | + | + | + | − | − | + | + | + | + | + | UD | + | − | − | 13 |
| Andrade et al., 2018 [36] | + | + | + | − | − | − | − | UD | − | − | − | + | + | + | + | UD | UD | − | − | 7 |
| Araújo & Ribeiro, 2011 [37] | + | − | − | − | − | + | − | + | − | − | − | − | + | UD | − | − | + | − | − | 5 |
| Arroyo et al., 2007 [38] | + | + | − | + | − | + | + | + | − | + | + | + | + | + | + | UD | + | − | − | 13 |
| Barberger et al., 1992 [39] | + | + | + | + | − | + | + | + | − | + | + | + | + | + | + | UD | + | − | − | 14 |
| Chang & Dong., 2014 [40] | + | + | + | − | − | + | + | − | − | + | + | + | + | + | + | UD | + | + | − | 13 |
| Chao et al., 2013 [41] | + | + | + | + | − | + | − | − | + | + | + | + | + | + | + | UD | − | − | − | 12 |
| Chen et al., 2012 [42] | + | + | + | − | − | + | + | + | + | + | + | + | + | + | + | UD | UD | − | − | 13 |
| Choudhary, 2020 [43] | + | + | − | − | − | + | + | + | + | − | − | + | + | + | + | UD | + | − | − | 11 |
| Chung et al., 2016 [44] | + | + | + | + | + | + | + | + | − | − | − | + | + | + | + | + | − | + | − | 14 |
| Confortin & Barbosa, 2015 [45] | + | + | − | + | − | + | − | + | + | − | − | + | + | + | + | UD | + | − | − | 11 |
| Desrosiers et al., 2009 [46] | + | + | + | + | − | + | + | + | + | + | + | + | + | + | + | UD | + | + | + | 17 |
| Dodge et al., 2008 [47] | + | + | + | + | + | + | + | + | + | + | + | + | + | + | + | + | + | − | − | 17 |
| Dong et al., 2014 [48] | + | + | + | + | − | + | + | + | − | − | + | + | + | + | + | UD | + | + | − | 14 |
| Ekström et al., 2016 [49] | + | + | − | + | − | + | + | + | + | − | − | + | + | + | + | UD | + | − | − | 12 |
| Fastame et al., 2020 [50] | + | + | + | − | + | + | + | UD | + | − | − | + | + | + | + | + | UD | − | − | 12 |
| Furuna et al., 1998 [51] | + | + | − | + | + | + | + | + | − | + | + | + | + | + | + | + | − | − | − | 14 |
| Ghinescu et al., 2014 [52] | + | + | − | + | − | + | + | + | − | + | + | + | + | + | + | UD | + | − | − | 13 |
| Grassi et al., 2020 [53] | + | + | + | + | − | + | + | + | + | + | − | + | + | + | + | UD | + | − | − | 14 |
| Hara et al., 2020 [54] | + | + | + | − | − | − | − | − | + | + | − | + | + | + | + | UD | + | + | + | 12 |
| Hayashi et al., 2002 [55] | + | + | − | + | − | + | + | + | − | − | − | + | + | + | + | UD | + | + | − | 12 |
| Hershman et al., 1995 [56] | + | + | + | + | + | + | + | + | − | UD | UD | + | + | + | − | + | + | − | − | 13 |
| Ignasiak et al., 2020 [57] | + | + | + | + | + | + | + | − | + | − | − | + | + | + | + | + | UD | − | − | 13 |
| Incel et al., 2009 [58] | + | + | + | + | − | + | + | + | − | − | − | + | + | + | + | UD | UD | − | − | 11 |
| Jansen et al., 2008 [59] | + | + | + | + | − | + | + | + | + | − | − | + | + | + | + | UD | + | + | − | 14 |
| Makizako et al., 2017 [60] | + | + | + | + | + | + | + | + | + | − | − | + | + | + | + | + | + | + | − | 16 |
| Marques et al., 2014 [61] | + | + | − | + | + | + | + | − | + | − | − | + | + | + | + | + | − | + | − | 13 |
| Moreira et al., 2016 [62] | + | + | + | + | − | + | + | + | + | + | + | + | + | + | + | UD | + | − | − | 15 |
| Nakagawa et al., 2017 [63] | + | + | + | + | + | + | + | + | + | − | − | + | + | + | + | + | UD | + | − | 15 |
| Nakamura et al., 2017 [64] | + | + | + | + | − | + | + | + | + | − | − | + | + | + | + | UD | + | − | − | 13 |
| Neri et al., 2012 [65] | + | + | + | − | − | + | + | + | + | − | − | + | + | + | + | UD | + | − | − | 12 |
| Papadakis et al., 1995 [66] | + | + | + | + | + | + | + | + | + | − | − | + | + | + | + | + | + | + | − | 16 |
| Pinheiro et al., 2013 [67] | + | + | + | + | + | + | + | + | + | + | + | + | + | + | + | + | + | − | − | 1 |
| Pisciottano et al., 2014 [68] | + | + | + | + | − | + | + | + | − | − | − | + | + | + | + | UD | + | + | − | 13 |
| Poon et al., 1992 [69] | + | + | − | − | − | − | − | − | + | − | − | + | + | + | + | UD | − | − | − | 7 |
| Prata & Scheicher, 2012 [70] | + | + | + | + | − | + | + | + | + | − | − | + | + | + | + | UD | + | + | − | 14 |
| Romero-Ortuno et al., 2009 [71] | + | + | + | + | + | + | + | + | + | − | − | + | + | + | + | + | + | + | − | 16 |
| Salvà et al., 2005 [72] | + | + | + | + | − | − | − | + | − | + | + | + | + | + | + | UD | + | − | − | 12 |
| Sarvestan et al., 2021 [73] | + | + | + | − | + | + | + | + | + | − | − | + | + | + | + | + | + | + | − | 15 |
| Sauvel et al., 1994 [74] | + | + | − | − | − | + | − | + | − | + | + | + | + | + | + | UD | + | − | − | 11 |
| Sherman and Reuben, 1998 [75] | + | + | + | + | − | + | + | + | − | − | − | + | + | UD | + | UD | + | − | − | 11 |
| Smee et al., 2012 [76] | + | + | + | − | − | + | + | + | − | − | − | − | + | + | + | UD | + | + | − | 11 |
| Tomita & Burns, 2013 [77] | + | + | + | + | + | + | − | + | + | + | + | + | + | − | + | + | + | − | − | 15 |
| Tomsone et al., 2013 [78] | + | + | + | + | + | + | + | + | + | − | + | + | + | + | + | + | + | − | − | 16 |
| Turner et al., 2016 [79] | + | + | + | + | + | + | + | + | + | − | + | + | + | + | + | + | + | − | − | 16 |
| Uttl et al., 2001 [80] | + | + | + | − | + | + | + | + | + | + | + | + | + | + | + | + | + | + | − | 17 |
| Zunzunegui et al., 2006 [81] | + | + | + | + | + | + | − | + | − | + | + | + | + | + | + | + | + | − | − | 15 |
| % positive description by item | 100 | 98 | 76 | 73 | 39 | 92 | 80 | 84 | 61 | 37 | 41 | 96 | 100 | 94 | 96 | 39 | 78 | 37 | 4 | |
| ICF | Indicator | Instrument/Test | Included Studies | ||||||||
| Objective Measures | Body Structures | Body composition | Digital scale/Dual-emission X-ray densitometer | [42] * | [57] * | [60] | [61] * | [68] * | |||
| Teeth/Masticatory function | Dentist, Ultrasonic diagnostic apparatus | [54] * | |||||||||
| Body Functions | Physical Performance | Senior Fitness Test | [44] | [57] * | [61] * | ||||||
| Physical Performance Test | [66] | [75] * | |||||||||
| National Institute on Aging Battery | [75] * | ||||||||||
| Continuous-Scale Physical Functional Performance | [76] * | ||||||||||
| Height, reaction time | Jump test | [55] * | |||||||||
| Mobility | Get up to sit on a chair | [55] * | |||||||||
| Sit and reach distance | [55] * | ||||||||||
| Pick-up-a-pen test | [67] | ||||||||||
| Endurance | 6 min step test | [43] * | |||||||||
| Shuttle walking test | [33] | ||||||||||
| Chair stand test | [45] * | [48] | [60] | [67] | |||||||
| Gait speed | Walk test | [34] * | [51] | [60] | [67] | [71] | [73] | ||||
| Timed up and go | [34] * | [47] | [68] * | ||||||||
| 8-foot timed walk | [48] | ||||||||||
| Gait parameters | Dynamic gait index | [68] * | |||||||||
| Performance Oriented Mobility Assessment | [72] * | [73] | |||||||||
| Handgrip strength | Grip test | [44] | [45] * | [51] | [55] * | [59] | [60] | [66] | [67] | ||
| Pinch force | Pinch gauges | [59] | |||||||||
| Manual speed | Maximum finger-tapping rate test | [51] | [55] * | ||||||||
| Lower limb Strength | Hip/Knee/Ankle maximum concentric and eccentric voluntary contraction | [34] * | [66] | [68] * | [73] | ||||||
| Balance | Four Square Step | [43] * | |||||||||
| Berg Balance Scale | [63] | [68] * | [70] | ||||||||
| Static balance | Single leg stance test | [44] | [51] | [55] * | |||||||
| Tandem stand | [48] | [67] | |||||||||
| Postural control parameters | Standing test (eyes open) | [73] | |||||||||
| Cardiorespiratory function: Respiration rate, Breath-holding, Lung capacity, Diastolic blood pressure | TruZone Peak FlowMeter; sphygmomanometer; number of occurrences in a 60-s period | [42] * | |||||||||
| Cognitive function | Mini-Mental State Exam | [40] * | [47] | [64] * | [65] * | [66] | [70] | [80] | |||
| Episodic memory | East Boston Memory Test-Immediate and delayed recall | [40] * | |||||||||
| Prospective, Retrospective memory | The name, letter, and the check task Buschke Recall 1-3; RAVLT A1-6, B1 | [80] | |||||||||
| Attention and Visuospatial ability | Cancel H, Card sorting | [80] | |||||||||
| Digit from Wechsler Memory Scale-Revised | [40] * | [47] | [66] | [80] | |||||||
| Executive function | 11-item Symbol Digit Modalities Test | [40] * | |||||||||
| Psychomotor and executive function | Trail-making A, B from Halstead-Reitan Neuropsychological Test | [47] | [66] | ||||||||
| Semantic Fluency | Verbal fluency, Animal and Picture naming | [80] | |||||||||
| Occupational aptitude | General Aptitude Test Battery | [55] * | |||||||||
| Environmental Factors | Overall morbidity | Total number of medications | [47] | [56] * | |||||||
| Mobility | Use of mobility devices | [49] * | |||||||||
| Subjective measures | Body Functions | Health status | Short Form Health Survey (SF-36), SF20 | [34] * | [52] * | ||||||
| Elderly Health Assessment Scale | [41] * | ||||||||||
| Relative health | Self-reported question | [39] | |||||||||
| Overall health status | Self-reported question | [39] | [48] | [52] * | [53] | ||||||
| Functioning | World Health Organization Disability Assessment Schedule II | [53] | |||||||||
| Mental health status | General Health Questionnaire | [55] * | |||||||||
| Functional independence | Self-reported question | [39] | [49] * | ||||||||
| Health changes over the last year | Self-reported question | [48] | |||||||||
| Morbidity | Number of Chronic conditions | [52] * | |||||||||
| Vision/Audition ability | Self-reported question | [39] | [47] * | ||||||||
| Dyspnea/Joint pain | Self-reported question | [39] | |||||||||
| Head symptoms | Checklist | [49] * | |||||||||
| Mobility | Mobility assessed by a six-level scale | [39] | |||||||||
| Self-reported question | [77] | ||||||||||
| Psychological functioning | Ryff scales of Psychological Wellbeing | [49] * | |||||||||
| Psychological Well-Being and Aging Questionnaire | [50] * | ||||||||||
| Sleep | Questionnaire | [79] | |||||||||
| Activities and Participation | Functional ability/Physical function ADL | Katz Index of ADL | [39] | [48] | [62] | [74] * | |||||
| ADL Staircase | [49] * | [78] | |||||||||
| Barthel index | [42] * | [63] | [70] | ||||||||
| Likert-type scale for different tasks | [42] * | ||||||||||
| Self-reported question | [77] | ||||||||||
| Functional ability IADL | Lawton and Brody Instrumental Activities of Daily Living Scale | [39] | [48] | [62] | [74] * | ||||||
| Tokyo Metropolitan Institute of Gerontology Index of Competence | [47] | [51] | |||||||||
| Self-reported question | [77] | ||||||||||
| Functional ability AADL/Mobility | Index of Mobility scale (Rosow and Breslau) | [38] * | [39] | [48] | |||||||
| Confinement scale | [74] * | ||||||||||
| Functional ability ADL and IADL | Groningen Activity Restriction Scale | [52] * | |||||||||
| Basic physical activities | Index of basic physical activities-Nagi | [48] | [38] * | ||||||||
| Social participation | Assessment of Life Habits | [46] | |||||||||
| Leisure activities | List of leisure activities | [47] | |||||||||
| Environmental Factors | Environmental barriers and accessibility problems | Housing Enabler instrument + Functional limitations total number | [49] * | ||||||||
| Attachment to home | 28-item Meaning of home questionnaire | [49] * | |||||||||
| External control beliefs in relation to home | Housing-related Control Beliefs Questionnaire | [49] * | |||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreira, J.S.; Melo, A.; Santos, R.; Sousa, A.S.P. Indicators and Instruments to Assess Components of Disability in Community-Dwelling Older Adults: A Systematic Review. Sensors 2022, 22, 8270. https://doi.org/10.3390/s22218270
Moreira JS, Melo A, Santos R, Sousa ASP. Indicators and Instruments to Assess Components of Disability in Community-Dwelling Older Adults: A Systematic Review. Sensors. 2022; 22(21):8270. https://doi.org/10.3390/s22218270
Chicago/Turabian StyleMoreira, Juliana Santos, Ana Melo, Rubim Santos, and Andreia S. P. Sousa. 2022. "Indicators and Instruments to Assess Components of Disability in Community-Dwelling Older Adults: A Systematic Review" Sensors 22, no. 21: 8270. https://doi.org/10.3390/s22218270
APA StyleMoreira, J. S., Melo, A., Santos, R., & Sousa, A. S. P. (2022). Indicators and Instruments to Assess Components of Disability in Community-Dwelling Older Adults: A Systematic Review. Sensors, 22(21), 8270. https://doi.org/10.3390/s22218270








