Modulation Spectral Signal Representation for Quality Measurement and Enhancement of Wearable Device Data: A Technical Note
Abstract
:1. Introduction
2. Modulation Spectrum Signal Representation
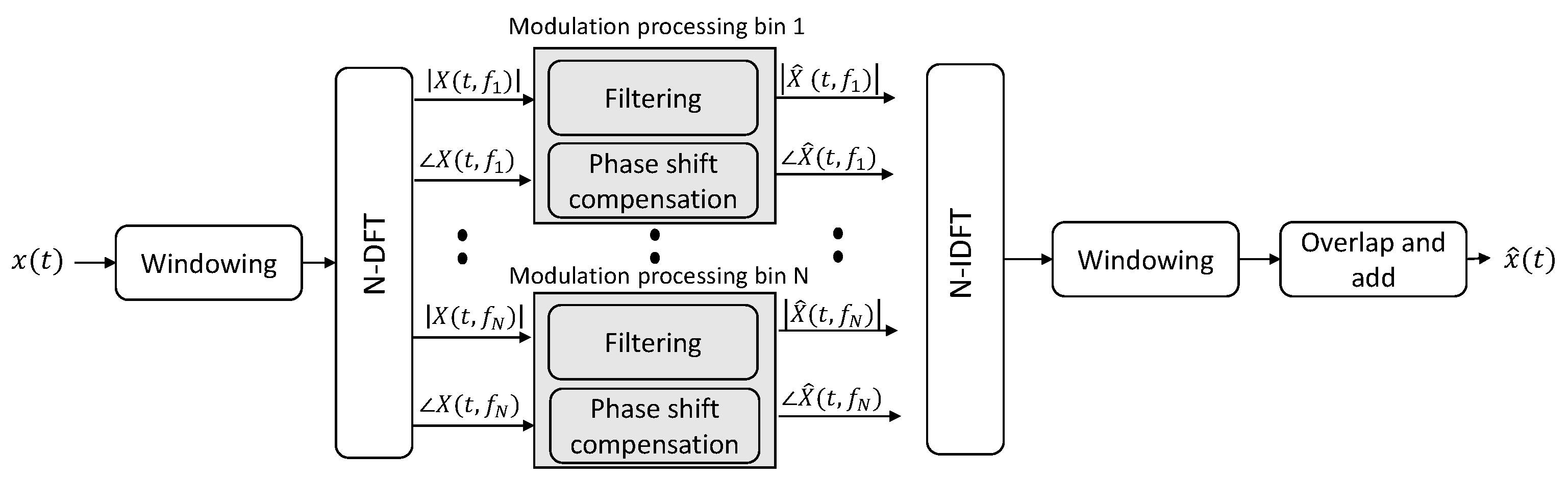
2.1. Signal Processing
2.2. Quality Assessment
2.3. Signal Enhancement
2.4. Blind Source Separation
2.5. Noise-Robust Feature Extraction
2.6. Disease Characterization
3. Applications
3.1. Quality Assessment
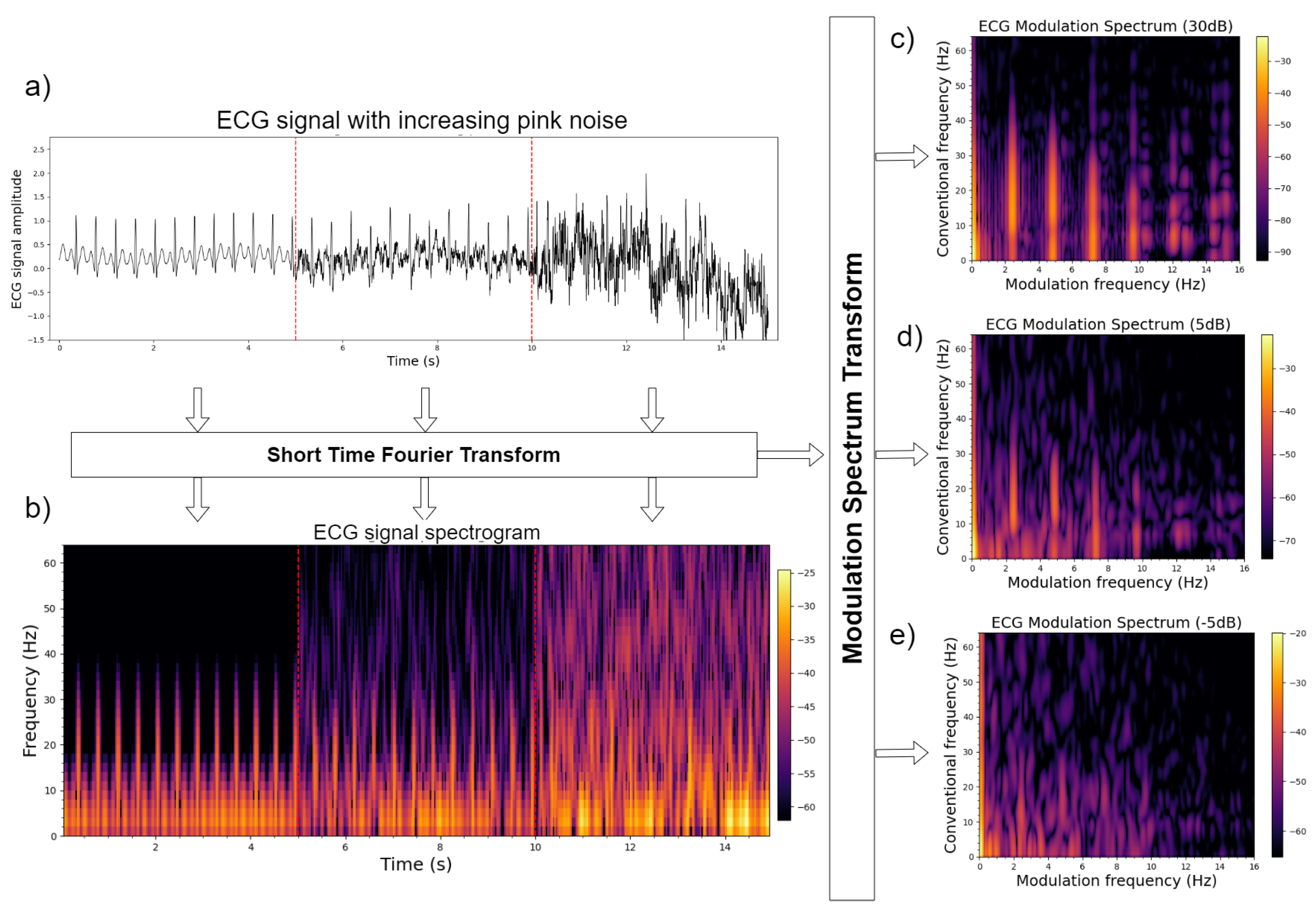
3.1.1. Electrocardiograms
3.1.2. Speech Signals
3.2. Signal Enhancement
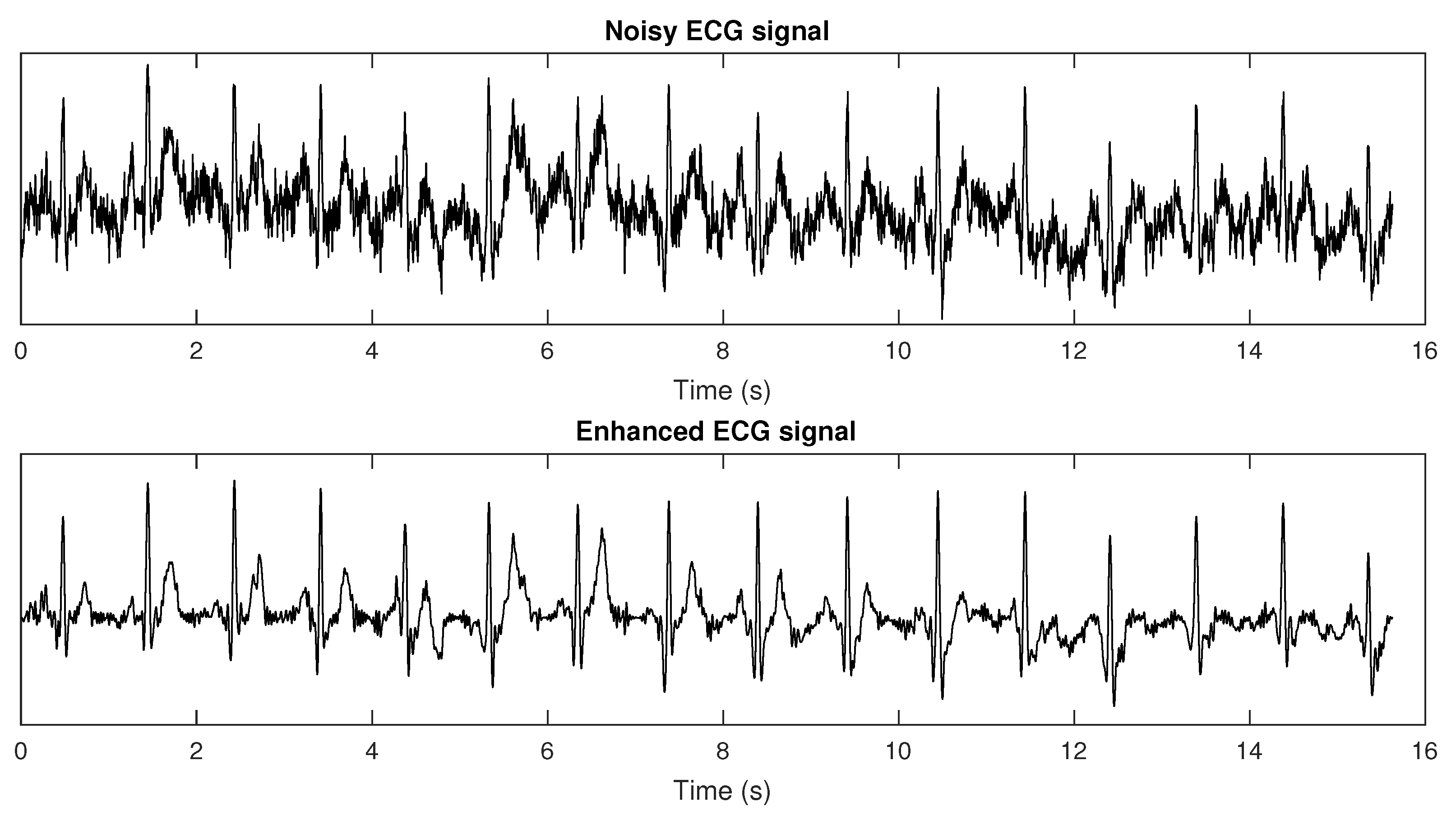
3.2.1. Electrocardiograms
3.2.2. Speech Signals
3.2.3. Electroencephalograms
3.3. Blind Source Separation
3.3.1. Measuring Breathing Rate from ECGs
3.3.2. Heart and Lung Sound Separation from Breath Sound Recordings
3.4. Noise-Robust Feature Extraction
3.4.1. Electrocardiograms
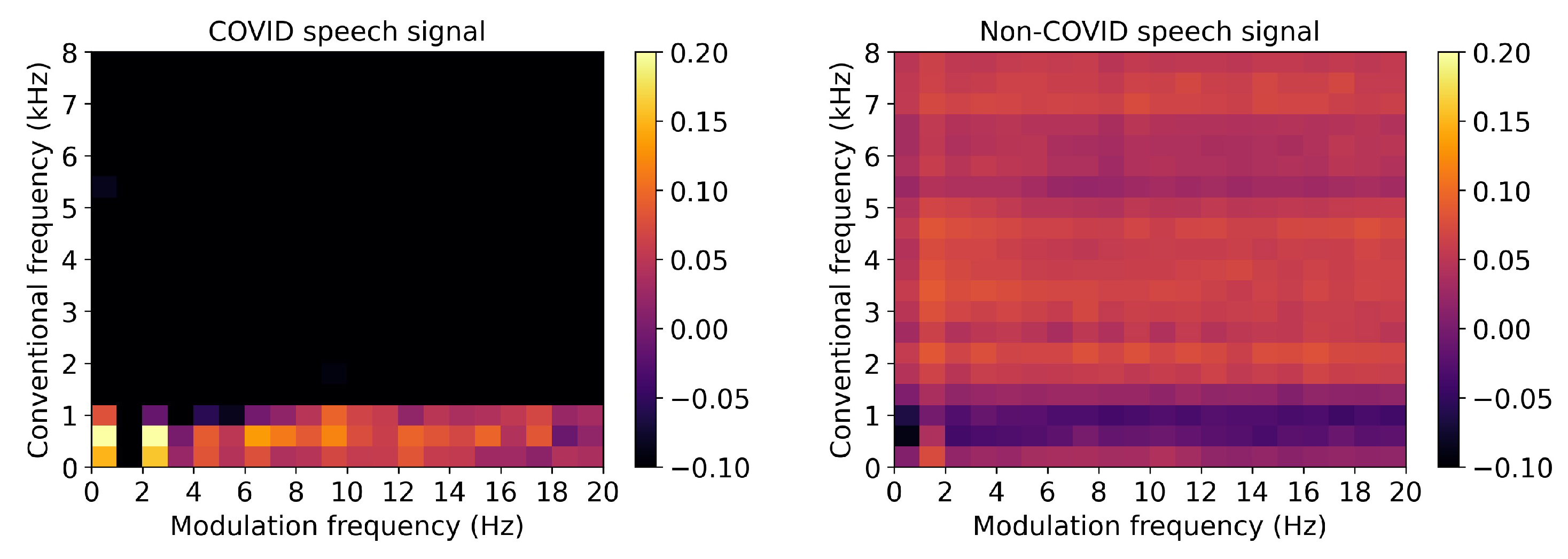
3.4.2. Speech Signals
3.4.3. Electroencephalograms
3.4.4. Accelerometry
3.5. Disease Characterization
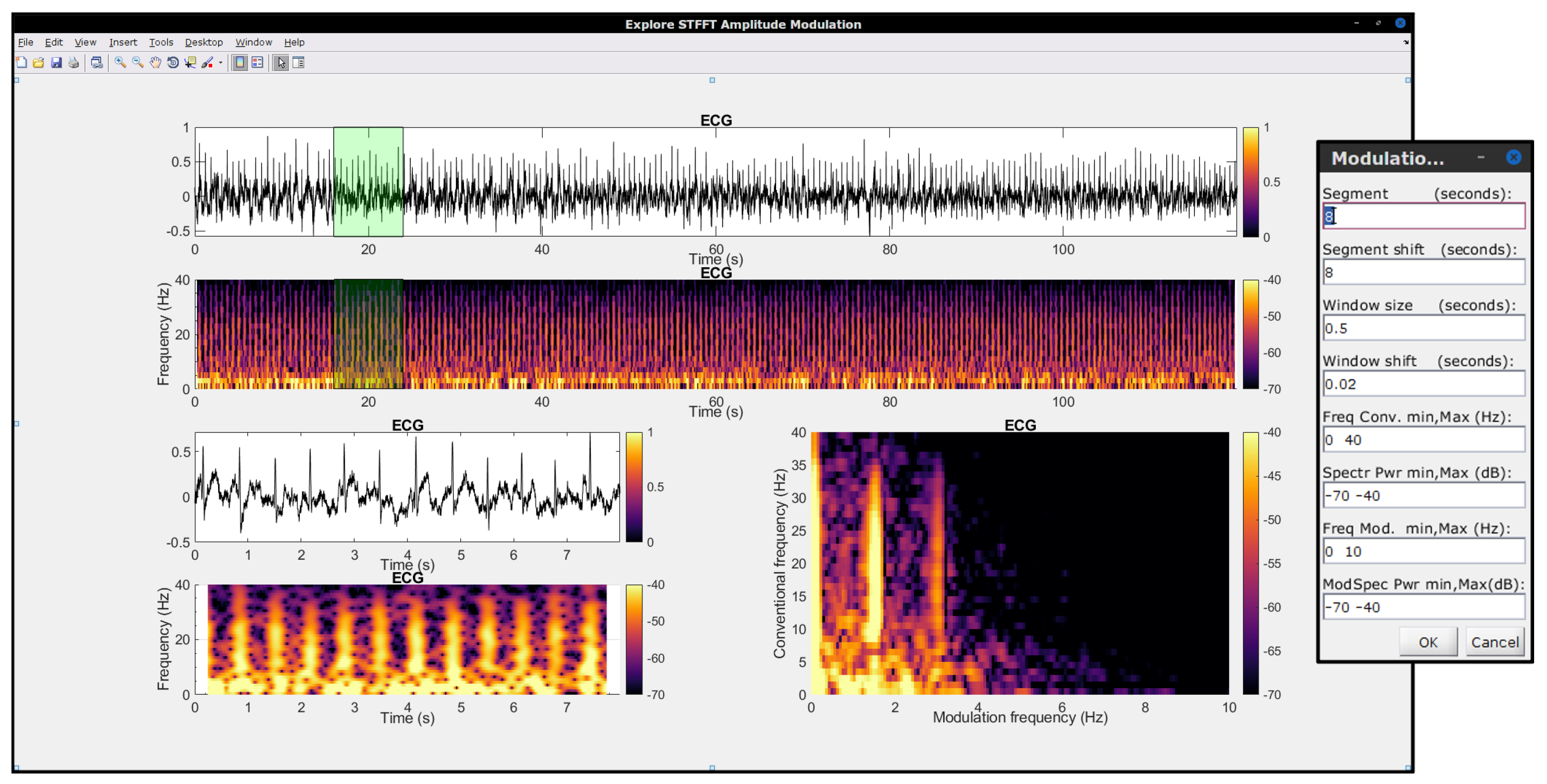
3.6. In-House Developed Software
4. Future Research Possibilities
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Markets And Markets. Wearable Technology Market by Product, Type, Application, and Geography—Global Forecast to 2026; Technical Report; Markets and Markets: Northbrook, IL, USA, 2021. [Google Scholar]
- Natarajan, A.; Su, H.W.; Heneghan, C. Assessment of physiological signs associated with COVID-19 measured using wearable devices. NPJ Digit. Med. 2020, 3, 156. [Google Scholar] [CrossRef] [PubMed]
- Smarr, B.L.; Aschbacher, K.; Fisher, S.M.; Chowdhary, A.; Dilchert, S.; Puldon, K.; Rao, A.; Hecht, F.M.; Mason, A.E. Feasibility of continuous fever monitoring using wearable devices. Sci. Rep. 2020, 10, 21640. [Google Scholar] [CrossRef] [PubMed]
- Bogu, G.K.; Snyder, M.P. Deep learning-based detection of COVID-19 using wearables data. MedRxiv 2021. [Google Scholar] [CrossRef]
- Dagher, L.; Shi, H.; Zhao, Y.; Marrouche, N.F. Wearables in cardiology: Here to stay. Heart Rhythm 2020, 17, 889–895. [Google Scholar] [CrossRef] [PubMed]
- Celik, Y.; Stuart, S.; Woo, W.L.; Godfrey, A. Gait analysis in neurological populations: Progression in the use of wearables. Med. Eng. Phys. 2020, 87, 9–29. [Google Scholar] [CrossRef]
- Feng, T.; Booth, B.M.; Baldwin-Rodríguez, B.; Osorno, F.; Narayanan, S. A multimodal analysis of physical activity, sleep, and work shift in nurses with wearable sensor data. Sci. Rep. 2021, 11, 1–12. [Google Scholar]
- Beauchamp, U.L.; Pappot, H.; Holländer-Mieritz, C. The use of wearables in clinical trials during cancer treatment: Systematic review. JMIR MHealth UHealth 2020, 8, e22006. [Google Scholar] [CrossRef]
- Smith, E.N.; Santoro, E.; Moraveji, N.; Susi, M.; Crum, A.J. Integrating wearables in stress management interventions: Promising evidence from a randomized trial. Int. J. Stress Manag. 2020, 27, 172. [Google Scholar] [CrossRef]
- Shah, R.V.; Grennan, G.; Zafar-Khan, M.; Alim, F.; Dey, S.; Ramanathan, D.; Mishra, J. Personalized machine learning of depressed mood using wearables. Transl. Psychiatry 2021, 11, 1–18. [Google Scholar]
- Sun, W.; Liu, J.; Zhang, H. When smart wearables meet intelligent vehicles: Challenges and future directions. IEEE Wirel. Commun. 2017, 24, 58–65. [Google Scholar] [CrossRef]
- Jiang, Y.; Li, Z.; Wang, J. Ptrack: Enhancing the applicability of pedestrian tracking with wearables. IEEE Trans. Mob. Comput. 2018, 18, 431–443. [Google Scholar] [CrossRef]
- Buruk, O.; Salminen, M.; Xi, N.; Nummenmaa, T.; Hamari, J. Towards the Next Generation of Gaming Wearables. In Proceedings of the 2021 CHI Conference on Human Factors in Computing Systems, Yokohama, Japan, 8–13 May 2021; pp. 1–15. [Google Scholar]
- Buruk, O.; Hamari, J. Towards the Next Generation of Extended Reality Wearables. In Proceedings of the Extended Abstracts of the 2021 CHI Conference on Human Factors in Computing Systems, Yokohama, Japan, 8–13 May 2021; pp. 1–7. [Google Scholar]
- Aun, N.F.M.; Soh, P.J.; Al-Hadi, A.A.; Jamlos, M.F.; Vandenbosch, G.A.; Schreurs, D. Revolutionizing wearables for 5G: 5G technologies: Recent developments and future perspectives for wearable devices and antennas. IEEE Microw. Mag. 2017, 18, 108–124. [Google Scholar] [CrossRef]
- Ahn, C.R.; Lee, S.; Sun, C.; Jebelli, H.; Yang, K.; Choi, B. Wearable sensing technology applications in construction safety and health. J. Constr. Eng. Manag. 2019, 145, 03119007. [Google Scholar] [CrossRef]
- Svertoka, E.; Rusu-Casandra, A.; Marghescu, I. State-of-the-Art of Industrial Wearables: A Systematic Review. In Proceedings of the 2020 13th International Conference on Communications (COMM), Bucharest, Romania, 18–20 June 2020; pp. 411–415. [Google Scholar]
- Haddad, M.; Hermassi, S.; Aganovic, Z.; Dalansi, F.; Kharbach, M.; Mohamed, A.O.; Bibi, K.W. Ecological validation and reliability of hexoskin wearable body metrics tool in measuring pre-exercise and peak heart rate during shuttle run test in professional handball players. Front. Physiol. 2020, 11, 957. [Google Scholar] [CrossRef]
- Cho, S.; Ensari, I.; Weng, C.; Kahn, M.G.; Natarajan, K. Factors Affecting the Quality of Person-Generated Wearable Device Data and Associated Challenges: Rapid Systematic Review. JMIR MHealth UHealth 2021, 9, e20738. [Google Scholar] [CrossRef] [PubMed]
- Martinez, G.J.; Mattingly, S.M.; Mirjafari, S.; Nepal, S.K.; Campbell, A.T.; Dey, A.K.; Striegel, A.D. On the quality of real-world wearable data in a longitudinal study of information workers. In Proceedings of the 2020 IEEE International Conference on Pervasive Computing and Communications Workshops (PerCom Workshops), Austin, TX, USA, 23–27 March 2020; pp. 1–6. [Google Scholar]
- Bizzego, A.; Gabrieli, G.; Furlanello, C.; Esposito, G. Comparison of wearable and clinical devices for acquisition of peripheral nervous system signals. Sensors 2020, 20, 6778. [Google Scholar] [CrossRef]
- Baek, H.J.; Shin, J. Effect of missing inter-beat interval data on heart rate variability analysis using wrist-worn wearables. J. Med. Syst. 2017, 41, 1–9. [Google Scholar] [CrossRef]
- Barrios, L.; Oldrati, P.; Santini, S.; Lutterotti, A. Evaluating the accuracy of heart rate sensors based on photoplethysmography for in-the-wild analysis. In Proceedings of the 13th EAI International Conference on Pervasive Computing Technologies for Healthcare, Trento, Italy, 20–23 May 2019; pp. 251–261. [Google Scholar]
- Liu, H.; Chen, F.; Hartmann, V.; Khalid, S.G.; Hughes, S.; Zheng, D. Comparison of different modulations of photoplethysmography in extracting respiratory rate: From a physiological perspective. Physiol. Meas. 2020, 41, 094001. [Google Scholar] [CrossRef]
- Yang, S.Y.; Lin, Y.P. Validating a LEGO-Like EEG Headset for a Simultaneous Recording of Wet-and Dry-Electrode Systems During Treadmill Walking. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; pp. 4055–4058. [Google Scholar]
- Fuller, D.; Colwell, E.; Low, J.; Orychock, K.; Tobin, M.A.; Simango, B.; Buote, R.; Van Heerden, D.; Luan, H.; Cullen, K.; et al. Reliability and validity of commercially available wearable devices for measuring steps, energy expenditure, and heart rate: Systematic review. JMIR MHealth UHealth 2020, 8, e18694. [Google Scholar] [CrossRef]
- Daly, I.; Pichiorri, F.; Faller, J.; Kaiser, V.; Kreilinger, A.; Scherer, R.; Müller-Putz, G. What does clean EEG look like? In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; pp. 3963–3966. [Google Scholar]
- Zaman, M.S.; Morshed, B.I. Estimating Reliability of Signal Quality of Physiological Data from Data Statistics Itself for Real-time Wearables. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; pp. 5967–5970. [Google Scholar]
- Behar, J.; Oster, J.; Li, Q.; Clifford, G. A single channel ECG quality metric. In 2012 Computing in Cardiology; IEEE: Piscataway, NJ, USA, 2012; pp. 381–384. [Google Scholar]
- Mahdid, Y.; Lee, U.; Blain-Moraes, S. Assessing the Quality of Wearable EEG Systems Using Functional Connectivity. IEEE Access 2020, 8, 193214–193225. [Google Scholar] [CrossRef]
- Wang, Z.; Zhu, J.; Yan, T.; Yang, L. A new modified wavelet-based ECG denoising. Comput. Assist. Surg. 2019, 24, 174–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nasseri, M.; Nurse, E.; Glasstetter, M.; Böttcher, S.; Gregg, N.M.; Laks Nandakumar, A.; Joseph, B.; Pal Attia, T.; Viana, P.F.; Bruno, E.; et al. Signal quality and patient experience with wearable devices for epilepsy management. Epilepsia 2020, 61, S25–S35. [Google Scholar] [CrossRef] [PubMed]
- Mammone, N.; La Foresta, F.; Morabito, F.C. Automatic artifact rejection from multichannel scalp EEG by wavelet ICA. IEEE Sens. J. 2011, 12, 533–542. [Google Scholar] [CrossRef]
- Qi, Y.; Yao, F.; Yu, M. A Single-Channel ICA-R Method for Speech Signal Denoising combining EMD and Wavelet. J. Comput. 2014, 9, 2082–2090. [Google Scholar] [CrossRef]
- Vullings, R.; De Vries, B.; Bergmans, J.W. An adaptive Kalman filter for ECG signal enhancement. IEEE Trans. Biomed. Eng. 2010, 58, 1094–1103. [Google Scholar] [CrossRef] [Green Version]
- Rosanne, O.; Albuquerque, I.; Cassani, R.; Gagnon, J.F.; Tremblay, S.; Falk, T.H. Adaptive filtering for improved eeg-based mental workload assessment of ambulant users. Front. Neurosci. 2021, 15, 341. [Google Scholar] [CrossRef]
- Arsene, C.T.; Hankins, R.; Yin, H. Deep learning models for denoising ECG signals. In Proceedings of the 2019 27th European Signal Processing Conference (EUSIPCO), Coruna, Spain, 2–6 September 2019; pp. 1–5. [Google Scholar]
- Singh, P.; Pradhan, G. A new ECG denoising framework using generative adversarial network. IEEE/ACM Trans. Comput. Biol. Bioinform. 2020, 18, 759–764. [Google Scholar] [CrossRef]
- Michelsanti, D.; Tan, Z.H.; Zhang, S.X.; Xu, Y.; Yu, M.; Yu, D.; Jensen, J. An overview of deep-learning-based audio-visual speech enhancement and separation. arXiv 2021, arXiv:2008.09586. [Google Scholar] [CrossRef]
- Xu, H.; Yan, W.; Lan, K.; Ma, C.; Wu, D.; Wu, A.; Yang, Z.; Wang, J.; Zang, Y.; Yan, M.; et al. Assessing Electrocardiogram and Respiratory Signal Quality of a Wearable Device (SensEcho): Semisupervised Machine Learning-Based Validation Study. JMIR MHealth UHealth 2021, 9, e25415. [Google Scholar] [CrossRef]
- Minguillon, J.; Lopez-Gordo, M.A.; Pelayo, F. Trends in EEG-BCI for daily-life: Requirements for artifact removal. Biomed. Signal Process. Control 2017, 31, 407–418. [Google Scholar] [CrossRef]
- Gabard-Durnam, L.J.; Mendez Leal, A.S.; Wilkinson, C.L.; Levin, A.R. The Harvard Automated Processing Pipeline for Electroencephalography (HAPPE): Standardized processing software for developmental and high-artifact data. Front. Neurosci. 2018, 12, 97. [Google Scholar] [CrossRef]
- Han, X.; Hu, Y.; Foschini, L.; Chinitz, L.; Jankelson, L.; Ranganath, R. Deep learning models for electrocardiograms are susceptible to adversarial attack. Nat. Med. 2020, 26, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Cassani, R.; Falk, T.H. Spectrotemporal modeling of biomedical signals: Theoretical foundation and applications. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Tobon, D.; Falk, T.H.; Maier, M. MS-QI: A modulation spectrum-based ECG quality index for telehealth applications. IEEE Trans. Biomed. Eng. 2014, 63, 1613–1622. [Google Scholar] [CrossRef] [PubMed]
- Cassani, R.; Falk, T.H.; Horai, A.; Gheorghe, L.A. Evaluating the measurement of driver heart and breathing rates from a sensor-equipped steering wheel using spectrotemporal signal processing. In Proceedings of the 2019 IEEE Intelligent Transportation Systems Conference (ITSC), Auckland, New Zeland, 27–30 October 2019; pp. 2843–2847. [Google Scholar]
- Tobón, D.P.; Falk, T.H. Online ECG quality assessment for context-aware wireless body area networks. In Proceedings of the 2015 IEEE 28th Canadian Conference on Electrical and Computer Engineering (CCECE), Halifax, NS, Canada, 3–6 May 2015; pp. 587–592. [Google Scholar]
- Drullman, R.; Festen, J.M.; Plomp, R. Effect of reducing slow temporal modulations on speech reception. J. Acoust. Soc. Am. 1994, 95, 2670–2680. [Google Scholar] [CrossRef] [PubMed]
- Plomp, R.; Houtgast, T.; Steeneken, H.J. The modulation transfer function in audition. In Limits in Perception; CRC Press: Boca Raton, FL, USA, 2020; pp. 117–138. [Google Scholar]
- Falk, T.H.; Zheng, C.; Chan, W.Y. A non-intrusive quality and intelligibility measure of reverberant and dereverberated speech. IEEE Trans. Audio Speech Lang. Process. 2010, 18, 1766–1774. [Google Scholar] [CrossRef]
- Falk, T.H.; Chan, W.Y. Temporal dynamics for blind measurement of room acoustical parameters. IEEE Trans. Instrum. Meas. 2010, 59, 978–989. [Google Scholar] [CrossRef] [Green Version]
- Braun, S.; Santos, J.F.; Habets, E.A.; Falk, T.H. Dual-channel modulation energy metric for direct-to-reverberation ratio estimation. In Proceedings of the 2018 IEEE International Conference on Acoustics, Speech and Signal Processing (ICASSP), Calgary, AB, Canada, 15–20 April 2018; pp. 206–210. [Google Scholar]
- Santos, J.F.; Senoussaoui, M.; Falk, T.H. An improved non-intrusive intelligibility metric for noisy and reverberant speech. In Proceedings of the 2014 14th International Workshop on Acoustic Signal Enhancement (IWAENC), Juan-les-Pins, France, 8–11 September 2014; pp. 55–59. [Google Scholar]
- Santos, J.F.; Cosentino, S.; Hazrati, O.; Loizou, P.C.; Falk, T.H. Objective speech intelligibility measurement for cochlear implant users in complex listening environments. Speech Commun. 2013, 55, 815–824. [Google Scholar] [CrossRef] [Green Version]
- Senoussaoui, M.; Santos, J.F.; Falk, T.H. SRMR variants for improved blind room acoustics characterization. arXiv 2015, arXiv:1510.04707. [Google Scholar]
- Cauchi, B.; Siedenburg, K.; Santos, J.F.; Falk, T.H.; Doclo, S.; Goetze, S. Non-intrusive speech quality prediction using modulation energies and lstm-network. IEEE/ACM Trans. Audio Speech Lang. Process. 2019, 27, 1151–1163. [Google Scholar] [CrossRef]
- Santos, J.F.; Bouserhal, R.; Voix, J.; Falk, T.H. Objective speech quality estimation of in-ear microphone speech. In Proceedings of the 5th ISCA/DEGA Workshop on Perceptual Quality of Systems (PQS 2016), Berlin, Germany, 29–31 August 2016. [Google Scholar]
- Kinoshita, K.; Delcroix, M.; Gannot, S.; Habets, E.A.; Haeb-Umbach, R.; Kellermann, W.; Leutnant, V.; Maas, R.; Nakatani, T.; Raj, B.; et al. A summary of the REVERB challenge: State-of-the-art and remaining challenges in reverberant speech processing research. EURASIP J. Adv. Signal Process. 2016, 2016, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Eaton, J.; Gaubitch, N.D.; Moore, A.H.; Naylor, P.A. Estimation of room acoustic parameters: The ACE challenge. IEEE/ACM Trans. Audio Speech Lang. Process. 2016, 24, 1681–1693. [Google Scholar] [CrossRef] [Green Version]
- Falk, T.H.; Parsa, V.; Santos, J.F.; Arehart, K.; Hazrati, O.; Huber, R.; Kates, J.M.; Scollie, S. Objective quality and intelligibility prediction for users of assistive listening devices: Advantages and limitations of existing tools. IEEE Signal Process. Mag. 2015, 32, 114–124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tobón, D.P.; Falk, T.H. Adaptive spectro-temporal filtering for electrocardiogram signal enhancement. IEEE J. Biomed. Health Informatics 2016, 22, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Tompkins, W.J. A real-time QRS detection algorithm. In IEEE Transactions on Biomedical Engineering; IEEE: Piscataway, NJ, USA, 1985; pp. 230–236. [Google Scholar]
- Tiwari, A.; Albuquerque, I.; Parent, M.; Gagnon, J.F.; Lafond, D.; Tremblay, S.; Falk, T.H. A Comparison of Two ECG Inter-beat Interval Measurement Methods for HRV-Based MentalWorkload Prediction of Ambulant Users. In Proceedings of the CMBES Proceedings, Ottawa, ON, Canada, 21–24 May 2019; Volume 42. [Google Scholar]
- Hermansky, H.; Wan, E.A.; Avendano, C. Speech enhancement based on temporal processing. In Proceedings of the 1995 International Conference on Acoustics, Speech, and Signal Processing, Detroit, MI, USA, 9–12 May 1995; Volume 1, pp. 405–408. [Google Scholar]
- Avendano, C.; Hermansky, H.; Vis, M.; Bayya, A. Adaptive speech enhancement using frequency-specific SNR estimates. In Proceedings of the IVTTA’96 Workshop on Interactive Voice Technology for Telecommunications Applications, Basking Ridge, NJ, USA, 30 September–1 October 1996; pp. 65–68. [Google Scholar]
- Hermansky, H.; Morgan, N. RASTA processing of speech. IEEE Trans. Speech Audio Process. 1994, 2, 578–589. [Google Scholar] [CrossRef] [Green Version]
- Falk, T.H.; Stadler, S.; Kleijn, W.B.; Chan, W.Y. Noise suppression based on extending a speech-dominated modulation band. In Proceedings of the Eighth Annual Conference of the International Speech Communication Association, Antwerp, Belgium, 27–31 August 2007. [Google Scholar]
- Karimian-Azari, S.; Falk, T.H. Modulation spectrum based beamforming for speech enhancement. In Proceedings of the 2017 IEEE Workshop on Applications of Signal Processing to Audio and Acoustics (WASPAA), New Paltz, NY, USA, 15–18 October 2017; pp. 91–95. [Google Scholar]
- Dos Santos, E.M.; Cassani, R.; Falk, T.H.; Fraga, F.J. Improved motor imagery brain-computer interface performance via adaptive modulation filtering and two-stage classification. Biomed. Signal Process. Control 2020, 57, 101812. [Google Scholar] [CrossRef]
- Cassani, R.; Narayanan, S.; Falk, T.H. Respiration rate estimation from noisy electrocardiograms based on modulation spectral analysis. In Proceedings of the CMBES Proceedings, Charlottetown, PEI, Canada, 8–11 May 2018; Volume 41. [Google Scholar]
- Falk, T.H.; Chan, W.Y.; Sejdic, E.; Chau, T. Spectro-temporal analysis of auscultatory sounds. In New Developments in Biomedical Engineering; IntechOpen: London, UK, 2010; pp. 93–104. [Google Scholar]
- Tobon, D.P.; Jayaraman, S.; Falk, T.H. Spectro-temporal electrocardiogram analysis for noise-robust heart rate and heart rate variability measurement. IEEE J. Transl. Eng. Health Med. 2017, 5, 1–11. [Google Scholar] [CrossRef]
- Falk, T.H.; Chan, W.Y. Modulation spectral features for robust far-field speaker identification. IEEE Trans. Audio Speech Lang. Process. 2009, 18, 90–100. [Google Scholar] [CrossRef] [Green Version]
- Avila, A.R.; Akhtar, Z.; Santos, J.F.; O’Shaughnessy, D.; Falk, T.H. Feature pooling of modulation spectrum features for improved speech emotion recognition in the wild. IEEE Trans. Affect. Comput. 2018, 12, 177–188. [Google Scholar] [CrossRef]
- Avila, A.R.; Kshirsagar, S.R.; Tiwari, A.; Lafond, D.; O’Shaughnessy, D.; Falk, T.H. Speech-based stress classification based on modulation spectral features and convolutional neural networks. In Proceedings of the 2019 27th European Signal Processing Conference (EUSIPCO), Coruna, Spain, 2–6 September 2019; pp. 1–5. [Google Scholar]
- Sarria-Paja, M.; Falk, T.H. Whispered speech detection in noise using auditory-inspired modulation spectrum features. IEEE Signal Process. Lett. 2013, 20, 783–786. [Google Scholar] [CrossRef]
- Sarria-Paja, M.; Falk, T.H. Fusion of bottleneck, spectral and modulation spectral features for improved speaker verification of neutral and whispered speech. Speech Commun. 2018, 102, 78–86. [Google Scholar] [CrossRef]
- Drouin-Picaro, A.; Albuquerque, I.; Gagnon, J.F.; Lafond, D.; Falk, T.H. EEG coupling features: Towards mental workload measurement based on wearables. In Proceedings of the 2017 IEEE International Conference on Systems, Man, and Cybernetics (SMC), Banff, AB, USA, 5–8 October 2017; pp. 28–33. [Google Scholar]
- Albuquerque, I.; Rosanne, O.; Gagnon, J.F.; Tremblay, S.; Falk, T.H. Fusion of spectral and spectro-temporal EEG features for mental workload assessment under different levels of physical activity. In Proceedings of the 2019 9th International IEEE/EMBS Conference on Neural Engineering (NER), San Francisco, CA, USA, 20–23 March 2019; pp. 311–314. [Google Scholar]
- Clerico, A.; Tiwari, A.; Gupta, R.; Jayaraman, S.; Falk, T.H. Electroencephalography amplitude modulation analysis for automated affective tagging of music video clips. Front. Comput. Neurosci. 2018, 11, 115. [Google Scholar] [CrossRef] [PubMed]
- Trambaiolli, L.R.; Cassani, R.; Falk, T.H. EEG spectro-temporal amplitude modulation as a measurement of cortical hemodynamics: An EEG-fNIRS study. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; pp. 3481–3484. [Google Scholar]
- Trambaiolli, L.; Cassani, R.; Biazoli, C.; Cravo, A.; Sato, J.; Falk, T. Resting-awake EEG amplitude modulation can predict performance of an fNIRS-based neurofeedback task. In Proceedings of the 2018 IEEE International Conference on Systems, Man, and Cybernetics (SMC), Miyazaki, Japan, 7–10 October 2018; pp. 1128–1132. [Google Scholar]
- Tobon, D.; Garudadri, H.; Godino, J.; Godbole, S.; Patrick, K.; Falk, T.H. Improved gait speed calculation via modulation spectral analysis of noisy accelerometer data. IEEE Sens. J. 2020, 21, 520–528. [Google Scholar] [CrossRef]
- Khandelwal, S.; Wickström, N. Evaluation of the performance of accelerometer-based gait event detection algorithms in different real-world scenarios using the MAREA gait database. Gait Posture 2017, 51, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Fraga, F.J.; Falk, T.H.; Kanda, P.A.; Anghinah, R. Characterizing Alzheimer’s disease severity via resting-awake EEG amplitude modulation analysis. PLoS ONE 2013, 8, e72240. [Google Scholar] [CrossRef] [PubMed]
- Cassani, R.; Falk, T.H.; Fraga, F.J.; Kanda, P.A.; Anghinah, R. The effects of automated artifact removal algorithms on electroencephalography-based Alzheimer’s disease diagnosis. Front. Aging Neurosci. 2014, 6, 55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cassani, R.; Falk, T.H.; Fraga, F.J.; Cecchi, M.; Moore, D.K.; Anghinah, R. Towards automated electroencephalography-based Alzheimer’s disease diagnosis using portable low-density devices. Biomed. Signal Process. Control 2017, 33, 261–271. [Google Scholar] [CrossRef]
- Cassani, R.; Falk, T.H. Alzheimer’s disease diagnosis and severity level detection based on electroencephalography modulation spectral “patch” features. IEEE J. Biomed. Health Inform. 2019, 24, 1982–1993. [Google Scholar] [CrossRef]
- Lopes, M.K.S.; Cassani, R.; Falk, T.H. On the use of saliency maps and convolutional neural networks for improved Alzheimer’s disease assessment. In Proceedings of the 2021 Alzheimer’s Association International Conference, Denver, CO, USA, 26–30 July 2021. [Google Scholar]
- Falk, T.H.; Chan, W.Y.; Shein, F. Characterization of atypical vocal source excitation, temporal dynamics and prosody for objective measurement of dysarthric word intelligibility. Speech Commun. 2012, 54, 622–631. [Google Scholar] [CrossRef]
- Paja, M.S.; Falk, T.H. Automated dysarthria severity classification for improved objective intelligibility assessment of spastic dysarthric speech. In Proceedings of the Thirteenth Annual Conference of the International Speech Communication Association, Portland, OR, USA, 9–13 September 2012. [Google Scholar]
- Bedoya, S.; Katz, N.B.; Brian, J.; O’Shaughnessy, D.; Falk, T.H. Acoustic and prosodic analysis of vocalizations of 18-month-old toddlers with autism spectrum disorder. In Acoustic Analysis of Pathologies; De Gruyter: Berlin, Germany, 2020; pp. 93–126. [Google Scholar]
- Zhu, Y.; Falk, T.H. Fusion of Modulation Spectral and Spectral Features with Symptom Metadata for Improved Speech-Based Covid-19 Detection. In Proceedings of the 2022 IEEE International Conference on Acoustics, Speech and Signal Processing (ICASSP), Singapore, 22–27 May 2022; pp. 8997–9001. [Google Scholar]
- Atlas, L.; Clark, P.; Schimmel, S. Modulation Toolbox Version 2.1 for Matlab; University of Washington: Seattle, WA, USA, 2010. [Google Scholar]
- Cassani, R.; Albuquerque, I.; Monteiro, J.; Falk, T.H. AMA: An Open-source Amplitude Modulation Analysis Toolkit for Signal Processing Applications. In Proceedings of the 2019 IEEE Global Conference on Signal and Information Processing (GlobalSIP), Ottawa, ON, Canada, 11–14 November 2019; pp. 1–4. [Google Scholar]
- Pimentel, A.; Tiwari, A.; Falk, T.H. Human Mental State Monitoring in the Wild: Are We Better Off with Deeper Neural Networks or Improved Input Features? In Proceedings of the CMBES Proceedings, Virtual, 11–13 May 2021; Volume 44. [Google Scholar]
- Shah, S.A.; Yang, X.; Abbasi, Q.H. Cognitive health care system and its application in pill-rolling assessment. Int. J. Numer. Model. Electron. Netw. Devices Fields 2019, 32, e2632. [Google Scholar] [CrossRef]
- Saeed, U.; Yaseen Shah, S.; Aziz Shah, S.; Liu, H.; Alhumaidi Alotaibi, A.; Althobaiti, T.; Ramzan, N.; Ullah Jan, S.; Ahmad, J.; H Abbasi, Q. Multiple Participants’ Discrete Activity Recognition in a Well-Controlled Environment Using Universal Software Radio Peripheral Wireless Sensing. Sensors 2022, 22, 809. [Google Scholar] [CrossRef]
- Yagoubi, M.R.; Serir, A.; Beghdadi, A. Joint enhancement-compression of handwritten document images through DjVu encoder. J. Vis. Commun. Image Represent. 2016, 41, 324–338. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tiwari, A.; Cassani, R.; Kshirsagar, S.; Tobon, D.P.; Zhu, Y.; Falk, T.H. Modulation Spectral Signal Representation for Quality Measurement and Enhancement of Wearable Device Data: A Technical Note. Sensors 2022, 22, 4579. https://doi.org/10.3390/s22124579
Tiwari A, Cassani R, Kshirsagar S, Tobon DP, Zhu Y, Falk TH. Modulation Spectral Signal Representation for Quality Measurement and Enhancement of Wearable Device Data: A Technical Note. Sensors. 2022; 22(12):4579. https://doi.org/10.3390/s22124579
Chicago/Turabian StyleTiwari, Abhishek, Raymundo Cassani, Shruti Kshirsagar, Diana P. Tobon, Yi Zhu, and Tiago H. Falk. 2022. "Modulation Spectral Signal Representation for Quality Measurement and Enhancement of Wearable Device Data: A Technical Note" Sensors 22, no. 12: 4579. https://doi.org/10.3390/s22124579
APA StyleTiwari, A., Cassani, R., Kshirsagar, S., Tobon, D. P., Zhu, Y., & Falk, T. H. (2022). Modulation Spectral Signal Representation for Quality Measurement and Enhancement of Wearable Device Data: A Technical Note. Sensors, 22(12), 4579. https://doi.org/10.3390/s22124579





