Sandwich Integration Technique for the Pressure Sensor Detection of Occlusal Force In Vitro
Abstract
:1. Introduction
2. Materials and Methods
2.1. Portable Biosensor Design by Sandwich Integration Technique
2.1.1. CAD Designing and 3D Printing
2.1.2. Conjunction of Resin Specimens and Piezoresistive-Film Sensors
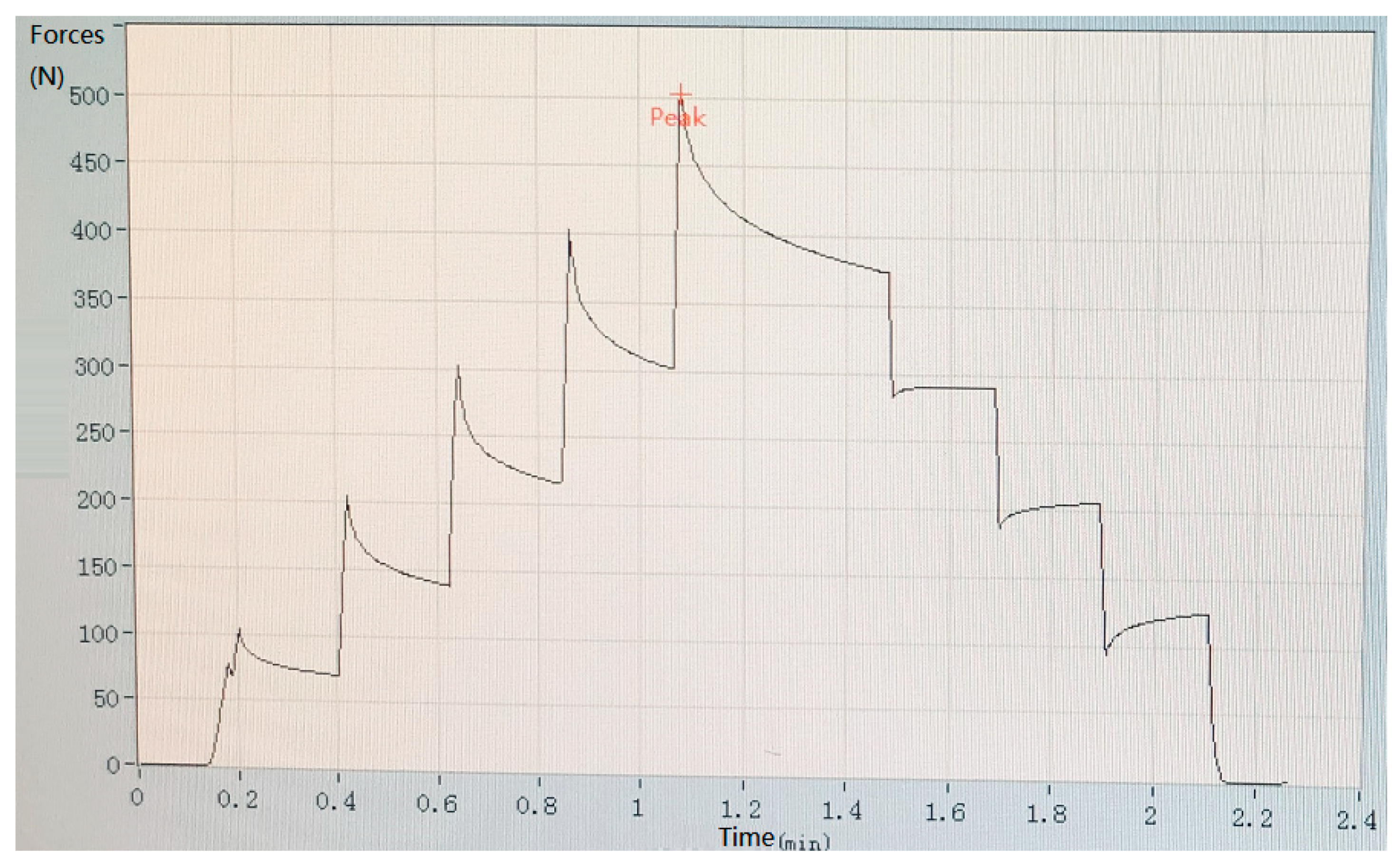
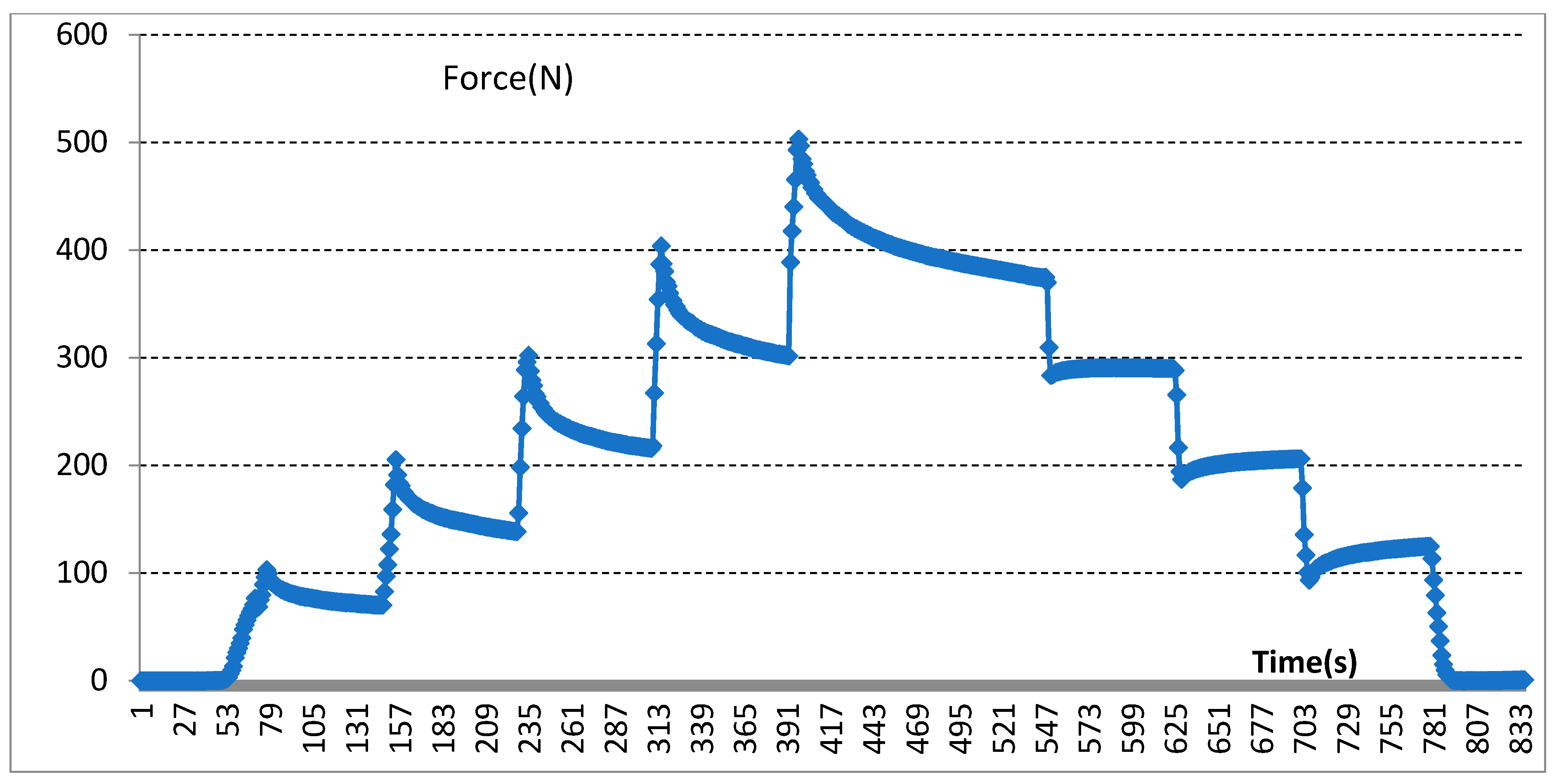
2.2. Mechanical Pressure Loading Test
2.3. A Customized Pressure Signal Acquisition Controller
3. Results
3.1. Results of Sandwich Technique
3.2. Results of Mechanical Pressure Loading Test
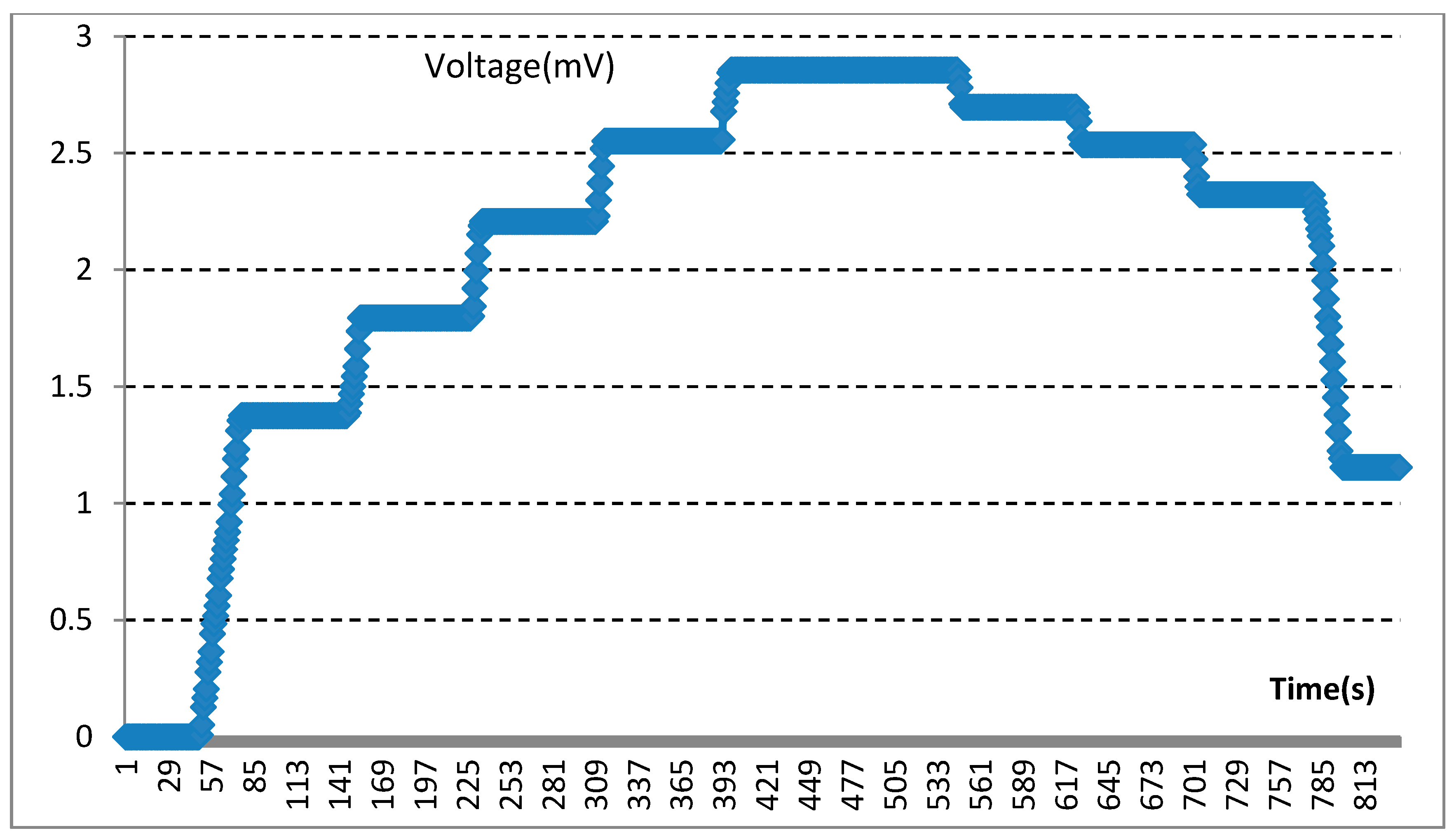
3.3. Results of Electrical Measurements of the Sensors
4. Discussion
4.1. Main Results
4.2. Integration Challenges of 3D Printing Devices and Piezo-Electric Sensors
4.3. Detection of Mechanical Signals
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Buvinic, S.; Balanta-Melo, J.; Kupczik, K.; Vásquez, W.; Beato, C.; Toro-Ibacache, V. Muscle-Bone Crosstalk in the Masticatory System: From Biomechanical to Molecular Interactions. Front. Endocrinol. 2020, 11, 606947. [Google Scholar] [CrossRef]
- Umesh, S.; Padma, S.; Asokan, S.; Srinivas, T. Fiber Bragg Grating based bite force measurement. J. Biomech. 2016, 49, 2877–2881. [Google Scholar] [CrossRef] [PubMed]
- Lepley, C.R.; Throckmorton, G.S.; Ceen, R.F.; Buschang, P.H. Relative contributions of occlusion, maximum bite force, and chewing cycle kinematics to masticatory performance. Am. J. Orthod. Dentofac. Orthop. 2011, 139, 606–613. [Google Scholar] [CrossRef]
- Bertolini, M.M.; Del Bel Cury, A.A.; Pizzoloto, L.; Acapa, I.R.H.; Shibli, J.A.; Bordin, D. Does traumatic occlusal forces lead to peri-implant bone loss? A systematic review. Braz. Oral Res. 2019, 33, e069. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Abreu, R.A.M.; Pereira, M.D.; Furtado, F.; Prado, G.P.R.; Mestriner, W.; Ferreira, L.M. Masticatory efficiency and bite force in individuals with normal occlusion. Arch. Oral Biol. 2014, 59, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Röhrle, O.; Saini, H.; Ackland, D.C. Occlusal loading during biting from an experimental and simulation point of view. Dent. Mater. 2018, 34, 58–68. [Google Scholar] [CrossRef]
- Bakke, M. Bite Force and Occlusion. Semin. Orthod. 2006, 12, 120–126. [Google Scholar] [CrossRef]
- Verma, T.P.; Kumathalli, K.I.; Jain, V.; Kumar, R. Bite Force Recording Devices—A Review. J. Clin. Diagn. Res. 2017, 11, ZE01–ZE05. [Google Scholar] [CrossRef] [PubMed]
- Nota, A.; Tecco, S.; Cioffi, C.; Beraldi, A.; Padulo, J.; Baldini, A. Occlusion time analysis in military pilots affected by bruxism. Sci. Rep. 2019, 9, 1408. [Google Scholar] [CrossRef] [Green Version]
- Shiga, H.; Komino, M.; Uesugi, H.; Sano, M.; Yokoyama, M.; Nakajima, K.; Ishikawa, A. Comparison of two dental prescale systems used for the measurement of occlusal force. Odontology 2020, 108, 676–680. [Google Scholar] [CrossRef]
- Gu, Y.Z.; Bai, Y.X.; Xie, X.J. Bite Force Transducers and Measurement Devices. Front. Bioeng. Biotechnol. 2021, 9, 665081. [Google Scholar] [CrossRef]
- D’Attilio, M.; Di Carlo, B.; Caroccia, F.; Moscagiuri, F.; D’Angelo, D.; Chiarelli, F.; Festa, F.; Breda, L. Clinical and Instrumental TMJ Evaluation in Children and Adolescents with Juvenile Idiopathic Arthritis: A Case—Control Study. Appl. Sci. 2021, 11, 5380. [Google Scholar] [CrossRef]
- Kim, J.J.; Stafford, G.R.; Beauchamp, C.; Kim, S.A. Development of a Dental Implantable Temperature Sensor for Real-Time Diagnosis of Infectious Disease. Sensors 2020, 20, 3953. [Google Scholar] [CrossRef] [PubMed]
- Quadir, N.A.; Albasha, L.; Taghadosi, M.; Qaddoumi, N.; Hatahet, B. Low-Power Implanted Sensor for Orthodontic Bond Failure Diagnosis and Detection. IEEE Sens. J. 2018, 18, 3003–3009. [Google Scholar] [CrossRef]
- Kim, J.; Imani, S.; de Araujo, W.R.; Warchall, J.; Valdés-Ramírez, G.; Paixao, T.R.L.C.; Mercier, P.P.; Wang, J. Wearable salivary uric acid mouthguard biosensor with integrated wireless electronics. Biosens. Bioelectron. 2015, 74, 1061–1068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tseng, P.; Napier, B.; Garbarini, L.; Kaplan, D.L.; Omenetto, F.G. Functional, RF-Trilayer Sensors for Tooth-Mounted, Wireless Monitoring of the Oral Cavity and Food Consumption. Adv. Mater. 2018, 30, e1703257. [Google Scholar] [CrossRef]
- Song, J.K.; Cho, T.H.; Pan, H.; Song, Y.M.; Kim, I.S.; Lee, T.H.; Hwang, S.J.; Kim, S.J. An electronic device for accelerating bone formation in tissues surrounding a dental implant. Bioelectromagnetics 2009, 30, 374–384. [Google Scholar] [CrossRef]
- Albasha, L.; Qaddoumi, N.; Hatahet, B.; Quadir, N.; Taghadosi, M. Design Challenges in Wireless Sensors for Dental Applications. In The IoT Physical Layer; Elfadel, I., Ismail, M., Eds.; Springer: Cham, Switzerland, 2019; pp. 105–126. [Google Scholar] [CrossRef]
- Lin, K.-R.; Chang, C.-H.; Liu, T.-H.; Lin, S.-W.; Lin, C.-H. Experimental and numerical estimations into the force distribution on an occlusal surface utilizing a flexible force sensor array. J. Biomech. 2011, 44, 1879–1884. [Google Scholar] [CrossRef]
- Fastier-Wooller, J.; Phan, H.-P.; Dinh, T.; Nguyen, T.-K.; Cameron, A.; Oechsner, A.; Dao, D.V. Novel Low-Cost Sensor for Human Bite Force Measurement. Sensors 2016, 16, 1244. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, C.P.; Glantz, P.O.J.; Svensson, S.A.; Bergmark, A. A novel sensor for bite force determinations. Dent. Mater. 2003, 19, 118–126. [Google Scholar] [CrossRef]
- Lantada, A.D.; Bris, C.G.; Morgado, P.L.; Maudes, J.S. Novel System for Bite-Force Sensing and Monitoring Based on Magnetic Near Field Communication. Sensors 2012, 12, 11544–11558. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, J.X.; Liu, L.J.; Gao, P.; Zheng, Y.H.; Hou, W.X.; Wang, J.H. Intelligent Occlusion Stabilization Splint with Stress-Sensor System for Bruxism Diagnosis and Treatment. Sensors 2019, 20, 89. [Google Scholar] [CrossRef] [Green Version]
- Imai, A.; Takamizawa, T.; Sugimura, R.; Tsujimoto, A.; Ishii, R.; Kawazu, M.; Saito, T.; Miyazaki, M. Interrelation among the handling, mechanical, and wear properties of the newly developed flowable resin composites. J. Mech. Behav. Biomed. Mater. 2019, 89, 72–80. [Google Scholar] [CrossRef] [PubMed]
- AlGhazzawi, T.F. Advancements in CAD/CAM technology: Options for practical implementation. J. Prosthodont. Res. 2016, 60, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Nesic, D.; Schaefer, B.M.; Sun, Y.; Saulacic, N.; Sailer, I. 3D Printing Approach in Dentistry: The Future for Personalized Oral Soft Tissue Regeneration. J. Clin. Med. 2020, 9, 2238. [Google Scholar] [CrossRef]
- Pillai, S.; Upadhyay, A.; Khayambashi, P.; Farooq, I.; Sabri, H.; Tarar, M.; Lee, K.T.; Harb, I.; Zhou, S.; Wang, Y.; et al. Dental 3D-Printing: Transferring Art from the Laboratories to the Clinics. Polymers 2021, 13, 157. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, T.A. Materials in digital dentistry—A review. J. Esthet. Restor. Dent. 2020, 32, 171–181. [Google Scholar] [CrossRef]
- Rafiee, M.; Farahani, R.D.; Therriault, D. Multi-Material 3D and 4D Printing: A Survey. Adv. Sci. 2020, 7, 1902307. [Google Scholar] [CrossRef]
- Seo, M.; Hwang, S.; Hwang, T.; Yeo, J. Fabrication of Soft Sensor Using Laser Processing Techniques: For the Alternative 3D Printing Process. Materials 2019, 12, 2955. [Google Scholar] [CrossRef] [Green Version]
- Corbani, K.; Hardan, L.; Skienhe, H.; Ozcan, M.; Alharbi, N.; Salameh, Z. Effect of material thickness on the fracture resistance and failure pattern of 3D-printed composite crowns. Int. J. Comput. Dent. 2020, 23, 225–233. [Google Scholar]
- Zhao, Y.; Liu, Y.; Li, Y.; Hao, Q. Development and Application of Resistance Strain Force Sensors. Sensors 2020, 20, 5826. [Google Scholar] [CrossRef] [PubMed]
- Ilie, N.; Hilton, T.J.; Heintze, S.D.; Hickel, R.; Watts, D.C.; Silikas, N.; Stansbury, J.W.; Cadenaro, M.; Ferracane, J.L. Academy of Dental Materials guidance—Resin composites: Part I—Mechanical properties. Dent. Mater. 2017, 33, 880–894. [Google Scholar] [CrossRef] [PubMed]
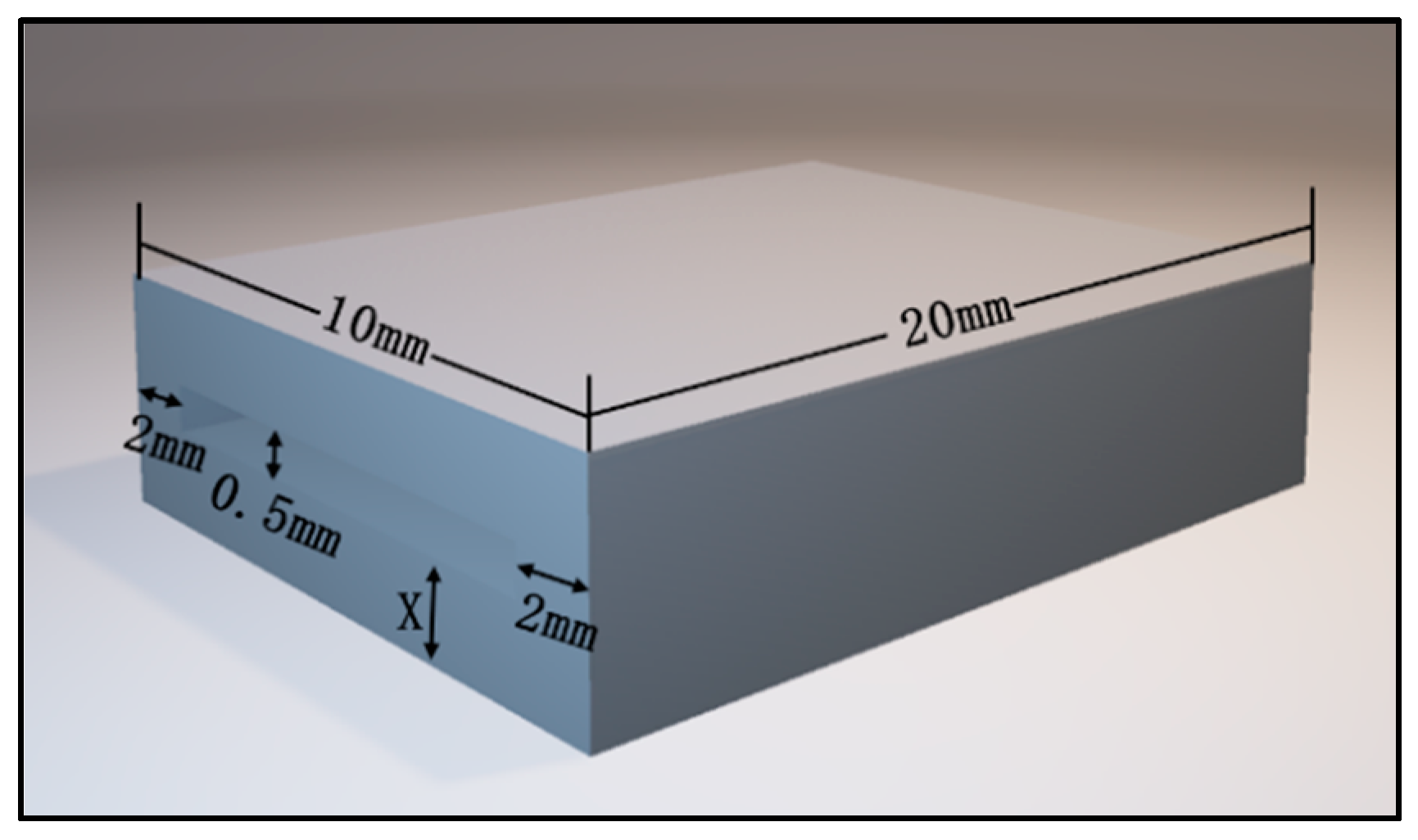







| Item | Parameter | ||
|---|---|---|---|
| Length | Width | Thickness | |
| Upper layer | 20 mm | 10 mm | 0.5 mm, 1.0 mm, 1.5 mm, 2.0 mm, 2.5 mm |
| Lower layer | 20 mm | 10 mm | 0.5 mm, 1.0 mm, 1.5 mm, 2.0 mm, 2.5 mm |
| Intermediate (piezoresistive-film sensor) | 16 mm | 6 mm | 0.5 mm (0.3 mm) |
| Device size | 20 mm | 10 mm | 1.5 mm, 2.5 mm, 3.5 mm, 4.5 mm, 5.5 mm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, J.; Liu, L.; Su, Z.; Wang, H. Sandwich Integration Technique for the Pressure Sensor Detection of Occlusal Force In Vitro. Sensors 2022, 22, 220. https://doi.org/10.3390/s22010220
Gao J, Liu L, Su Z, Wang H. Sandwich Integration Technique for the Pressure Sensor Detection of Occlusal Force In Vitro. Sensors. 2022; 22(1):220. https://doi.org/10.3390/s22010220
Chicago/Turabian StyleGao, Jinxia, Longjun Liu, Zhiwen Su, and Haitao Wang. 2022. "Sandwich Integration Technique for the Pressure Sensor Detection of Occlusal Force In Vitro" Sensors 22, no. 1: 220. https://doi.org/10.3390/s22010220
APA StyleGao, J., Liu, L., Su, Z., & Wang, H. (2022). Sandwich Integration Technique for the Pressure Sensor Detection of Occlusal Force In Vitro. Sensors, 22(1), 220. https://doi.org/10.3390/s22010220





