Early Detection of Fluid Retention in Patients with Advanced Heart Failure: A Review of a Novel Multisensory Algorithm, HeartLogicTM
Abstract
1. Introduction
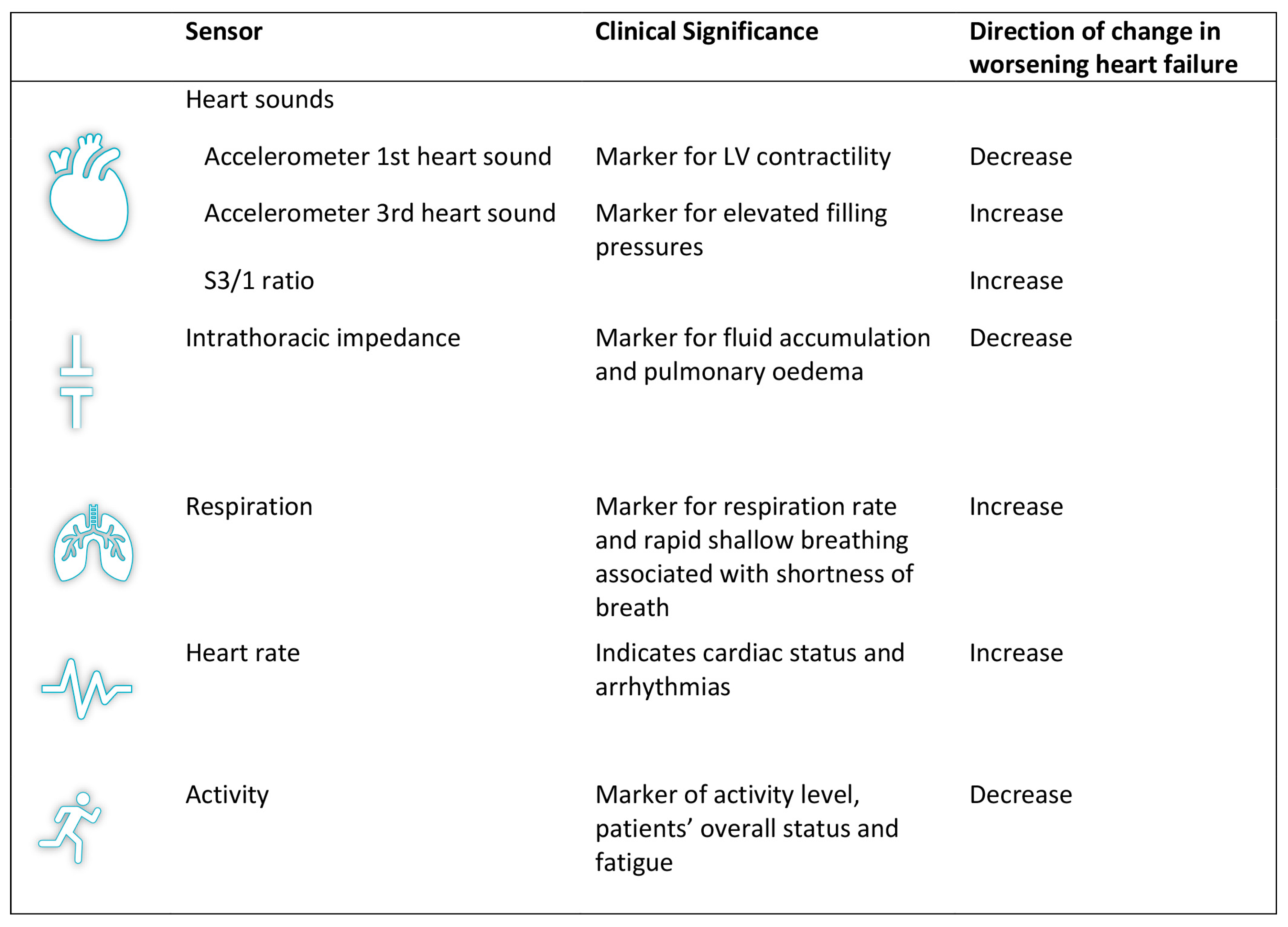
2. The HeartLogicTM Algorithm and the Sensors behind It
3. Evidence to Date
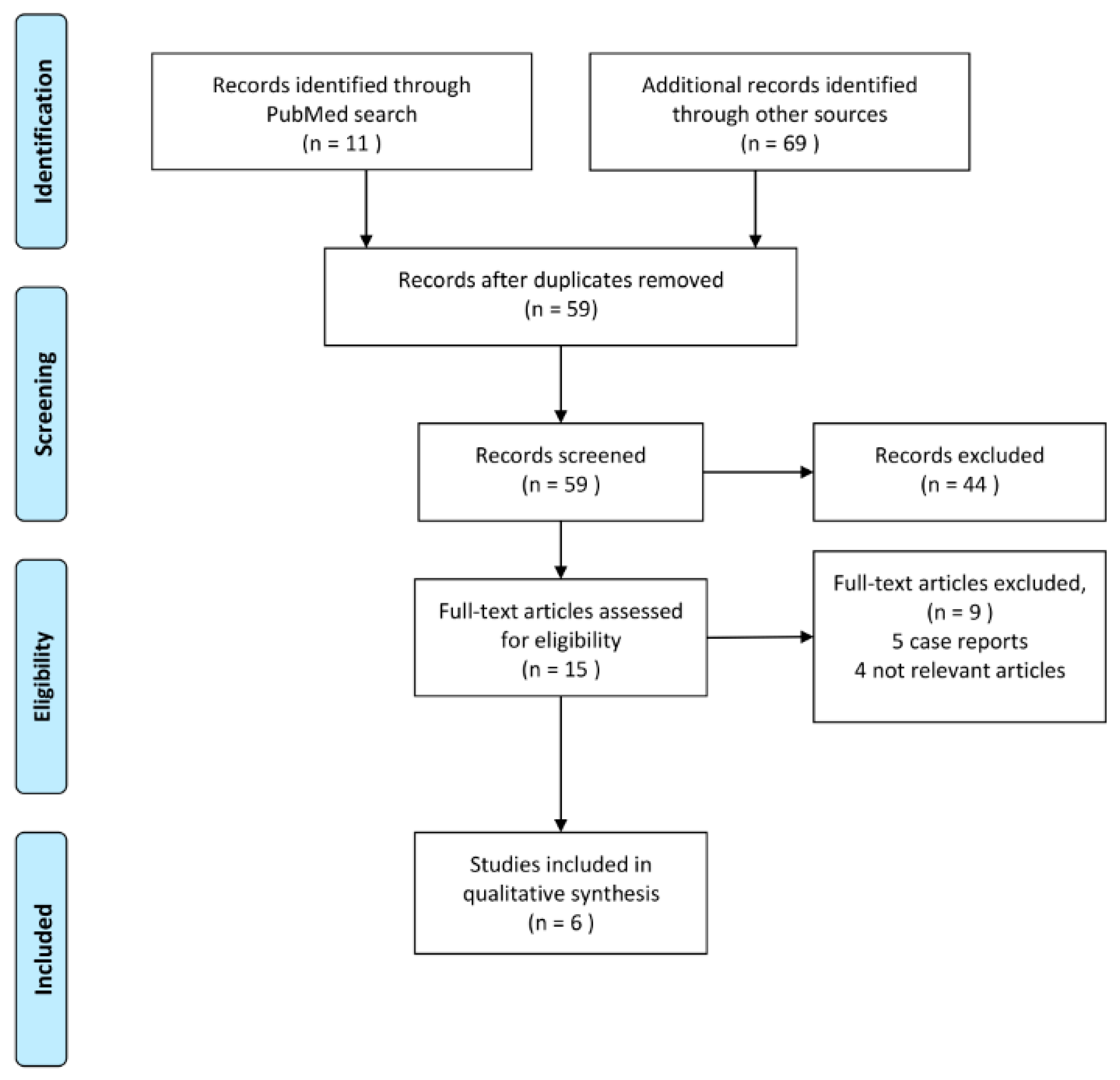
3.1. Literature Search and Selection
3.2. Studies Published to Date
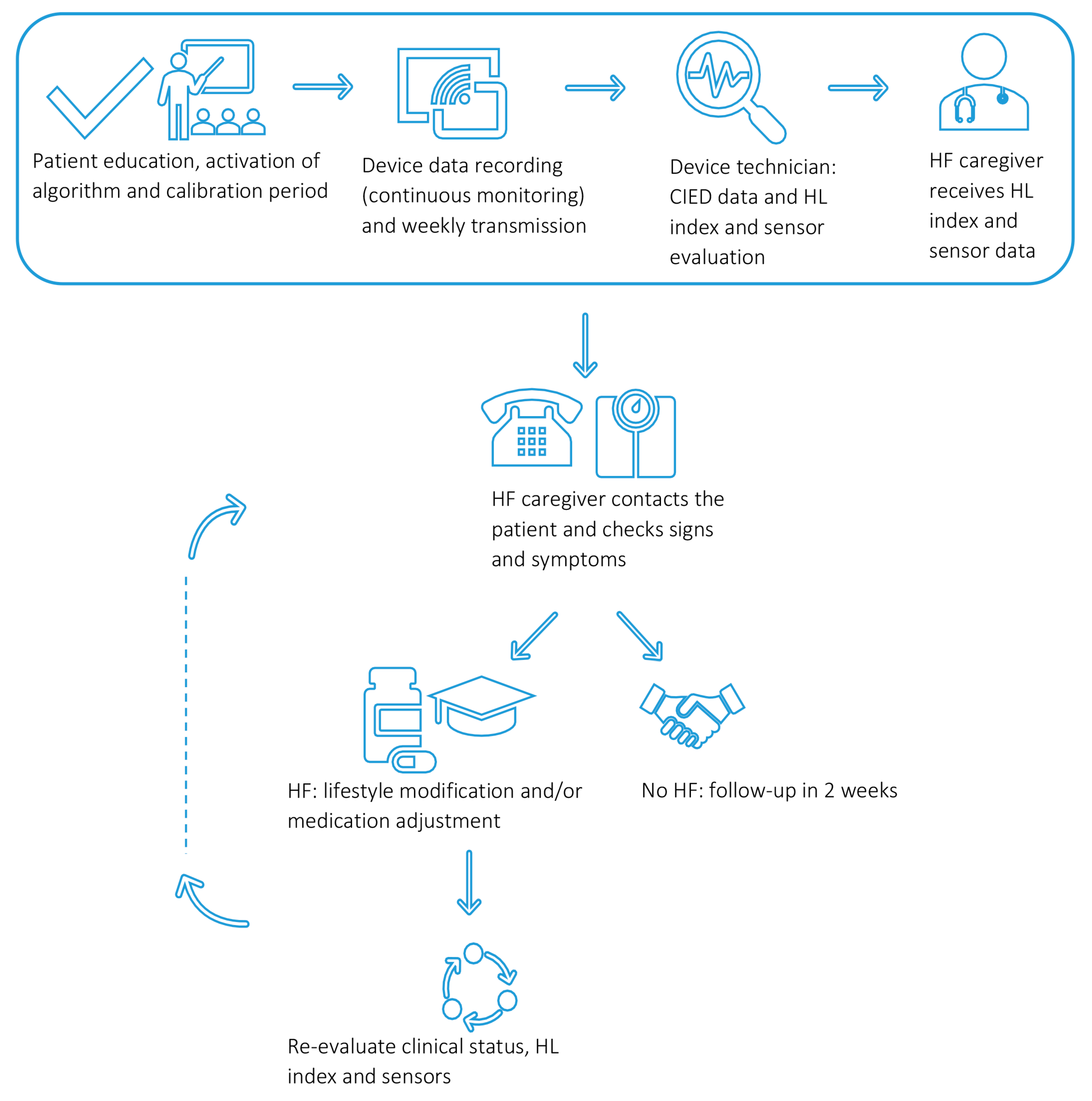
4. HeartLogic in Daily Clinical Practice: Logistics and Course of Action
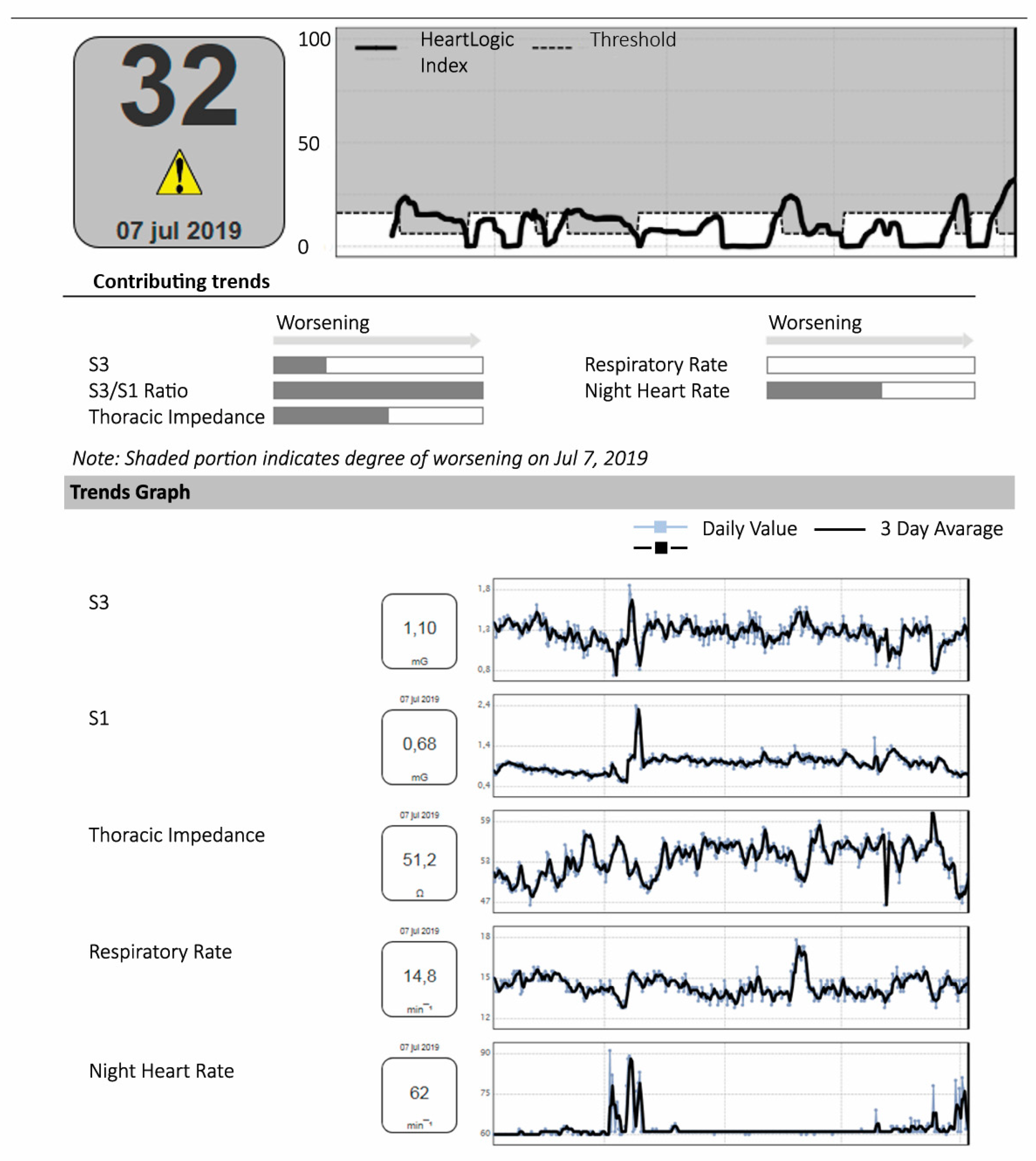
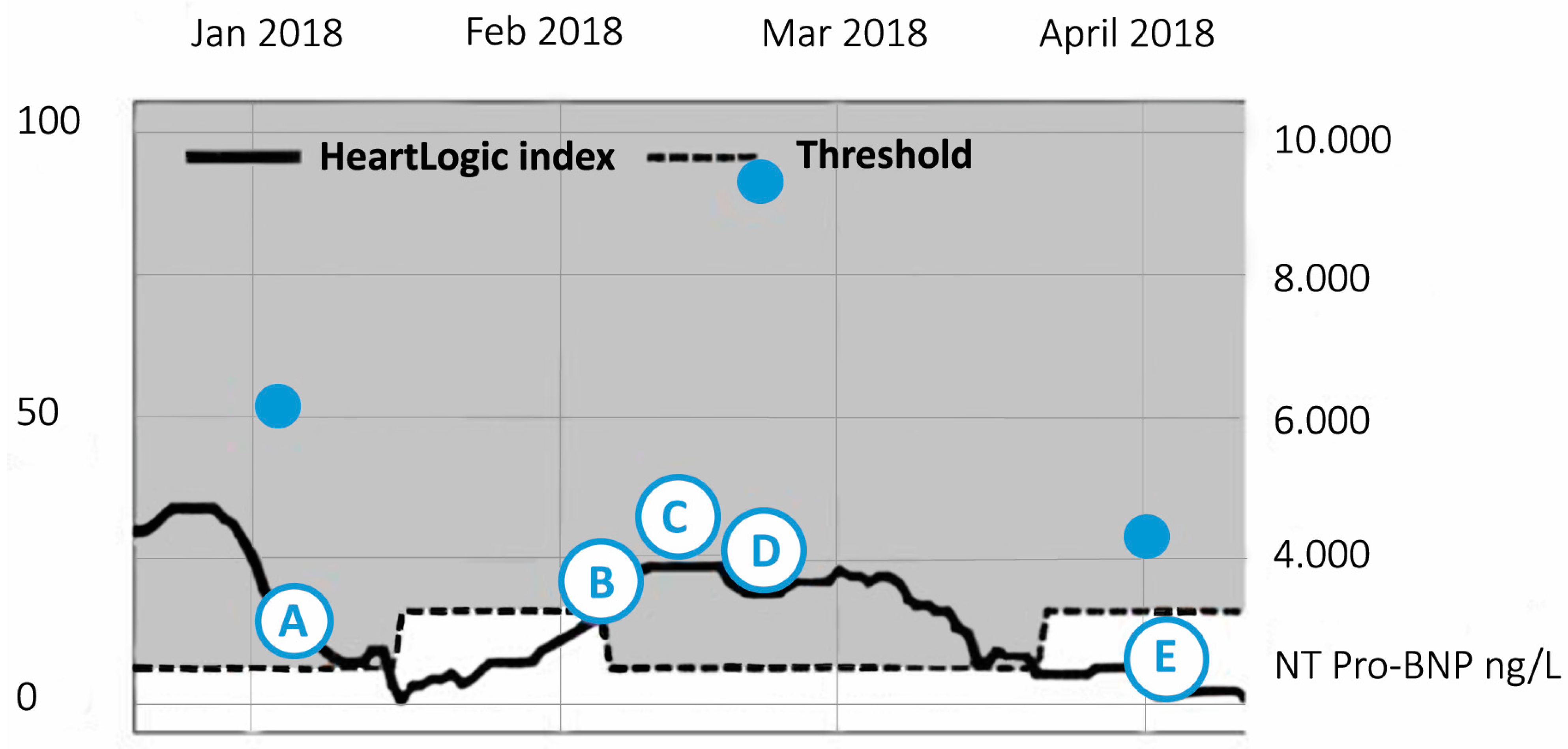
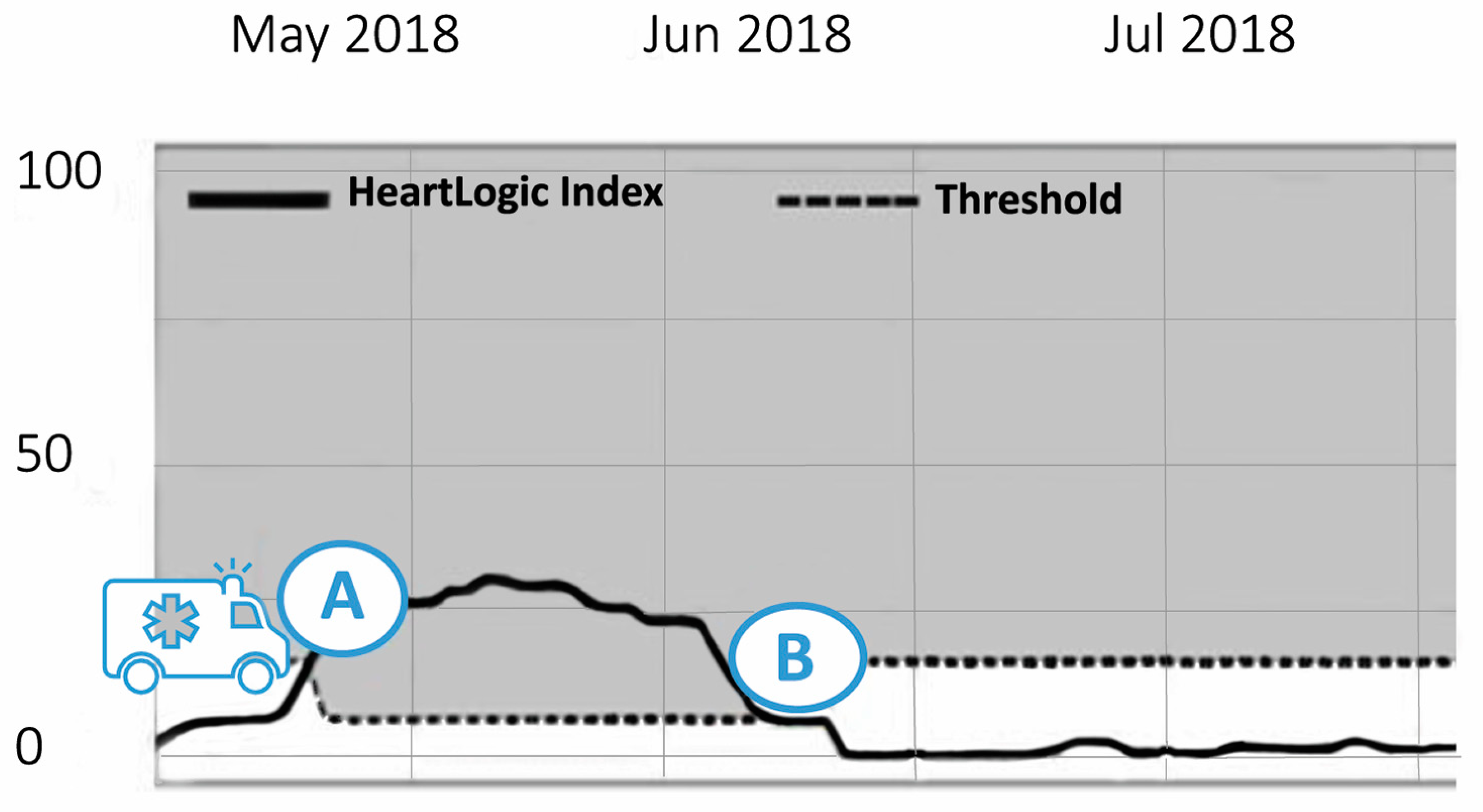
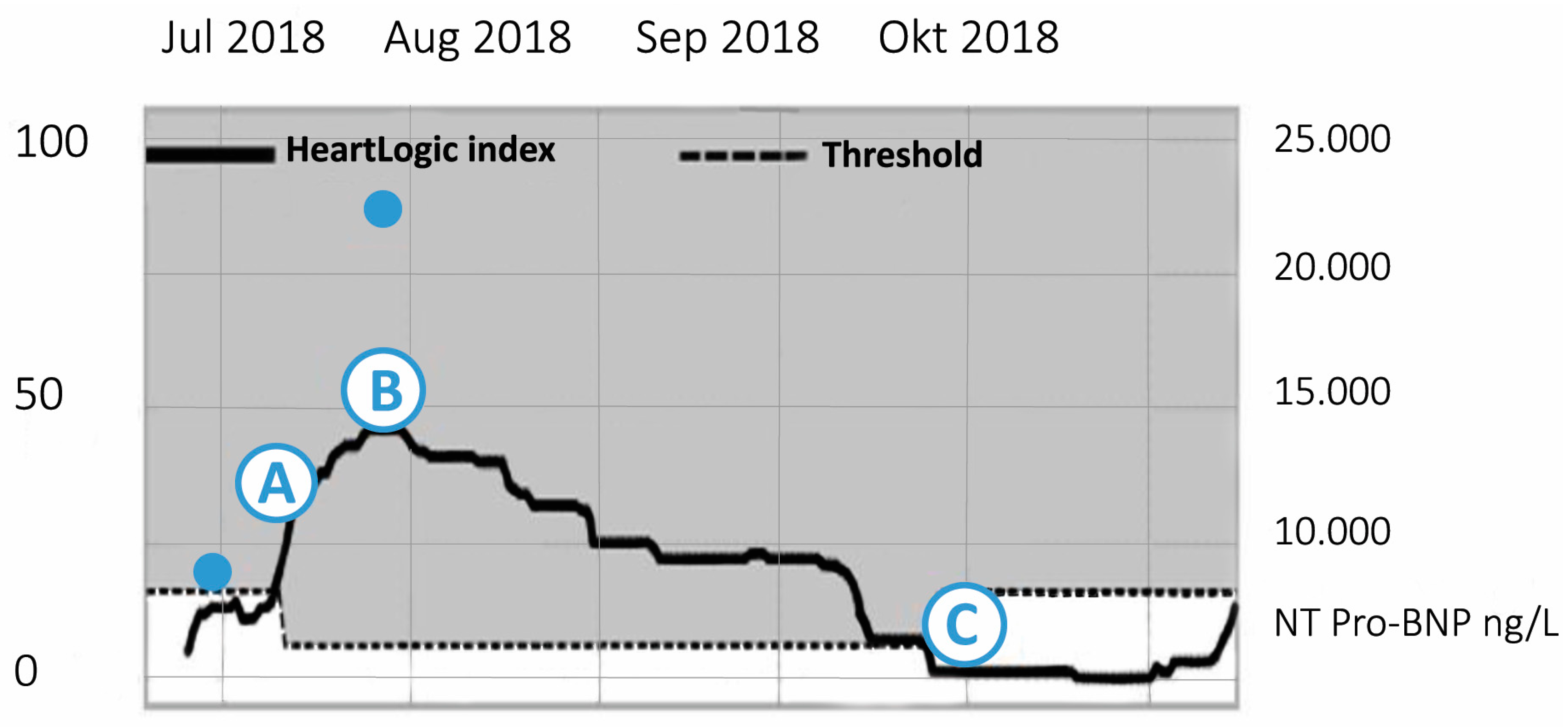
4.1. Case 1: HeartLogicTM Index as Part of a Heart Failure Care Path
4.2. Case 2: HeartLogicTM Index as an Accurate Reflection of the Fluid (and Not Arrythmia) Status
4.3. Case 3: HeartLogicTM Index in a Complex Clinical Scenario of Decompensated Cardiorenal Failure
5. Ongoing Studies
5.1. Literature Search of Ongoing Studies
5.2. The MANAGE-HF Study
5.3. The PREEMPT-HF Study
5.4. Other Ongoing Studies
6. Future Perspectives
6.1. Clinical Implementation
6.2. Patient Selection
6.3. Logistics
6.4. Limitations
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.; Coats, A.J.; Falk, V.; Gonzalez-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 2016, 18, 891–975. [Google Scholar] [CrossRef]
- Adams, K.F., Jr.; Fonarow, G.C.; Emerman, C.L.; LeJemtel, T.H.; Costanzo, M.R.; Abraham, W.T.; Berkowitz, R.L.; Galvao, M.; Horton, D.P.; Committee, A.S.A.; et al. Characteristics and outcomes of patients hospitalized for heart failure in the United States: Rationale, design, and preliminary observations from the first 100,000 cases in the Acute Decompensated Heart Failure National Registry (ADHERE). Am. Heart J. 2005, 149, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Fonarow, G.C. The Hospital Readmissions Reduction Program: Evidence for Harm. JACC Heart Fail. 2018, 6, 607–609. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.F.; Li, Y.; Pfeffer, M.A.; Solomon, S.D.; Weinfurt, K.P.; Velazquez, E.J.; Califf, R.M.; Rouleau, J.L.; Kober, L.; White, H.D.; et al. Impact of cardiovascular events on change in quality of life and utilities in patients after myocardial infarction: A VALIANT study (valsartan in acute myocardial infarction). JACC Heart Fail. 2014, 2, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Cleland, J.G.; Abraham, W.T.; Linde, C.; Gold, M.R.; Young, J.B.; Claude Daubert, J.; Sherfesee, L.; Wells, G.A.; Tang, A.S. An individual patient meta-analysis of five randomized trials assessing the effects of cardiac resynchronization therapy on morbidity and mortality in patients with symptomatic heart failure. Eur. Heart J. 2013, 34, 3547–3556. [Google Scholar] [CrossRef]
- Hjalmarson, A.; Goldstein, S.; Fagerberg, B.; Wedel, H.; Waagstein, F.; Kjekshus, J.; Wikstrand, J.; El Allaf, D.; Vitovec, J.; Aldershvile, J.; et al. Effects of controlled-release metoprolol on total mortality, hospitalizations, and well-being in patients with heart failure: The Metoprolol CR/XL Randomized Intervention Trial in congestive heart failure (MERIT-HF). MERIT-HF Study Group. JAMA 2000, 283, 1295–1302. [Google Scholar] [CrossRef]
- Investigators, S.; Yusuf, S.; Pitt, B.; Davis, C.E.; Hood, W.B., Jr.; Cohn, J.N. Effect of enalapril on mortality and the development of heart failure in asymptomatic patients with reduced left ventricular ejection fractions. N. Engl. J. Med. 1992, 327, 685–691. [Google Scholar] [CrossRef]
- McMurray, J.J.; Packer, M.; Desai, A.S.; Gong, J.; Lefkowitz, M.P.; Rizkala, A.R.; Rouleau, J.L.; Shi, V.C.; Solomon, S.D.; Swedberg, K.; et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 2014, 371, 993–1004. [Google Scholar] [CrossRef]
- Moss, A.J.; Zareba, W.; Hall, W.J.; Klein, H.; Wilber, D.J.; Cannom, D.S.; Daubert, J.P.; Higgins, S.L.; Brown, M.W.; Andrews, M.L.; et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N. Engl. J. Med. 2002, 346, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Pitt, B.; Zannad, F.; Remme, W.J.; Cody, R.; Castaigne, A.; Perez, A.; Palensky, J.; Wittes, J. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N. Engl. J. Med. 1999, 341, 709–717. [Google Scholar] [CrossRef]
- Writing Group, M.; Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; de Ferranti, S.; Despres, J.P.; et al. Executive Summary: Heart Disease and Stroke Statistics--2016 Update: A Report From the American Heart Association. Circulation 2016, 133, 447–454. [Google Scholar] [CrossRef]
- Virani, S.S.; Alonso, A.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Delling, F.N.; et al. Heart Disease and Stroke Statistics-2020 Update: A Report From the American Heart Association. Circulation 2020, 141, e139–e596. [Google Scholar] [CrossRef] [PubMed]
- Adamson, P.B. Pathophysiology of the transition from chronic compensated and acute decompensated heart failure: New insights from continuous monitoring devices. Curr Heart Fail. Rep. 2009, 6, 287–292. [Google Scholar] [CrossRef]
- Chaudhry, S.I.; Mattera, J.A.; Curtis, J.P.; Spertus, J.A.; Herrin, J.; Lin, Z.; Phillips, C.O.; Hodshon, B.V.; Cooper, L.S.; Krumholz, H.M. Telemonitoring in patients with heart failure. N. Engl. J. Med. 2010, 363, 2301–2309. [Google Scholar] [CrossRef]
- Abraham, W.T.; Stevenson, L.W.; Bourge, R.C.; Lindenfeld, J.A.; Bauman, J.G.; Adamson, P.B.; Group, C.T.S. Sustained efficacy of pulmonary artery pressure to guide adjustment of chronic heart failure therapy: Complete follow-up results from the CHAMPION randomised trial. Lancet 2016, 387, 453–461. [Google Scholar] [CrossRef]
- Angermann, C.E.; Stork, S.; Gelbrich, G.; Faller, H.; Jahns, R.; Frantz, S.; Loeffler, M.; Ertl, G.; Competence Network Heart, F. Mode of action and effects of standardized collaborative disease management on mortality and morbidity in patients with systolic heart failure: The Interdisciplinary Network for Heart Failure (INH) study. Circ. Heart Fail. 2012, 5, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Boyne, J.J.; Vrijhoef, H.J.; Crijns, H.J.; De Weerd, G.; Kragten, J.; Gorgels, A.P.; TEHAF investigators. Tailored telemonitoring in patients with heart failure: Results of a multicentre randomized controlled trial. Eur. J. Heart Fail. 2012, 14, 791–801. [Google Scholar] [CrossRef]
- Brahmbhatt, D.H.; Cowie, M.R. Remote Management of Heart Failure: An Overview of Telemonitoring Technologies. Card Fail. Rev. 2019, 5, 86–92. [Google Scholar] [CrossRef]
- Clark, R.A.; Inglis, S.C.; McAlister, F.A.; Cleland, J.G.; Stewart, S. Telemonitoring or structured telephone support programmes for patients with chronic heart failure: Systematic review and meta-analysis. BMJ 2007, 334, 942. [Google Scholar] [CrossRef]
- Clarke, M.; Shah, A.; Sharma, U. Systematic review of studies on telemonitoring of patients with congestive heart failure: A meta-analysis. J. Telemed Telecare 2011, 17, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Inglis, S.C.; Clark, R.A.; McAlister, F.A.; Ball, J.; Lewinter, C.; Cullington, D.; Stewart, S.; Cleland, J.G. Structured telephone support or telemonitoring programmes for patients with chronic heart failure. Cochrane Database Syst. Rev. 2010, CD007228. [Google Scholar] [CrossRef]
- Klersy, C.; De Silvestri, A.; Gabutti, G.; Regoli, F.; Auricchio, A. A meta-analysis of remote monitoring of heart failure patients. J. Am. Coll. Cardiol. 2009, 54, 1683–1694. [Google Scholar] [CrossRef]
- Koehler, F.; Winkler, S.; Schieber, M.; Sechtem, U.; Stangl, K.; Bohm, M.; Boll, H.; Kim, S.S.; Koehler, K.; Lucke, S.; et al. Telemedical Interventional Monitoring in Heart Failure (TIM-HF), a randomized, controlled intervention trial investigating the impact of telemedicine on mortality in ambulatory patients with heart failure: Study design. Eur. J. Heart Fail. 2010, 12, 1354–1362. [Google Scholar] [CrossRef]
- Lynga, P.; Persson, H.; Hagg-Martinell, A.; Hagglund, E.; Hagerman, I.; Langius-Eklof, A.; Rosenqvist, M. Weight monitoring in patients with severe heart failure (WISH). A randomized controlled trial. Eur. J. Heart Fail. 2012, 14, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, K.; Bennett, D.; Conrad, N.; Williams, T.M.; Basu, J.; Dwight, J.; Woodward, M.; Patel, A.; McMurray, J.; MacMahon, S. Risk prediction in patients with heart failure: A systematic review and analysis. JACC Heart Fail. 2014, 2, 440–446. [Google Scholar] [CrossRef]
- Ritzema, J.; Troughton, R.; Melton, I.; Crozier, I.; Doughty, R.; Krum, H.; Walton, A.; Adamson, P.; Kar, S.; Shah, P.K.; et al. Physician-directed patient self-management of left atrial pressure in advanced chronic heart failure. Circulation 2010, 121, 1086–1095. [Google Scholar] [CrossRef]
- Brugts, J.J.; Manintveld, O.C.; van Mieghem, N. Remote monitoring of pulmonary artery pressures with CardioMEMS in patients with chronic heart failure and NYHA class III: First experiences in the Netherlands. Neth. Heart J. 2018, 26, 55–57. [Google Scholar] [CrossRef]
- Conraads, V.M.; Tavazzi, L.; Santini, M.; Oliva, F.; Gerritse, B.; Yu, C.M.; Cowie, M.R. Sensitivity and positive predictive value of implantable intrathoracic impedance monitoring as a predictor of heart failure hospitalizations: The SENSE-HF trial. Eur. Heart J. 2011, 32, 2266–2273. [Google Scholar] [CrossRef]
- Palfy, J.A.; Benezet-Mazuecos, J.; Martinez Milla, J.; Iglesias, J.A.; de la Vieja, J.J.; Sanchez-Borque, P.; Miracle, A.; Rubio, J.M. CorVue algorithm efficacy to predict heart failure in real life: Unnecessary and potentially misleading information? Pacing Clin. Electrophysiol. 2018. [Google Scholar] [CrossRef]
- van Veldhuisen, D.J.; Braunschweig, F.; Conraads, V.; Ford, I.; Cowie, M.R.; Jondeau, G.; Kautzner, J.; Aguilera, R.M.; Lunati, M.; Yu, C.M.; et al. Intrathoracic impedance monitoring, audible patient alerts, and outcome in patients with heart failure. Circulation 2011, 124, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.M.; Wang, L.; Chau, E.; Chan, R.H.; Kong, S.L.; Tang, M.O.; Christensen, J.; Stadler, R.W.; Lau, C.P. Intrathoracic impedance monitoring in patients with heart failure: Correlation with fluid status and feasibility of early warning preceding hospitalization. Circulation 2005, 112, 841–848. [Google Scholar] [CrossRef]
- Boehmer, J.P.; Hariharan, R.; Devecchi, F.G.; Smith, A.L.; Molon, G.; Capucci, A.; An, Q.; Averina, V.; Stolen, C.M.; Thakur, P.H.; et al. A Multisensor Algorithm Predicts Heart Failure Events in Patients With Implanted Devices: Results From the MultiSENSE Study. JACC Heart Fail. 2017, 5, 216–225. [Google Scholar] [CrossRef]
- Capucci, A.; Santini, L.; Favale, S.; Pecora, D.; Petracci, B.; Calo, L.; Molon, G.; Cipolletta, L.; Bianchi, V.; Schirripa, V.; et al. Preliminary experience with the multisensor HeartLogic algorithm for heart failure monitoring: A retrospective case series report. ESC Heart Fail. 2019, 6, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Wang, L. Fundamentals of intrathoracic impedance monitoring in heart failure. Am. J. Cardiol. 2007, 99, 3G–10G. [Google Scholar] [CrossRef]
- Forleo, G.B.; Santini, L.; Campoli, M.; Malavasi, M.; Scaccia, A.; Menichelli, M.; Riva, U.; Lamberti, F.; Carreras, G.; Orazi, S.; et al. Long-term monitoring of respiratory rate in patients with heart failure: The Multiparametric Heart Failure Evaluation in Implantable Cardioverter-Defibrillator Patients (MULTITUDE-HF) study. J. Interv. Card Electrophysiol. 2015, 43, 135–144. [Google Scholar] [CrossRef]
- Abraham, W.T.; Adamson, P.B.; Bourge, R.C.; Aaron, M.F.; Costanzo, M.R.; Stevenson, L.W.; Strickland, W.; Neelagaru, S.; Raval, N.; Krueger, S.; et al. Wireless pulmonary artery haemodynamic monitoring in chronic heart failure: A randomised controlled trial. Lancet 2011, 377, 658–666. [Google Scholar] [CrossRef]
- McMurray, J.J.V.; DeMets, D.L.; Inzucchi, S.E.; Kober, L.; Kosiborod, M.N.; Langkilde, A.M.; Martinez, F.A.; Bengtsson, O.; Ponikowski, P.; Sabatine, M.S.; et al. A trial to evaluate the effect of the sodium-glucose co-transporter 2 inhibitor dapagliflozin on morbidity and mortality in patients with heart failure and reduced left ventricular ejection fraction (DAPA-HF). Eur. J. Heart Fail. 2019, 21, 665–675. [Google Scholar] [CrossRef]
- Gardner, R.S.; Singh, J.P.; Stancak, B.; Nair, D.G.; Cao, M.; Schulze, C.; Thakur, P.H.; An, Q.; Wehrenberg, S.; Hammill, E.F.; et al. HeartLogic Multisensor Algorithm Identifies Patients During Periods of Significantly Increased Risk of Heart Failure Events: Results From the MultiSENSE Study. Circ. Heart Fail. 2018, 11, e004669. [Google Scholar] [CrossRef]
- Calo, L.; Capucci, A.; Santini, L.; Pecora, D.; Favale, S.; Petracci, B.; Molon, G.; Bianchi, V.; Cipolletta, L.; De Ruvo, E.; et al. ICD-measured heart sounds and their correlation with echocardiographic indexes of systolic and diastolic function. J. Interv. Card Electrophysiol. 2020, 58, 95–101. [Google Scholar] [CrossRef]
- Santini, L.; D’Onofrio, A.; Dello Russo, A.; Calo, L.; Pecora, D.; Favale, S.; Petracci, B.; Molon, G.; Bianchi, V.; De Ruvo, E.; et al. Prospective evaluation of the multisensor HeartLogic algorithm for heart failure monitoring. Clin. Cardiol. 2020, 43, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Mitter, S.S.; Alvarez-Garcia, J.; Miller, M.A.; Moss, N.; Lala, A. Insights From HeartLogic Multisensor Monitoring During the COVID-19 Pandemic in New York City. JACC Heart Fail. 2020, 8, 1053–1055. [Google Scholar] [CrossRef] [PubMed]
- Multiple Cardiac Sensors for the Management of Heart Failure (MANAGE-HF); NCT03237858; U.S. National Library of Medicine: Bethesda, MD, USA, 2017.
- Precision Event Monitoring for Patients With Heart Failure Using HeartLogic (PREEMPT-HF); NCT03579641; U.S. National Library of Medicine: Bethesda, MD, USA, 2018.
- Jukema, J.W.; Timal, R.J.; Rotmans, J.I.; Hensen, L.C.R.; Buiten, M.S.; de Bie, M.K.; Putter, H.; Zwinderman, A.H.; van Erven, L.; Krol-van Straaten, M.J.; et al. Prophylactic Use of Implantable Cardioverter-Defibrillators in the Prevention of Sudden Cardiac Death in Dialysis Patients. Circulation 2019, 139, 2628–2638. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, R.S.; Tu, J.V.; Lee, D.S.; Austin, P.C.; Fang, J.; Haouzi, A.; Gong, Y.; Liu, P.P. Outcome of heart failure with preserved ejection fraction in a population-based study. N. Engl. J. Med. 2006, 355, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Owan, T.E.; Hodge, D.O.; Herges, R.M.; Jacobsen, S.J.; Roger, V.L.; Redfield, M.M. Trends in prevalence and outcome of heart failure with preserved ejection fraction. N. Engl. J. Med. 2006, 355, 251–259. [Google Scholar] [CrossRef]
- Tkachenko, R.; Izonin, I.; Kryvinska, N.; Dronyuk, I.; Zub, K. An Approach towards Increasing Prediction Accuracy for the Recovery of Missing IoT Data Based on the GRNN-SGTM Ensemble. Sensors 2020, 20, 2625. [Google Scholar] [CrossRef]
| Year | Study Design | Nr Pts | Mean Age in Years | Gender (Male in %) | Aetiology HF (ICM in %) | NYHA Class (I/II/III/IV in %) | LVEF (in %) | FU Time in Months | Primary Endpoint | Secondary Endpoint | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehmer et al. (Multi SENSE study) | 2017 | Multicentre non-randomised trial | 974 | 67 | 71 | 52 | 4/64/25/1 | 30 | 12 | Predict sensitivity of 70% for heart failure event Alert 34 days prior to a HF event | Unexplained alert rate of 1.47 per patient year |
| Gardner et al. | 2018 | MultiSENSE study post hoc analysis | 900 | 67 | 73 | 51 | 5/67/27/<1 | 30 | 12.9 | IN alert state associated with 0.80 events per patient year vs. OUT of alert with an event rate of 0.08 events per patient year Event ratio IN/OUT 10.6. | 50-fold risk of HF event when high NT-proBNP and IN alert vs. low NT-pro BNP and OUT alert state |
| Capucci et al. | 2019 | Retrospective case series | 67 | 71 | 81 | 37 | 4/50/44/2 | 30 | 5 ± 3 |
| Unexplained alert rate of 0.41 per patient year |
| Calò et al. | 2020 | Multicentre prospective registry | 104 | 71 | 73 | 40 | 2/44/51/3 | 29 | - | S3 detects a restrictive filling pattern with 85% sensitivity and 82% specificity S1 detects LVEF < 35% with a 28% sensitivity and an 88% specificity | More impairment of systolic and diastolic function was associated with more frequent signs of functional limitation and congestion |
| Santini et al. | 2020 | Multicentre prospective registry | 104 | 71 | 73 | 40 | 2/44/51/3 | 29 | 13 | 60% (60/100) of alerts were clinically meaningful 80% (48/60) of the clinical meaningful alerts were newly signalled by the algorithm 90% (43/48) of alerts triggered clinical action | Alert-based management strategy more efficient than a scheduled monthly remote (phone call) follow-up scheme |
| Mitter et al. | 2020 | Retrospective case series | 38 | 60 | 76 | - | 18/63/18/0 | 32 | 3 | A significant drop in activity may have resulted in less congestion |
| Start of Study | (Expected) Completion of Study | Study Design | Estimated Enrolment, (n) | Primary Endpoint | Secondary Endpoint | |
|---|---|---|---|---|---|---|
| MANAGE-HF | 2017 | 2025 | Randomised open label trial | 2700 | All-cause mortality | Hospitalisation for heart failure |
| PREEMPT-HF | 2018 | 2026 | Prospective observational trial | 2184 (current, recruitment terminated in June 2020) | Association of HeartLogicTM sensors with 30-day heart failure re-admission | |
| Treskes et al. | 2018 | 2020 (completed) | Multicentre non-blinded single-arm cohort | 74 | Total number of hospitalisations for decompensated heart failure, comparison pre- and post-activation | Number of patients hospitalised for heart failure Number of heart failure admissions per patient Duration of hospital admission |
| HeartLogicTM France study | 2020 | 2023 | Prospective cohort study | 300 | Hospitalisation for heart failure | Annual cardiovascular mortality rate Annual mortality rate due to heart failure Annual rate of unplanned hospitalisations due to ventricular arrhythmia Annual rate of unplanned hospitalisations due to atrial arrhythmia Annual rate of hospitalisation due to heart failure, ventricular or atrial arrhythmia Patient quality of life using the Kansas City Cardiomyopathy Questionnaire Weekly evolution of the HeartLogicTM index during a 12-month period |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feijen, M.; Egorova, A.D.; Beeres, S.L.M.A.; Treskes, R.W. Early Detection of Fluid Retention in Patients with Advanced Heart Failure: A Review of a Novel Multisensory Algorithm, HeartLogicTM. Sensors 2021, 21, 1361. https://doi.org/10.3390/s21041361
Feijen M, Egorova AD, Beeres SLMA, Treskes RW. Early Detection of Fluid Retention in Patients with Advanced Heart Failure: A Review of a Novel Multisensory Algorithm, HeartLogicTM. Sensors. 2021; 21(4):1361. https://doi.org/10.3390/s21041361
Chicago/Turabian StyleFeijen, Michelle, Anastasia D. Egorova, Saskia L. M. A. Beeres, and Roderick W. Treskes. 2021. "Early Detection of Fluid Retention in Patients with Advanced Heart Failure: A Review of a Novel Multisensory Algorithm, HeartLogicTM" Sensors 21, no. 4: 1361. https://doi.org/10.3390/s21041361
APA StyleFeijen, M., Egorova, A. D., Beeres, S. L. M. A., & Treskes, R. W. (2021). Early Detection of Fluid Retention in Patients with Advanced Heart Failure: A Review of a Novel Multisensory Algorithm, HeartLogicTM. Sensors, 21(4), 1361. https://doi.org/10.3390/s21041361






