Non-Invasive Method to Detect Infection with Mycobacterium tuberculosis Complex in Wild Boar by Measurement of Volatile Organic Compounds Obtained from Feces with an Electronic Nose System
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Samples
2.2. Electronic Nose and Sensing Measurements
- (i)
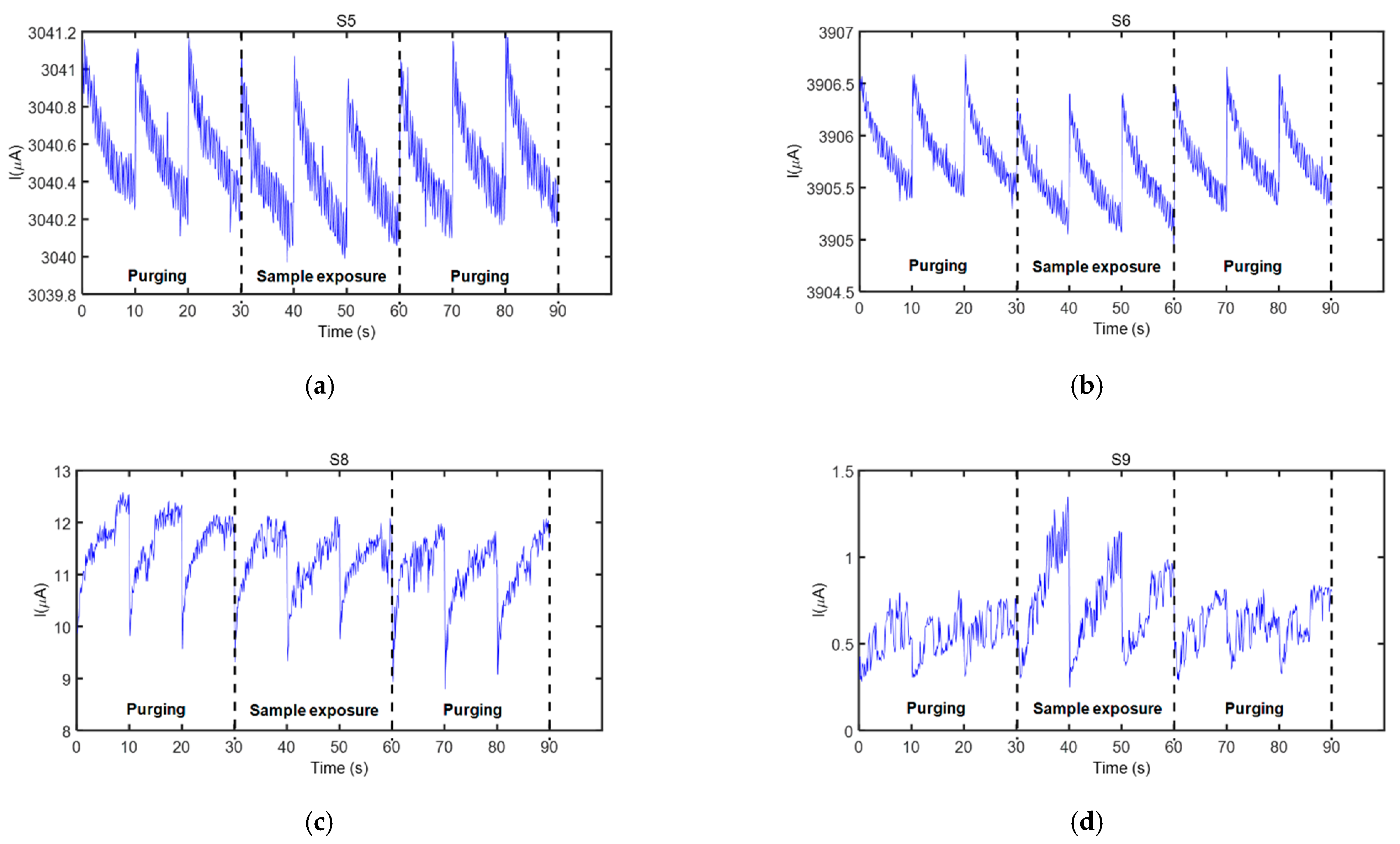
- 5 min of continuous N2 flow (delivered from a commercial N2 gas bottle, Cryogas S.A., Colombia—Figure 2a(F)) passed at 5 L/min flow rate through the sensor test chamber for purging purposes before the sample measurement,
- (ii)
- 5 min of exposure to the fecal VOCs carried by continuous N2 flow that passed at 100 mL/min flow rate at first through the thermal desorption unit for taking the thermally released VOCs (see Figure 2c) and then through the sensor test chamber together with the fecal VOCs,
- (iii)
- 5 min of continuous N2 flow passed at 5 L/min flow rate through the sensor test chamber for purging purposes after the sample measurement.
2.3. Data Analysis
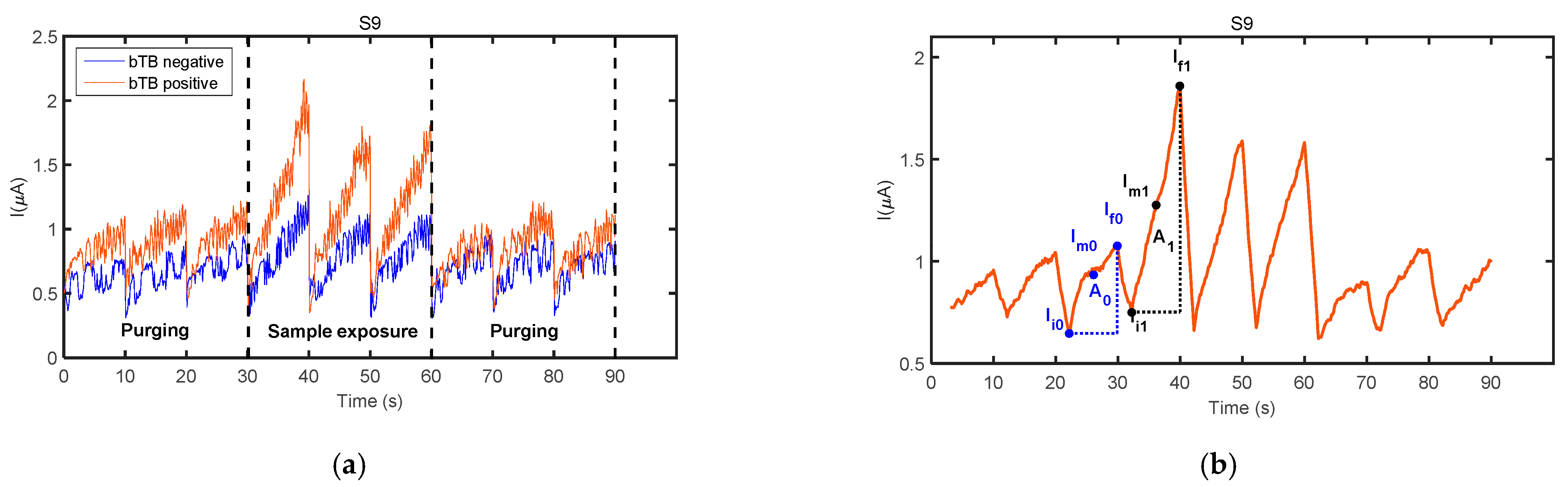
- F1: A1/A0, where A1 is the area under the curve calculated from the first 70 data points of the filtered signal for the first operation period of the sensor exposure to the fecal VOC sample, and A0 is the area under the curve calculated from the first 70 data points of the filtered signal obtained in the last operation period of the same sensor during the purging process immediately prior to sample exposure,
- F2: (Im1 − Im0)/Im0, where Im1 is the averaged current calculated with the first 70 current values of the filtered signal for the first operation period of the sensor exposure to the fecal VOC sample, and Im0 is the average current calculated with the first 70 current values of the filtered signal obtained in the last operation period of the same sensor during the purging process immediately prior to sample exposure,
- F3: (∆I1 − ∆I0)/∆I0, where ∆I1 = If1 − Ii1 and ∆I0 = If0 − Ii0, and where If1 and Ii1 are the 70th and 1st current values, respectively, of the filtered signal for the first operation period of the sensor exposure to the fecal VOC sample, and If0 and Ii0 are the 70th and 1st current values, respectively, of the filtered signal obtained in the last operation period of the same sensor during the purging process immediately prior to sample exposure.
3. Results
3.1. Sensor Responses
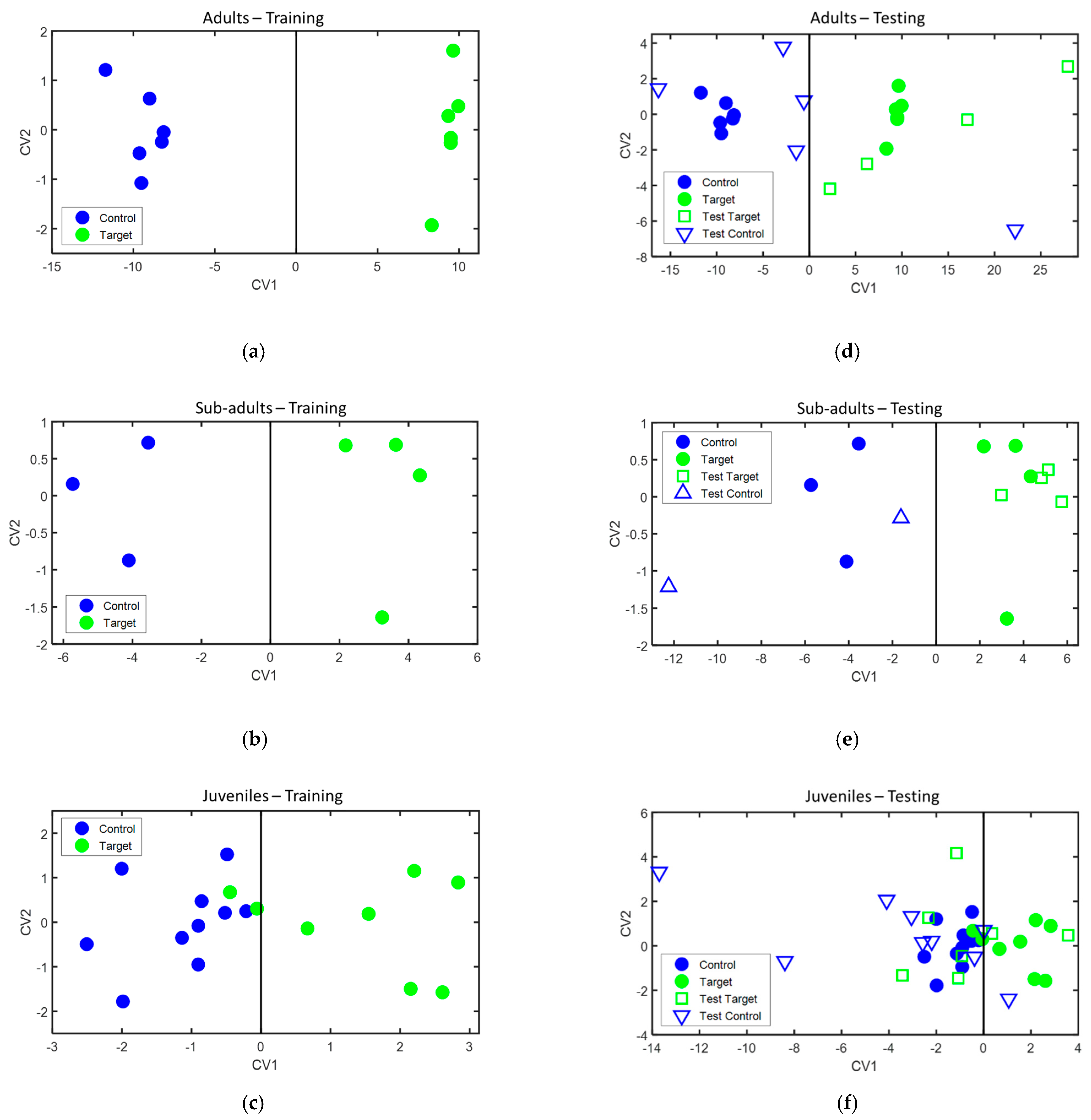
3.2. Classification Results
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Di Marco, V.; Mazzone, P.; Capucchio, M.T.; Boniotti, M.B.; Aronica, V.; Russo, M.; Fiasconaro, M.; Cifani, N.; Corneli, S.; Biasibetti, E.; et al. Epidemiological significance of the domestic black pig (Sus scrofa) in maintenance of bovine tuberculosis in Sicily. J. Clin. Microbiol. 2012, 50, 1209–1218. [Google Scholar] [CrossRef]
- Bapat, P.R.; Dodkey, R.S.; Shekhawat, S.D.; Husain, A.A.; Nayak, A.R.; Kawle, A.P.; Daginawala, H.F.; Singh, L.K.; Kashyap, R.S. Prevalence of zoonotic tuberculosis and associated risk factors in Central Indian populations. J. Epidemiol. Glob. Health 2017, 7, 277–283. [Google Scholar] [CrossRef]
- Müller, B.; Dürr, S.; Alonso, S.; Hattendorf, J.; Laisse, C.J.M.; Parsons, S.D.C.; Van Helden, P.D.; Zinsstag, J. Induced tuberculosis in humans. Emergy Infect. Dis. 2013, 19, 899–908. [Google Scholar] [CrossRef]
- Palmer, M.V.; Waters, W.R. Advances in bovine tuberculosis diagnosis and pathogenesis: What policy makers need to know. Vet. Microbiol. 2006, 112, 181–190. [Google Scholar] [CrossRef][Green Version]
- Richeldi, L. Rapid identification of Mycobacterium tuberculosis infection. Clin. Microbiol. Infect. 2006, 12, 34–36. [Google Scholar] [CrossRef][Green Version]
- Lerche, N.W.; Yee, J.A.L.; Capuano, S.V.; Flynn, J.L. New approaches to tuberculosis surveillance in nonhuman primates. ILAR J. 2008, 49, 170–178. [Google Scholar] [CrossRef]
- Dugal, C.J.; Van Beest, F.M.; Vander Wal, E.; Brook, R.K. Targeting hunter distribution based on host resource selection and kill sites to manage disease risk. Ecol. Evol. 2013, 3, 4265–4277. [Google Scholar] [CrossRef]
- Sandoval Barron, E.; Swift, B.; Chantrey, J.; Christley, R.; Gardner, R.; Jewell, C.; McGrath, I.; Mitchell, A.; O’Cathail, C.; Prosser, A.; et al. A study of tuberculosis in road traffic-killed badgers on the edge of the British bovine TB epidemic areafile. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef]
- Wobeser, G. Bovine tuberculosis in Canadian wildlife: An updated history. Can. Vet. J. La Rev. Vet. Can. 2009, 50, 1169–1176. [Google Scholar]
- Rodríguez-Hernández, P.; Cardador, M.J.; Arce, L.; Rodríguez-Estévez, V. Analytical tools for disease diagnosis in animals via fecal volatilome. Crit. Rev. Anal. Chem. 2020. [Google Scholar] [CrossRef]
- Kataoka, H.; Saito, K.; Kato, H.; Masuda, K. Noninvasive analysis of volatile biomarkers in human emanations for health and early disease diagnosis. Bioanalysis 2013, 5, 1443–1459. [Google Scholar] [CrossRef]
- Wilson, A.D. Applications of electronic-nose technologies for noninvasive early detection of plant, animal and human diseases. Chemosensors 2018, 6, 45. [Google Scholar] [CrossRef]
- Ellis, C.K.; Rice, S.; Maurer, D.; Stahl, R.; Waters, W.R.; Palmer, M.V.; Nol, P.; Rhyan, J.C.; VerCauteren, K.C.; Koziel, J.A. Use of fecal volatile organic compound analysis to discriminate between non-vaccinated and BCG—Vaccinated cattle prior to and after Mycobacterium bovis challenge. PLoS ONE 2017, 12, 1–25. [Google Scholar] [CrossRef]
- Stahl, R.S.; Ellis, C.K.; Nol, P.; Waters, W.R.; Palmer, M.; VerCauteren, K.C. Fecal volatile organic ccompound profiles from white-tailed deer (Odocoileus virginianus) as indicators of Mycobacterium bovis exposure or Mycobacterium bovis Bacille Calmette-Guerin (BCG) vaccination. PLoS ONE 2015, 10, 1–20. [Google Scholar] [CrossRef]
- Purkhart, R.; Köhler, H.; Liebler-Tenorio, E.; Meyer, M.; Becher, G.; Kikowatz, A.; Reinhold, P. Chronic intestinal Mycobacteria infection: Discrimination via VOC analysis in exhaled breath and headspace of feces using differential ion mobility spectrometry. J. Breath Res. 2011, 5, 027103. [Google Scholar] [CrossRef]
- Bergmann, A.; Trefz, P.; Fischer, S.; Klepik, K.; Walter, G.; Steffens, M.; Ziller, M.; Schubert, J.K.; Reinhold, P.; Köhler, H.; et al. In vivo volatile organic compound signatures of Mycobacterium avium subsp. paratuberculosis. PLoS ONE 2015, 10, 1–20. [Google Scholar] [CrossRef]
- Nol, P.; Ionescu, R.; Welearegay, T.G.; Barasona, J.A.; Vicente, J.; Beleño-Sáenz, K.J.; Barrenetxea, I.; Torres, M.J.; Ionescu, F.; Rhyan, J. Evaluation of volatile organic compounds obtained from breath and feces to detect mycobacterium tuberculosis complex in wild boar (Sus scrofa) in Doñana National Park, Spain. Pathogens 2020, 9, 346. [Google Scholar] [CrossRef]
- Ellis, C.K.; Volker, S.F.; Griffin, D.L.; VerCauteren, K.C.; Nichols, T.A. Use of faecal volatile organic compound analysis for ante-mortem discrimination between CWD-positive, -negative exposed, and -known negative white-tailed deer (Odocoileus virginianus). Prion 2019, 13, 94–105. [Google Scholar] [CrossRef]
- Cho, Y.S.; Jung, S.C.; Oh, S. Diagnosis of bovine tuberculosis using a metal oxide-based electronic nose. Lett. Appl. Microbiol. 2015, 60, 513–516. [Google Scholar] [CrossRef]
- Peled, N.; Ionescu, R.; Nol, P.; Barash, O.; McCollum, M.; Vercauteren, K.; Koslow, M.; Stahl, R.; Rhyan, J.; Haick, H. Detection of volatile organic compounds in cattle naturally infected with Mycobacterium bovis. Sens. Actuators B Chem. 2012, 171–172, 588–594. [Google Scholar] [CrossRef]
- Barroso, P.; Barasona, J.A.; Acevedo, P.; Palencia, P.; Carro, F.; Negro, J.J.; Torres, M.J.; Gortázar, C.; Soriguer, R.C.; Vicente, J. Long-term determinants of tuberculosis in the ungulate host community of Doñana National Park. Pathogens 2020, 9, 445. [Google Scholar] [CrossRef] [PubMed]
- Barasona, J.A.; López-Olvera, J.R.; Beltrán-Beck, B.; Gortázar, C.; Vicente, J. Trap-effectiveness and response to tiletamine-zolazepam and medetomidine anaesthesia in Eurasian wild boar captured with cage and corral traps. BMC Vet. Res. 2013, 9, 107. [Google Scholar] [CrossRef]
- Sáenz de Buruaga, M.; Lucio, A.; Purroy, J. Reconocimiento de Sexo y Edad en Especies Cinegéticas; Edilesa: León, Spain, 1991. [Google Scholar]
- Safianowska, A.; Walkiewicz, R.; Nejman-Gryz, P.; Chazan, R.; Grubek-Jaworska, H. Diagnostic utility of the molecular assay GenoType MTBC (HAIN Lifescience, Germany) for identification of tuberculous mycobacteria. Pneumonol. Alergol. Pol. 2009, 77, 517–520. [Google Scholar] [PubMed]
- Welearegay, T.G.; Cindemir, U.; Österlund, L.; Ionescu, R. Fabrication and characterisation of ligand-functionalised ultrapure monodispersed metal nanoparticle nanoassemblies employing advanced gas deposition technique. Nanotechnology 2018, 29, 065603. [Google Scholar] [CrossRef]
- Ionescu, R.; Llobet, E.; Vilanova, X.; Brezmes, J.; Sueiras, J.E.; Calderer, J.; Correig, X. Quantitative analysis of NO2 in the presence of CO using a single tungsten oxide semiconductor sensor and dynamic signal processing. Analyst 2002, 127, 1237–1246. [Google Scholar] [CrossRef]
- Zollanvari, A.; Braga-Neto, U.M.; Dougherty, E.R. Applications of electronic-nose technologies for noninvasive early detection of plant, animal and human diseases. Pattern Recognit. 2009, 42, 2705–2723. [Google Scholar] [CrossRef]
- Tisch, U.; Haick, H. Nanomaterials for cross-reactive sensor arrays. MRS Bull. 2010, 35, 797–803. [Google Scholar] [CrossRef]
- Peng, G.; Tisch, U.; Adams, O.; Hakim, M.; Shehada, N.; Broza, Y.Y.; Billan, S.; Abdah-Bortnyak, R.; Kuten, A.; Haick, H. Diagnosing lung cancer in exhaled breath using gold nanoparticles. Nat. Nanotechnol. 2009, 4, 669–673. [Google Scholar] [CrossRef]
- Durán-Acevedo, C.M.; Jaimes-Mogollón, A.L.; Gualdrón-Guerrero, O.E.; Welearegay, T.G.; Martinez-Marín, J.D.; Caceres-Tarazona, J.M.; Sánchez- Acevedo, Z.C.; Beleño-Saenz, K.J.; Cindemir, U.; österlund, L.; et al. Exhaled breath analysis for gastric cancer diagnosis in Colombian patients. Oncotarget 2018, 9, 28805–28817. [Google Scholar] [CrossRef]
- Ionescu, R.; Cindemir, U.; Welearegay, T.G.; Calavia, R.; Haddi, Z.; Topalian, Z.; Granqvist, C.-G.; Llobet, E. Fabrication of ultra-pure gold nanoparticles capped with dodecanethiol for Schottky-diode chemical gas sensing devices. Sens. Actuators B Chem. 2017, 239, 455–461. [Google Scholar] [CrossRef]
- Welearegay, T.G.; Diouani, M.F.; Österlund, L.; Borys, S.; Khaled, S.; Smadhi, H.; Ionescu, F.; Bouchekoua, M.; Aloui, D.; Laouini, D.; et al. Diagnosis of human echinococcosis via exhaled breath analysis: A promise for rapid diagnosis of infectious diseases caused by helminths. J. Infect. Dis. 2019, 219, 101–109. [Google Scholar] [CrossRef] [PubMed]


| Animal No. | M. bovis Infection | Sex | Age 1 | Location | Number of Samples 2 |
|---|---|---|---|---|---|
| 1 | Positive | Male | Adult | Martinazo | 2 |
| 2 | Negative | Male | Juvenile | Santa Olalla | 2 |
| 3 | Positive | Male | Juvenile | Palacio | 2 |
| 4 | Positive | Male | Juvenile | Palacio | 2 |
| 5 | Positive | Female | Adult | Palacio | 2 |
| 6 | Positive | Female | Sub-adult | Palacio | 2 |
| 7 | Negative | Male | Sub-adult | Santa Olalla | 2 |
| 8 | Negative | Female | Sub-adult | Santa Olalla | 2 |
| 9 | Positive | Male | Adult | Santa Olalla | 2 |
| 10 | Negative | Male | Juvenile | Santa Olalla | 2 |
| 11 | Negative | Female | Adult | Santa Olalla | 2 |
| 12 | Positive | Male | Adult | Santa Olalla | 2 |
| 13 | Negative | Male | Adult | Santa Olalla | 2 |
| 14 | Negative | Female | Adult | Martinazo | 2 |
| 15 | Positive | Female | Juvenile | Martinazo | 2 |
| 16 | Negative | Male | Juvenile | Martinazo | 2 |
| 17 | Positive | Male | Sub-adult | Martinazo | 2 |
| 18 | Positive | Male | Sub-adult | Martinazo | 2 |
| 19 | Negative | Female | Juvenile | Palacio | 2 |
| 20 | Negative | Female | Juvenile | Palacio | 2 |
| 21 | Negative | Female | Juvenile | Martinazo | 2 |
| 22 | Negative | Female | Juvenile | Santa Olalla | 2 |
| 23 | Negative | Female | Adult | Santa Olalla | 2 |
| 24 | Negative | Female | Juvenile | Santa Olalla | 2 |
| 25 | Positive | Female | Juvenile | Martinazo | 2 |
| 26 | Positive | Male | Juvenile | Martinazo | 2 |
| 27 | Positive | Female | Juvenile | Martinazo | 2 |
| 28 | Positive | Female | Juvenile | Martinazo | 1 |
| 29 | Positive | Male | Adult | Santa Olalla | 1 |
| 30 | Positive | Female | Adult | Santa Olalla | 1 |
| 31 | Positive | Male | Juvenile | Martinazo | 2 |
| 32 | Negative | Female | Adult | Martinazo | 1 |
| 33 | Negative | Male | Adult | Santa Olalla | 2 |
| 34 | Positive | Male | Sub-adult | Santa Olalla | 2 |
| 35 | Negative | Female | Juvenile | Santa Olalla | 2 |
| 36 | Negative | Male | Juvenile | Fuente del Duque | 1 |
| 37 | Negative | Female | Sub-adult | Martinazo | 1 |
| Age | Location | bTB Negative | bTB Positive | ||
|---|---|---|---|---|---|
| Male | Female | Male | Female | ||
| Adult | Santa Olalla | 2 | 2 | 3 | 1 |
| Martinazo | - | 2 | 1 | - | |
| Palacio | - | - | - | 1 | |
| Total | 2 | 4 | 4 | 2 | |
| Sub-adult | Santa Olalla | 1 | 1 | 1 | - |
| Martinazo | - | 1 | 2 | - | |
| Palacio | - | - | - | 1 | |
| Total | 1 | 2 | 3 | 1 | |
| Juvenile | Santa Olalla | 2 | 3 | - | - |
| Martinazo | 1 | 1 | 2 | 4 | |
| Palacio | - | 2 | 2 | - | |
| Fuente del Duque | 1 | - | - | - | |
| Total | 4 | 6 | 4 | 4 | |
| Sensor No. | Organic Functionality | Electrical Resistance |
|---|---|---|
| S1 | 2-Mercaptobenzoxazole | 347 kΩ |
| S2 | Methyl-3-mercaptopropionate | 253 kΩ |
| S3 | 1-Decanethiol | 506 kΩ |
| S4 | 1-Decanethiol | 641 kΩ |
| S5 | 2-Mercaptobenzoxazole | 1.5 kΩ |
| S6 | 11-Mercaptoundecanoic acid | 1.6 kΩ |
| S7 | 4-Methoxy-α-toluenethiol | 11 kΩ |
| S8 | 4-Methoxy-α-toluenethiol | 6.8 MΩ |
| S9 | 1-Butanethiol | 759 kΩ |
| S10 | Octadecylamine | 6.2 MΩ |
| Sensors | |||||
|---|---|---|---|---|---|
| S5 | S6 | S8 | S9 | ||
| Features | F1 | - | a, s | a, j | - |
| F2 | a, j | s | s, j | j | |
| F3 | a, j | j | a, s, j | j | |
| Phase | Age Group | Accuracy (%) | Sensitivity (%) | Specificity (%) | TP | TN | FN | FP |
|---|---|---|---|---|---|---|---|---|
| Training | Adult | 100 | 100 | 100 | 6 | 6 | 0 | 0 |
| Sub-adult | 100 | 100 | 100 | 4 | 3 | 0 | 0 | |
| Juvenile | 88.9 | 75 | 100 | 6 | 10 | 2 | 0 | |
| Testing | Adult | 88.9 | 100 | 80 | 4 | 4 | 0 | 1 |
| Sub-adult | 100 | 100 | 100 | 4 | 2 | 0 | 0 | |
| Juvenile | 62.5 | 28.6 | 88.9 | 2 | 8 | 5 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Jesús Beleño-Sáenz, K.; Cáceres-Tarazona, J.M.; Nol, P.; Jaimes-Mogollón, A.L.; Gualdrón-Guerrero, O.E.; Durán-Acevedo, C.M.; Barasona, J.A.; Vicente, J.; Torres, M.J.; Welearegay, T.G.; et al. Non-Invasive Method to Detect Infection with Mycobacterium tuberculosis Complex in Wild Boar by Measurement of Volatile Organic Compounds Obtained from Feces with an Electronic Nose System. Sensors 2021, 21, 584. https://doi.org/10.3390/s21020584
de Jesús Beleño-Sáenz K, Cáceres-Tarazona JM, Nol P, Jaimes-Mogollón AL, Gualdrón-Guerrero OE, Durán-Acevedo CM, Barasona JA, Vicente J, Torres MJ, Welearegay TG, et al. Non-Invasive Method to Detect Infection with Mycobacterium tuberculosis Complex in Wild Boar by Measurement of Volatile Organic Compounds Obtained from Feces with an Electronic Nose System. Sensors. 2021; 21(2):584. https://doi.org/10.3390/s21020584
Chicago/Turabian Stylede Jesús Beleño-Sáenz, Kelvin, Juan Martín Cáceres-Tarazona, Pauline Nol, Aylen Lisset Jaimes-Mogollón, Oscar Eduardo Gualdrón-Guerrero, Cristhian Manuel Durán-Acevedo, Jose Angel Barasona, Joaquin Vicente, María José Torres, Tesfalem Geremariam Welearegay, and et al. 2021. "Non-Invasive Method to Detect Infection with Mycobacterium tuberculosis Complex in Wild Boar by Measurement of Volatile Organic Compounds Obtained from Feces with an Electronic Nose System" Sensors 21, no. 2: 584. https://doi.org/10.3390/s21020584
APA Stylede Jesús Beleño-Sáenz, K., Cáceres-Tarazona, J. M., Nol, P., Jaimes-Mogollón, A. L., Gualdrón-Guerrero, O. E., Durán-Acevedo, C. M., Barasona, J. A., Vicente, J., Torres, M. J., Welearegay, T. G., Österlund, L., Rhyan, J., & Ionescu, R. (2021). Non-Invasive Method to Detect Infection with Mycobacterium tuberculosis Complex in Wild Boar by Measurement of Volatile Organic Compounds Obtained from Feces with an Electronic Nose System. Sensors, 21(2), 584. https://doi.org/10.3390/s21020584










