Fabrication, Potentiometric Characterization, and Application of Screen-Printed RuO2 pH Electrodes for Water Quality Testing
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of RuO2 Paste
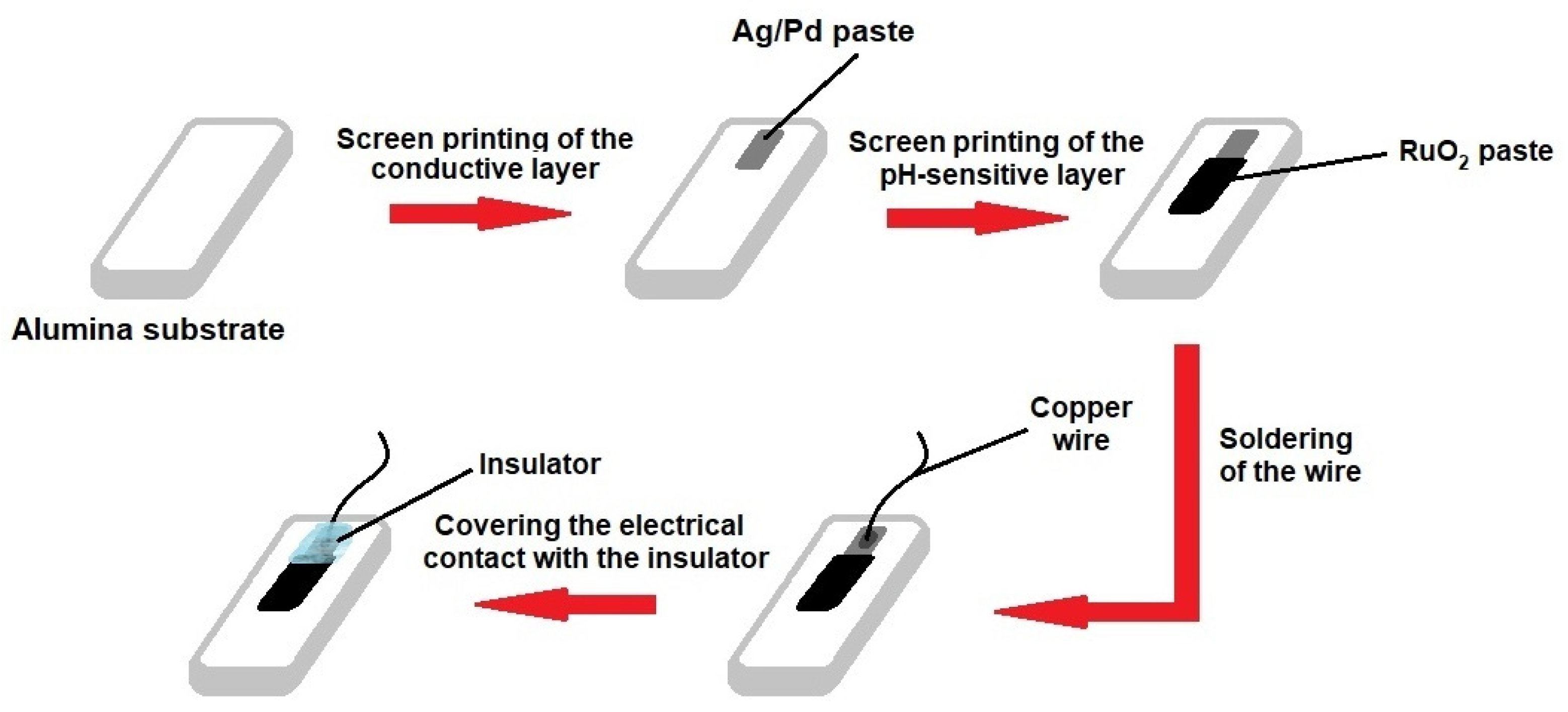
2.2. Fabrication of RuO2 Electrodes for Potentiometric Sensors
2.3. Microstructural Studies
2.4. Electrochemical Studies
2.4.1. pH Measurements
2.4.2. Response Time, Drift Rate, and Hysteresis
2.4.3. Cross-Sensitivity
2.5. Measurements of Real-Life Samples
3. Results and Discussion
3.1. Microstructure of RuO2 Electrodes
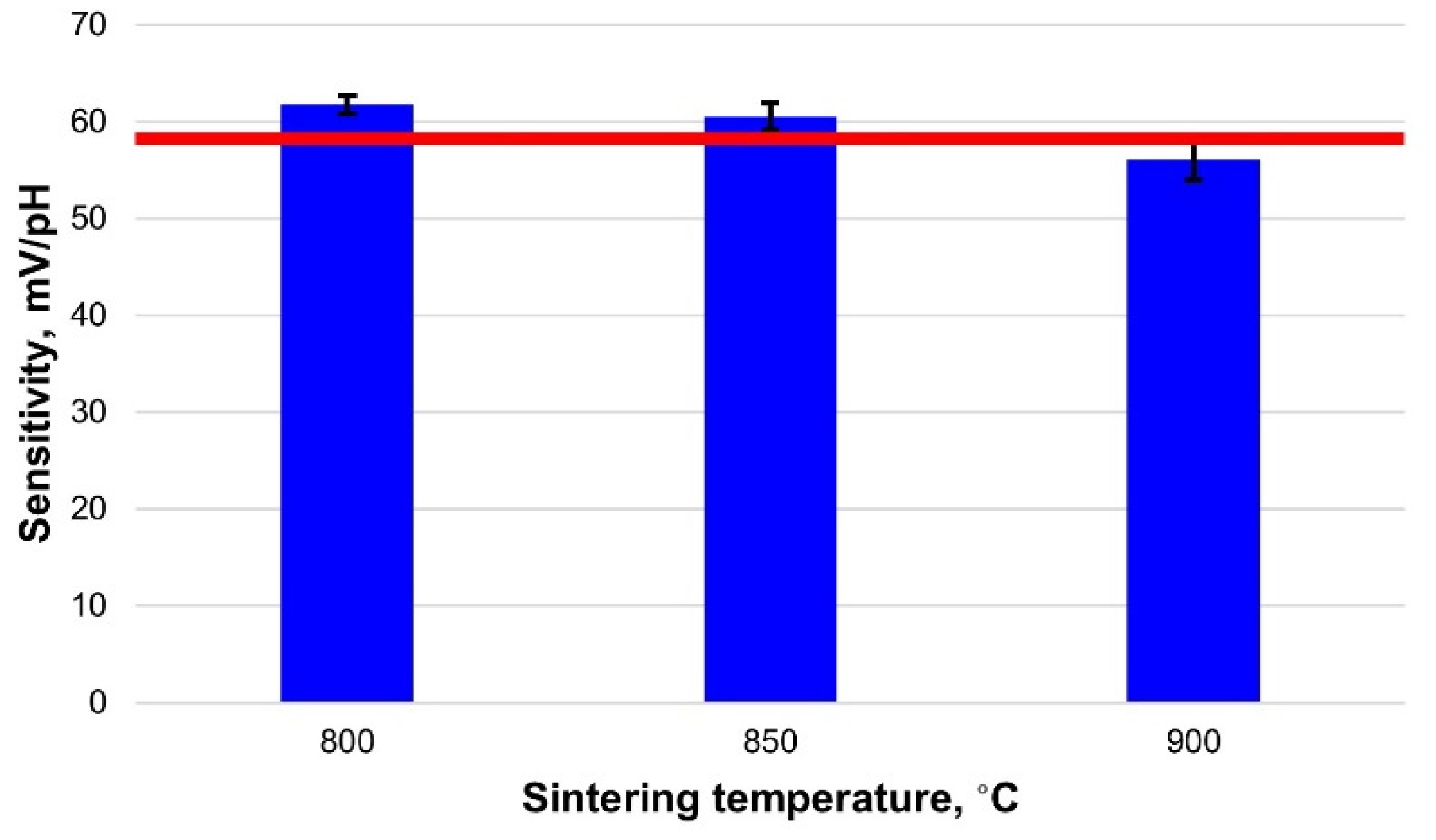
3.2. Sensitivity of the Fabricated Electrodes
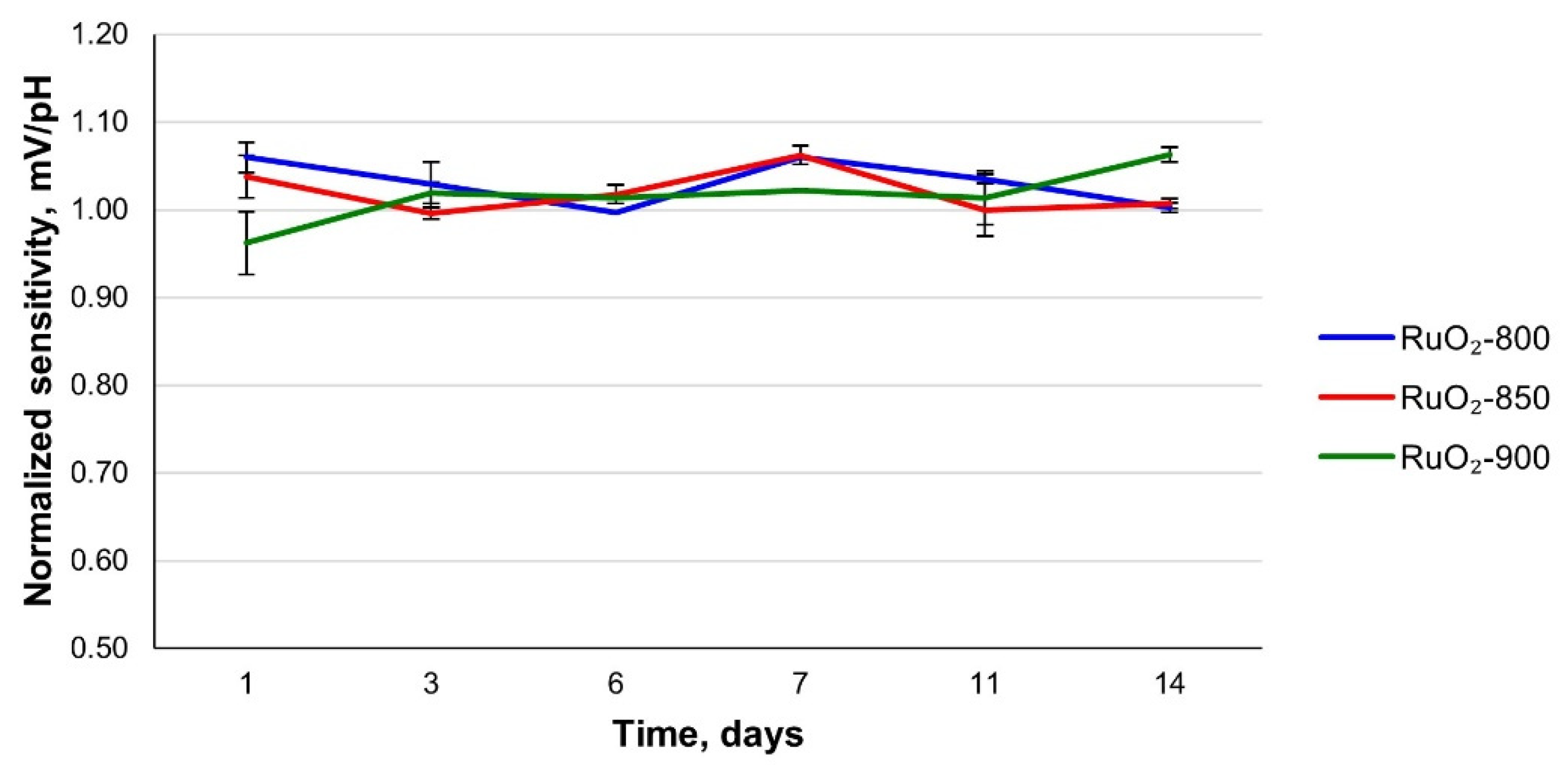
3.3. Long-Term Stability and Repeatability
3.4. Drift and Response Time
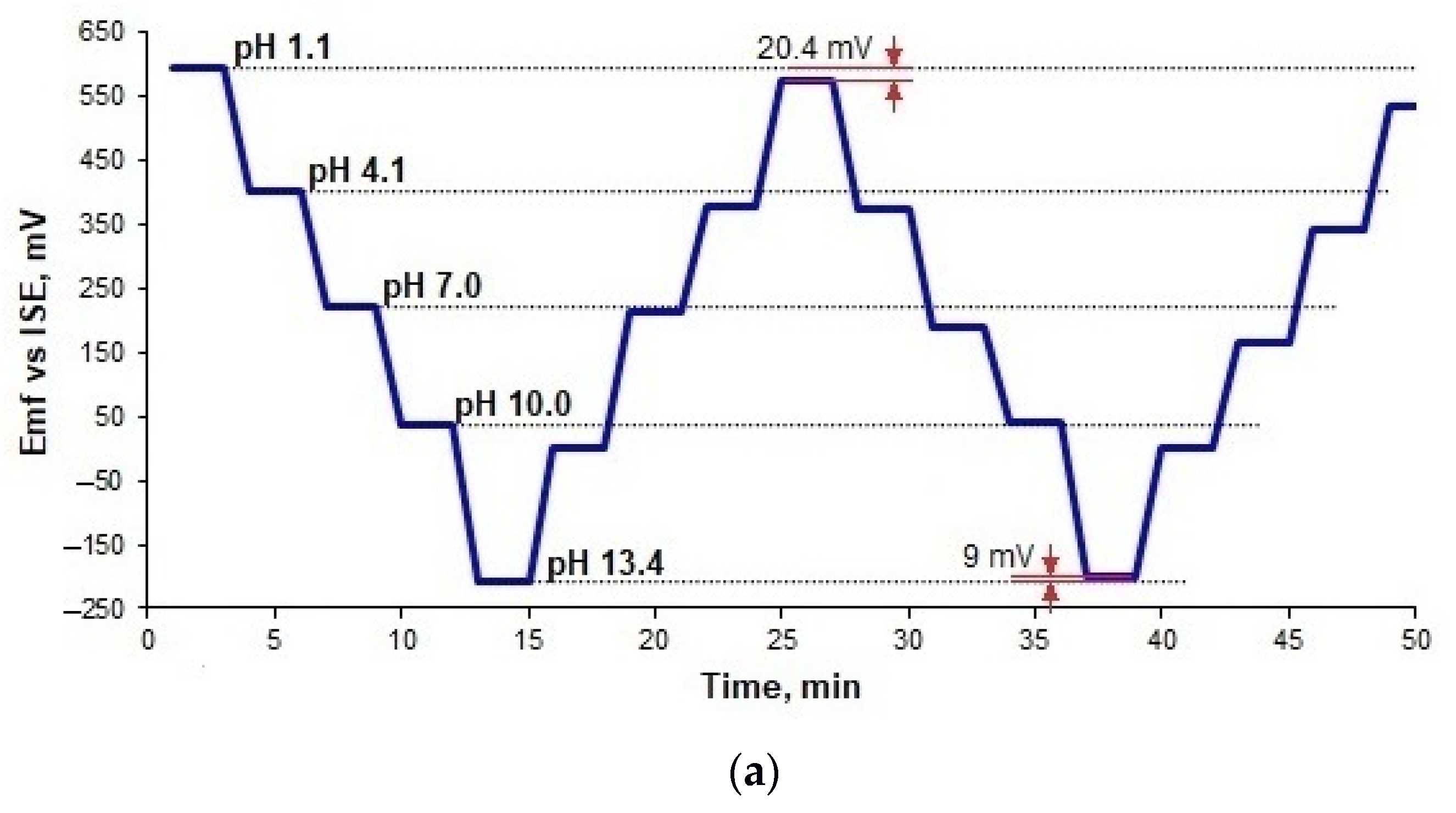
3.5. Hysteresis
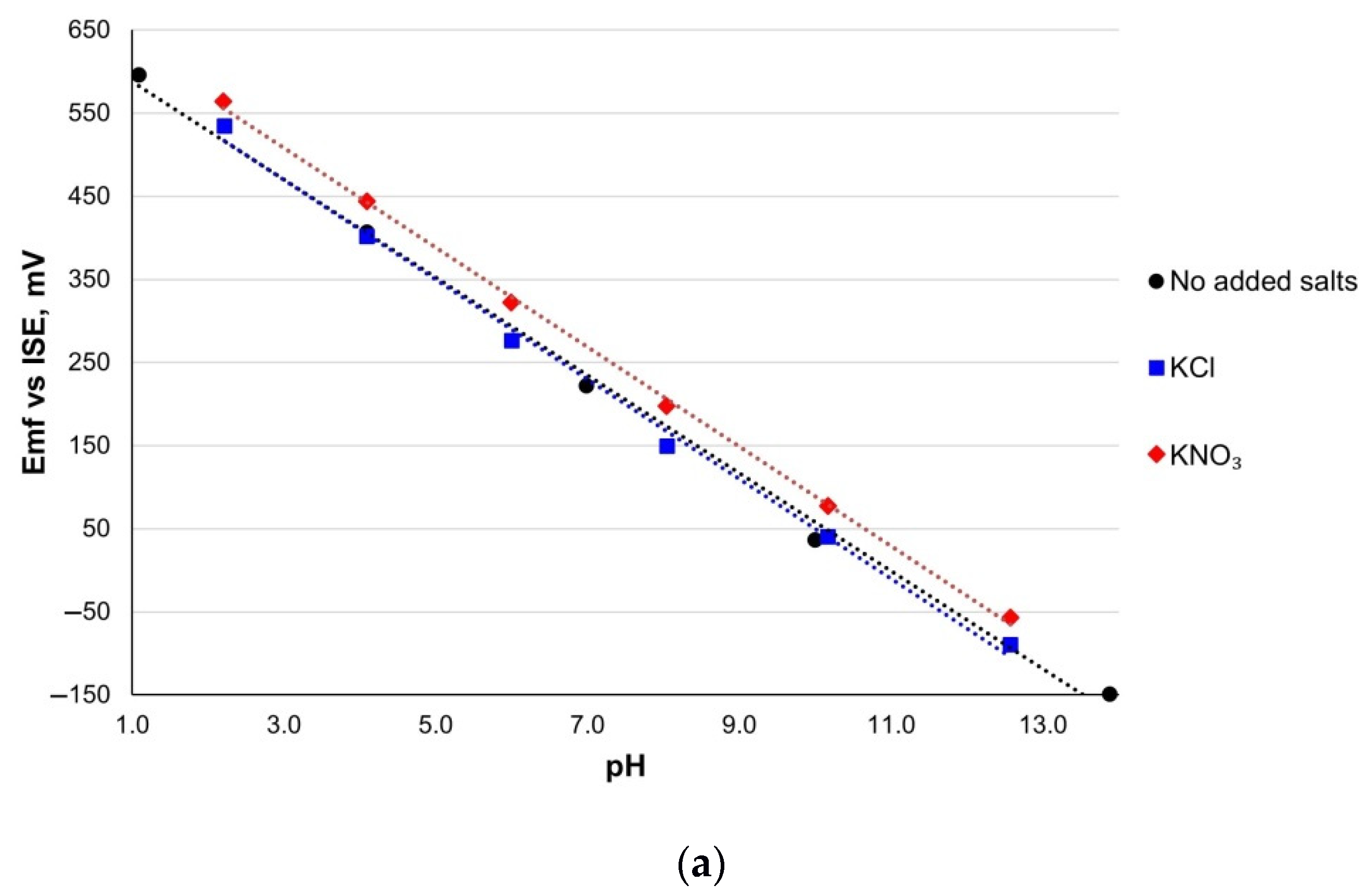
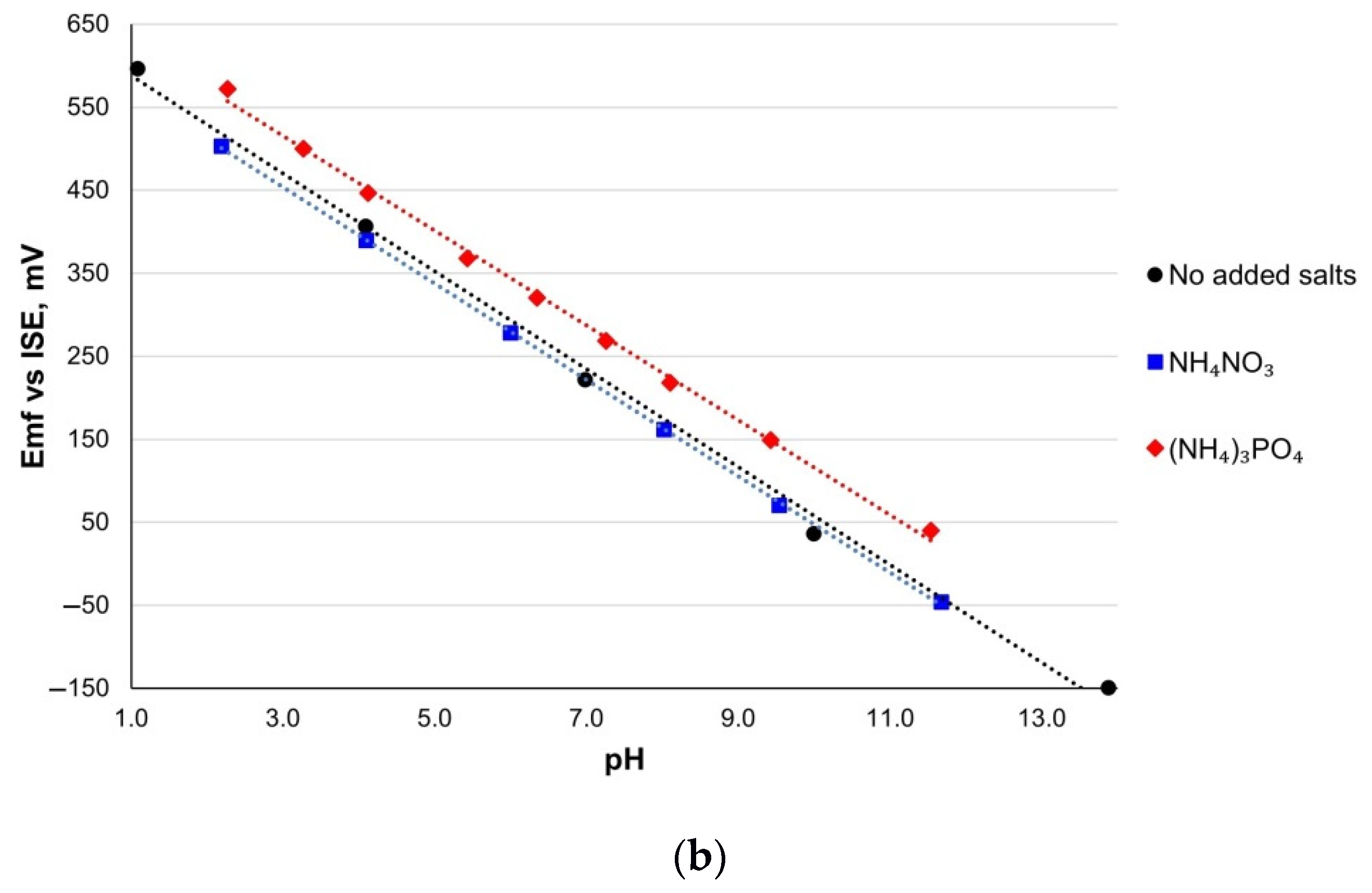
3.6. Cross-Sensitivity
3.7. Performance of pH Electrodes in Real-Life Water Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hassan Omer, N. Water quality parameters. In Water Quality-Science, Assessments and Policy; IntechOpen Ltd.: London, UK, 2019; pp. 1–18. [Google Scholar]
- Boyd, C.E.; Tucker, C.S.; Viriyatum, R. Interpretation of pH, Acidity, and Alkalinity in Aquaculture and Fisheries. N. Am. J. Aquac. 2011, 73, 403–408. [Google Scholar] [CrossRef]
- Poghossian, A.; Baade, A.; Emons, H.; Schöning, M. Application of ISFETs for pH measurement in rain droplets. Sens. Actuators B: Chem. 2001, 76, 634–638. [Google Scholar] [CrossRef]
- Chan, L.C.Z.; Moghaddam, G.K.; Wang, Z.; Lowe, C.R. Miniaturized pH Holographic Sensors for the Monitoring of Lactobacillus casei Shirota Growth in a Microfluidic Chip. ACS Sens. 2019, 4, 456–463. [Google Scholar] [CrossRef] [PubMed]
- John, G.T.; Goelling, D.; Klimant, I.; Schneider, H.; Heinzle, E. PH-sensing 96-well microtitre plates for the characterization of acid production by dairy starter cultures. J. Dairy Res. 2003, 70, 327–333. [Google Scholar] [CrossRef]
- Manjakkal, L.; Szwagierczak, D.; Dahiya, R. Metal oxides based electrochemical pH sensors: Current progress and future perspectives. Prog. Mater. Sci. 2020, 109, 100635. [Google Scholar] [CrossRef]
- Oh, H.; Lee, K.J.; Baek, J.; Yang, S.S.; Lee, K. Development of a high sensitive pH sensor based on shear horizontal surface acoustic wave with ZnO nanoparticles. Microelectron. Eng. 2013, 111, 154–159. [Google Scholar] [CrossRef]
- Khan, M.I.; Mukherjee, K.; Shoukat, R.; Dong, H. A review on pH sensitive materials for sensors and detection methods. Microsyst. Technol. 2017, 23, 4391–4404. [Google Scholar] [CrossRef]
- Yuqing, M.; Jianrong, C.; Keming, F. New technology for the detection of pH. J. Biochem. Biophys. Methods 2005, 63, 1–9. [Google Scholar] [CrossRef]
- Karastogianni, S.; Girousi, S.; Sotiropoulos, S. pH: Principles and Measurement, 1st ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2016; ISBN 9780123849533. [Google Scholar]
- Kurzweil, P. Metal Oxides and Ion-Exchanging Surfaces as pH Sensors in Liquids: State-of-the-Art and Outlook. Sensors 2009, 9, 4955–4985. [Google Scholar] [CrossRef] [Green Version]
- Hall, D.G. Ion-Selective Membrane Electrodes: A General Limiting Treatment of Interference Effects. J. Phys. Chem. 1996, 100, 7230–7236. [Google Scholar] [CrossRef]
- Fog, A.; Buck, R.P. Electronic semiconducting oxides as pH sensors. Sens. Actuators 1984, 5, 137–146. [Google Scholar] [CrossRef]
- Yates, D.E.; Levine, S.; Healy, T.W. Site-binding model of the electrical double layer at the oxide/water interface. J. Chem. Soc. Faraday Trans. 1 Phys. Chem. Condens. Phases 1974, 70, 1807–1818. [Google Scholar] [CrossRef]
- Kurzweil, P. Precious metal oxides for electrochemical energy converters: Pseudocapacitance and pH dependence of redox processes. J. Power Sources 2009, 190, 189–200. [Google Scholar] [CrossRef]
- Jadon, A.; Rossi, C.; Djafari-Rouhani, M.; Estève, A.; Pech, D. Interaction of hydrogen with the bulk, surface and subsurface of crystalline RuO2 from first principles. Phys. Open 2021, 7, 100059. [Google Scholar] [CrossRef]
- Zhuiykov, S. Morphology of Pt-doped nanofabricated RuO2 sensing electrodes and their properties in water quality monitoring sensors. Sens. Actuators B Chem. 2009, 136, 248–256. [Google Scholar] [CrossRef]
- Zhuiykov, S.; Kats, E.; Marney, D.; Kalantar-Zadeh, K. Improved antifouling resistance of electrochemical water quality sensors based on Cu2O-doped RuO2 sensing electrode. Prog. Org. Coat. 2011, 70, 67–73. [Google Scholar] [CrossRef]
- Sardarinejad, A.; Maurya, D.; Khaled, M.; Alameh, K. Temperature effects on the performance of RuO2 thin-film pH sensor. Sens. Actuators A Phys. 2015, 233, 414–421. [Google Scholar] [CrossRef]
- Xu, B.; Zhang, W.-D. Modification of vertically aligned carbon nanotubes with RuO2 for a solid-state pH sensor. Electrochim. Acta 2010, 55, 2859–2864. [Google Scholar] [CrossRef]
- Lonsdale, W.; Wajrak, M.; Alameh, K. Effect of conditioning protocol, redox species and material thickness on the pH sensitivity and hysteresis of sputtered RuO2 electrodes. Sens. Actuators B Chem. 2017, 252, 251–256. [Google Scholar] [CrossRef]
- Lonsdale, W.; Shylendra, S.P.; Wajrak, M.; Alameh, K. Application of all solid-state 3D printed pH sensor to beverage samples using matrix matched standard. Talanta 2019, 196, 18–21. [Google Scholar] [CrossRef]
- Liao, Y.-H.; Chou, J.-C. Preparation and characteristics of ruthenium dioxide for pH array sensors with real-time measurement system. Sens. Actuators B Chem. 2008, 128, 603–612. [Google Scholar] [CrossRef]
- Armelao, L.; Barreca, D.; Moraru, B. A molecular approach to RuO2-based thin films: Sol–gel synthesis and characterisation. J. Non-Cryst. Solids 2003, 316, 364–371. [Google Scholar] [CrossRef]
- Kahram, M.; Asnavandi, M.; Dolati, A. Synthesis and electrochemical characterization of sol–gel-derived RuO2/carbon nanotube composites. J. Solid State Electrochem. 2013, 18, 993–1003. [Google Scholar] [CrossRef]
- Pocrifka, L.; Gonçalves, C.; Grossi, P.; Colpa, P.; Pereira, E. Development of RuO2–TiO2 (70–30)mol% for pH measurements. Sens. Actuators B Chem. 2006, 113, 1012–1016. [Google Scholar] [CrossRef]
- Sadig, H.R.; Li, C. Applying a Novel Polymeric Precursor Derived by Capillary-Gravitational Coating in Fabrication of Nanostructured Tri- Metal Oxide-Based pH Sensing Electrode. IEEE Sens. J. 2020, 20, 12512–12521. [Google Scholar] [CrossRef]
- Shim, J.H.; Kang, M.; Lee, Y.; Lee, C. A nanoporous ruthenium oxide framework for amperometric sensing of glucose and potentiometric sensing of pH. Microchim. Acta 2012, 177, 211–219. [Google Scholar] [CrossRef]
- Manjakkal, L.; Cvejin, K.; Kulawik, J.; Zaraska, K.; Szwagierczak, D.; Socha, R.P. Fabrication of thick film sensitive RuO2-TiO2 and Ag/AgCl/KCl reference electrodes and their application for pH measurements. Sens. Actuators B Chem. 2014, 204, 57–67. [Google Scholar] [CrossRef]
- Manjakkal, L.; Cvejin, K.; Kulawik, J.; Zaraska, K.; Socha, R.P.; Szwagierczak, D. X-ray photoelectron spectroscopic and electrochemical impedance spectroscopic analysis of RuO2-Ta2O5 thick film pH sensors. Anal. Chim. Acta 2016, 931, 47–56. [Google Scholar] [CrossRef]
- Manjakkal, L.; Cvejin, K.; Kulawik, J.; Zaraska, K.; Szwagierczak, D.; Stojanovic, G. Sensing mechanism of RuO2–SnO2 thick film pH sensors studied by potentiometric method and electrochemical impedance spectroscopy. J. Electroanal. Chem. 2015, 759, 82–90. [Google Scholar] [CrossRef]
- Zhuiykov, S.; Marney, D.; Kats, E. Investigation of Electrochemical Properties of La2O3-RuO2 Thin-Film Sensing Electrodes Used in Sensors for the Analysis of Complex Solutions. Int. J. Appl. Ceram. Technol. 2011, 8, 1192–1200. [Google Scholar] [CrossRef]
- McMurray, H.; Douglas, P.; Abbot, D. Novel thick-film pH sensors based on ruthenium dioxide-glass composites. Sens. Actuators B Chem. 1995, 28, 9–15. [Google Scholar] [CrossRef]
- Lazouskaya, M.; Tamm, M.; Scheler, O.; Uppuluri, K.; Zaraska, K. Nafion as a protective membrane for screen-printed pH-sensitive ruthenium oxide electrodes. In Proceedings of the 2020 17th Biennial Baltic Electronics Conference (BEC), Tallinn, Estonia, 6–8 October 2020; pp. 1–4. [Google Scholar]
- Manjakkal, L.; Synkiewicz, B.; Zaraska, K.; Cvejin, K.; Kulawik, J.; Szwagierczak, D. Development and characterization of miniaturized LTCC pH sensors with RuO2 based sensing electrodes. Sens. Actuators B Chem. 2016, 223, 641–649. [Google Scholar] [CrossRef]
- Li, M.; Li, Y.-T.; Li, D.-W.; Long, Y.-T. Recent developments and applications of screen-printed electrodes in environmental assays—A review. Anal. Chim. Acta 2012, 734, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Antuña-Jiménez, D.; González-García, M.B.; Hernández-Santos, D.; Fanjul-Bolado, P. Screen-Printed Electrodes Modified with Metal Nanoparticles for Small Molecule Sensing. Biosensors 2020, 10, 9. [Google Scholar] [CrossRef] [Green Version]
- Labrador, R.H.; Soto, J.; Martínez-Máñez, R.; Coll, C.; Benito, A.; Ibáñez, J.; García-Breijo, E.; Gil, L. An electrochemical characterization of thick-film electrodes based on RuO2-containing resistive pastes. J. Electroanal. Chem. 2007, 611, 175–180. [Google Scholar] [CrossRef]
- Council of Europe. Potentiometric determination of pH. In European Pharmacopoeia 5.0; Council of Europe: Strasbourg, France, 2005; pp. 26–27. ISBN 978-9287152817. [Google Scholar]
- Lonsdale, W.; Wajrak, M.; Alameh, K. RuO2 pH Sensor with Super-Glue-Inspired Reference Electrode. Sensors 2017, 17, 2036. [Google Scholar] [CrossRef] [Green Version]
- Yao, X.; Vepsäläinen, M.; Isa, F.; Martin, P.; Munroe, P.; Bendavid, A. Advanced RuO2 Thin Films for pH Sensing Application. Sensors 2020, 20, 6432. [Google Scholar] [CrossRef]
- Mingels, R.; Kalsi, S.; Cheong, Y.; Morgan, H. Iridium and Ruthenium oxide miniature pH sensors: Long-term performance. Sens. Actuators B Chem. 2019, 297, 126779. [Google Scholar] [CrossRef]
- Uppuluri, K.; Lazouskaya, M.; Szwagierczak, D.; Zaraska, K. Influence of temperature on the performance of Nafion coated RuO2 based pH electrodes. In Proceedings of the 2021 IEEE International Conference on Flexible and Printable Sensors and Systems (FLEPS), Manchester, UK, 20–23 June 2021; pp. 1–4. [Google Scholar]
- Hrovat, M.; Bencan, A.; Belavič, D.; Holc, J.; Dražič, G. The influence of firing temperature on the electrical and microstructural characteristics of thick-film resistors for strain gauge applications. Sens. Actuators A Phys. 2003, 103, 341–352. [Google Scholar] [CrossRef]
- Osman, J.R.; Crayston, J.A.; Pratt, A.; Richens, D.T. RuO2–TiO2 mixed oxides prepared from the hydrolysis of the metal alkoxides. Mater. Chem. Phys. 2008, 110, 256–262. [Google Scholar] [CrossRef]
- Vitosh, M.L.; Fertilizers, N.-P.-K. Types, Uses and Characteristics; Extension Bulletin E—Cooperative Extension Service; Michigan State University: East Lansing, MI, USA, 1983. [Google Scholar]
- Jachimowski, A. Factors Affecting Water Quality in a Water Supply Network. J. Ecol. Eng. 2017, 18, 110–117. [Google Scholar] [CrossRef]
- Aleksander-Kwaterczak, U.; Plenzler, D. Contamination of small urban watercourses on the example of a stream in Krakow (Poland). Environ. Earth Sci. 2019, 78, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Galas, J. Limnological study on a lake formed in a limestone quarry (Kraków, Poland). I. Water chemistry. Pol. J. Environ. Stud. 2003, 12, 297–300. [Google Scholar]
- Sobczyk, W.; Mateusz, W.; Małgorzata, P. Physical and Chemical Indicators of Surface Waters of the Tatra National Park. J. Ecol. Eng. 2020, 21, 174–179. [Google Scholar] [CrossRef]



| Electrode Type | Sensitivity, mV/pH | E0, mV | R2 | Response Time, s | Drift Rate, mV/h | ||
|---|---|---|---|---|---|---|---|
| Theoretical | Observed | pH ≤ 7 | pH > 7 | ||||
| RuO2-800 | 58.9 | 61.8 ± 1.0 | 681.9 ± 5.0 | 0.996 | 2 ± 0 | 36 ± 1 | 0.1–0.2 |
| RuO2-850 | 60.5 ± 1.4 | 664.2 ± 11.3 | 0.994 | 2 ± 0 | 34 ± 0 | 0.1–0.9 | |
| RuO2-900 | 56.1 ± 2.1 | 624.0 ± 27.9 | 0.996 | 2 ± 0 | 26 ± 2 | 0.1–0.4 | |
| Sensitivity, mV/pH | E0, mV | R2 | |
|---|---|---|---|
| RuO2-800 | |||
| No added salt | 59.1 ± 0.2 | 647.0 ± 0.1 | 0.982 |
| KCl | 58.9 ± 1.1 | 630.3 ± 18.9 | 0.992 |
| KNO3 | 59.7 ± 0.2 | 684.3 ± 3.8 | 0.999 |
| NH4NO3 | 58.5 ± 0.6 | 633.9 ± 6.5 | 1.000 |
| (NH4)3PO4 | 58.0 ± 2.3 | 686.15 ± 0.0 | 0.998 |
| RuO2-850 | |||
| No added salt | 59.0 ± 0.4 | 642.4 ± 0.7 | 0.998 |
| KCl | 55.8 ± 0.2 | 585.3 ± 41.7 | 0.996 |
| KNO3 | 54.4 ± 2.2 | 550.8 ± 100.1 | 0.997 |
| NH4NO3 | 59.0 ± 0.8 | 642.0 ± 0.2 | 1.000 |
| (NH4)3PO4 | 57.0 ± 1.4 | 654.3 ± 16.1 | 0.995 |
| RuO2-900 | |||
| No added salt | 61.5 ± 1.1 | 645.6 ± 17.0 | 0.998 |
| KCl | 56.4 ± 0.6 | 642.3 ± 14.5 | 0.999 |
| KNO3 | 58.0 ± 0.7 | 674.9 ± 14.2 | 0.999 |
| NH4NO3 | 57.9 ± 0.3 | 629.4 ± 1.3 | 0.999 |
| (NH4)3PO4 | 59.9 ± 1.0 | 695.2 ± 13.1 | 0.996 |
| Water Sample | pH Values Measured with | |||
|---|---|---|---|---|
| Glass Electrode | RuO2-800 | RuO2-850 | RuO2-900 | |
| Distilled water | 7.30 | 7.29 ± 0.04 | 7.29 ± 0.01 | 7.27 ± 0.00 |
| Mineral water | 5.74 | 5.75 ± 0.01 | 5.74 ± 0.01 | 5.75 ± 0.00 |
| Tap water | 8.32 | 8.35 ± 0.05 | 8.39 ± 0.01 | 8.33 ± 0.07 |
| River water | 8.05 | 8.12 ± 0.01 | 8.11 ± 0.00 | 8.12 ± 0.00 |
| Lake Z water (surface) | 8.04 | 7.97 ± 0.11 | 8.04 ± 0.01 | 7.93 ± 0.02 |
| Lake Z water (deep) | 8.12 | 8.06 ± 0.07 | 8.13 ± 0.01 | 8.08 ± 0.11 |
| Lake M water (surface) | 6.75 | 6.84 ± 0.08 | 6.83 ± 0.11 | 6.86 ± 0.13 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uppuluri, K.; Lazouskaya, M.; Szwagierczak, D.; Zaraska, K.; Tamm, M. Fabrication, Potentiometric Characterization, and Application of Screen-Printed RuO2 pH Electrodes for Water Quality Testing. Sensors 2021, 21, 5399. https://doi.org/10.3390/s21165399
Uppuluri K, Lazouskaya M, Szwagierczak D, Zaraska K, Tamm M. Fabrication, Potentiometric Characterization, and Application of Screen-Printed RuO2 pH Electrodes for Water Quality Testing. Sensors. 2021; 21(16):5399. https://doi.org/10.3390/s21165399
Chicago/Turabian StyleUppuluri, Kiranmai, Maryna Lazouskaya, Dorota Szwagierczak, Krzysztof Zaraska, and Martti Tamm. 2021. "Fabrication, Potentiometric Characterization, and Application of Screen-Printed RuO2 pH Electrodes for Water Quality Testing" Sensors 21, no. 16: 5399. https://doi.org/10.3390/s21165399
APA StyleUppuluri, K., Lazouskaya, M., Szwagierczak, D., Zaraska, K., & Tamm, M. (2021). Fabrication, Potentiometric Characterization, and Application of Screen-Printed RuO2 pH Electrodes for Water Quality Testing. Sensors, 21(16), 5399. https://doi.org/10.3390/s21165399






