Wearable Technology to Detect Motor Fluctuations in Parkinson’s Disease Patients: Current State and Challenges
Abstract
1. Introduction
2. Methods
2.1. PRISMA Statement
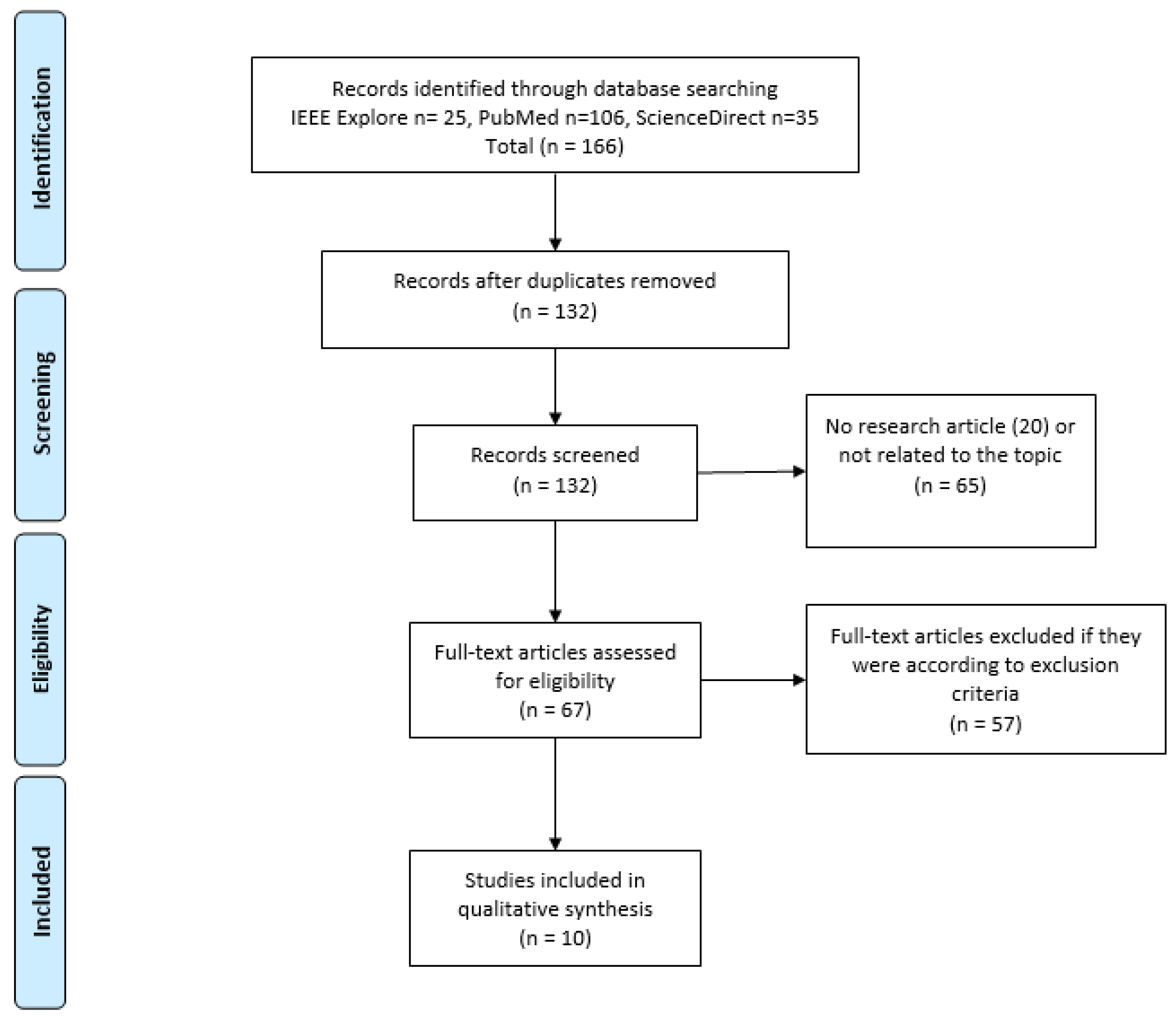
2.2. Identification: Search Strategy and Sources
2.3. Screening and Eligibility
2.4. Data Extraction and Analysis
- -
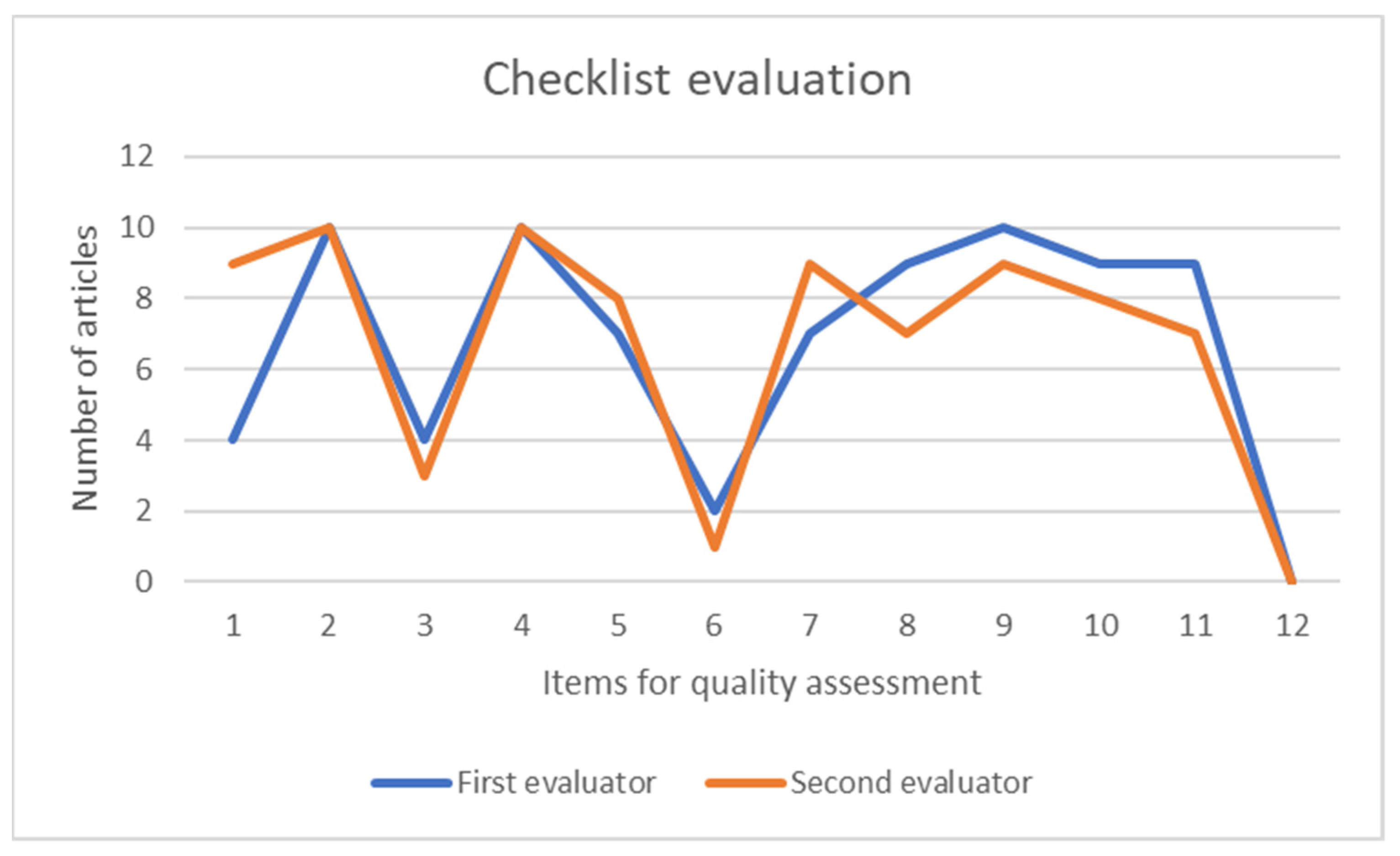
- Items 1, 2: focused on reviewing the title and the abstract.
- -
- Items 3, 4: evaluate the information provided in the introduction.
- -
- Items 5, 6: evaluate the description and completeness of the dataset and the main method/s analyzed in the research.
- -
- Item 7: this point evaluates the data pre-processing method, if any, and, in general, any step to prepare the data for analysis.
- -
- Item 8: this item is related to the steps that form the predictive model.
- -
- Item 9: this is focused on evaluating the performance on the evaluated model.
- -
- Items 10, 11 and 12: these items are related to the quality of the discussion. They evaluate the existence and quality of the clinical implications, the limitations of the study, and unexpected results.
2.5. ML Techniques
3. Results
3.1. Eligibility According to PRISMA Flow Diagram
3.2. Analysis of the Quality of the Articles
| (a) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Authors | References | Country | Publication Year | Sample Size | Sex (F/M) | Stage (UPDRS or H&Y) | Sensor | Features | Classifier | Performance Indices and Outcome |
| A Kinematic Sensor and Algorithm to Detect Motor Fluctuations in Parkinson Disease: Validation Study Under Real Conditions of Use | Rodriguez-Molinero, A. et al. | [22] | Spain | 2018 | 23 | 7/16 | 21 ± 16 UPDRS | IMU | Spatiotemporal gait | Own machine learning algorithm | Accuracy (92.2%) |
| A Supervised Machine Learning Approach to Detect the On/Off State in Parkinson’s Disease Using Wearable Based Gait Signals | Aich, S. et al. | [23] | South Korea | 2020 | 20 | 8/12 | 15.8 ± 10.13 UPDRS | Accelerometer | Statistical features + spatiotemporal gait features | Random forest, kNN, SVM and naïve Bayes | Accuracy (96.72%), recall (97.35%), precision (96.92%) |
| A Treatment-Response Index from Wearable Sensors for Quantifying Parkinson’s Disease Motor States | Thomas, I. et al. | [24] | Sweden | 2017 | 19 | 5/14 | Advanced stage | Accelerometer and gyroscope | Spatiotemporal features | SVM, decision tree, random forest, linear regression | Classification accuracy (89%, 74%, 84%, 81%) |
| (b) | |||||||||||
| Analysis of Correlation between an Accelerometer-Based Algorithm for Detecting Parkinsonian Gait and UPDRS Subscales | Rodriguez-Molinero, A. et al. | [25] | Spain, Italy, Israel, Ireland, | 2017 | 75 | 27/48 | 15 ± 13 UPDRS | IMU | Spatiotemporal gait features | SVM | Correlation (rho −0.73; p < 0.001) |
| Assessing Motor Fluctuations in Parkinson’s Disease Patients Based on a Single Inertial Sensor | Pérez-López, C. | [26] | Spain | 2016 | 15 | 5/10 | 2.66 H&Y | IMU | Spatiotemporal, frequential gait features | hierarchical algorithm | Specificity (92%), sensitivity (92%) |
| Assessment of response to medication in individuals with Parkinson’s disease | Hssayeni, M.D. et al. | [27] | United States | 2019 | 19 | 5/14 | 14 ± 8 UPDRS | Gyroscope and accelerometer | Spatiotemporal, frequential gait features | SVM | Accuracy (90.5%), sensitivity (94.2%), specificity (85.4%) |
| High-Resolution Motor State Detection in Parkinson’s Disease Using Convolutional Neural Networks | Pfister, F.M.J. et al. | [28] | Germany | 2020 | 30 | 10/20 | 21.6 ± 15.3 UPDRS | IMU | Spatiotemporal gait | CNN | Sensitivity (64%), specificity (89%) |
| (c) | |||||||||||
| Multilevel Features for Sensor-Based Assessment of Motor Fluctuation in Parkinson’s Disease Subjects | Ghoraani, B. et al. | [29] | United States | 2019 | 19 | 5/14 | UPDRS: 14 ± 8 | Gyroscope | Time-domain features, frequency-domain features | SVM | Accuracy (83.56%), sensitivity (78.51%), specificity (92.02%) |
| Unsupervised home monitoring of Parkinson’s disease motor symptoms using body-worn accelerometers | Fisher, J.M. et al. | [30] | United Kingdom | 2016 | 34 | Not specified | H&R I-IV | Accelerometer | Temporal features | ANN | Sensitivity (51%), specificity (87%) |
| Validation of a portable device for mapping motor and gait disturbances in Parkinson’s disease | Rodriguez-Molinero, A. et al. | [31] | Spain | 2015 | 35 | 8/27 | H&Y III | Accelerometer | Frequential and spatiotemporal parameters | SVM | Sensitivity (96%), specificity (94%) |
3.3. Analysis of the Selected Articles
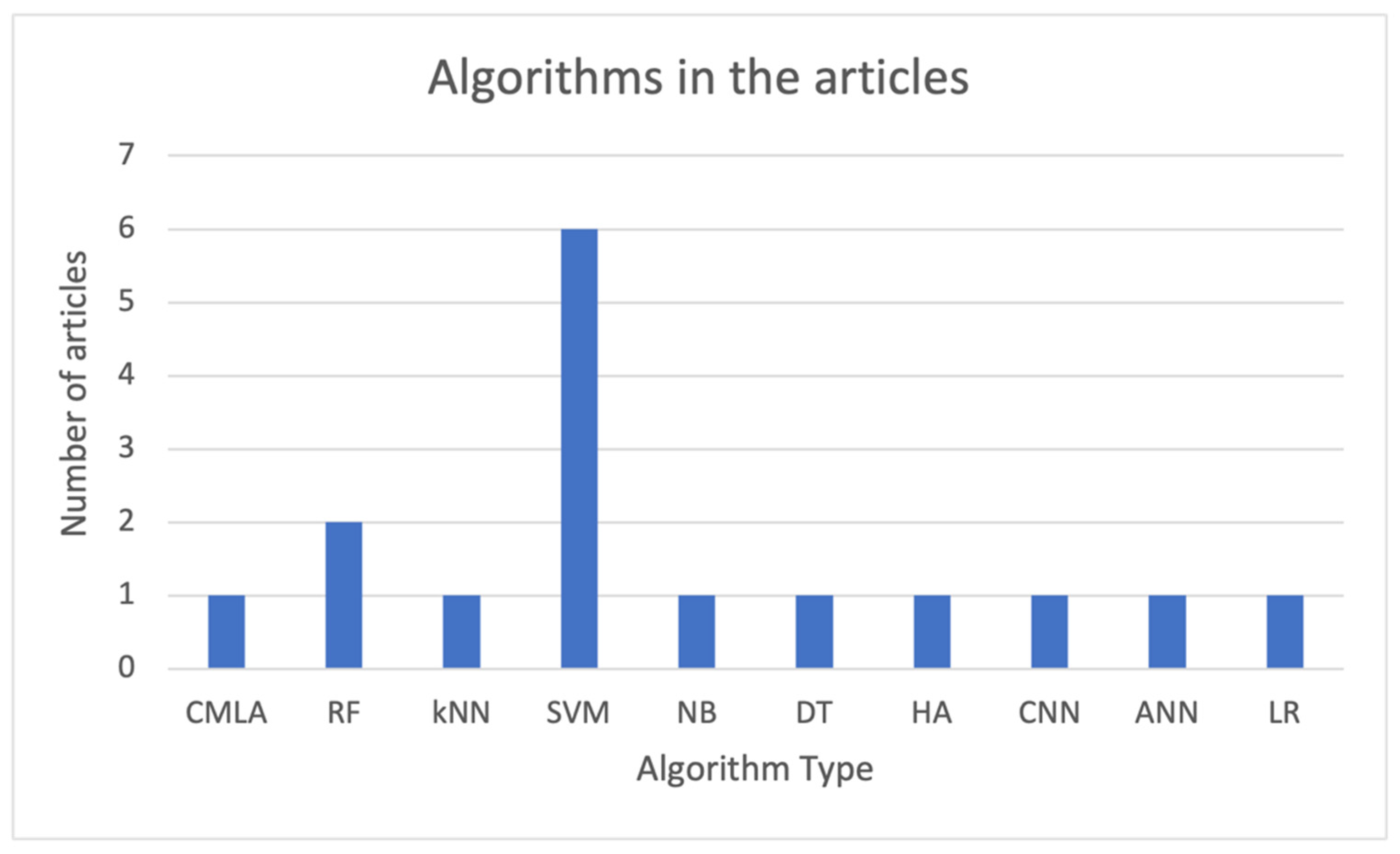
3.3.1. Types of Models Considered
3.3.2. Type of Data Collected
Daily Living Activities
Specific Activities
Combination of Both (Daily Activity + Specific Activities)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Monteagudo, J.L.; Serrano, L.; Hernández Salvador, C. La telemedicina: ¿ciencia o ficción? Anales del Sistema Sanitario de Navarra 2005, 28, 309–323. [Google Scholar] [CrossRef] [PubMed][Green Version]
- De Lau, L.M.; Breteler, M.M. Epidemiology of Parkinson’s disease. Lancet Neurol. 2006, 5, 525–535. [Google Scholar] [CrossRef]
- Gelb, D.J.; Oliver, E.; Gilman, S. Diagnostic criteria for Parkinson disease. Arch. Neurol. 1999, 56, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Katzenschlager, R.; Lees, A.J. Treatment of Parkinson’s disease: Levodopa as the first choice. J. Neurol. 2002, 249, 19–24. [Google Scholar] [CrossRef]
- National Institute of Neurological Disorders and Stroke. Parkinson’s disease: Challenges, progress and promise; NIH Publ.: Bethesda, MY, USA, 2015; p. 15. [Google Scholar]
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov. Disord. 2008, 23, 2129–2170. [Google Scholar] [CrossRef]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression, and mortality. Neurology 1967, 17, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Pastorino, M.; Cancela, J.; Arredondo, M.T.; Pastor-Sanz, L.; Contardi, S.; Valzania, F. Preliminary results of ON/OFF detection using an integrated system for Parkinson’s disease monitoring. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; Volume 941. [Google Scholar]
- Tzallas, A.T.; Tsipouras, M.G.; Rigas, G. PERFORM: A system for monitoring, assessment and management of patients with Parkinson’s disease. Sensor 2014, 14, 21329–21357. [Google Scholar] [CrossRef]
- Patel, S.; Chen, B.R.; Mancinelli, C.; Paganoni, S.; Shih, L.; Welsh, M. Longitudinal monitoring of patients with Parkinson’s disease via wearable sensor technology in the home setting. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; pp. 552–555. [Google Scholar]
- Son, H.; Park, W.S.; Kim, H. Mobility monitoring using smart technologies for Parkinson’s disease in free-living environment. Collegian 2018, 25, 549–560. [Google Scholar] [CrossRef]
- Ossig, C.; Antonini, A.; Buhmann, C.; Classen, J.; Csoti, I.; Falkenburger, B. Wearable sensor-based objective assessment of motor symptoms in Parkinson’s disease. J. Neural Transm. 2016, 123, 57–64. [Google Scholar] [CrossRef]
- Godinho, C.; Domingos, J.; Cunha, G.; Santos, A.T.; Fernandes, R.M.; Abreu, D. A systematic review of the characteristics and validity of monitoring technologies to assess Parkinson’s disease. J. Neuroeng. Rehabil. 2016, 13, 24. [Google Scholar] [CrossRef] [PubMed]
- Hasan, H.; Athauda, D.S.; Foltynie, T.; Noyce, A.J. Technologies assessing limb bradykinesia in Parkinson’s disease. J. Parkinsons Dis. 2017, 7, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Au, L. Recent scientific/intellectual movements in biomedicine. Soc. Sci. Med. 2021, 278, 113950. [Google Scholar] [CrossRef]
- Nalinipriya, G.; Geetha, M.; Cristin, R.; Maram, B. Biomedical data mining for improved clinical Diagnosis. In Artificial Intelligence in Data Mining; Academic Press: London, UK, 2021. [Google Scholar]
- Hayward, J. Wearable Sensors 2021–2031: Global Market Forecasts, Key Players and Technology Appraisal; IDTechEx Research: Cambridge, UK, 2020. [Google Scholar]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; The PRISMA-P Group. Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015: Statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; The PRISMA-P Group. Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015: Elaboration and explanation. BMJ 2015, 349, g7647, Erratum in 2016, 354, i4086. [Google Scholar] [CrossRef]
- Luo, W.; Phung, D.; Tran, T.; Gupta, S.; Rana, S.; Karmakar, C.; Shilton, A.; Yearwood, J.; Dimitrova, N.; Ho, T.B.; et al. Guidelines for Developing and Reporting Machine Learning Predictive Models in Biomedical Research: A Multidisciplinary View. J. Med. Internet Res. 2016, 18. [Google Scholar] [CrossRef] [PubMed]
- McHugh, M.L. Interrater reliability: The kappa statistic. Biochem. Med. 2012, 22, 276–282. [Google Scholar] [CrossRef]
- Rodríguez-Molinero, A.; Pérez-López, C.; Samà, A.; de Mingo, E.; Rodríguez-Martín, D.; Hernández-Vara, J.; Bayés, À.; Moral, A.; Álvarez, R.; Pérez-Martínez, D.A.; et al. A Kinematic Sensor and Algorithm to Detect Motor Fluctuations in Parkinson Disease: Validation Study Under Real Condi-tions of Use. JMIR Rehabil. Assist. Technol. 2018, 5, e8. [Google Scholar] [CrossRef]
- Aich, S.; Youn, J.; Chakraborty, S.; Pradhan, P.M.; Park, J.H.; Park, S.; Park, J. A Supervised Machine Learning Approach to Detect the On/Off State in Parkinson’s Disease Using Wearable Based Gait Signals. Diagnostics 2020, 10, 421. [Google Scholar] [CrossRef]
- Thomas, I.; Westin, J.; Alam, M.; Bergquist, F.; Nyholm, D.; Senek, M.; Memedi, M. A Treatment-Response Index From Wearable Sensors for Quantifying Parkinson’s Disease Motor States. J. Biomed. Health Inform. 2018, 22, 1341–1349. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Molinero, A.; Samà, A.; Pérez-López, C.; Rodríguez-Martín, D.; Quinlan, L.R.; Alcaine, S. Analysis of Correlation between an Accelerometer-Based Algorithm for Detecting Parkinsonian Gait and UPDRS Subscales. Front. Neurol. 2017, 8, 431. [Google Scholar] [CrossRef]
- Pérez-López, C.; Samà, A.; Rodríguez-Martín, D.; Català, A.; Cabestany, J.; Moreno-Arostegui, J.M.; de Mingo, E.; Rodríguez-Mlinero, A. Assessing Motor Fluctuations in Parkinson’s Disease Patients Based on a Single Inertial Sensor. Sensors 2016, 16, 2132. [Google Scholar] [CrossRef]
- Hssayeni, M.D.; Burack, M.A.; Jimenez-Shahed, J. Assessment of response to medication in individuals with Parkinson’s disease. Med. Eng. Phys. 2019, 67, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Pfister, F.M.J.; Um, T.T.; Pichler, D.C. High-Resolution Motor State Detection in Parkinson’s Disease Using Convolutional Neural Networks. Sci. Rep. 2020, 10, 5860. [Google Scholar] [CrossRef] [PubMed]
- Ghoraani, B.; Hssayeni, M.D.; Bruack, M.M.; Jimenez-Shahed, J. Multilevel Features for Sensor-Based Assessment of Motor Fluctuation in Parkinson’s Disease Subjects. IEEE J. Biomed. Health Inform. 2019, 24, 1284–1295. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.M.; Hammerla, N.Y.; Ploetz, T.; Andras, P.; Rochester, L.; Walker, R.W. Unsupervised home monitoring of Parkinson’s disease motor symptoms using body-worn accelerometers. Parkinsonism Relat. Disord. 2016, 33, 44–50. [Google Scholar] [CrossRef]
- Rodríguez-Molinero, A.; Samà, A.; Pérez-Martínez, D.A.; Pérez López, C.; Romagosa, J.; Bayés, À.; Sanz, P.; Calopa, M.; Gálvez-Barrón, C.; de Mingo, E.; et al. Validation of a portable device for mapping motor and gait disturbances in Parkinson’s disease. JMIR Mhealth Uhealth 2015, 3, e9. [Google Scholar] [CrossRef] [PubMed]
- Espay, A.J.; Bonato, P.; Nahab, F.B.; Maetzler, W.; Dean, J.M.; Klucken, J.; Eskofier, B.M.; Merola, A.; Horak, F.; Lang, A.E.; et al. Technology in Parkinson’s disease: Challenges and opportunities. Mov. Disord. 2016, 31, 1272–1282. [Google Scholar] [CrossRef]
- Cabestany, J.; Pérez-López, C.; Sama, A.; Moreno, J.M.; Bayes, A.; Rodriguez-Molinero, A. REMPARK: When AI and Technology Meet Parkinson Disease Assessment. In Proceedings of the 20th International Conference Mixed Design of Integrated Circuits and Systems (MIXDES) 2013, Gdynia, Poland, 20–22 June 2013; pp. 562–567. [Google Scholar]
- Ahlrichs, C.; Sam´a, A.; Rovira, J.; Herrlich, S.; Rodríguez Molinero, A. HELP: Optimizing Treatment of Parkinson’s Disease Patients. In Proceedings of the 3rd International Conference on Elderly and New Technologies; Universitat per a Majors de la Universitat Jaume I: Castellón de la Plana, Spain, 2013; pp. 17–24. [Google Scholar]
- European Comission i-PROGNOSIS Project Builds Early Detection Test of Parkinsons’ Disease. 2016. Available online: https://ec.europa.eu/programmes/horizon2020/en/news/i-prognosis-project-builds-early-detection-test-parkinsons%E2%80%99-disease (accessed on 10 November 2020).
- Maitín, A.M.; García-Tejedor, A.J.; Muñoz, J.P.R. Machine Learning Approaches for Detecting Parkinson’s Disease from EEG Analysis: A Systematic Review. Appl. Sci. 2020, 10, 8662. [Google Scholar] [CrossRef]
- Belić, M.; Bobić, V.; Badža, M.; Solaja, N.; Đurić-Jovičić, M.; Kostić, V.S. Artificial intelligence for assisting diagnostics and assessment of Parkinson’s disease—A review. Clin. Neurol. Neurosurg. 2019, 184, 105442. [Google Scholar] [CrossRef] [PubMed]
- Attia, Z.I.; Noseworthy, P.A.; Lopez-Jimenez, F.; Asirvatham, S.J.; Deshmukh, A.J.; Gersh, B.J. An artificial intelligence-enabled ECG algorithm for the identification of patients with atrial fibrillation during sinus rhythm: A retrospective analysis of outcome prediction. Lancet 2019, 394, 861–867. [Google Scholar] [CrossRef]
- Raza, M.; Awais, M.; Ellahi, W.; Aslam, N.; Nguyen, H.; Le-Minh, H. Diagnosis and Monitoring of Alzheimer’s Patients Using Classical and Deep Learning Techniques. Expert Syst. Appl. 2019, 136, 353–364. [Google Scholar] [CrossRef]
- Sharma, S.; Dudeja, R.K.; Aujla, G.S.; Bali, R.S.; Kumar, N. DeTrAs: Deep learning-based healthcare framework for IoT-based assistance of Alzheimer patients. Neural Comput. Appl. 2020. [Google Scholar] [CrossRef]
- Simblet, S.K.; Greer, B.; Matcham, F.; Curtis, H.; Polhemus, A.; Ferrao, J.; Gamble, P.; Wykes, T. Barriers and facilitators to engagement with remote measurement technology for managinghealth: A systematic review and content analysis of findings. J. Med. Internet Res. 2018, 20, e10480. [Google Scholar] [CrossRef] [PubMed]
| ML Type | Purpose | Typical Algorithms | Description |
|---|---|---|---|
| Supervised algorithm | Classification | Naïve Bayes, logistic regression, support vector machines | The main purpose of these algorithms is to classify data into the different predefined classes |
| Regression | Linear and non-linear regression | The main purpose of these algorithms is to find the relation between different variables | |
| Both | Decision trees, random forest, k-nearest neighbors, neural networks | These have classification properties but also the ability to find the relation between different variables | |
| Unsupervised algorithm | Clustering | K-means, neural networks, hidden Markov model | The main purpose of these types of algorithm is to discover groups in the input data |
| Refs | Year | Features | Cleaning Method | Results | Classifier | Perf. Indicator |
|---|---|---|---|---|---|---|
| [22] | 2018 | Spatiotemporal characteristics | Not specified | 92.20% | Own machine learning algorithm | Accuracy |
| [23] | 2020 | Statistical features + spatiotemporal features | Low pass BW filter | RF: 96.72%, 97.35%, 96.92%; SVM: 93%, 02%, 93%; KNN: 86%, 84%, 85%; NB: 88%, 86%, 85% | Random forest, kNN, SVM, and Naive Bayes | Accuracy, recall, precision |
| [24] | 2017 | Spatiotemporal features | ApEn method for motion removing | SVM:0.89, DT: 0.84, RF: 0.81, LR: 0.74 | SVM, decision tree, RF, linear regression | Classification accuracy |
| [25] | 2017 | Spatiotemporal features | Not specified | Correlation between the algorithm outputs gait status (rho −0.73; p < 0.001) | SVM | Correlation with UPDRS-III |
| [26] | 2016 | Spatiotemporal features + frequency features | Not specified | 92%, 92% | Hierarchical algorithm | Specificity and sensitivity |
| [27] | 2019 | Spatiotemporal + frequential features | Bandpass FIR filter | 90.5%, 94.2%, 85.4% | SVM | Accuracy, sensitivity, specificity |
| [28] | 2020 | Spatiotemporal features | Two direction BW filter | 64%, 89% | CNN | sensitivity, specificity |
| [29] | 2019 | Time-domain features and frequency-domain features | Bandpass filter | 83.56%, 78.51%, 92.02% | SVM | Accuracy, sensitivity and specificity |
| [30] | 2016 | Temporal features | Not specified | 51%, 87% | ANN | Sensitivity, specificity |
| [31] | 2015 | Frequency parameters (spectral power) | Not specified | 96%, 94% | SVM | Sensitivity, specificity |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barrachina-Fernández, M.; Maitín, A.M.; Sánchez-Ávila, C.; Romero, J.P. Wearable Technology to Detect Motor Fluctuations in Parkinson’s Disease Patients: Current State and Challenges. Sensors 2021, 21, 4188. https://doi.org/10.3390/s21124188
Barrachina-Fernández M, Maitín AM, Sánchez-Ávila C, Romero JP. Wearable Technology to Detect Motor Fluctuations in Parkinson’s Disease Patients: Current State and Challenges. Sensors. 2021; 21(12):4188. https://doi.org/10.3390/s21124188
Chicago/Turabian StyleBarrachina-Fernández, Mercedes, Ana María Maitín, Carmen Sánchez-Ávila, and Juan Pablo Romero. 2021. "Wearable Technology to Detect Motor Fluctuations in Parkinson’s Disease Patients: Current State and Challenges" Sensors 21, no. 12: 4188. https://doi.org/10.3390/s21124188
APA StyleBarrachina-Fernández, M., Maitín, A. M., Sánchez-Ávila, C., & Romero, J. P. (2021). Wearable Technology to Detect Motor Fluctuations in Parkinson’s Disease Patients: Current State and Challenges. Sensors, 21(12), 4188. https://doi.org/10.3390/s21124188








