Two-Dimensional Nanostructures for Electrochemical Biosensor
Abstract
1. Introduction
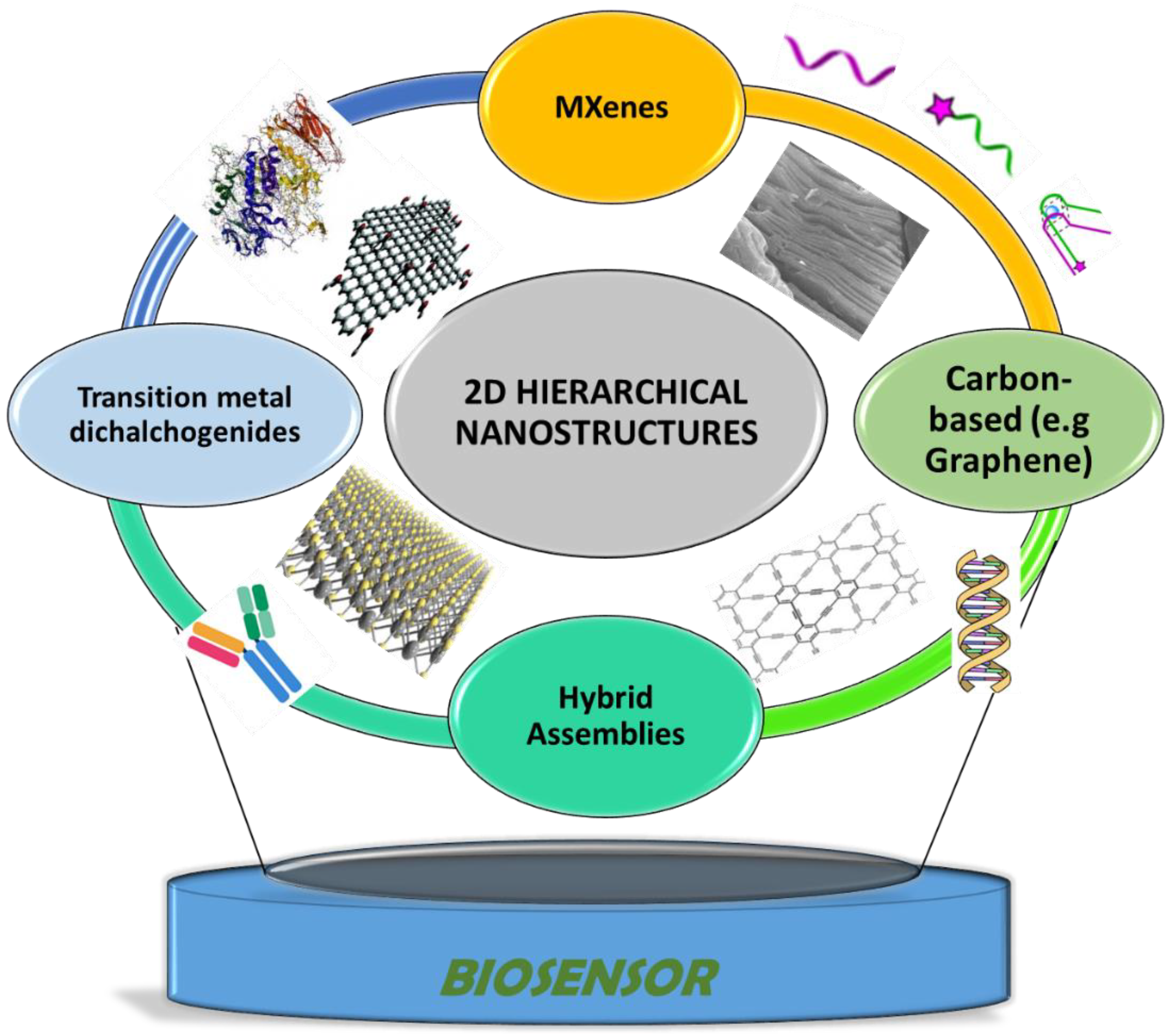
2. 2D Nanostructures: Synthesis, Properties, and Integration in Biosensing Design
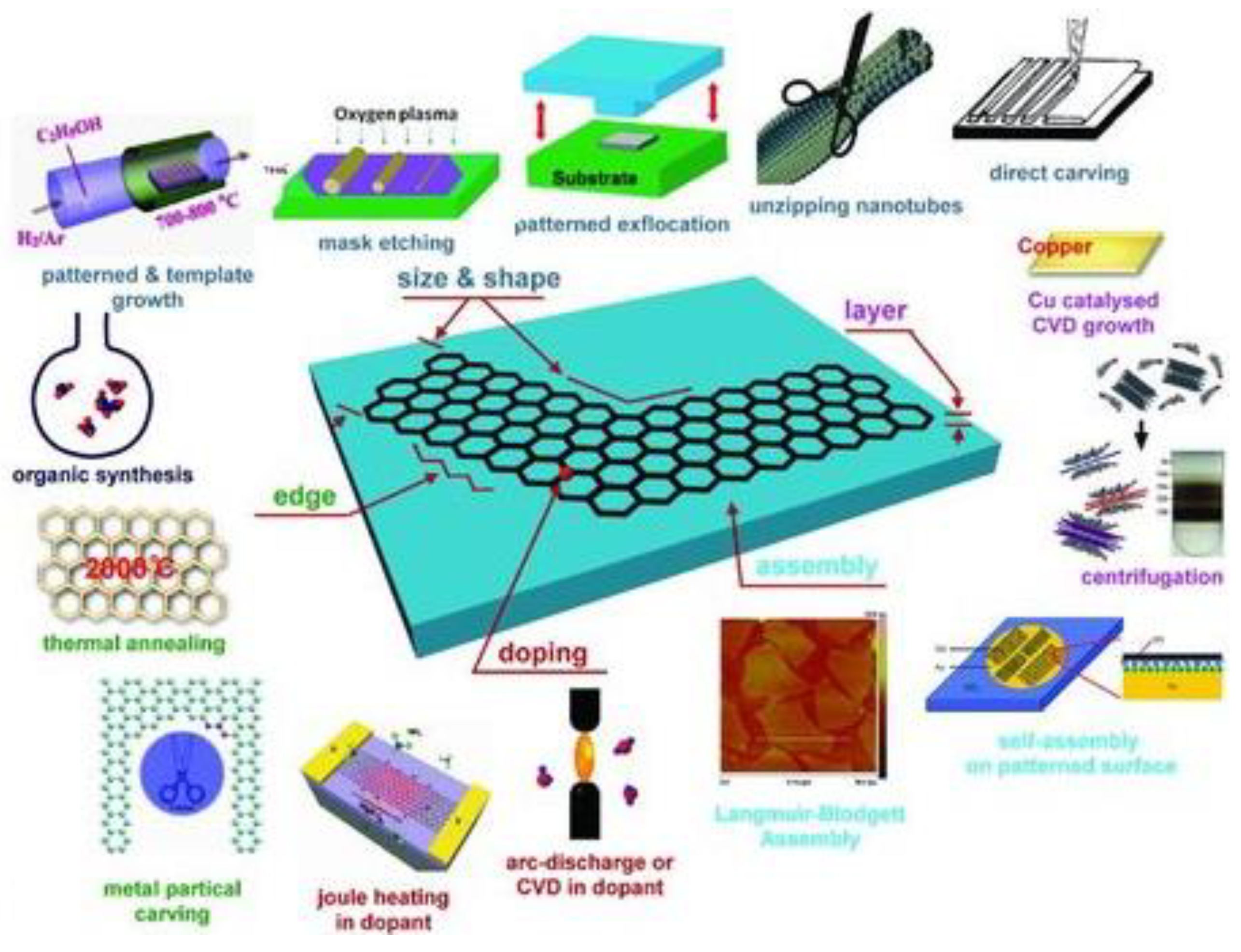
2.1. 2D Carbon-Based Nanostructures: Graphene, Graphynes, and Graphidyne
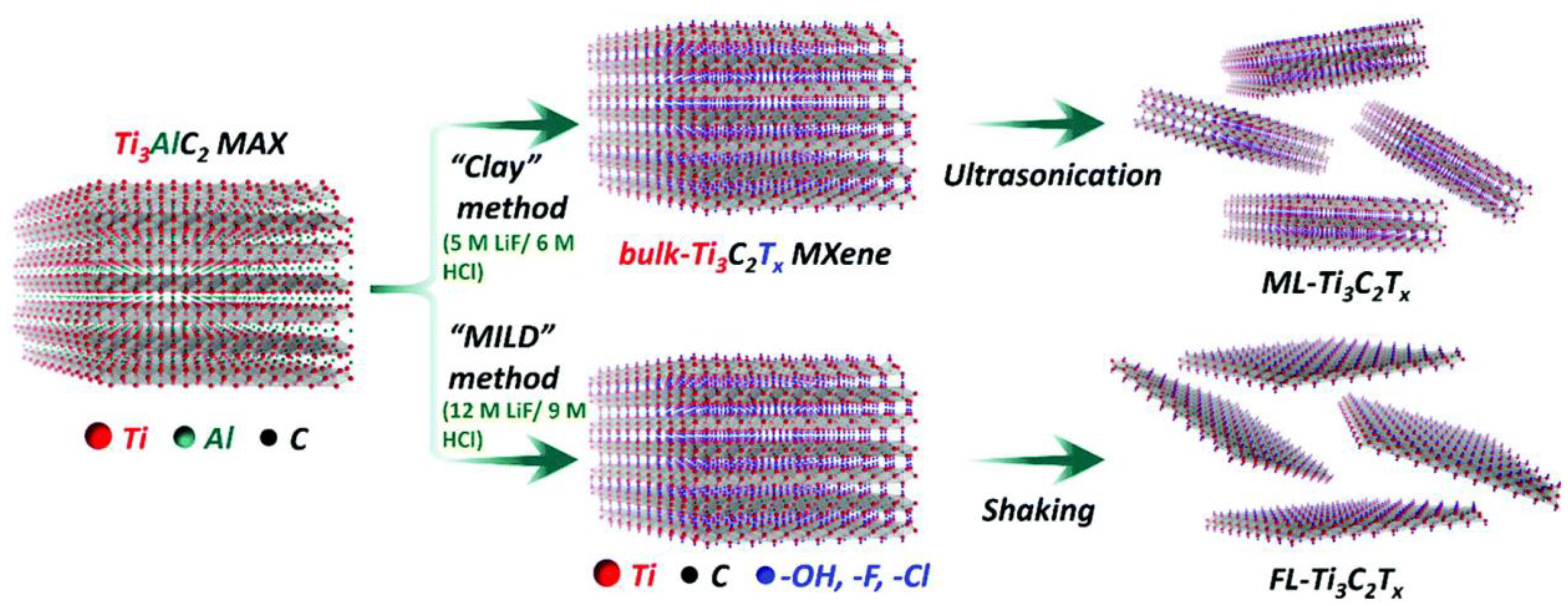
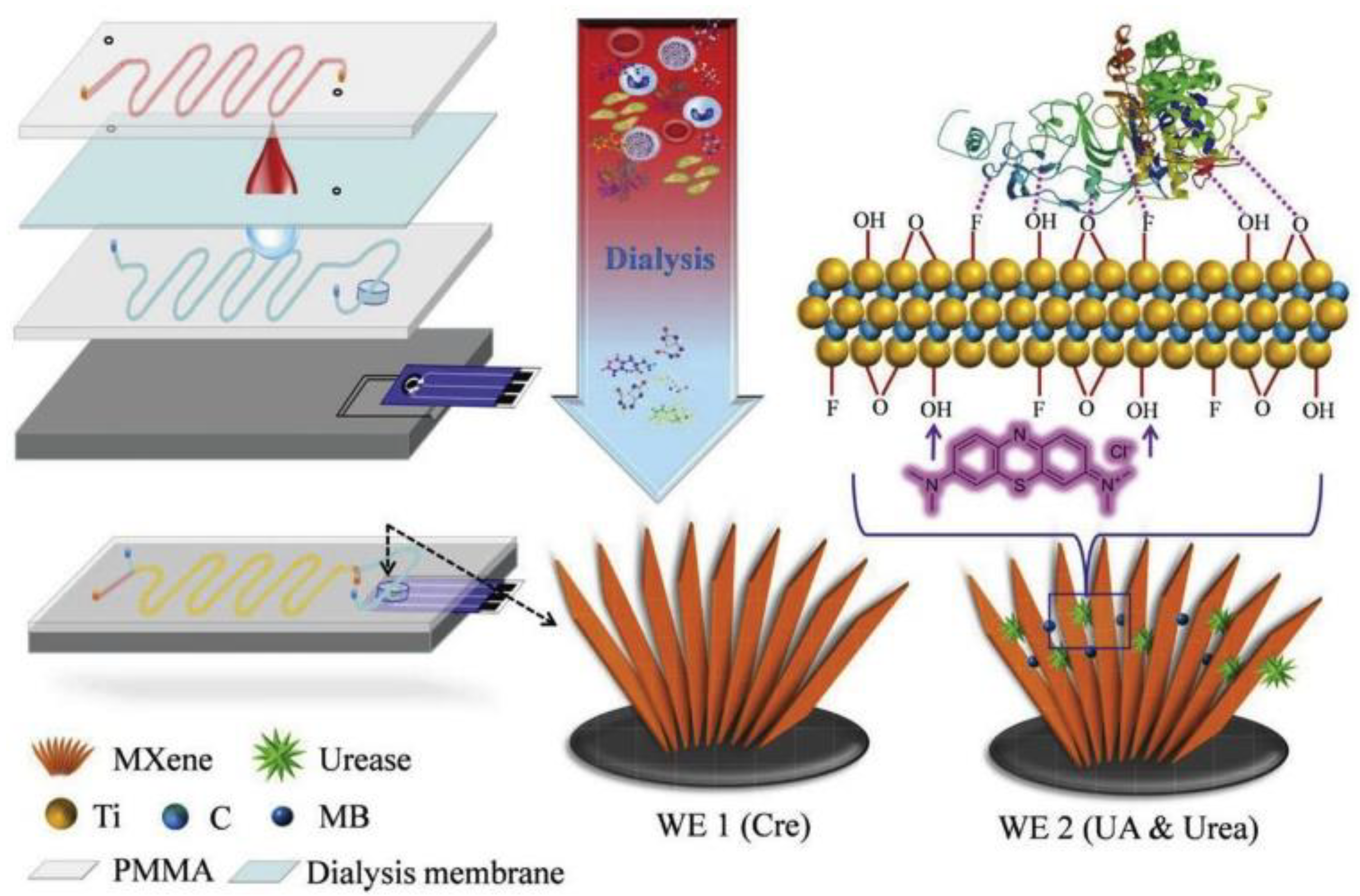
2.2. MXenes
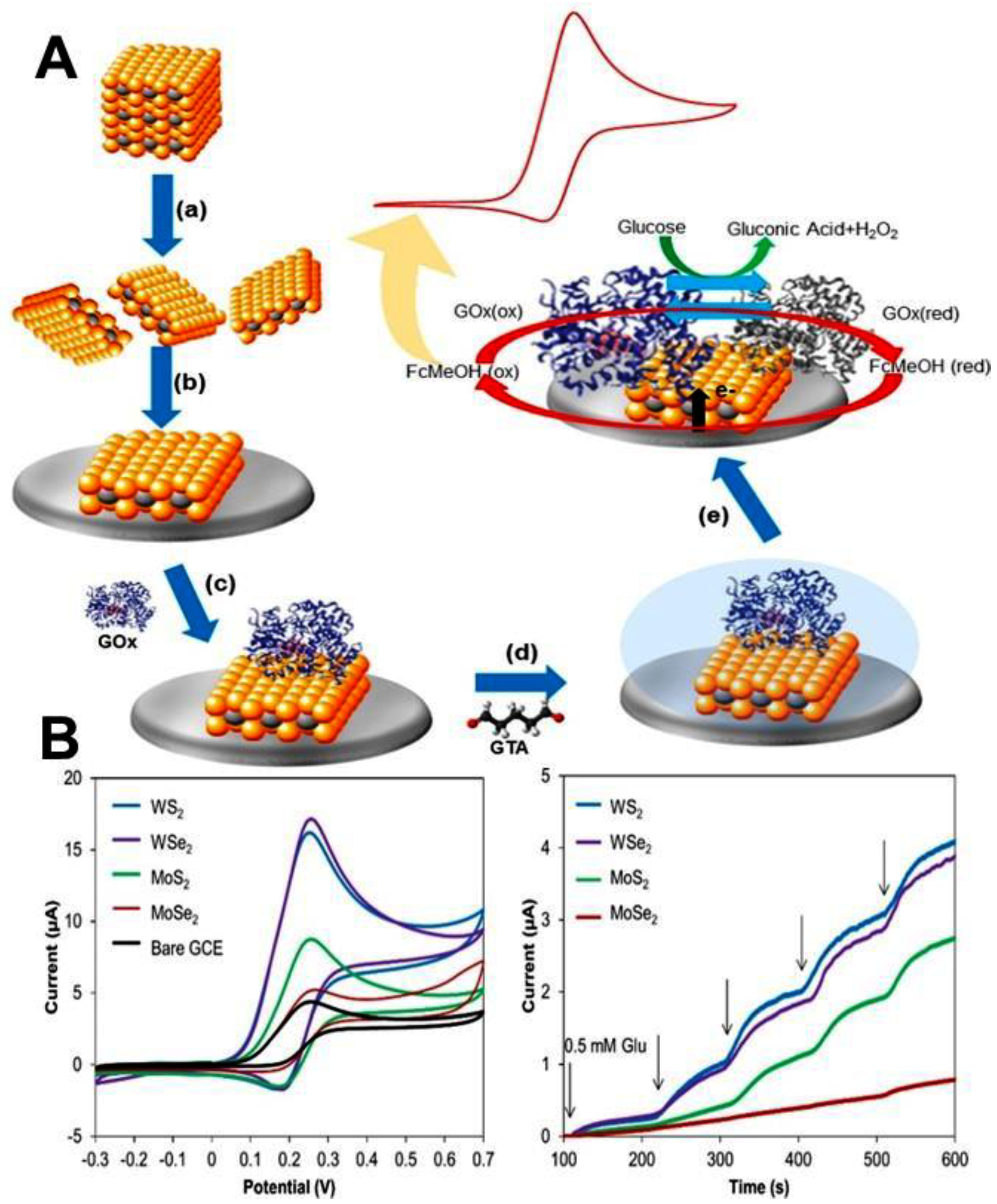
2.3. Transition Metal Dichalcogenides (TMDs)
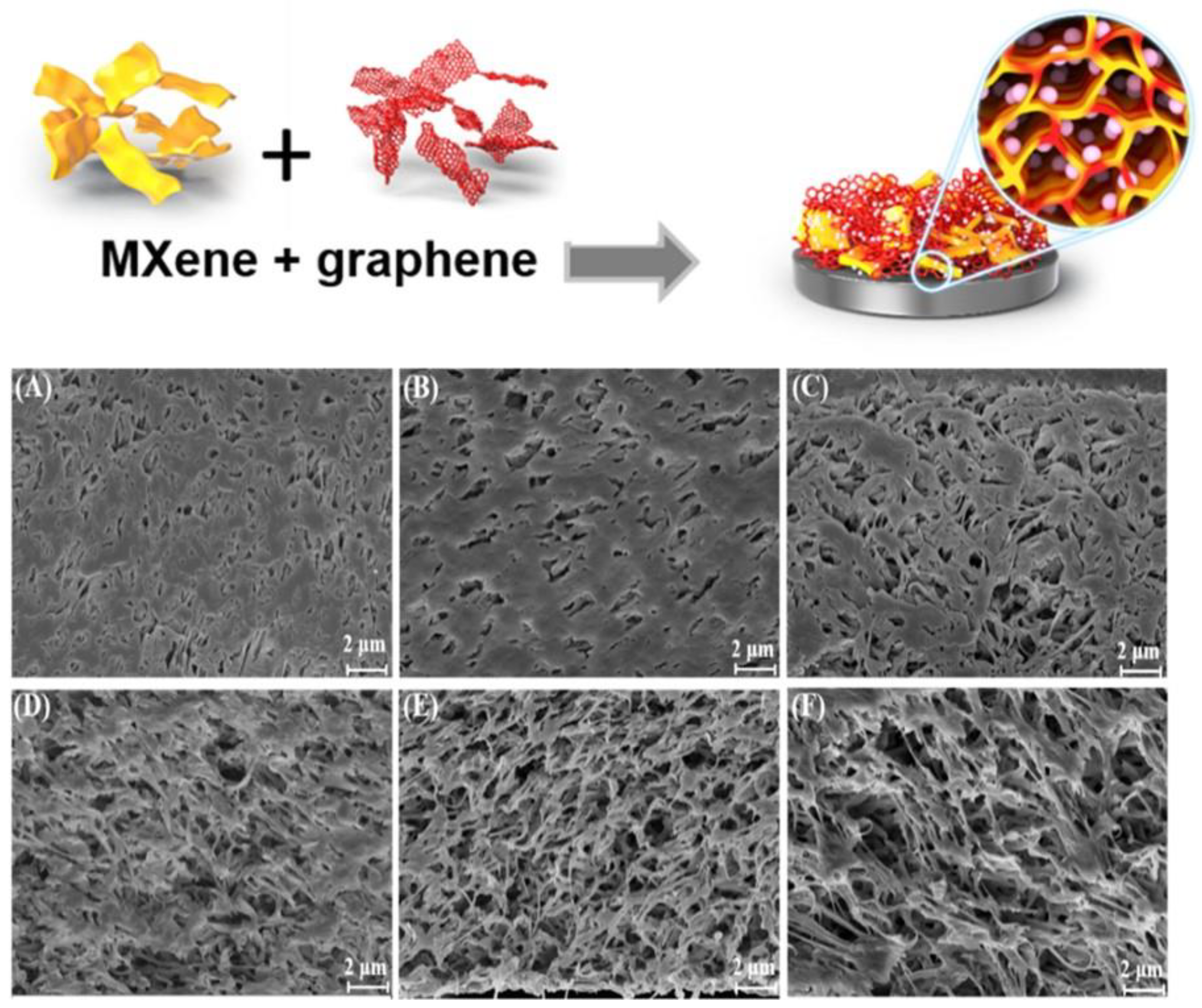
2.4. Hybrid Hierarchical Assemblies Based on Combined Heterogeneous Nanostructures
3. Role of 2D Nanomaterials in Electrochemical Sensing Platforms
3.1. Improvement in Electron Transfer Kinetics with 2D Nanomaterials
3.2. 2D Nanostructures as Electrode Material Modifier
3.3. 2D Nanostructures as an Electrochemically Active Label and Support for Electroactive Probes
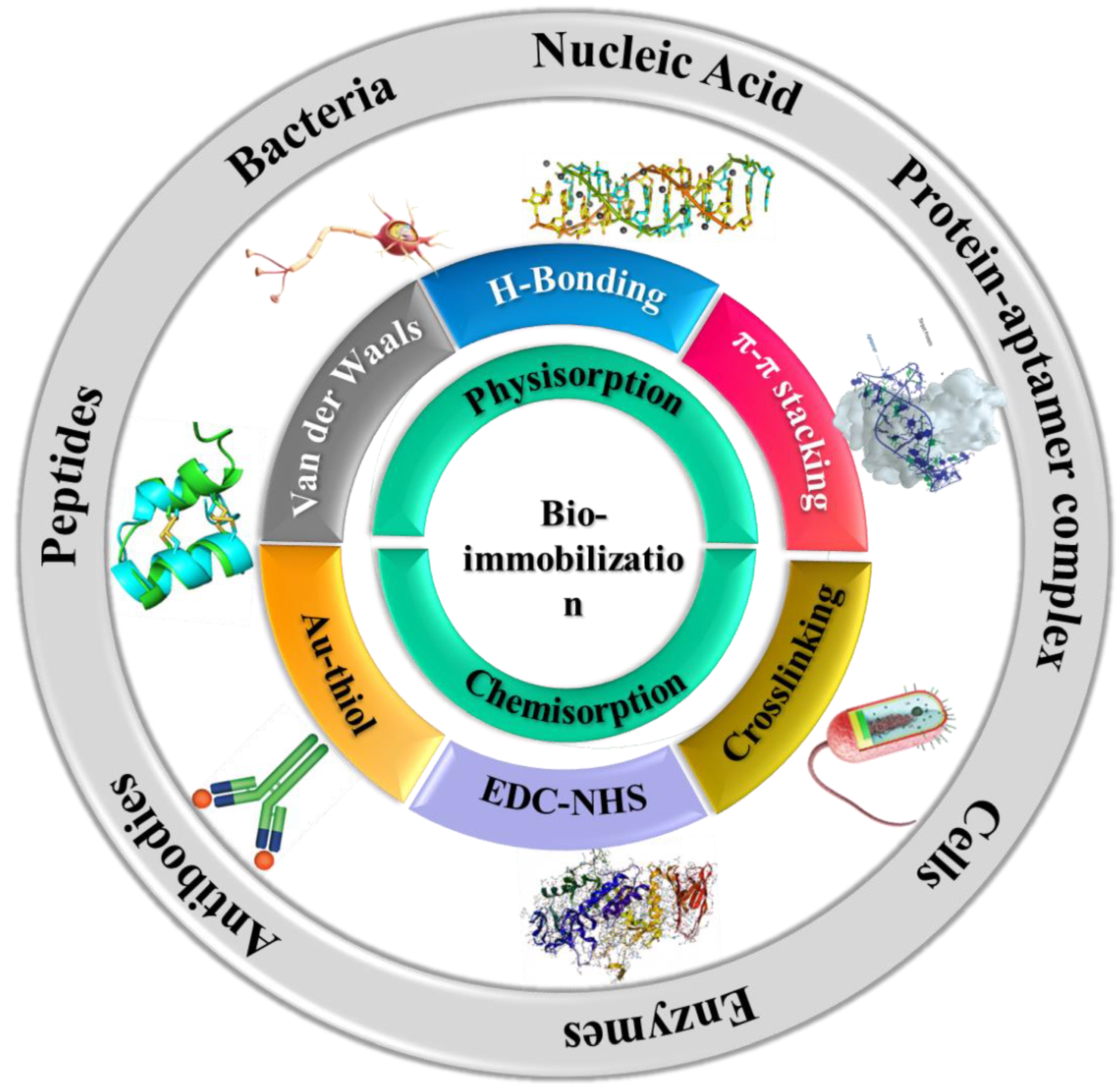
4. Integration of Biomolecules with 2D Nanostructures
4.1. Bacteriophages
4.2. Nucleic Acids
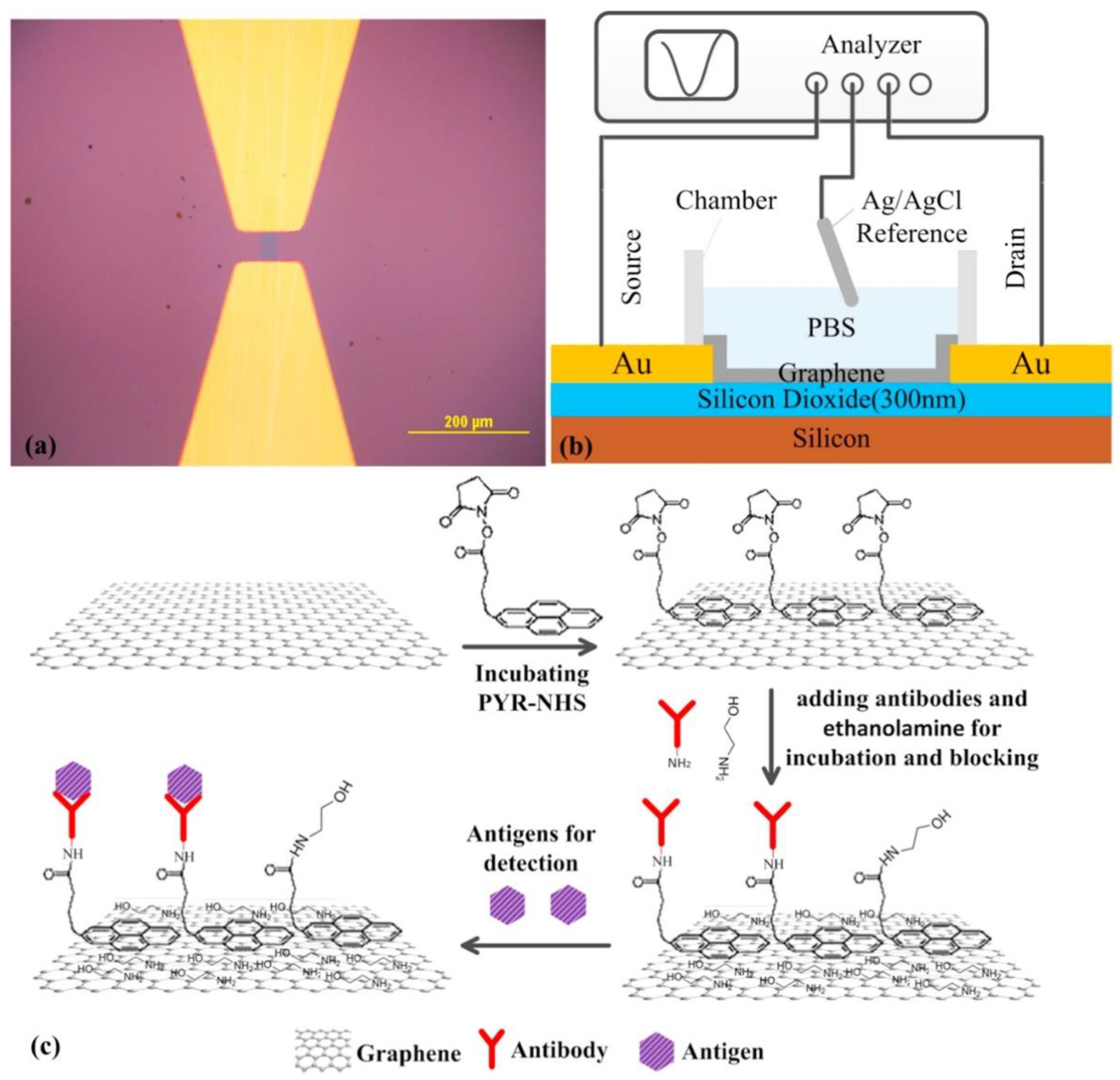
4.3. Antibodies
4.4. Peptides
4.5. Enzymes and Proteins
5. Biosensing Applications of 2D Nanomaterials for Food and Environmental Analysis
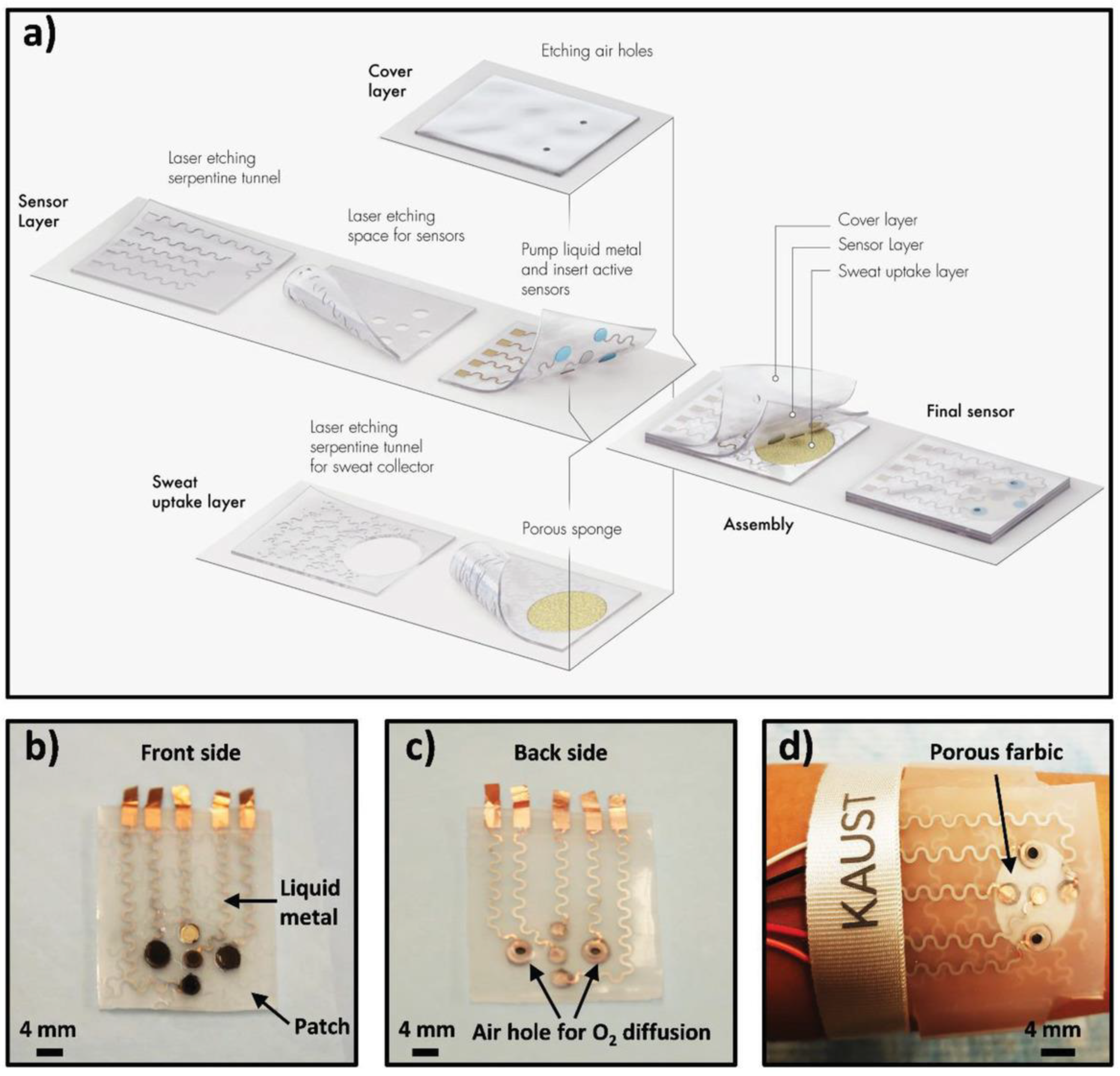
6. 2D-Based Biosensors for Wearable Devices
7. Conclusions and Future Outlook
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Lou, Z.; Wang, L.; Jiang, K.; Wei, Z.; Shen, G. Reviews of wearable healthcare systems: Materials, devices and system integration. Mater. Sci. Eng. R Rep. 2020, 140, 100523. [Google Scholar] [CrossRef]
- Wang, L.; Chen, D.; Jiang, K.; Shen, G. New insights and perspectives into biological materials for flexible electronics. Chem. Soc. Rev. 2017, 46, 6764–6815. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lou, Z.; Shen, G. 2D Nanomaterials with Hierarchical Architecture for Flexible Sensor Application. ACS Symp. Ser. 2020, 1353, 93–116. [Google Scholar]
- Keshavarz, M.; Chowdhury, A.R.H.; Kassanos, P.; Tan, B.; Venkatakrishnan, K. Self-assembled N-doped Q-dot carbon nanostructures as a SERS-active biosensor with selective therapeutic functionality. Sens. Actuators B Chem. 2020, 323, 128703. [Google Scholar] [CrossRef]
- Othman, A.; Karimi, A.; Andreescu, S. Functional nanostructures for enzyme based biosensors: Properties, fabrication and applications. J. Mater. Chem. B 2016, 4, 7178–7203. [Google Scholar] [CrossRef]
- Khan, R.; Sherazi, T.A.; Catanante, G.; Rasheed, S.; Marty, J.L.; Hayat, A. Switchable fluorescence sensor toward PAT via CA-MWCNTs quenched aptamer-tagged carboxyfluorescein. Food Chem. 2020, 312, 126048. [Google Scholar] [CrossRef]
- Glavin, N.R.; Rao, R.; Varshney, V.; Bianco, E.; Apte, A.; Roy, A.; Ringe, E.; Ajayan, P.M. Emerging Applications of Elemental 2D Materials. Adv. Mater. 2020, 32, 2070052. [Google Scholar] [CrossRef]
- Yuhara, J.; He, B.; Matsunami, N.; Nakatake, M.; le Lay, G. Graphene’s Latest Cousin: Plumbene Epitaxial Growth on a “Nano WaterCube”. Adv. Mater. 2019, 31, 1901017. [Google Scholar] [CrossRef]
- Wang, J.; Tang, J.; Ding, B.; Malgras, V.; Chang, Z.; Hao, X.; Wang, Y.; Dou, H.; Zhang, X.; Yamauchi, Y. Hierarchical porous carbons with layer-by-layer motif architectures from confined soft-template self-assembly in layered materials. Nat. Commun. 2017, 8, 15717. [Google Scholar] [CrossRef]
- Lee, W.-K.; Odom, T.W. Designing Hierarchical Nanostructures from Conformable and Deformable Thin Materials. ACS Nano 2019, 13, 6170–6177. [Google Scholar] [CrossRef] [PubMed]
- Egea, J.; Fabregat, I.; Frapart, Y.M.; Ghezzi, P.; Görlach, A.; Kietzmann, T.; Kubaichuk, K.; Knaus, U.G.; Lopez, M.G.; Olaso-Gonzalez, G.; et al. Corrigendum to European contribution to the study of ROS: A summary of the findings and prospects for the future from the COST action BM1203 (EU-ROS) [Redox Biol. 13 (2017) 94–162]. Redox Biol. 2018, 14, 694–696. [Google Scholar] [CrossRef]
- Cheng, M.M.-C.; Cuda, G.; Bunimovich, Y.L.; Gaspari, M.; Heath, J.R.; Hill, H.D.; Mirkin, C.A.; Nijdam, A.J.; Terracciano, R.; Thundat, T. Nanotechnologies for biomolecular detection and medical diagnostics. Curr. Opin. Chem. Biol. 2006, 10, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Park, S.; Yogeesh, M.N.; McNicholas, K.M.; Bank, S.R.; Akinwande, D. Black Phosphorus Flexible Thin Film Transistors at Gighertz Frequencies. Nano Lett. 2016, 16, 2301–2306. [Google Scholar] [CrossRef] [PubMed]
- Joshi, N.; Hayasaka, T.; Liu, Y.; Liu, H.; Oliveira, O.N.; Lin, L. A review on chemiresistive room temperature gas sensors based on metal oxide nanostructures, graphene and 2D transition metal dichalcogenides. Microchim. Acta 2018, 185, 213. [Google Scholar] [CrossRef]
- Gan, X.; Zhao, H.; Wong, K.-Y.; Lei, D.Y.; Zhang, Y.; Quan, X. Covalent functionalization of MoS2 nanosheets synthesized by liquid phase exfoliation to construct electrochemical sensors for Cd (II) detection. Talanta 2018, 182, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Lou, Z.; Wang, L.; Zhao, L.; Zhao, S.; Wang, D.; Han, W.; Jiang, K.; Shen, G. Bioinspired Interlocked Structure-Induced High Deformability for Two-Dimensional Titanium Carbide (MXene)/Natural Microcapsule-Based Flexible Pressure Sensors. ACS Nano 2019, 13, 9139–9147. [Google Scholar] [CrossRef]
- Cheng, L.; Wang, X.; Gong, F.; Liu, T.; Liu, Z. 2D Nanomaterials for Cancer Theranostic Applications. Adv. Mater. 2020, 32, e1902333. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, L.; Jiang, K.; Shen, G. Programmable three-dimensional advanced materials based on nanostructures as building blocks for flexible sensors. Nano Today 2019, 26, 176–198. [Google Scholar] [CrossRef]
- Yang, T.; Wang, W.; Huang, Y.; Jiang, X.; Zhao, X. Accurate Monitoring of Small Strain for Timbre Recognition via Ductile Fragmentation of Functionalized Graphene Multilayers. ACS Appl. Mater. Interfaces 2020, 12, 57352–57361. [Google Scholar] [CrossRef] [PubMed]
- Bolotsky, A.; Butler, D.; Dong, C.; Gerace, K.; Glavin, N.R.; Muratore, C.; Robinson, J.A.; Ebrahimi, A. Two-Dimensional Materials in Biosensing and Healthcare: From In Vitro Diagnostics to Optogenetics and Beyond. ACS Nano 2019, 13, 9781–9810. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Liu, H.; Wang, D.; Zhang, J. Graphdiyne: Synthesis, properties, and applications. Chem. Soc. Rev. 2019, 48, 908–936. [Google Scholar] [CrossRef]
- Khan, R.; Andreescu, S. MXenes-Based Bioanalytical Sensors: Design, Characterization, and Applications. Sensors 2020, 20, 5434. [Google Scholar] [CrossRef]
- Yanbing, Y.; Xiangdong, Y.; Yujie, Y.; Quan, Y. Aptamer-functionalized carbon nanomaterials electrochemical sensors for detecting cancer relevant biomolecules. Carbon 2018, 129, 380–395. [Google Scholar] [CrossRef]
- Gupta, A.; Sakthivel, T.; Seal, S. Recent development in 2D materials beyond graphene. Prog. Mater. Sci. 2015, 73, 44–126. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef]
- Yi, M.; Shen, Z. A review on mechanical exfoliation for the scalable production of graphene. J. Mater. Chem. A 2015, 3, 11700–11715. [Google Scholar] [CrossRef]
- Hernandez, Y.; Nicolosi, V.; Lotya, M.; Blighe, F.M.; Sun, Z.; De, S.; McGovern, I.T.; Holland, B.; Byrne, M.; Gun’Ko, Y.K.; et al. High-yield production of graphene by liquid-phase exfoliation of graphite. Nat. Nanotechnol. 2008, 3, 563–568. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, Y.; Abidi, N.; Cabrales, L. Wettability and Surface Free Energy of Graphene Films. Langmuir 2009, 25, 11078–11081. [Google Scholar] [CrossRef]
- Zhao, H.; Xu, B.; Ding, J.; Wang, Z.; Yu, H. Natural Amino Acids: High-Efficiency Intercalants for Graphene Exfoliation. ACS Sustain. Chem. Eng. 2019, 7, 18819–18825. [Google Scholar] [CrossRef]
- Wei, D.; Liu, Y. Controllable synthesis of graphene and its applications. Adv. Mater. 2010, 2010. 22, 3225–3241. [Google Scholar] [CrossRef]
- Suvarnaphaet, P.; Pechprasarn, S. Graphene-Based Materials for Biosensors: A Review. Sensors 2017, 17, 2161. [Google Scholar] [CrossRef] [PubMed]
- Peña-Bahamonde, J.; Nguyen, H.N.; Fanourakis, S.K.; Rodrigues, D.F. Recent advances in graphene-based biosensor technology with applications in life sciences. J. Nanobiotechnology 2018, 16, 1–17. [Google Scholar] [CrossRef]
- Hao, Y.; Fang, M.; Xu, C.; Ying, Z.; Wang, H.; Zhang, R.; Cheng, H.-M.; Zeng, Y. A graphene-laminated electrode with high glucose oxidase loading for highly-sensitive glucose detection. J. Mater. Sci. Technol. 2021, 66, 57–63. [Google Scholar] [CrossRef]
- Yoo, E.-H.; Lee, S.-Y. Glucose Biosensors: An Overview of Use in Clinical Practice. Sensors 2010, 10, 4558–4576. [Google Scholar] [CrossRef]
- Wu, L.; Gao, J.; Lu, X.; Huang, C.; Dhanjai; Chen, J. Graphdiyne: A new promising member of 2D all-carbon nanomaterial as robust electrochemical enzyme biosensor platform. Carbon 2020, 156, 568–575. [Google Scholar] [CrossRef]
- Deshmukh, K.; Kovářík, T.; Pasha, S.K. State of the art recent progress in two dimensional MXenes based gas sensors and biosensors: A comprehensive review. Co-Ord. Chem. Rev. 2020, 424, 213514. [Google Scholar] [CrossRef]
- Naguib, M.; Mashtalir, O.; Carle, J.; Presser, V.; Lu, J.; Hultman, L.; Gogotsi, Y.; Barsoum, M.W. Two-Dimensional Transition Metal Carbides. ACS Nano 2012, 6, 1322–1331. [Google Scholar] [CrossRef]
- Liu, F.; Zhou, J.; Wang, S.; Wang, B.; Shen, C.; Wang, L.; Hu, Q.; Huang, Q.; Zhou, A. Preparation of High-Purity V2C MXene and Electrochemical Properties as Li-Ion Batteries. J. Electrochem. Soc. 2017, 164, A709–A713. [Google Scholar] [CrossRef]
- Naguib, M.; Kurtoglu, M.; Presser, V.; Lu, J.; Niu, J.; Heon, M.; Hultman, L.; Gogotsi, Y.; Barsoum, M.W. Two-Dimensional Nanocrystals Produced by Exfoliation of Ti3AlC2. Adv. Mater. 2011, 23, 4248–4253. [Google Scholar] [CrossRef]
- Peng, J.; Chen, X.; Ong, W.-J.; Zhao, X.; Li, N. Surface and Heterointerface Engineering of 2D MXenes and Their Nanocomposites: Insights into Electro- and Photocatalysis. Chem 2019, 5, 18–50. [Google Scholar] [CrossRef]
- Liu, J.; Jiang, X.; Zhang, R.; Zhang, Y.; Wu, L.; Lu, W.; Li, J.; Li, Y.; Zhang, H. MXene-Enabled Electrochemical Microfluidic Biosensor: Applications toward Multicomponent Continuous Monitoring in Whole Blood. Adv. Funct. Mater. 2019, 29, 1807326. [Google Scholar] [CrossRef]
- Chia, H.L.; Mayorga-Martinez, C.C.; Antonatos, N.; Sofer, Z.; Gonzalez-Julian, J.J.; Webster, R.D.; Pumera, M. MXene Titanium Carbide-based Biosensor: Strong Dependence of Exfoliation Method on Performance. Anal. Chem. 2020, 92, 2452–2459. [Google Scholar] [CrossRef] [PubMed]
- Nayak, P.; Jiang, Q.; Mohanraman, R.; Anjum, D.H.; Hedhili, M.N.; Alshareef, H.N. Inherent electrochemistry and charge transfer properties of few-layered two-dimensional Ti3C2TxMXene. Nanoscale 2018, 10, 17030–17037. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Lei, Y.; Alshareef, N.H.; Quevedo-Lopez, M.; Salama, K.N. Biofunctionalized two-dimensional Ti3C2 MXenes for ultrasensitive detection of cancer biomarker. Biosens. Bioelectron. 2018, 121, 243–249. [Google Scholar] [CrossRef]
- Wang, H.; Li, H.; Huang, Y.; Xiong, M.; Wang, F.; Li, C. A label-free electrochemical biosensor for highly sensitive detection of gliotoxin based on DNA nanostructure/MXene nanocomplexes. Biosens. Bioelectron. 2019, 142, 111531. [Google Scholar] [CrossRef]
- Koyappayil, A.; Chavan, S.G.; Mohammadniaei, M.; Go, A.; Hwang, S.Y.; Lee, M.-H. β-Hydroxybutyrate dehydrogenase decorated MXene nanosheets for the amperometric determination of β-hydroxybutyrate. Microchim. Acta 2020, 187, 1–7. [Google Scholar] [CrossRef]
- Wu, L.; Lu, X.; Dhanjai; Wu, Z.-S.; Dong, Y.; Wang, X.; Zheng, S.; Chen, J. 2D transition metal carbide MXene as a robust biosensing platform for enzyme immobilization and ultrasensitive detection of phenol. Biosens. Bioelectron. 2018, 107, 69–75. [Google Scholar] [CrossRef]
- Manzeli, S.; Ovchinnikov, D.; Pasquier, D.; Yazyev, O.V.; Kis, A. 2D transition metal dichalcogenides. Nat. Rev. Mater. 2017, 2. [Google Scholar] [CrossRef]
- Hu, H.; Zavabeti, A.; Quan, H.; Zhu, W.; Wei, H.; Chen, D.; Ou, J.Z. Recent advances in two-dimensional transition metal dichalcogenides for biological sensing. Biosens. Bioelectron. 2019, 142, 111573. [Google Scholar] [CrossRef]
- Huang, K.-J.; Liu, Y.-J.; Zhang, J.-Z. Aptamer-based electrochemical assay of 17β-estradiol using a glassy carbon electrode modified with copper sulfide nanosheets and gold nanoparticles, and applying enzyme-based signal amplification. Microchim. Acta 2015, 182, 409–417. [Google Scholar] [CrossRef]
- Wang, Q.H.; Kalantar-Zadeh, K.; Kis, A.; Coleman, J.N.; Strano, M.S. Electronics and optoelectronics of two-dimensional transition metal dichalcogenides. Nat. Nanotechnol. 2012, 7, 699–712. [Google Scholar] [CrossRef]
- Geim, A.K.; Grigorieva, I.V. Van der Waals heterostructures. Nat. Cell Biol. 2013, 499, 419–425. [Google Scholar] [CrossRef]
- Chhowalla, M.; Liu, Z.; Zhang, H. Two-dimensional transition metal dichalcogenide (TMD) nanosheets. Chem. Soc. Rev. 2015, 44, 2584–2586. [Google Scholar] [CrossRef]
- Ganatra, R.; Zhang, Q. Few-Layer MoS2: A Promising Layered Semiconductor. ACS Nano 2014, 8, 4074–4099. [Google Scholar] [CrossRef] [PubMed]
- Heine, T. Transition Metal Chalcogenides: Ultrathin Inorganic Materials with Tunable Electronic Properties. Acc. Chem. Res. 2015, 48, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.J.; Koski, K.J.; Huang, K.C.Y.; Wang, K.X.; Luo, W.; Kong, D.; Yu, Z.; Fan, S.; Brongersma, M.L.; Cui, Y. Two-Dimensional Chalcogenide Nanoplates as Tunable Metamaterials via Chemical Intercalation. Nano Lett. 2013, 13, 5913–5918. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-H.; Huang, K.-J.; Wu, X. Recent advances in transition-metal dichalcogenides based electrochemical biosensors: A review. Biosens. Bioelectron. 2017, 97, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Jiang, D.; Schedin, F.; Booth, T.J.; Khotkevich, V.V.; Morozov, S.V.; Geim, A.K. Two-dimensional atomic crystals. Proc. Natl. Acad. Sci. USA 2005, 102, 10451–10453. [Google Scholar] [CrossRef]
- Matte, H.S.S.R.; Gomathi, A.; Manna, A.K.; Late, D.J.; Datta, R.; Pati, S.K.; Rao, C.N.R. MoS2 and WS2 Analogues of Graphene. Angew. Chem. Int. Ed. 2010, 49, 4059–4062. [Google Scholar] [CrossRef]
- Magda, G.Z.; Pető, J.; Dobrik, G.; Hwang, C.; Biró, L.P.; Tapasztó, L. Exfoliation of large-area transition metal chalcogenide single layers. Sci. Rep. 2015, 5, 14714. [Google Scholar] [CrossRef] [PubMed]
- Nicolosi, V.; Chhowalla, M.; Kanatzidis, M.G.; Strano, M.S.; Coleman, J.N. Liquid Exfoliation of Layered Materials. Science 2013, 340, 1226419. [Google Scholar] [CrossRef]
- Khan, U.; May, P.; O’Neill, A.; Bell, A.P.; Boussac, E.; Martin, A.; Semple, J.; Coleman, J.N. Polymer reinforcement using liquid-exfoliated boron nitride nanosheets. Nanoscale 2013, 5, 581–587. [Google Scholar] [CrossRef]
- Yu, X.-Y.; Hu, H.; Wang, Y.; Chen, H.; Lou, X.W. (David). Ultrathin MoS2Nanosheets Supported on N-doped Carbon Nanoboxes with Enhanced Lithium Storage and Electrocatalytic Properties. Angew. Chem. 2015, 127, 7503–7506. [Google Scholar] [CrossRef]
- Radisavljevic, B.; Radenovic, A.; Brivio, J.; Giacometti, V.; Kis, A. Single-layer MoS2 transistors. Nat. Nanotechnol. 2011, 6, 147–150. [Google Scholar] [CrossRef]
- Liu, N.; Kim, P.; Kim, J.H.; Ye, J.H.; Kim, S.; Lee, C.J. Large-Area Atomically Thin MoS2 Nanosheets Prepared Using Electrochemical Exfoliation. ACS Nano 2014, 8, 6902–6910. [Google Scholar] [CrossRef]
- Zeng, Z.; Yin, Z.; Huang, X.; Li, H.; He, Q.; Lu, G.; Boey, F.; Zhang, H. Single-Layer Semiconducting Nanosheets: High-Yield Preparation and Device Fabrication. Angew. Chem. Int. Ed. 2011, 50, 11093–11097. [Google Scholar] [CrossRef] [PubMed]
- Veeramalai, C.P.; Xu, H.; Guo, T.; Li, F.; Kim, T.W. One pot hydrothermal synthesis of graphene like MoS2 nanosheets for application in high performance lithium ion batteries. RCS Adv. 2015, 5, 57666–57670. [Google Scholar] [CrossRef]
- Li, X.; Zhu, H. Two-dimensional MoS2: Properties, preparation, and applications. J. Mater. 2015, 1, 33–44. [Google Scholar] [CrossRef]
- Eng, A.Y.S.; Ambrosi, A.; Sofer, Z.; Šimek, P.; Pumera, M. Electrochemistry of Transition Metal Dichalcogenides: Strong Dependence on the Metal-to-Chalcogen Composition and Exfoliation Method. ACS Nano 2014, 8, 12185–12198. [Google Scholar] [CrossRef]
- Rohaizad, N.; Mayorga-Martinez, C.C.; Sofer, Z.; Pumera, M. 1T-Phase Transition Metal Dichalcogenides (MoS2, MoSe2, WS2, and WSe2) with Fast Heterogeneous Electron Transfer: Application on Second-Generation Enzyme-Based Biosensor. ACS Appl. Mater. Interfaces 2017, 9, 40697–40706. [Google Scholar] [CrossRef] [PubMed]
- Riazi, H.; Taghizadeh, G.; Soroush, M. MXene-Based Nanocomposite Sensors. ACS Omega 2021, 6, 11103–11112. [Google Scholar] [CrossRef]
- Yin, P.T.; Kim, T.-H.; Choi, J.-W.; Lee, K.-B. Prospects for graphene–nanoparticle-based hybrid sensors. Phys. Chem. Chem. Phys. 2013, 15, 12785–12799. [Google Scholar] [CrossRef]
- Artiles, M.S.; Rout, C.S.; Fisher, T.S. Graphene-based hybrid materials and devices for biosensing. Adv. Drug Deliv. Rev. 2011, 63, 1352–1360. [Google Scholar] [CrossRef]
- Ren, Z.; Guo, Y.; Liu, C.-H.; Gao, P.-X. Hierarchically nanostructured materials for sustainable environmental applications. Front. Chem. 2013, 1. [Google Scholar] [CrossRef]
- Zhang, S.; Geryak, R.; Geldmeier, J.; Kim, S.; Tsukruk, V.V. Synthesis, Assembly, and Applications of Hybrid Nanostructures for Biosensing. Chem. Rev. 2017, 117, 12942–13038. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Zhang, X.; Sun, Y.; Yang, S.; Song, X.; Yang, Z. Hierarchical CuO nanoflowers: Water-required synthesis and their application in a nonenzymatic glucose biosensor. Phys. Chem. Chem. Phys. 2013, 15, 10904–10913. [Google Scholar] [CrossRef]
- Cheng, S.; Dela-Cruz, S.; Chen, C.; Tang, Z.; Shi, T.; Carraro, C.; Maboudian, R. Hierarchical Co3O4/CuO nanorod array supported on carbon cloth for highly sensitive non-enzymatic glucose biosensing. Sens. Actuators B Chem. 2019, 298, 126860. [Google Scholar] [CrossRef]
- Tran, T.D.; Kim, M.I. Organic-Inorganic Hybrid Nanoflowers as Potent Materials for Biosensing and Biocatalytic Applications. Biochip J. 2018, 12, 268–279. [Google Scholar] [CrossRef]
- Gu, H.; Xing, Y.; Xiong, P.; Tang, H.; Li, C.; Chen, S.; Zeng, R.; Han, K.; Shi, G. Three-Dimensional Porous Ti3C2Tx MXene–Graphene Hybrid Films for Glucose Biosensing. ACS Appl. Nano Mater. 2019, 2, 6537–6545. [Google Scholar] [CrossRef]
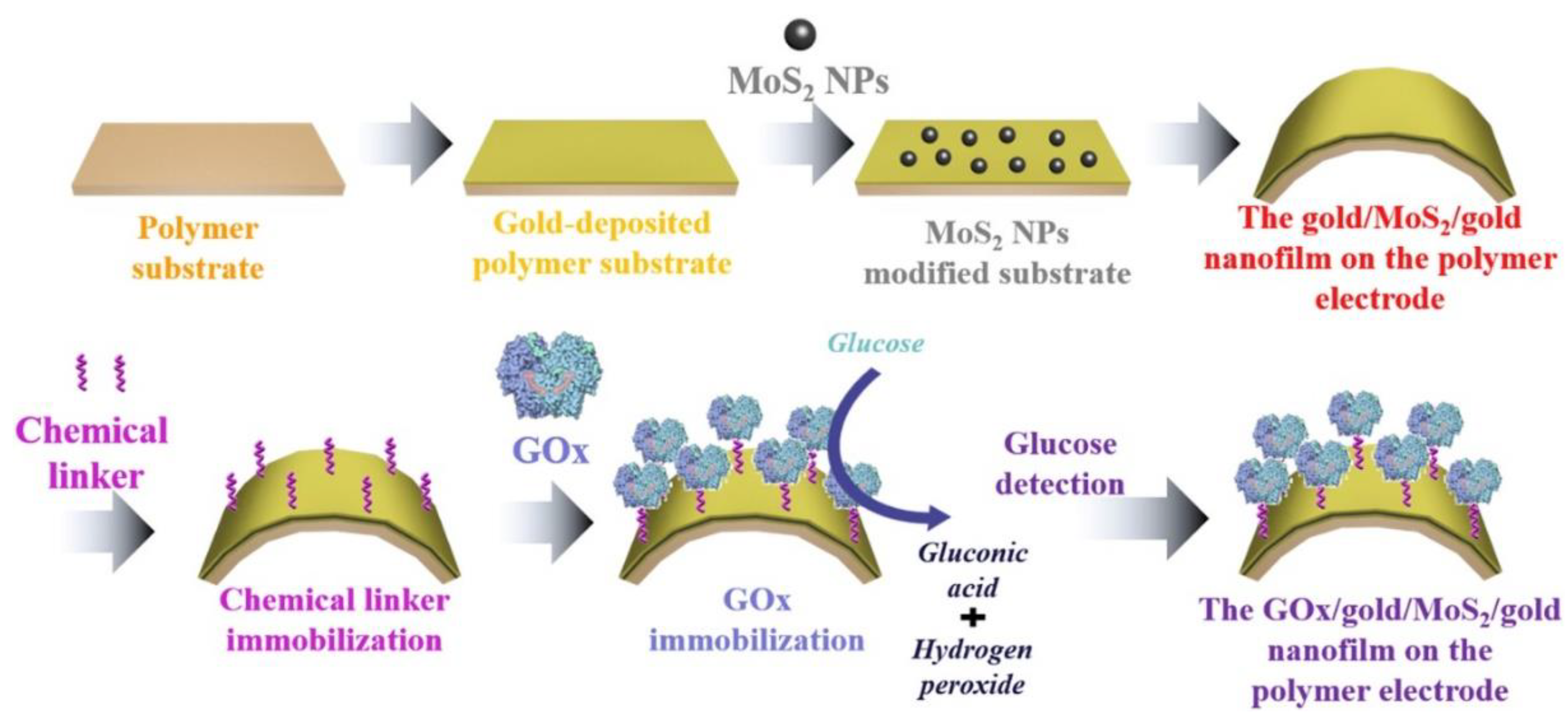
- Yoon, J.; Lee, S.N.; Shin, M.K.; Kim, H.-W.; Choi, H.K.; Lee, T.; Choi, J.-W. Flexible electrochemical glucose biosensor based on GOx/gold/MoS2/gold nanofilm on the polymer electrode. Biosens. Bioelectron. 2019, 140, 111343. [Google Scholar] [CrossRef]
- Wang, K.; Li, H.-N.; Wu, J.; Ju, C.; Yan, J.-J.; Liu, Q.; Qiu, B. TiO2-decorated graphene nanohybrids for fabricating an amperometric acetylcholinesterase biosensor. Analyst 2011, 136, 3349–3354. [Google Scholar] [CrossRef]
- Wang, F.; Yang, C.; Duan, M.; Tang, Y.; Zhu, J. TiO2 nanoparticle modified organ-like Ti3C2 MXene nanocomposite encapsulating hemoglobin for a mediator-free biosensor with excellent performances. Biosens. Bioelectron. 2015, 74, 1022–1028. [Google Scholar] [CrossRef]
- Zhang, J.-J.; Dong, S. Superconductivity of monolayer Mo2C: The key role of functional groups. J. Chem. Phys. 2017, 146, 034705. [Google Scholar] [CrossRef]
- Velický, M.; Bissett, M.A.; Toth, P.S.; Patten, H.V.; Worrall, S.D.; Rodgers, A.N.J.; Hill, E.W.; Kinloch, I.A.; Novoselov, K.S.; Georgiou, T.; et al. Electron transfer kinetics on natural crystals of MoS2 and graphite. Phys. Chem. Chem. Phys. 2015, 17, 17844–17853. [Google Scholar] [CrossRef]
- Li, W.; Tan, C.; Lowe, M.A.; Abruña, H.D.; Ralph, D.C. Electrochemistry of Individual Monolayer Graphene Sheets. ACS Nano 2011, 5, 2264–2270. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Yan, H.; Wu, F.; Zhao, L.; Yu, P.; Liu, H.; Li, Y.; Mao, L. Graphdiyne as Electrode Material: Tuning Electronic State and Surface Chemistry for Improved Electrode Reactivity. Anal. Chem. 2017, 89, 13008–13015. [Google Scholar] [CrossRef]
- Srivastava, S.; Abraham, S.; Singh, C.; Ali, A.; Srivastava, A.; Sumana, G.; Malhotra, B.D. Protein conjugated carboxylated gold@reduced graphene oxide for aflatoxin B1 detection. RCS Adv. 2014, 5, 5406–5414. [Google Scholar] [CrossRef]
- Srivastava, S.; Kumar, V.; Ali, A.; Solanki, P.R.; Srivastava, A.; Sumana, G.; Saxena, P.S.; Joshi, A.G.; Malhotra, B.D. Electrophoretically deposited reduced graphene oxide platform for food toxin detection. Nanoscale 2013, 5, 3043–3051. [Google Scholar] [CrossRef]
- Srivastava, S.; Kumar, V.; Arora, K.; Singh, C.; Ali, A.; Puri, N.K.; Malhotra, B.D. Antibody conjugated metal nanoparticle decorated graphene sheets for a mycotoxin sensor. RCS Adv. 2016, 6, 56518–56526. [Google Scholar] [CrossRef]
- Loo, A.H.; Bonanni, A.; Pumera, M. Mycotoxin Aptasensing Amplification by using Inherently Electroactive Graphene-Oxide Nanoplatelet Labels. ChemElectroChem 2015, 2, 743–747. [Google Scholar] [CrossRef]
- Beheshti-Marnani, A.; Hatefi-Mehrjardi, A.; Es’Haghi, Z. A sensitive biosensing method for detecting of ultra-trace amounts of AFB1 based on “Aptamer/reduced graphene oxide” nano-bio interaction. Colloids Surf. B Biointerfaces 2019, 175, 98–105. [Google Scholar] [CrossRef]
- Wang, Y.; Ning, G.; Bi, H.; Wu, Y.; Liu, G.; Zhao, Y. A novel ratiometric electrochemical assay for ochratoxin A coupling Au nanoparticles decorated MoS2 nanosheets with aptamer. Electrochim. Acta 2018, 285, 120–127. [Google Scholar] [CrossRef]
- Zhou, L.; Mao, H.; Wu, C.; Tang, L.; Wu, Z.; Sun, H.; Zhang, H.; Zhou, H.; Jia, C.; Jin, Q.; et al. Label-free graphene biosensor targeting cancer molecules based on non-covalent modification. Biosens. Bioelectron. 2017, 87, 701–707. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Li, Y.-T.; Zhang, H.; Xiao, M.-M.; Ning, Y.; Zhang, Z.-Y.; Zhang, G.-J. Molybdenum disulfide field-effect transistor biosensor for ultrasensitive detection of DNA by employing morpholino as probe. Biosens. Bioelectron. 2018, 110, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.W.; Kang, D.-H.; Cho, J.H.; Lee, S.; Jun, D.-H.; Park, J.-H. Highly Sensitive and Reusable Membraneless Field-Effect Transistor (FET)-Type Tungsten Diselenide (WSe2) Biosensors. ACS Appl. Mater. Interfaces 2018, 10, 17639–17645. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Bhardwaj, S.K.; Mehta, J.; Mohanta, G.C.; Deep, A. Bacteriophage immobilized graphene electrodes for impedimetric sensing of bacteria (Staphylococcus arlettae). Anal. Biochem. 2016, 505, 18–25. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Yan, X. An amperometric zearalenone aptasensor based on signal amplification by using a composite prepared from porous platinum nanotubes, gold nanoparticles and thionine-labelled graphene oxide. Microchim. Acta 2019, 186, 383. [Google Scholar] [CrossRef]
- Qian, J.; Jiang, L.; Yang, X.; Yan, Y.; Mao, H.; Wang, K. Highly sensitive impedimetric aptasensor based on covalent binding of gold nanoparticles on reduced graphene oxide with good dispersity and high density. Analyst 2014, 139, 5587–5593. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Li, X.; Cui, F.; Qiu, Q.; Chen, X.; Huang, H. Aflatoxin B1 Electrochemical Aptasensor Based on Tetrahedral DNA Nanostructures Functionalized Three Dimensionally Ordered Macroporous MoS2–AuNPs Film. ACS Appl. Mater. Interfaces 2018, 10, 17551–17559. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, J.; Li, D.; Li, Y. An electrochemical aptasensor based on gold nanoparticles dotted graphene modified glassy carbon electrode for label-free detection of bisphenol A in milk samples. Food Chem. 2014, 162, 34–40. [Google Scholar] [CrossRef]
- Huang, K.-J.; Liu, Y.-J.; Zhang, J.-Z.; Liu, Y.-M. A novel aptamer sensor based on layered tungsten disulfide nanosheets and Au nanoparticles amplification for 17β-estradiol detection. Anal. Methods 2014, 6, 8011–8017. [Google Scholar] [CrossRef]
- Shi, Z.-Y.; Zheng, Y.-T.; Zhang, H.-B.; He, C.-H.; Wu, W.-D.; Zhang, H.-B. DNA Electrochemical Aptasensor for Detecting Fumonisins B1Based on Graphene and Thionine Nanocomposite. Electroanalysis 2015, 27, 1097–1103. [Google Scholar] [CrossRef]
- Jiang, L.; Qian, J.; Yang, X.; Yan, Y.; Liu, Q.; Wang, K.; Wang, K. Amplified impedimetric aptasensor based on gold nanoparticles covalently bound graphene sheet for the picomolar detection of ochratoxin A. Anal. Chim. Acta 2014, 806, 128–135. [Google Scholar] [CrossRef]
- Tang, J.; Huang, Y.; Cheng, Y.; Huang, L.; Zhuang, J.; Tang, D. Two-dimensional MoS2 as a nano-binder for ssDNA: Ultrasensitive aptamer based amperometric detection of Ochratoxin A. Microchim. Acta 2018, 185, 162. [Google Scholar] [CrossRef] [PubMed]
- Kaur, N.; Bharti, A.; Batra, S.; Rana, S.; Rana, S.; Bhalla, A.; Prabhakar, N. An electrochemical aptasensor based on graphene doped chitosan nanocomposites for determination of Ochratoxin A. Microchem. J. 2019, 144, 102–109. [Google Scholar] [CrossRef]
- Dai, G.; Li, Z.; Luo, F.; Ai, S.; Chen, B.; Wang, Q. Electrochemical determination of Salmonella typhimurium by using aptamer-loaded gold nanoparticles and a composite prepared from a metal-organic framework (type UiO-67) and graphene. Microchim. Acta 2019, 186, 620. [Google Scholar] [CrossRef] [PubMed]
- Eissa, S.; Ng, A.; Siaj, M.; Zourob, M. Label-Free Voltammetric Aptasensor for the Sensitive Detection of Microcystin-LR Using Graphene-Modified Electrodes. Anal. Chem. 2014, 86, 7551–7557. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Jiang, D.; Dai, L.; Zhou, L.; Hao, N.; Qian, J.; Qiu, B.; Wang, K. Fabricating photoelectrochemical aptasensor for selectively monitoring microcystin-LR residues in fish based on visible light-responsive BiOBr nanoflakes/N-doped graphene photoelectrode. Biosens. Bioelectron. 2016, 81, 242–248. [Google Scholar] [CrossRef]
- Zhao, Z.; Chen, H.; Ma, L.; Liu, D.; Wang, Z. A label-free electrochemical impedance aptasensor for cylindrospermopsin detection based on thionine–graphene nanocomposites. Analyst 2015, 140, 5570–5577. [Google Scholar] [CrossRef]
- Bulbul, G.; Hayat, A.; Andreescu, S. A generic amplification strategy for electrochemical aptasensors using a non-enzymatic nanoceria tag. Nanoscale 2015, 7, 13230–13238. [Google Scholar] [CrossRef]
- Goud, K.Y.; Hayat, A.; Catanante, G.; Satyanarayana, M.; Gobi, K.V.; Marty, J.L. An electrochemical aptasensor based on functionalized graphene oxide assisted electrocatalytic signal amplification of methylene blue for aflatoxin B1 detection. Electrochim. Acta 2017, 244, 96–103. [Google Scholar] [CrossRef]
- Lin, T.; Shen, Y. Fabricating electrochemical aptasensors for detecting aflatoxin B1 via layer-by-layer self-assembly. J. Electroanal. Chem. 2020, 870, 114247. [Google Scholar] [CrossRef]
- Shu, J.; Qiu, Z.; Wei, Q.; Zhuang, J.; Tang, D. Cobalt-Porphyrin-Platinum-Functionalized Reduced Graphene Oxide Hybrid Nanostructures: A Novel Peroxidase Mimetic System for Improved Electrochemical Immunoassay. Sci. Rep. 2015, 5, 15113. [Google Scholar] [CrossRef]
- Zhang, W.; Jia, B.; Furumai, H. Fabrication of graphene film composite electrochemical biosensor as a pre-screening algal toxin detection tool in the event of water contamination. Sci. Rep. 2018, 8, 10686. [Google Scholar] [CrossRef] [PubMed]
- Eissa, S.; L’Hocine, L.; Siaj, M.; Zourob, M. A graphene-based label-free voltammetric immunosensor for sensitive detection of the egg allergen ovalbumin. Analyst 2013, 138, 4378–4384. [Google Scholar] [CrossRef]
- Chan, C.-Y.; Guo, J.; Sun, C.; Tsang, M.-K.; Tian, F.; Hao, J.; Chen, S.; Yang, M. A reduced graphene oxide-Au based electrochemical biosensor for ultrasensitive detection of enzymatic activity of botulinum neurotoxin A. Sens. Actuators B Chem. 2015, 220, 131–137. [Google Scholar] [CrossRef]
- Antón-Millán, N.; Garcia-Tojal, J.; Marty-Roda, M.; Garroni, S.; Cuesta-López, S.; Tamayo-Ramos, J.A. Influence of Three Commercial Graphene Derivatives on the Catalytic Properties of a Lactobacillus plantarum α-l-Rhamnosidase When Used as Immobilization Matrices. ACS Appl. Mater. Interfaces 2018, 10, 18170–18182. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Deng, D.; Jin, J.; Lu, X.; Chen, J. Nanographene-based tyrosinase biosensor for rapid detection of bisphenol A. Biosens. Bioelectron. 2012, 35, 193–199. [Google Scholar] [CrossRef]
- Rakhi, R.B.; Nayak, P.; Xia, C.; Alshareef, H.N. Novel amperometric glucose biosensor based on MXene nanocomposite. Sci. Rep. 2016, 6, 36422. [Google Scholar] [CrossRef]
- Xu, Q.; Cai, L.; Zhao, H.; Tang, J.; Shen, Y.; Hu, X.; Zeng, H. Forchlorfenuron detection based on its inhibitory effect towards catalase immobilized on boron nitride substrate. Biosens. Bioelectron. 2015, 63, 294–300. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, Y.-H.; Zhuge, Z.; Tang, Y.-H.; Tao, J.-W.; Chen, Y. Synthesis of a Poly-l-Lysine/Black Phosphorus Hybrid for Biosensors. Anal. Chem. 2018, 90, 3149–3155. [Google Scholar] [CrossRef]
- Jin, D.; Gong, A.; Zhou, H. Visible-light-activated photoelectrochemical biosensor for the detection of the pesticide acetochlor in vegetables and fruit based on its inhibition of glucose oxidase. RCS Adv. 2017, 7, 17489–17496. [Google Scholar] [CrossRef]
- Mayorga-Martinez, C.C.; Sofer, Z.; Pumera, M. Binary Phosphorene Redox Behavior in Oxidoreductase Enzymatic Systems. ACS Nano 2019, 13, 13217–13224. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Ni, Y.; Kokot, S. Investigations of an electrochemical platform based on the layered MoS2–graphene and horseradish peroxidase nanocomposite for direct electrochemistry and electrocatalysis. Biosens. Bioelectron. 2014, 56, 137–143. [Google Scholar] [CrossRef]
- Vasilescu, I.; Eremia, S.A.; Kusko, M.; Radoi, A.; Vasile, E.; Radu, G.-L. Molybdenum disulphide and graphene quantum dots as electrode modifiers for laccase biosensor. Biosens. Bioelectron. 2016, 75, 232–237. [Google Scholar] [CrossRef]
- Albelda, J.A.; Uzunoglu, A.; Santos, G.N.C.; Stanciu, L.A. Graphene-titanium dioxide nanocomposite based hypoxanthine sensor for assessment of meat freshness. Biosens. Bioelectron. 2017, 89, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Nasir, M.Z.M.; Mayorga-Martinez, C.C.; Sofer, Z.; Pumera, M. Two-Dimensional 1T-Phase Transition Metal Dichalcogenides as Nanocarriers To Enhance and Stabilize Enzyme Activity for Electrochemical Pesticide Detection. ACS Nano 2017, 11, 5774–5784. [Google Scholar] [CrossRef]
- Sharma, A.; Rao, V.K.; Kamboj, D.V.; Gaur, R.; Shaik, M.; Shrivastava, A.R. Enzyme free detection of staphylococcal enterotoxin B (SEB) using ferrocene carboxylic acid labeled monoclonal antibodies: An electrochemical approach. New J. Chem. 2016, 40, 8334–8341. [Google Scholar] [CrossRef]
- Nodoushan, S.M.; Nasirizadeh, N.; Amani, J.; Halabian, R.; Fooladi, A.A.I. An electrochemical aptasensor for staphylococcal enterotoxin B detection based on reduced graphene oxide and gold nano-urchins. Biosens. Bioelectron. 2019, 127, 221–228. [Google Scholar] [CrossRef]
- Narayanan, J.; Sharma, M.K.; Ponmariappan, S.; Sarita; Shaik, M.; Upadhyay, S. Electrochemical immunosensor for botulinum neurotoxin type-E using covalently ordered graphene nanosheets modified electrodes and gold nanoparticles-enzyme conjugate. Biosens. Bioelectron. 2015, 69, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Appaturi, J.N.; Pulingam, T.; Thong, K.L.; Muniandy, S.; Ahmad, N.; Leo, B.F. Rapid and sensitive detection of Salmonella with reduced graphene oxide-carbon nanotube based electrochemical aptasensor. Anal. Biochem. 2020, 589, 113489. [Google Scholar] [CrossRef] [PubMed]
- Soares, R.R.A.; Hjort, R.G.; Pola, C.C.; Parate, K.; Reis, E.L.; Soares, N.F.F.; McLamore, E.S.; Claussen, J.C.; Gomes, C.L. Laser-Induced Graphene Electrochemical Immunosensors for Rapid and Label-Free Monitoring of Salmonella enterica in Chicken Broth. ACS Sens. 2020, 5, 1900–1911. [Google Scholar] [CrossRef]
- Afkhami, A.; Hashemi, P.; Bagheri, H.; Salimian, J.; Ahmadi, A.; Madrakian, T. Impedimetric immunosensor for the label-free and direct detection of botulinum neurotoxin serotype A using Au nanoparticles/graphene-chitosan composite. Biosens. Bioelectron. 2017, 93, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, Y.; Zhang, X.; Gao, Y.; Sun, C.; Ding, Y.; Feng, F.; Jin, W.; Yang, G. An impedimetric immunosensor for determination of porcine epidemic diarrhea virus based on the nanocomposite consisting of molybdenum disulfide/reduced graphene oxide decorated with gold nanoparticles. Microchim. Acta 2020, 187, 217–219. [Google Scholar] [CrossRef]
- Kaur, H.; Shorie, M.; Sharma, M.; Ganguli, A.K.; Sabherwal, P. Bridged Rebar Graphene functionalized aptasensor for pathogenic E. coli O78:K80:H11 detection. Biosens. Bioelectron. 2017, 98, 486–493. [Google Scholar] [CrossRef]
- Thakur, B.; Zhou, G.; Chang, J.; Pu, H.; Jin, B.; Sui, X.; Yuan, X.; Yang, C.-H.; Magruder, M.; Chen, J. Rapid detection of single E. coli bacteria using a graphene-based field-effect transistor device. Biosens. Bioelectron. 2018, 110, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Feng, R.; Zhang, Y.; Ma, H.; Wu, D.; Fan, H.; Wang, H.; Li, H.; Du, B.; Wei, Q. Ultrasensitive non-enzymatic and non-mediator electrochemical biosensor using nitrogen-doped graphene sheets for signal amplification and nanoporous alloy as carrier. Electrochim. Acta 2013, 97, 105–111. [Google Scholar] [CrossRef]
- Wang, D.; Hu, W.; Xiong, Y.; Xu, Y.; Li, C.M. Multifunctionalized reduced graphene oxide-doped polypyrrole/pyrrolepropylic acid nanocomposite impedimetric immunosensor to ultra-sensitively detect small molecular aflatoxin. Biosens. Bioelectron. 2015, 63, 185–189. [Google Scholar] [CrossRef]
- Tang, J.; Huang, Y.; Zhang, C.; Liu, H.; Tang, D. Amplified impedimetric immunosensor based on instant catalyst for sensitive determination of ochratoxin A. Biosens. Bioelectron. 2016, 86, 386–392. [Google Scholar] [CrossRef]
- Zhu, X.; Kou, F.; Xu, H.; Han, Y.; Yang, G.; Huang, X.; Chen, W.; Chi, Y.; Lin, Z. Label-free ochratoxin A electrochemical aptasensor based on target-induced noncovalent assembly of peroxidase-like graphitic carbon nitride nanosheet. Sens. Actuators B Chem. 2018, 270, 263–269. [Google Scholar] [CrossRef]
- Sharifuzzaman; Barman, S.C.; Zahed, A.; San, N.J.; Park, J.Y. Green Synthesis of Reduced Graphene Oxide Decorated with Few-Layered MoS2-Nanoroses and Au/Pd/Ag Trimetallic Nanoparticles for Ultrasensitive Label-Free Immunosensing Platforms. J. Electrochem. Soc. 2019, 166, B249–B257. [Google Scholar] [CrossRef]
- Huang, K.-J.; Shuai, H.-L.; Chen, Y.-X. Layered molybdenum selenide stacking flower-like nanostructure coupled with guanine-rich DNA sequence for ultrasensitive ochratoxin A aptasensor application. Sens. Actuators B Chem. 2016, 225, 391–397. [Google Scholar] [CrossRef]
- Li, D.-L.; Zhang, X.; Ma, Y.; Deng, Y.; Hu, R.; Yang, Y. Preparation of an OTA aptasensor based on a metal–organic framework. Anal. Methods 2018, 10, 3273–3279. [Google Scholar] [CrossRef]
- Tang, J.; Hou, L.; Tang, D.; Zhou, J.; Wang, Z.; Li, J.; Chen, G. Magneto-controlled electrochemical immunoassay of brevetoxin B in seafood based on guanine-functionalized graphene nanoribbons. Biosens. Bioelectron. 2012, 38, 86–93. [Google Scholar] [CrossRef]
- Bratakou, S.; Nikoleli, G.-P.; Siontorou, C.G.; Nikolelis, D.P.; Karapetis, S.; Tzamtzis, N. Development of an Electrochemical Biosensor for the Rapid Detection of Saxitoxin Based on Air Stable Lipid Films with Incorporated Anti-STX Using Graphene Electrodes. Electroanalysis 2017, 29, 990–997. [Google Scholar] [CrossRef]
- Eissa, S.; Zourob, M. A graphene-based electrochemical competitive immunosensor for the sensitive detection of okadaic acid in shellfish. Nanoscale 2012, 4, 7593–7599. [Google Scholar] [CrossRef]
- Gan, X.; Zhao, H.; Schirhagl, R.; Quan, X. Two-dimensional nanomaterial based sensors for heavy metal ions. Microchim. Acta 2018, 185, 478. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Jiang, D.; Li, H.; Hao, N.; You, T.; Wang, K. An intriguing signal-off responsive photoelectrochemical aptasensor for ultrasensitive detection of microcystin-LR and its mechanism study. Sens. Actuators B Chem. 2018, 259, 316–324. [Google Scholar] [CrossRef]
- Eissa, S.; N’Diaye, J.; Brisebois, P.; Izquierdo, R.; Tavares, A.C.; Siaj, M. Probing the influence of graphene oxide sheets size on the performance of label-free electrochemical biosensors. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, M.; Li, H.; Yan, F.; Pang, P.; Wang, H.; Wu, Z.; Yang, W. A molybdenum disulfide/gold nanorod composite-based electrochemical immunosensor for sensitive and quantitative detection of microcystin-LR in environmental samples. Sens. Actuators B Chem. 2017, 244, 606–615. [Google Scholar] [CrossRef]
- Pang, P.; Teng, X.; Chen, M.; Zhang, Y.; Wang, H.; Yang, C.; Yang, W.; Barrow, C.J. Ultrasensitive enzyme-free electrochemical immunosensor for microcystin-LR using molybdenum disulfide/gold nanoclusters nanocomposites as platform and Au@Pt core-shell nanoparticles as signal enhancer. Sens. Actuators B Chem. 2018, 266, 400–407. [Google Scholar] [CrossRef]
- Ramalingam, S.; Chand, R.; Singh, C.B.; Singh, A. Phosphorene-gold nanocomposite based microfluidic aptasensor for the detection of okadaic acid. Biosens. Bioelectron. 2019, 135, 14–21. [Google Scholar] [CrossRef]
- Chekin, F.; Singh, S.K.; Vasilescu, A.; Dhavale, V.M.; Kurungot, S.; Boukherroub, R.; Szunerits, S. Reduced Graphene Oxide Modified Electrodes for Sensitive Sensing of Gliadin in Food Samples. ACS Sens. 2016, 1, 1462–1470. [Google Scholar] [CrossRef]
- Qu, Y.; Ma, M.; Wang, Z.; Zhan, G.; Li, B.; Wang, X.; Fang, H.; Zhang, H.; Li, C. Sensitive amperometric biosensor for phenolic compounds based on graphene–silk peptide/tyrosinase composite nanointerface. Biosens. Bioelectron. 2013, 44, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Gu, Y.; Lan, H.; Sun, Y.; Gao, H. Functional graphene-gold nano-composite fabricated electrochemical biosensor for direct and rapid detection of bisphenol A. Anal. Chim. Acta 2015, 853, 297–302. [Google Scholar] [CrossRef]
- Li, R.; Wang, Y.; Deng, Y.; Liu, G.; Hou, X.; Huang, Y.; Li, C. Enhanced Biosensing of Bisphenol A Using a Nanointerface Based on Tyrosinase/Reduced Graphene Oxides Functionalized with Ionic Liquid. Electroanalysis 2016, 28, 96–102. [Google Scholar] [CrossRef]
- Fernandes, P.M.V.; Campiña, J.M.; Silva, A.F. A layered nanocomposite of laccase, chitosan, and Fe3O4 nanoparticles-reduced graphene oxide for the nanomolar electrochemical detection of bisphenol A. Microchim. Acta 2020, 187, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Wang, X.; Wu, L.; Wu, L.; Dhanjai; Fu, L.; Gao, Y.; Chen, J. Response Characteristics of Bisphenols on a Metal–Organic Framework-Based Tyrosinase Nanosensor. ACS Appl. Mater. Interfaces 2016, 8, 16533–16539. [Google Scholar] [CrossRef]
- Wang, X.; Lu, X.; Wu, L.; Chen, J. 3D metal-organic framework as highly efficient biosensing platform for ultrasensitive and rapid detection of bisphenol A. Biosens. Bioelectron. 2015, 65, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Li, R.; Liu, J.; Zhang, X.; Wang, P.; Zhou, B.; Li, H.; Wang, J.; Li, Z.; Sun, B. Promotion effect of Zn on 2D bimetallic NiZn metal organic framework nanosheets for tyrosinase immobilization and ultrasensitive detection of phenol. Anal. Chim. Acta 2020, 1127, 131–139. [Google Scholar] [CrossRef]
- Mayorga-Martinez, C.C.; Gusmão, R.; Sofer, Z.; Pumera, M. Pnictogen-Based Enzymatic Phenol Biosensors: Phosphorene, Arsenene, Antimonene, and Bismuthene. Angew. Chem. Int. Ed. 2019, 58, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Parate, K.; Pola, C.C.; Rangnekar, S.V.; Mendivelso-Perez, D.L.; Smith, E.A.; Hersam, M.C.; Gomes, C.L.; Claussen, J.C. Aerosol-jet-printed graphene electrochemical histamine sensors for food safety monitoring. 2D Mater. 2020, 7, 034002. [Google Scholar] [CrossRef]
- Devi, R.; Gogoi, S.; Barua, S.; Sankar, D.H.; Bordoloi, M.; Khan, R. Electrochemical detection of monosodium glutamate in foodstuffs based on Au@MoS2/chitosan modified glassy carbon electrode. Food Chem. 2019, 276, 350–357. [Google Scholar] [CrossRef]
- Zhang, J.; Han, D.; Wang, Y.; Wang, L.; Chen, X.; Qiao, X.; Yu, X. Synergy between nanozymes and natural enzymes on the hybrid MoS2 nanosheets/graphite microfiber for enhanced voltammetric determination of hydrogen peroxide. Microchim. Acta 2020, 187, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, X.; Hou, M.; Li, X.; Wu, X.; Ge, J. Immobilization on Metal-Organic Framework Engenders High Sensitivity for Enzymatic Electrochemical Detection. ACS Appl. Mater. Interfaces 2017, 9, 13831–13836. [Google Scholar] [CrossRef]
- Dong, S.; Peng, L.; Wei, W.; Huang, T. Three MOF-Templated Carbon Nanocomposites for Potential Platforms of Enzyme Immobilization with Improved Electrochemical Performance. ACS Appl. Mater. Interfaces 2018, 10, 14665–14672. [Google Scholar] [CrossRef]
- Wang, X.; Yang, C.; Zhu, S.; Yan, M.; Ge, S.; Yu, J. 3D origami electrochemical device for sensitive Pb2+ testing based on DNA func-tionalized iron-porphyrinic metal-organic framework. Biosens. Bioelectron. 2017, 87, 108–115. [Google Scholar] [CrossRef]
- Lei, Y.; Zhao, W.; Zhang, Y.; Jiang, Q.; He, J.; Baeumner, A.J.; Wolfbeis, O.S.; Wang, Z.L.; Salama, K.N.; Alshareef, H.N. A MXene-Based Wearable Biosensor System for High-Performance In Vitro Perspiration Analysis. Small 2019, 15, e1901190. [Google Scholar] [CrossRef]
| Analyte | Description/Immobilization Details | Performances | Selectivity | Real Sample Application | Ref |
|---|---|---|---|---|---|
| Porcine epidemic diarrhea virus (PEDV) | Ab/AuNP-MoS2-rGO/GCE. Detection by faradaic EIS | Range: 82.5–1.65 × 104 TCID50 mL−1. Incubation time with the virus: 140 min at 37.5 °C. Stability: 89.2% of activity after 2 months at 4 °C. | No interferences from Salmonella spp., Campylobacter jejuni, and E. coli | Spiked pig manure | [134] |
| Salmonella typhimurium | cDNA –MOF (type UiO−67) –Gr/GCE and APT-AuNP-HRP for signal amplification. Detection by DPV | Range: 2 × 101–2 × 108 cfu mL−1; DL: 5 cfu mL−1; Analysis time: 3 h; Stability: 91.3% of activity after 2 weeks at 4 °C | S. aureus, S. flexneri, E. coli, and Streptococcus not interfering | Spiked milk | [106] |
| Staphylococcus arlettae | Bacteriophages/Gr electrodes. Detection by faradaic EIS | Range: 2.0–2.0 × 106 cfu mL−1; DL: 2 cfu mL−1; Response time: 2 min incubation with bacteria at 37 °C; Stability: 3 months. | S. aureus, E. coli, and S. lentus not interfering | Spiked water and apple juice samples | [96] |
| Salmonella enterica serovar Typhimurium | Ab/Porous graphene Detection by faradaic EIS with ferri/ferro | Range: 25–105 cfu mL–1; DL: 13 cfu mL–1; Response time: 22 min; Stability: change of 3.36% in Rct after 7 days at −20 °C | Bacillus cereus, E. coli O157:H7, Listeria monocytogenes, Pseudomonas aeruginosa, and Staphylococcus aureus not interfering | Chicken broth | [132] |
| Salmonella Typhimurium | APT/rGO-CNT/GCE. | Range: 101–108 cfu mL−1; DL 101 cfu mL−1; Test time: 10 min; Stability: 20 days in water at 4 °C | E. coli, K. pneumonia, p. aeruginosa, S. aureus, and Enterococcus faecalis not interfere, but Salmonella enterica such as S. Albany, S. Enteritidis, S. Weltevreden, S. Typhi, and S. Derby interfere | Raw chicken meat | [131] |
| Detection by DPV using [Fe(CN)6]4−3− | |||||
| E. coli O78:K80:H11 | APT-PLL-Bridged rebar graphene Detection by faradaic EIS using [Fe(CN)6]4−/3− as a redox probe | Range: 101–106 cfu mL−1; Detection limit: 101 cfu mL−1 | E. coli DH5α, p. vulgaris, L. monocytogenes, S. boydii, S. flexneri, E. aerogenes, C. braakii, and B. Subtilis not interfering | Spiked fruit juice (guava, litchi, mango) and milk | [135] |
| E. Coli | rGO-FET, passivated with an ultrathin layer of Al2O3, then decorated with AuNPs onto which a specific Ab was immobilized | Range: 103 to 105 cfu mL−1; No significant change after 14 days at 4 °C; Response time: 50 s | Salmonella typhimurium and Streptococcus pneumonia are not interfering | Spiked river water; DL: 104 cfu mL−1 | [136] |
| Botulinum neurotoxin serotype A | Ab/Au-Gr-Cs/GCE Detection by faradaic EIS using [Fe(CN)6]4–/3– | Range: 0.27–268 pg mL−1; DL: 0.11 pg mL−1; Incubation time with the toxin: 60 min at 37 °C. Stability: 90% of response after 4 days at 4 °C in 0.1 MPBS buffer pH 7.4 with 0.02% NaN2 | BoNT serotypes E and B | Spiked milk and human serum, BoNT was not detected in an unspiked sample by ELISA | [133] |
| Botulinum neurotoxin A | SNAP−25- peptide/chemically reduced GO/Au; Detection of BoNT serotype A light chain protease activity by DPV using [Fe(CN)6]4–/3 | Range: 8.6 pg mL−1–1 ng mL−1; DL: 8.6 pg mL−1 | BoNT serotype B lightchain (BoNT-LcB) and heat-treated LCA (denatured enzyme) did not interfere. | Spiked skimmed milk | [116] |
| Staphylococcal enterotoxin B (SEB) | Ab1/GR–AuNPs/GCE + Fc–Ab2.. Detection by SWV of ferrocene | DL: 5 ng mL−1; Analysis time: 35 min; Stability: 3 weeks at 4 °C | BTX−2, Okadaic acid MC-LR; Na+, K+, Ca2+, Mg2+, Sr2+, Cl−, SO42−, Br−, HCO3−, and F− not interfering. −95% cross-reactivity for BTX−1 and BTX−3 | Spiked mollusk extract of S. constricta, M. senhousia, and T. granosa, | [128] |
| Staphylococcal enterotoxin B | APT/cDNA/rGO- AuNUs.; Detection of hematoxylin intercalator by DPV | Range: 5.0–500.0 fM; DL: 0.21 fM | Staphylococcal enterotoxin A, Staphylococcal enterotoxin C, BSA, and tryptophan not interfering | Spiked and simulated samples: milk, meat, and human serum | [129] |
| Botulinum Toxin E | Ab1/Gr-GCE + Ab2 + IgG-ALP/AuNPs. Detection by LSV–stripping of Ag deposited in the reaction of 3-IP and Ag+ catalyzed by ALP | Range: 10 pg mL−1–10 ng mL−1; DL: 5.0 pg mL−1; Analysis time: 65 min | BoNT/A, BoNT/B, and BoNT/F not interfering | Spiked orange juice and milk | [130] |
| Analyte | Description/Immobilization Details | Anal Performance | Selectivity | Application | Ref |
|---|---|---|---|---|---|
| Zearalenone | APT/AuNP/p-PtNTs/Au + Thionine-labeled GO Detection of thionine by DPV | Range: 5 × 10−13–5 × 10−7 g mL−1; DL: 1.67 × 10−13 g mL−1; Stability: decrease of 5.3% after storage for 7 days at 4 °C. | Aflatoxin B1, deoxynivalenol and patulin not interfering | Spiked maize extract | [97] |
| Fumonisin B1 | cDNA/AuNP/GCE + APT/Gr-Th. Detection of thionine by cyclic voltammetry | Range: 1–106 pg mL−1; DL: 1 pg mL−1; Incubation time with FB1: 25 min; Stability: no change in activity after 21 days at 4 °C | Fumonisin B1, Ochratoxin A, zearalenone and not interfering | Feed sample spiked at 5 concentration levels | [102] |
| Aflatoxin B1 (AFB1) | AFB1-BSA/AuNP/GCE + Ab/PtNP-CoTPP-rGO; Detection of H2O2 reduction by DPV | DL: 5.0 pg mL−1; Incubation time with sample and Ab-nanostructure conjugates: 25 min | Aflatoxins G1, FG2, alpha-fetoprotein, and thyroid-stimulating hormone not interfering. AFB2 interferes due to the cross-reactivity of the AFB1 antibody. | Spiked peanut and naturally contaminated peanut | [113] |
| Zearalenone | Ab1- N-GS/GCE + NP-PtCo-Ab2. Detection of H2O2 by amperometry at −0.4 V in phosphate buffer pH 5.8 | Range: 0.005–25 ng mL−1; DL: 2.1 pg mL−1; Incubation time: NA; Incubation time with NP-PtCo-Ab2: 1 h; Stability: decrease of 5.6% of activity after after7-days in phosphate buffer pH = 5.8 at 4 °C. | Aflatoxin, ochratoxin, zeranol, kanamycin, gentamicin not interfering | Spiked pig feed | [137] |
| Aflatoxin B1 | APT/c-PS/PDDA/Gr/GCE Detection by EIS | Range: 0.001–0.1 ng mL−1; DL: 0.002 ng mL−1; Stability: decrease to 85% response after 30 days at 4 °C | Ochratoxin A not interfering | Spiked oil and soy sauce | [112] |
| Aflatoxin B1 | Ab/PPy-PPa-rGO Direct detection by non-faradaic EIS | Range: 10 fg mL−1–10 pg mL−1; DL: 10 fg mL−1; Incubation with AFB1: 50 min | Fumonisin B2, Aflatoxins G1 and G2, deoxynivalenol, and ochratoxin A A not interfering | Spiked corn | [138] |
| Aflatoxin B1 | Ab/rGO–Ni. Detection by DPV using [Fe(CN)6]3−/4− | Range: 1–8 ng mL−1; DL: 0.16 ng mL−1; Stability: less than 10% decrease in signal after 6 weeks at 4 °C. | OTA not interfering | N/A | [89] |
| Ochratoxin A | APT/DNA1/Au + DNA2/AuNP–rGO Detection by EIS, using [Fe(CN)6]4−/3− | Range: 1 pg mL−1–50 ng mL−1; DL: 0.3 pg mL−1 (0.74 pM); Incubation time: 120 min (with-OTA). +60 min (with AuNPs–rGO-DNA2) | Ochratoxin B, Fumonisin B1 are not interfering | Spiked wines | [103] |
| Ochratoxin A | OTA-BSA/GCE + Ab-GO-PAMAM-Mn2+ Oxidation of 4-chloro−1-naphthol to an insoluble product. Detection by EIS using [Fe(CN)6]4−/3− | Range: 0.1 pg mL−1–30 ng mL−1; DL: 0.055 pg mL−1; Incubation time: 25 min with OTA and anti-OTA-GO-PAMAM-Mn2+, 1 min with KMnO4 and 25 min with 4-chloro−1-naphthol; Stability: 93.5% of activity after 20 days at 4 °C. | Ochratoxin B, Aflatoxin B1, B2 and G1; Na+, Cu2+, Fe3+, Mn2+, Zn2+, K+, Ca2+, Mg2+ not interfering | Spiked red wine | [139] |
| Aflatoxin B1 | APT/GCE + rGO Detection by DPV using [Fe(CN)6]4−/3− | Range: 0.5 nM−4 μM; DL: 0.07 nM; Incubation time: 1 h with AFN1 +1 h with rGO; Stability: decrease of less than 4% in response after 1 week at 4 °C. | NA | Spiked pasteurized cow milk and human blood plasma spiked | [91] |
| AFB1 | MB-APT/COOH-GO/SPCE. Detection of MB by DPV | Range: 0.05–6.0 ng mL−1; DL: 0.05 ng mL−1; Incubation time with AFB1: 1 h | AFM1, OTA not interfering | Spiked beer and wine | [111] |
| OTA | APT/Au-ATP-rGO/Au Detection by EIS using [Fe(CN)6]4−/3− | Range: 0.1–200 ng mL−1; DL: 0.03 ng mL−1; Incubation time with OTA: 90 min.; Stability: 1 month at 4 °C | Fumonisin B1, ochratoxin B not interfering | Spiked wine | [98] |
| OTA | APT/COOH-GO/SPCE. +Nanoceria (nCe)-OTA; Detection of H2O2 | Range: 0.15–50 nM; DL: 1 nM | OTB not interfering | Cereal | [110] |
| OTA | APT/STR/GR/ITO. Detection by DPV using [Fe(CN)6]4−/3− | Range: 0.01–1000 ng mL−1; DL: 1 fg mL−1 (buffer); 10 pg mL−1 (spiked sample); Incubation with OTA: 8 min; Stability: 91.4% of initial activity after 7days | Malathion and heavy metals tested; malathion appears to interfere | Spiked grape juice | [105] |
| OTA | APT/cDNA/Au + g-C3N4. Detection −0.8 V in the presence of H2O2, based on the peroxidase-like activity of g-C3N4. | Range: 0.2–500 nM; DL: 0.073 nM; Incubation with OTA: 1 h Incubation with 1 mg/mL g-C3N4 solution: 30 min | Ochratoxin B and aflatoxin B1 not interfering | Spiked red wines, juice and corn | [140] |
| OTA | Ab/AuPdAg/MoS2/rGO/GCE.; Detection by DPV using [Fe(CN)6]3−/4− | Range: 10 fg mL−1–150 ng mL−1; DL: 5 fg mL−1; Incubation with OTA: 40 min; Stability: 99.6% of activity after 10 weeks; storage conditions not specified | Ochratoxin B, aflatoxin B1 IgG and glucose not interfering | Spiked coffee and corn | [141] |
| OTA | APT/cDNA/Au + MoS2 as peroxidase mimic Detection by amperometry at −0.2 V | Range: 0.5 pg mL−1–1.0 ng mL−1.; DL: 0.23 pg mL−1; Incubation time: 25 min at 37 °C with OTA + 25 min at RT with MoS2 + 5 min with H2O2 and hydroquinone; Stability: 90% of activity after 21 days at 4 °C. | Ochratoxins B and C, Aflatoxins B1 and B2, Cu2+, Mg2+, Zn2+, and Mn2+ not interfering | Spiked red wine | [104] |
| OTA | MB-APT/β-CD/Au + MoS2/AuNP + ferrocenecarboxylic acid; Detection by DPV of MB and Fc | Range: 0.1 nM and 50 nM; DL: 0.06 nM; Incubation time with the aptamer: 1 h; Stability: 95.4% after 30 days of storage in the dark at 4 °C. | Aflatoxins M1 and B1, fumonisin M1, Ochratoxin B and C not interfering | Spiked red wine | [92] |
| Ochratoxin A | APT/AuNPs/MoSe2/GCE + cDNA + MB; Detection of MB by DPV | Range: 0.0001–1 nM; DL: 0.8 pM; Test time: 45 min; Stability: 3.2% decrease in activity after 15 days at 4 °C | Ochratoxin B and C and aflatoxin B1 not interfering | Spiked red wine | [142] |
| Aflatoxin B1 | 3DOM MoS2/AuNPs/Au aptamer-including tetrahedral DNA nanostructures + HRP-cDNA/AuNP-SiO2@Fe3O4 + thionine. Detection by DPV | Range: 0.1 fg mL−1–0.1 μg mL−1; DL: 0.01 fg mL−1; Incubation time with AFB1: 50 min at 37 °C; Incubation with HRP-cDNA/AuNP-SiO2@Fe3O4: 2.5 h Stability: 91% of initial activity after 1 month at 4 °C. | Aflatoxin B, M1, zearalenone and ochratoxin A are not interfering. | Spiked rice and wheat powder | [99] |
| OTA | b-APT/cDNA/AuNP-MoS2/GCE + STR- AuNP@Cd-MOF−74; Detection of Cd2+ by DPV | Range: 0.05–100 ng mL−1; DL: 10 pg mL−1; Incubation time with OTA: 25 min | Microcystin (MC)-LR MC-RR), thrombin, and Ochratoxin B not interfering | Spiked red wines | [143] |
| Analyte | Description/Immobilization Details | Analytical Performances | Selectivity | Application | Ref |
|---|---|---|---|---|---|
| Mycrocystin LR (MC-LR) | APT/Commercial graphene-modified screen-printed electrode; Detection by SWV using [Fe(CN)6]4–/3− | Range: 1.9 pM–1.0 nM; DL: 1.9 pM; Stability: 2.9% decrease in activity after 1 month at 4 °C; Incubation time with MC-LR: 45 min | Okadaic acid, microcystin-LA, and microcystin-YR not interfering | Spiked fish extract and spiked tap water | [107] |
| MC-LR | APT/BiOBr nanoflakes/n-doped graphene/ITO; Photoelectrochimical biosensor | Range: 1 pM–100 nM; DL: 3.3 × 102 pM; Incubation time with MC-LR: 30 min; Stability: no change in activity after 2 weeks at 4 °C | MC-LA, MC-YR not interfering | Spiked fish extract | [108] |
| Cylindrospermopsin | APT/Thionine–graphene; Detection by EIS using [Fe(CN)6] 4–/3− | Range: 0.39−78 ng mL−1 (1–200 nM); DL: 0.117 ng mL−1 (300 pM); Incubation time with CYN: 2 h; Stability: 88.2% and 74.7% of activity after 14 days and 30 days, respectively of storage in buffer at 4 °C | MC-LR, okadaic acid not interfering | Spiked lake water | [109] |
| Brevetoxin i | Ab-magnetic beads + BTX2-BSA-GGNR+ MCPE; Detection by SWV | Range: 1.0 pg mL−1–10 ng mL−1; DL: 1.0 pg mL−1; Incubation time with BTX2: 30 min | -Okadaic acid, MC-LR, Na+, K+, Ca2+, Mg2+, Sr2+, Cl−, SO4 2−, Br−, HCO3−, and F− not interfering. -BTX1, BTX3 interfere due to the cross-reactivity of the antibody for BTX2 | Spiked mollusks extracts | [144] |
| Saxitoxin | Ab/Graphene nanosheets—lipid films; Detection by potentiometry. | Range: 1 × 10−9 M∓1 × 10−6 M; DL: 1 nM; Response time: less than 20 min | Mg2+, Ca2+, HC3−, SO42−, Cl−, NO3−, NH4+ not interfering | Lake water and shellfish samples; Spiked mussels, oysters, and mollusks | [145] |
| Okadaic Acid | Ab/GSPE + okadaic acid-ovalbumin conjugate; Detection by SWV using [Fe(CN)6] 4−/3− | Range: up to 5000 ng L−1; DL: 19 ng L−1; Stability: 98% of activity after 40 days at 4 °C | Microcystin-LA not interfering | Spiked mussel extract | [146] |
| MC-LR | Ab-GO-IL -Au NP/GCE; Detection by DPV using [Fe(CN)6]4−/3− | Range: 0.1–1000 ng mL−1; DL: 0.1 ng mL−1; Incubation time with MC-LR: 50 min; Stability: 90.58% after 30 days in buffer at 4 °C | MC-LA, MC-RR, and MC-YR, as Na+, Ca 2+, K+,Cl−, and CO3 2− not interfering | Spiked river water | [147] |
| MC-LR | APT/AgI-NG/ITO; Photoelectrochemical aptasensor, | Range: 0.05 pM–5 nM; DL: 0.017 pM; Incubation time with MC-LR: 20 min; Stability: 94.5% of activity after 2 weeks at 4 °C | MC-LA and MC-YR are not interfering | Spiked fish | [148] |
| MC-LR | Ab/oxidized CVD graphene; Detection by EIS | Range: 0.005–10 μg L−1; DL: 2.3 ng L−1; Stability: 92.5% and 83.6% of activity after 1 and 2 weeks of storage at 4 °C. | Environmental water samples with TOC 0.53–8.99 mgL−1, total dissolved solid 118–170 mgL−1, sodium, (2.62–7.46 mgL−1), magnesium (4.9–14.67 mgL−1), aluminium (0.25–0.47 mgL−1), potassium (0.02–3.56 mgL−1), calcium (2.29–2.93 mgL−1), manganese (0.49–14.32 µgL−1), iron (0.86–174.2 µgL−1), copper (1.19–7.99 µgL−1) are not interfering | Spiked tap water, pond water, and lake water | [114] |
| MC-LR | APT/GO-modified printed electrode; (aptamer adsorbed or covalently immobilized; Detection by SWV using [Fe(CN)6]4−/3−. | Apt/Phys−300 μm; Range: 0.1 nM–1.0 μM DL: 0.038 nM; Apt/Phys−0.22 μm; Range: 0.1 nM–1.0 μM; DL: 0.088 nM; Apt/Cov−300 μm; Range: 1 nM−1.0 μM; DL:0.25 nM; Apt/Cov−0.22 μm: Range: 1 nM−1.0 μM; DL: 0.018 nM | Okadaic acid, MC-LA not interfering | N/A | [149] |
| Microcystin-L vccccR | MC-LR/AuNP@MoS2-TiONB/GCE + biotin-cDNA + Avidin-HRP; Detection by DPV | Range: 0.005–30 nM; DL: 0.002 nM; Incubation time: 120 min (60 min with the mixture of cDNA and MC-LR and 60 min with avidin -HRP; Stability: 90% activity after 10 days at 4 °C. | MC-LA, MC-YR, atrazine, and trichlorfon are not interfering | Spiked tap water, reservoir water and river water | [21] |
| MC-LR | Ab1/AuNR/MoS2/+HRP-Ab2; Detection by DPV | Range: 0.01–20 μg L−1; DL: 5 ng L−1; Incubation time with MC-LR: 1 h at 37 °C. and incubation with HRP-anti-MC-LR: 1 h at 37 °C; Stability: 99.46% and 95.62% after 1 week and 4 weeks of storage at 4 °C, respectively. | MC-RR, Okadaic acid, starch, ascorbic acid, Na+, NH4+, Ca2+, Cl−, SO42−, and CO32− not interfering | Spiked: lake water, tap water, and drinking water | [150] |
| MC-LR | Ab/BSA-stabilized Au nanoclusters/MoS2/Au electrode: +Au@Pt core-shell nanoparticles Detection by DPV | Range: 1.0 ngL−1–1.0 mgL−1; DL: 0.3 ngL−1; Incubation time with MC-LR: 1 h at 37 °C. and incubation with Au@Pt: 1 h at 37 °C; Stability: 98% and 92% of initial activity after 1 week and 4 weeks storage at 4 °C | MC-RR, MC-LA, dopamine, uric acid, ascorbic acid, Al3+, Ca2+, Mg2+, Na+, K+, NH4+, SO42−, CO32−, NO3− not interfering | Spiked water samples Recoveries: 99.6–101.3% | [151] |
| Okadaic acid | APT/phosphorene-gold NP/SPCE Detection by DPV using [Fe(CN)6]3−/4− | Range: 10 nM−250 nM; DL: 8 pM | Spiked mussel extract | [152] |
| Analyte | Description/Immobilization Details | Anal Perform | Selectivity | Application | Ref |
|---|---|---|---|---|---|
| Gliadin | Ab/prGO/GCE Detection by DPV using [Fe(CN)6]3−/4− | Range: 1.2–34 ng mL–1; DL: 1.2 ng mL−1; Stability: 5% decrease in activity after 2 months at 4 °C | Lysozyme, casein, rice flour, cornflour not interfering | Wheat flour, pasta, cereal, Quaker oats, Gluten-free wheat flour Spiked rice flour and gluten-free flour | [153] |
| Ovalbumin | Ab/Graphene-modified SPCE Detection by DPV using [Fe(CN)6]4−/3− | Range: 1 pg mL−1 - 0.5 μg mL−1; DL: 0.83 pg mL−1; Incubation time with ovalbumin: 45 min; Stability: less than 2% decrease in activity after 14 days at 4 °C. | β-lactoglobulin, BSA, egg lysozyme, and casein are not interfering | Spiked cake extract % | [115] |
| β-lactoglobulin | Ab adsorbed/GO-modified electrode and Ab covalent/GO-modified electrode. Detection by SWV using [Fe(CN)6]4−/3− | Ab/Phys−300 μm; Range: 0.001–1.0 μg mL−1; DL: 0.46 ng mL−1; Ab/Phys−0.22 μmL; Range: 0.001–1.0 μg mL−1; DL: 0.79 ng mL−1; Ab/Cov−300 μm: Range: 0.01–1.0 μg mL−1; DL: 2.6 ng mL−1; Ab/Cov−0.22 μm: Range: 0.01–1.0 μg mL−1; DL: 1.2 ng mL−1 | OVA, BSA not interfering | N/a | [149] |
| Catechol, Phenol, BPA | Tyrosinase/Silk peptide–graphene nanosheets/GCE Detection by amperometry at −0.10 V in PBS buffer pH 6 and 35 °C | Catechol; Range: 0.001–16.91 μM; DL: 0.23 nM-Phenol: Range: 0.0015–21.12 μM; DL: 0.35 nM–BPA: Range: 0.002–5.48 μM DL: 0.72 nM; Stability: 93.6% of activity after one month at 4 °C | Vitamin C, uric acid, m-dihydroxybenzene and p-nitrophenol not interfering. The sensor responds to dopamine, catechol, and phenol, besides BPA | BPA in plastic drinking bottles | [154] |
| BPA | Tyrosinase–graphydyne–chitosan/GCE Detection by amperometry at −0.04 V in 0.05 M PBS pH 7.0 | Range: 0.0 × 10−7 to 3.5 × 10−6 M; DL: 24 nM; Response time: 20s; Stability: 94% activity after 3 weeks when stored dry at 4 °C | Phtalates (dimethyl, octyl) and Bisphenol S (BPS) are not detected | Water bottle (PC);beverage bottle (Al); coffee spoon (PP);beverage bottle (tinplate), mineralvwater bottle (PET); tap water | [35] |
| BPA | Tyrosinase–hydrophilic nanographene–chitosan/GCE. Detection by amperometry at −0.1 V | Range: 0.1–2 µM, DL: 33 nM | Phthalates, dimethylphthalate, doctylphthalate KNO3, sodiumcitrate, sodium oxalate, urea, ethylacetate, diethylcarbonate, acetonitrile, n-hexane, benzene, hexachlorobenzene, naphthalene are not interfering | 5 samples: polycarbonate (PC) bottle, paper cup, PEGT water bottle and glass bottle. | [118] |
| BPA | Tyrosinase–graphene–Au/GCE. Detection by DPV, 0.1 M PBS pH 7 | Range: 0.025–3 µM, DL: 1 nM | NA | Spiked plastic cup and milk carton samples | [155] |
| BPA | APT/Au NP-G/GCE Detection by CV using ferricyanide | Range: 0.01 and 10 µM; LOD: 5 nM; Incubation time: 30 min; Stability: 2 weeks at 4 °C | BPB, 4,4′-biphenol and 6F-BPA are not interfering | Spiked milk | [100] |
| BPA | Tyrosinase-(rGO–DAPPT/GCE Detection by amperometry at 0.1 V in 0.1 M phosphate buffer (pH 7.0) | Range: 1.0 × 10−9–3.8 × 10−5 M DL: 3.5 × 10−10 M; Stability: 90% of initial activity after 1 month at 4 °C | m-dihydroxy-benzene, p-nitrophenol, ascorbic acid, and uric acid not interfering but, p-dihydroxybenzene, dopamine, phenol, catechol, and cysteine interfere at 50 times larger concentrations than BPA | Spiked plastic drinking bottles | [156] |
| BPA | LACC/rGO-Fe3O4; Detection by amperometry at +0.15 V | Range: 6–228 ppb; DL:18 nM(4 ppb); Storage: 18% decrease in current density after 1 month in buffer at 4 °C | Catechol, ascorbic acid uric acid, 1-naphthol 4-nitrophenol and benzene not interfering; Glucose interferes | Spiked bottled water | [157] |
| Bisphenols | Tyrosinase-chitosan-CuMOF/GCE:. Detection by amperometry at −0.1 V in 0.05 M PBS buffer pH 7 | BPE: Range: 5.0 × 10−8–3.0 × 10−6 M.; DL: 15 nM–BPF: Range: 5.0 × 10−8–3.0 × 10−6 M, DL:16 nM–BPA: Range: 5.0 × 10−8–3.0 × 10−6 M; DL: 13 nmM; BFB; Range: 1.25 × 10−7–8.0 × 10−6 M; DL: 56 nM–BFZ: Range: 2.5 × 10−7–5.0 × 10−6 M, DL: 33 nM | 81.8–98.7% activity when the biosensor was incubated 0.5 with 0.1 mM of Hg2+, Pd2+, Cu2+, Fe2+, Co2+, Ba2+, Zn2+, Cd2+, and Ni2+ due to enzyme inhibition | metals | [158] |
| BPA | 3D-CuMOF tyrosinase. Detection by amperometry at −0.1 V | BPA: Range: 5.0 × 10−8–3.0 × 10−6 M; DL: 13 nM; Storage stability: 90% of activity after 3 weeks at 4 °C in PBS buffer | K+, Na+, NO3−, HPO42−, Cl, and acetone, acetonitrile, methanol, ethanol not interfering- phenols chemicals (e.g., phenol and catechol) not used in PC products, and phthalates are not substrates of tyrosinase | Spiked plastics: water bottle (PC), nursing bottle (PP), coffee spoon(PP), mineral water bottle (PET) | [159] |
| Phenol | Tyrosinase–chitosn−2D NiZn/GCE; Detection by amperometry at −0.05 V | Range: 0.08–58.2 μM; DL: 6.5 nM; Storage stability: 93% of the original response after 5-weeks at 4 °C | K+, Mg2+, Ca2+, Fe3+, Zn2+, SO42−, PO43−, CO32−, NO3−, uric acid, ascorbic acid, glucose not interfering | Spiked tap water | [160] |
| 17β-estradiol | APT/WS2 Au NPs/GCE; Detection by DPV using [Fe(CN)6]4−/3− | Range: 1.0 × 10−11–5.0 × 10−9 M; DL: 2.0 × 10−12 M; Incubation time with 17β-estradiol: 3 h | Naphthalene and 1-aminoanthraquinone are not interfering | Spiked river water and serum | [101] |
| Phenol | Tyrosinase/2D layered pnictogens (phosphorene, arsenene, antimonene, and bismuthene; Detection by amperometry at −0.005 V in 0.1 M PBS buffer pH 6.5 | Antimonene- based biosensor: Range: 500–2500 nM and 7.5–27.5 μM; DL: 255 nM. | Ca, Mg, Cu, aniline, benzyl alcohol not interfering | Spiked tap water | [161] |
| Total polyphenol | LACC/GrQD/MoS2/SPCE; Detection by amperometry at +0.05 V in 0.1 M acetate buffer pH 5.00 | Caffeic acid; Range: 0.38–100 µM; DL: 0.32 µM–Chlorogenic acid; Range: 0.38–8.26 μM; DL: 0.19 μM (-) Epicatechin; Range: 2.86–100.00 μM; DL: 2.04 μM; Stability: 85% of initial activity after 4 weeks at 4 °C | Wines | [125] | |
| Histamine | Ab/Gr; Detection by EIS with [Fe(CN)6]4−/3− | Range: 6.25–200 ppm (56.25 μM–1.8 mM; DL: 3.41 ppm (30.7 μM); Incubation time: 30 min | Bovine serum albumine(BSA) goat serum, whey protein not interfering | Tuna broth samples | [162] |
| Hypoxanthine | XOD/Gr-TiO2./GCE Detecction by amperometry at 0.8 V | Range: 20−512 μM; DL: 9.5 μM; Stability: 77% of initial activity after 10 days at 4C in 0.05 M PBS, 50% activity after 30 days | -Uric acid, ascorbic acid, and glucose do not interfere—xanthine interferes | Pork meat is stored at room temperature for seven days | [126] |
| Monosodium glutamate | Ab/AuNP-MoS2-chitosan/GCE. Detection by DPV using [Fe(CN)6]4−/3− | Range: 0.05–200 μM; DL: 0.03 µM; Stability: 98.7% response after 15 days at 4 °C | Cysteine, arginine, aspartic acid, butylated hydroxyl toluene and bisphenol-A are not interfering | Spiked vegetable soup % | [163] |
| Glucose | Gox/Au- Ti3C2Tx MXene/GCE; Detection by amperometry at −0.402 V | Gox/Au- MXene/GCE: Range: 0.1–18 mM; DL: 5.9 µM; Gox/MXene//GCE; LR: 0.5–6 mM; DL: 100 μM; Storage: 93% activity after 2 months | Dopamine, uric acid, ascorbic acid not interfering | N/A | [119] |
| H2O2 | HRP/Phosphorene/GCE; Detection by amperometry at −0.1 V in PBS buffer pH 7.2 | Range: 5–275 µM; DL: 0.14 µM; Kmapp = 164 µM; Stability:93% and 69% of activity after 7 and 15 days, respectively | Dopamine ascorbic acid and uric acid do not interfere | N/A | [123] |
| H2O2 | HRP/MB/chitosan/MoS2/graphite fiber Detection by amperometry at −0.3 V in 0.1 M phosphate buffer pH 7.0 | Range: 0.1 to 90 μM; DL: 30 nM; Stability: 89% of initial activity after 60 days at 4 °C in buffer | Ascorbic acid, uric acid, dopamine, Na+, K+, Mg2+, Ca2+, Cl− are not interfering | N/A | [164] |
| H2O2 | Hemoglobin/poly-l-lysine-black phosphorus/GCE; Detection by cyclic voltammetry | Range: 10–700 µM | Uric acid and ascorbic acid are not interfering | N/A | [121] |
| H2O2 | HRP-MoS2–Gr/GCE Detection by amperometry at −0.08 V | Range: 0.2 μM–1.103 mM; DL: 0.049 μM; Stability: 91.5% and 84.2% of initial activity after 2 weeks and 1 month, respectively at 4 °C. | Ascorbic acid, dopamine, cysteine, and lysine do not interfere | N/a | [124] |
| H2O2 | Cytochrome c/ZIF−8-MOFs/SPCE screen-printed electrode; Detection by amperometry at −0.05 V | Range: 0.09–3.6 mM | Glucose, dopamine, and bovine serum albumin are not interfering | Spiked milk and beer | [165] |
| Acetochlor | GOx/CS/NH2-MIL−125(Ti)/TiO2-MOF/GCE Photoelectrochemical sensor, inhibition of glucose oxidase | Range: 0.02–200 nM; DL: 0.003 nM. Stability: 92.5% activity after 30 days at 4 °C | Sucrose, glycine, citric acid, K+, Na+, Ca2+, prometryn, clethodim, cycloxydim, and sethoxydim not interfering | Spiked strawberry, tomato, cucumber, and greens | [122] |
| Fenitrothion | AChE-BSA/TMDs (MoS2, MoSe2, WS2, WSe2/GCE Detection by amperometry at 0.1 V | 1T-Phase WS2 based biosensor; Range: 1–1000 nM; DL:2.86 nM; Incubation time: 5 min | Fe2+, Cu2+, ascorbic acid and phenol: not interfering | Spiked apple juice | [127] |
| Forchlorfenuron | Catalase/boron nitride/GCE; Detection by amperometry at −0.35 in 1 M phosphate buffer pH 7.0 V | Range: 0.5–10.0 µM; DL: 0.07 μM; Stability: 91.3% of initial activity after 2 months at 4 °C | Glucose, sucrose, glycine, citric acid, Na+ and Ca2+ not interfering | Spiked orange juice, kiwi, watermelon, grape | [120] |
| Methyl parathion | Nafion/AChE/MOF/electrode detection by DPV | [Fe-MOF-NH2]N2: Range 10−12–10−8 g mL−1; DL: 3.2 × 10−13 g mL−1 (1.2 × 10−12 M); Zr-MOF-NH2]N2: Range: 5.0 × 10−13–5.0 × 10−9 g mL−1, DL: 1.8 × 10−13 g mL−1 (6.9 × 10−13 M) [La-MOF-NH2]N2 Range: 1.0 × 10−13–5.0 × 10−9 g mL−1; DL: 5.8 × 10−14 g mL−1 (2.2 × 10−13 M) Incubation time: 12 min; Stability: 81%, 83% and 84% after 4 weeks at 4 °C in PBS buffer pH 7. | No data reported | N/a | [166] |
| Pb 2+ | DNA functionalized iron-porphyrinic metal–organic framework ((Fe-p)n-MOF-Au-GR/Au-PWE. Detection by DPV | Range: 0.03–1000 nM.; DL: 0.02 nM; Stability: 95% and 50% of activity after 20 and 60 days at room temperature, respectively; 95% and 94% of activity after 60 days of storage in the refrigerator and freezer, respectively | Fe3+, Cd2+, Co2+, Zn2+, Mn2+, Ni2+, Cu2+, Hg2+ and Ag+ not interfering | Industrial waste water, river water, fruit juice (orange and apple), solid samples, serum | [167] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, R.; Radoi, A.; Rashid, S.; Hayat, A.; Vasilescu, A.; Andreescu, S. Two-Dimensional Nanostructures for Electrochemical Biosensor. Sensors 2021, 21, 3369. https://doi.org/10.3390/s21103369
Khan R, Radoi A, Rashid S, Hayat A, Vasilescu A, Andreescu S. Two-Dimensional Nanostructures for Electrochemical Biosensor. Sensors. 2021; 21(10):3369. https://doi.org/10.3390/s21103369
Chicago/Turabian StyleKhan, Reem, Antonio Radoi, Sidra Rashid, Akhtar Hayat, Alina Vasilescu, and Silvana Andreescu. 2021. "Two-Dimensional Nanostructures for Electrochemical Biosensor" Sensors 21, no. 10: 3369. https://doi.org/10.3390/s21103369
APA StyleKhan, R., Radoi, A., Rashid, S., Hayat, A., Vasilescu, A., & Andreescu, S. (2021). Two-Dimensional Nanostructures for Electrochemical Biosensor. Sensors, 21(10), 3369. https://doi.org/10.3390/s21103369









