Quantitative Measurement of Rigidity in Parkinson’s Disease: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Methods
2.2. Data Extraction and Analysis
2.3. Assessment of the Methodological Rigor of the Included Studies
3. Results
3.1. Description of the Selection Process
3.2. Description of the Included Studies
3.2.1. Methods Evaluating the Mechanical or Neurophysiological Properties of the Muscle
3.2.2. Methods Using Sensors for the Capture and Analysis of Movement
3.2.3. Electro-Mechanized Methods Adhered to the Body for Joint Mobilization
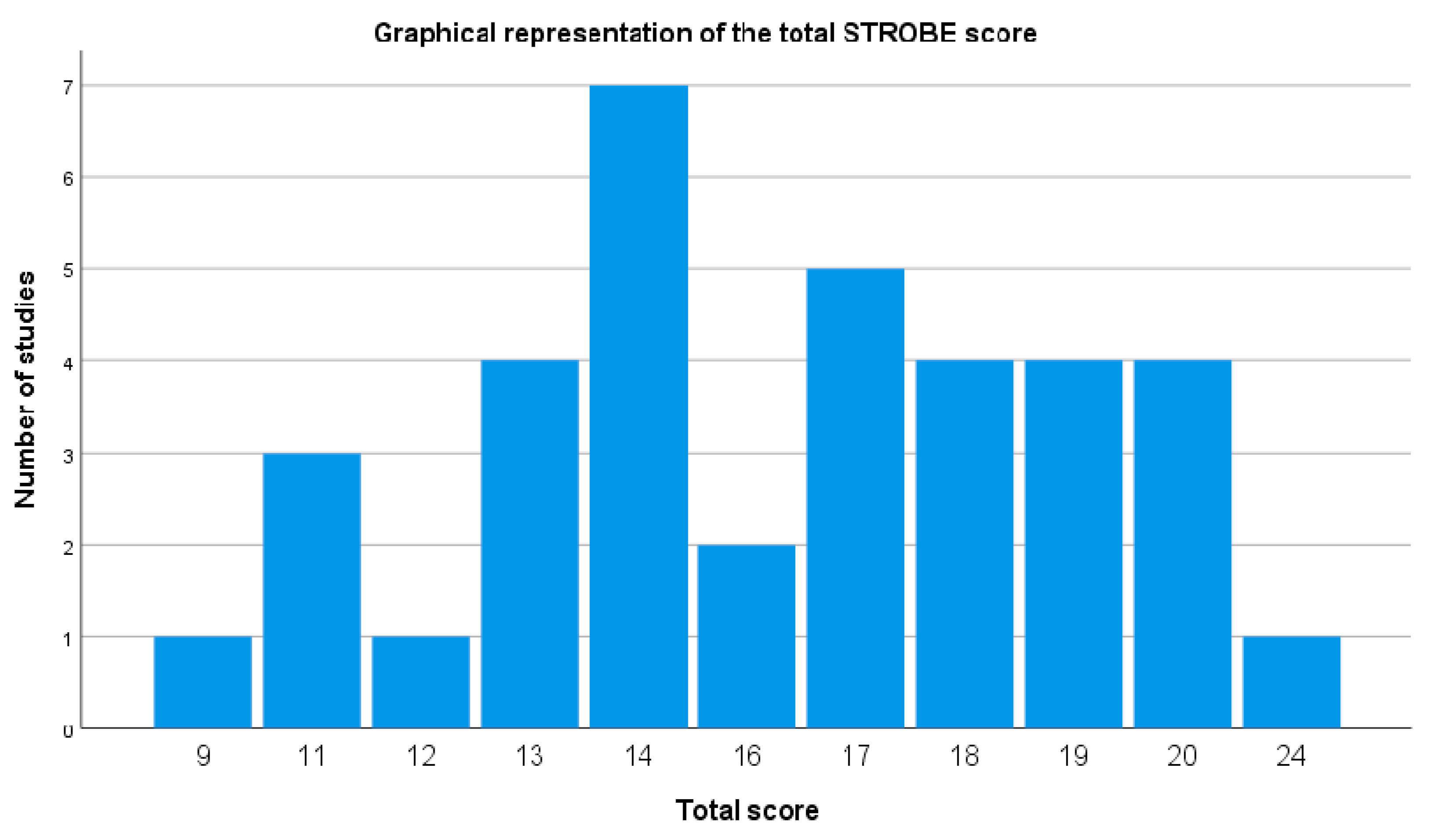
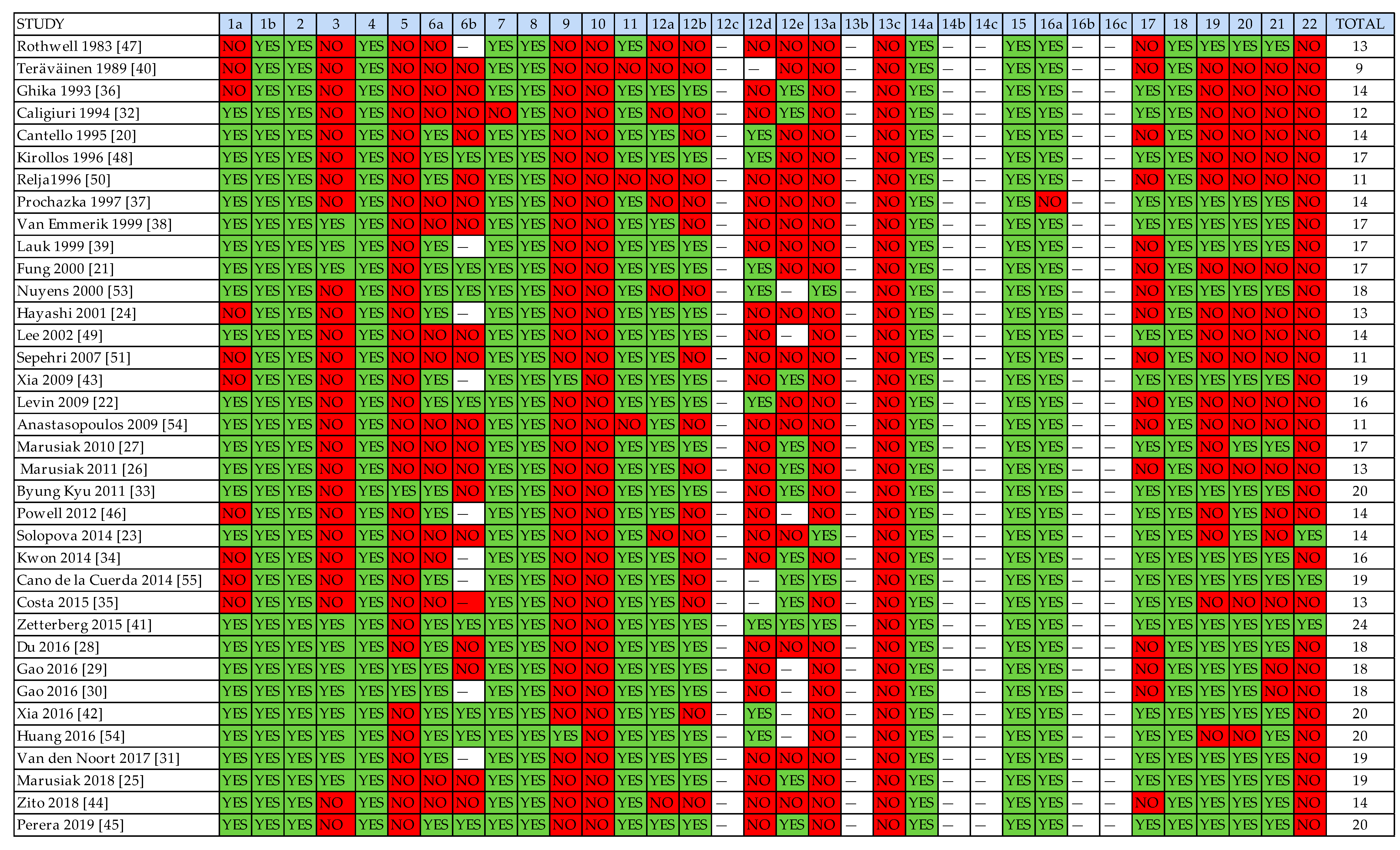
3.3. Methodological Rigor of Included Studies
4. Discussion
4.1. Neurophysiological Mechanisms of Rigidity
4.2. Relevant Parameters in the Evaluation of Rigidity: Angular Speed, ROM, and Other Factors
4.3. Dopaminergic Medication and Rigidity
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tysnes, O.B.; Storstein, A. Epidemiology of Parkinson’s disease. J. Neural Transm. 2017, 124, 901–905. [Google Scholar] [CrossRef] [PubMed]
- Mutch, W.; Strudwick, A.; Roy, S.; Downie, A. Parkinson’s disease: Disability, review, and management. Br. Med. J. (Clin. Res. Ed.) 1986, 293, 675–677. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, A.A.; Chakravarthy, S.; Phillips, J.R.; Gupta, A.; Keri, S.; Polner, B.; Frank, M.J.; Jahanshahi, M. Motor symptoms in Parkinson’s disease: A unified framework. Neurosci. Biobehav. Rev. 2016, 68, 727–740. [Google Scholar] [CrossRef] [PubMed]
- Berardelli, A.; Sabra, A.; Hallett, M. Physiological mechanisms of rigidity in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1983, 46, 45–53. [Google Scholar] [CrossRef]
- Patrick, S.K.; Denington, A.A.; Gauthier, M.J.A.; Member, A.; Gillard, D.M.; Prochazka, A.; Member, S. Quantification of the UPDRS Rigidity Scale. IEEE Trans. Neural Syst. Rehabil. Eng. 2001, 9, 31–41. [Google Scholar] [CrossRef]
- Di Biase, L.; Summa, S.; Tosi, J.; Taffoni, F.; Marano, M.; Cascio Rizzo, A.; Vecchio, F.; Formica, D.; Di Lazzaro, V.; Di Pino, G.; et al. Quantitative Analysis of Bradykinesia and Rigidity in Parkinson’s Disease. Front. Neurol. 2018, 9, 121. [Google Scholar] [CrossRef]
- Mak, M.K.; Wong, E.C.; Hui-Chan, C.W. Quantitative measurement of trunk rigidity in parkinsonian patients. J. Neurol. 2007, 254, 202–209. [Google Scholar] [CrossRef]
- Wright, W.; Gurfinkel, V.; Nutt, J.; Horak, F.; Cordo, P. Axial hypertonicity in Parkinson’s disease: Direct measurements of trunk and hip torque. Exp. Neurol. 2007, 208, 38–46. [Google Scholar] [CrossRef]
- Rinalduzzi, S.; Trompetto, C.; Marinelli, L.; Alibardi, A.; Missori, P.; Fattapposta, F.; Pierelli, F.; Currà, A. Balance Dysfunction in Parkinson’s Disease. BioMed Res. Int. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Sánchez-Ferro, Á.; Elshehabi, M.; Godinho, C.; Salkovic, D.; Hobert, M.A.; Domingos, J.; van Uem, J.M.; Ferreira, J.J.; Maetzler, W. New methods for the assessment of Parkinson’s disease (2005 to 2015): A systematic review. Mov. Disord. 2016, 31, 1283–1292. [Google Scholar] [CrossRef]
- Eisen, A. Electromyography in disorders of muscle tone. Can. J. Neurol. Sci. 1987, 14, 501–505. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Battista, L.; Romaniello, A. A novel device for continuous monitoring of tremor and other motor symptoms. Neurol. Sci. 2018, 39, 1333–1343. [Google Scholar] [CrossRef]
- Bravo, M.; Bermeo, A.; Huerta, M.; Llumiguano, C.; Bermeo, J.; Clotet, R.; Soto, A. A system for finger tremor quantification in patients with Parkinson’s disease. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, EMBS, Seogwipo, Korea, 11–15 July 2017; Institute of Electrical and Electronics Engineers Inc.: Piscataway, NJ, USA, 2017; pp. 3549–3552. [Google Scholar]
- Sanchez-Perez, L.A.; Sanchez-Fernandez, L.P.; Shaout, A.; Martinez-Hernandez, J.M.; Alvarez-Noriega, M.J. Rest tremor quantification based on fuzzy inference systems and wearable sensors. Int. J. Med. Inform. 2018, 114, 6–17. [Google Scholar] [CrossRef] [PubMed]
- Memar, S.; Delrobaei, M.; Pieterman, M.; McIsaac, K.; Jog, M. Quantification of whole-body bradykinesia in Parkinson’s disease participants using multiple inertial sensors. J. Neurol. Sci. 2018, 387, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Teshuva, I.; Hillel, I.; Grazit, E.; Giladi, N.; Mirelman, A.; Hausdorff, J. Using wearables to assess bradykinesia and rigidity in patients with Parkinson’s disease: A focused, narrative review of the literature. J. Neural Transm. 2019, 126, 669–710. [Google Scholar] [CrossRef] [PubMed]
- Palakurthi, B.; Burugupally, S.P. Postural instability in parkinson’s disease: A review. Brain Sci. 2019, 9, 239. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Altman, D.; Antes, G.; Atkins, D.; Barbour, V.; Barrowman, N.; Berlin, J.A.; et al. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Lancet 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- Cantello, R.; Gianelli, M.; Civardi, C.; Mutani, R. Parkinson’s disease rigidity: EMG in a small hand muscle at ‘rest’. Electroencephalogr. Clin. Neurophysiol. 1995, 97, 215–222. [Google Scholar] [CrossRef]
- Fung, V.; Burne, J.; Morris, J. Objective quantification of resting and activated parkinsonian rigidity: A comparison of angular impulse and work scores. Mov. Disord. 2000, 15, 48–55. [Google Scholar] [CrossRef]
- Levin, J.; Krafczyk, S.; Valkovic, P.; Eggert, T.; Claassen, J.; Bötzel, K. Objective measurement of muscle rigidity in Parkinsonian patients treated with subthalamic stimulation. Mov. Disord. 2009, 24, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Solopova, I.; Selionov, V.; Zhvansky, D.; Ivanenko, Y.; Chernikova, L. Investigation of muscle tone in patients with Parkinson’s disease in unloading conditions. Hum. Physiol. 2014, 40, 125–131. [Google Scholar] [CrossRef]
- Hayashi, R.; Hashimoto, T.; Tada, T.; Ikeda, S. Relation between changes in long-latency stretch reflexes and muscle stiffness in Parkinson’s disease—Comparison before and after unilateral pallidotomy. Clin. Neurophysiol. 2001, 112, 1814–1821. [Google Scholar] [CrossRef]
- Marusiak, J.; Jarocka, E.; Jaskólska, A.; Jaskólski, A. Influence of number of records on reliability of myotonometric measurements of muscle stiffness at rest and contraction. Acta Bioeng. Biomech. 2018, 20, 123–131. [Google Scholar] [PubMed]
- Marusiak, J.; Jaskólska, A.; Budrewicz, S.; Koszewicz, M.; Jaskólski, A. Increased muscle belly and tendon stiffness in patients with Parkinson’s disease, as measured by myotonometry. Mov. Disord. 2011, 26, 2119–2122. [Google Scholar] [CrossRef]
- Marusiak, J.; Kisiel-Sajewicz, K.; Jaskólska, A.; Jaskólski, A. Higher Muscle Passive Stiffness in Parkinson’s Disease Patients Than in Controls Measured by Myotonometry. Arch. Phys. Med. Rehabil. 2010, 91, 800–802. [Google Scholar] [CrossRef]
- Du, L.-J.; He, W.; Cheng, L.-G.; Li, S.; Pan, Y.-S.; Gao, J. Ultrasound shear wave elastography in assessment of muscle stiffness in patients with Parkinson’s disease: A primary observation. Clin. Imaging 2016, 40, 1075–1080. [Google Scholar] [CrossRef]
- Gao, J.; He, W.; Du, L.-J.; Li, S.; Cheng, L.-G.; Shih, G.; Rubin, J. Ultrasound strain elastography in assessment of resting biceps brachii muscle stiffness in patients with Parkinson’s disease: A primary observation. Clin. Imaging 2016, 40, 440–444. [Google Scholar] [CrossRef]
- Gao, J.; Du, L.-J.; He, W.; Li, S.; Cheng, L.-G. Ultrasound Strain Elastography in Assessment of Muscle Stiffness in Acute Levodopa Challenge Test: A Feasibility Study. Ultrasound Med. Biol. 2016, 42, 1084–1089. [Google Scholar] [CrossRef]
- van den Noort, J.C.; Verhagen, R.; van Dijk, K.J.; Veltink, P.H.; Vos, M.C.P.M.; de Bie, R.M.A.; Bour, L.J.; Heida, C.T. Quantification of Hand Motor Symptoms in Parkinson’s Disease: A Proof-of-Principle Study Using Inertial and Force Sensors. Ann. Biomed. Eng. 2017, 45, 2423–2436. [Google Scholar] [CrossRef]
- Caligiuri, M.P. Portable device for quantifying parkinsonian wrist rigidity. Mov. Disord. 1994, 9, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Park, B.K.; Kwon, Y.; Kim, J.-W.; Lee, J.-H.; Eom, G.-M.; Koh, S.-B.; Jun, J.-H.; Hong, J. Analysis of Viscoelastic Properties of Wrist Joint for Quantification of Parkinsonian Rigidity. IEEE Trans. Neural Syst. Rehabil. Eng. 2011, 19, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.; Park, S.-H.; Kim, J.-W.; Ho, Y.; Jeon, H.-M.; Bang, M.-J.; Koh, S.-B.; Kim, J.-H.; Eom, G.-M. Quantitative evaluation of parkinsonian rigidity during intra-operative deep brain stimulation. Biomed. Mater. Eng. 2014, 24, 2273–2281. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.; Rosas, M.J.; Vaz, R.; Cunha, J.P. Wrist rigidity assessment during Deep Brain Stimulation surgery. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2015, 2015, 3423–3426. [Google Scholar]
- Ghika, J.; Wiegner, A.; Fang, J.; Davies, L.; Young, R.; Growdon, J. Portable system for quantifying motor abnormalities in Parkinson’s disease. IEEE Trans. Biomed. Eng. 1993, 40, 276–283. [Google Scholar] [CrossRef]
- Prochazka, A.; Bennett, D.J.; Stephens, M.J.; Patrick, S.K.; Sears-Duru, R.; Roberts, T.; Jhamandas, J.H. Measurement of rigidity in Parkinson’s disease. Mov. Disord. 1997, 12, 24–32. [Google Scholar] [CrossRef]
- Van Emmerik, R.E.; Wagenaar, R.C.; Winogrodzka, A.; Wolters, E.C. Identification of axial rigidity during locomotion in Parkinson disease. Arch. Phys. Med. Rehabil. 1999, 80, 186–191. [Google Scholar] [CrossRef]
- Lauk, M.; Chow, C.C.; Lipsitz, L.A.; Mitchell, S.L.; Collins, J.J. Assessing muscle stiffness from quiet stance in Parkinson’s disease. Muscle Nerve 1999, 22, 635–639. [Google Scholar] [CrossRef]
- Teräväinen, H.; Tsui, J.; Mak, E.; Calne, D. Optimal indices for testing parkinsonian rigidity. Can. J. Neurol. Sci. 1989, 16, 180–183. [Google Scholar] [CrossRef]
- Zetterberg, H.; Frykberg, G.; Gäverth, J.; Lindberg, P. Neural and nonneural contributions to wrist rigidity in Parkinson’s disease: An explorative study using the NeuroFlexor. BioMed Res. Int. 2015, 2015, 276182. [Google Scholar] [CrossRef]
- Xia, R.; Muthumani, A.; Mao, Z.-H.; Powell, D.W. Quantification of neural reflex and muscular intrinsic contributions to parkinsonian rigidity. Exp. Brain Res. 2016, 234, 3587–3595. [Google Scholar] [CrossRef] [PubMed]
- Xia, R.; Sun, J.; Threlkeld, A.J. Analysis of interactive effect of stretch reflex and shortening reaction on rigidity in Parkinson’s disease. Clin. Neurophysiol. 2009, 120, 1400–1407. [Google Scholar] [CrossRef] [PubMed]
- Zito, G.A.; Gerber, S.M.; Urwyler, P.; Shamsollahi, M.J.; Pal, N.; Benninger, D.; Nef, T. Development and Pilot Testing of a Novel Electromechanical Device to Measure Wrist Rigidity in Parkinson’s Disease. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; pp. 4885–4888. [Google Scholar]
- Perera, T.; Lee, W.-L.; Jones, M.; Tan, J.L.; Proud, E.L.; Begg, A.; Sinclair, N.C.; Peppard, R.; McDermott, H.J. A palm-worn device to quantify rigidity in Parkinson’s disease. J. Neurosci. Methods 2019, 317, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Powell, D.; Threlkeld, A.J.; Fang, X.; Muthumani, A.; Xia, R. Amplitude- and velocity-dependency of rigidity measured at the wrist in Parkinson’s disease. Clin. Neurophysiol. 2012, 123, 764–773. [Google Scholar] [CrossRef] [PubMed]
- Rothwell, J.; Obeso, J.; Traub, M.; Marsden, C. The behaviour of the long-latency stretch reflex in patients with Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1983, 46, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Kirollos, C.; Charlett, A.; O’Neill, C.; Kosik, R.; Mozol, K.; Purkiss, A.; Bowes, S.; Nicholson, P.; Hunt, W.B.; Weller, C.; et al. Objective measurement of activation of rigidity: Diagnostic, pathogenetic and therapeutic implications in parkinsonism. Br. J. Clin. Pharmacol. 1996, 41, 557–564. [Google Scholar] [CrossRef]
- Lee, H.; Huang, Y.; Chen, J.J.; Hwang, I. Quantitative analysis of the velocity related pathophysiology of spasticity and rigidity in the elbow flexors. J. Neurol. Neurosurg. Psychiatry 2002, 72, 621–629. [Google Scholar] [CrossRef]
- Relja, M.; Petravic, D.; Kolaj, M. Quantifying rigidity with a new computerized elbow device. Clin. Neuropharmacol. 1996, 19, 148–156. [Google Scholar] [CrossRef]
- Sepehri, B.; Esteki, A.; Ebrahimi-Takamjani, E.; Shahidi, G.-A.; Khamseh, F.; Moinodin, M. Quantification of Rigidity in Parkinson’s Disease. Ann. Biomed. Eng. 2007, 35, 2196–2203. [Google Scholar] [CrossRef]
- Huang, H.-W.; Ju, M.-S.; Lin, C.-C.K. Flexor and extensor muscle tone evaluated using the quantitative pendulum test in stroke and parkinsonian patients. J. Clin. Neurosci. 2016, 27, 48–52. [Google Scholar] [CrossRef]
- Nuyens, G.; De Weerdt, W.; Dom, R.; Nieuwboer, A.; Spaepen, A. Torque variations during repeated passive isokinetic movements of the knee in subjects with Parkinson’s disease and healthy control subjects. Parkinsonism Relat. Disord. 2000, 6, 87–93. [Google Scholar] [CrossRef]
- Anastasopoulos, D.; Maurer, C.; Nasios, G.; Mergner, T. Neck rigidity in Parkinson’s disease patients is related to incomplete suppression of reflexive head stabilization. Exp. Neurol. 2009, 217, 336–346. [Google Scholar] [CrossRef] [PubMed]
- Cano-de-la-Cuerda, R.; Vela-Desojo, L.; Miangolarra-Page, J.C.; Macías-Macías, Y. Isokinetic dynamometry as a technologic assessment tool for trunk rigidity in Parkinson’s disease patients. NeuroRehabilitation 2014, 35, 493–501. [Google Scholar] [CrossRef]
- Delwaide, P.J. Parkinsonian rigidity. Funct. Neurol. 2001, 16, 147–156. [Google Scholar] [PubMed]
- Kwon, Y.; Kim, J.W.; Kim, J.S.; Koh, S.B.; Eom, G.M.; Lim, T.H. Comparison of EMG during passive stretching and shortening phases of each muscle for the investigation of parkinsonian rigidity. Biomed. Mater. Eng. 2015, 26, S2155–S2163. [Google Scholar] [CrossRef] [PubMed]
- Xia, R.; Rymer, W.Z. The role of shortening reaction in mediating rigidity in Parkinson’s disease. Exp. Brain Res. 2004, 156, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Diener, C.; Scholz, E.; Guschlbauer, B.; Dichagans, J. Increased shortening reaction in Parkinson’s disease reflects a difficulty in modulating long loop reflexes. Mov. Disord. 1987, 2, 31–36. [Google Scholar] [CrossRef]
- Forgaard, C.J.; Franks, I.M.; Maslovat, D.; Chua, R. Perturbation predictability can influence the long-latency stretch response. PLoS ONE 2016, 11, e0163854. [Google Scholar] [CrossRef]
- Lewis, G.N.; Polych, M.A.; Byblow, W.D. Proposed cortical and sub-cortical contributions to the long-latency stretch reflex in the forearm. Exp. Brain Res. 2004, 156, 72–79. [Google Scholar]
- Eklund, G.; Hagbarth, K.; Hägglund, J.; Wallin, E. The ‘late’ reflex responses to muscle stretch: The ‘resonance hypothesis’ versus the ‘long-loop hypothesis’. J. Physiol. 1982, 326, 79–90. [Google Scholar] [CrossRef]
- Rossi, B.; Siciliano, G.; Carboncini, M.C.; Manca, M.L.; Massetani, R.; Viacava, P.; Muratorio, A. Muscle modifications in Parkinson’s disease: Myoelectric manifestations. Electroencephalogr. Clin. Neurophysiol. Electromyogr. Mot. Control 1996, 101, 211–218. [Google Scholar] [CrossRef]
- Raza, C.; Anjum, R.; Shakeel, A. Parkinson’s disease: Mechanisms, translational models and management strategies. Life Sci. 2019, 226, 77–90. [Google Scholar] [CrossRef] [PubMed]

| Database | Specific Filters | Search Strategy and Key Words |
|---|---|---|
| Web of Science |
| [“rigidity” OR “hypertonia” OR “muscle stiffness”] AND [“evaluation” OR “assessment” OR “quantification”] AND [“Parkinson” OR “parkinsonian”] |
| PubMed |
| |
| Scopus |
|
| Study | Study Design | Sample | Assessment Method | Joint Explored | Evaluation Protocol | Results |
|---|---|---|---|---|---|---|
| Cantello, 1995 [20] | Case–control | 8 people with PD 8 healthy controls | Surface EMG on hand muscles. | Hand | Tested on the most affected side. Basal muscle activity was measured (at rest) with a maintained contraction for 16 s. Number of action potentials by motor unit, firing rate, and recruitment order were obtained. | EMG activity at rest was significantly higher in PD in comparison with healthy controls after relaxation time. This was a consequence of the discharge of a greater number of different motor units (5.9 ± 2.7 in control group vs. 20.9 ± 5.3 in PD group) (excessive recruitment). Firing rate was not significantly different between groups. |
| Fung, 2000 [21] | Case–control | 20 people with PD 10 healthy controls | Surface EMG on flexor carpi radialis and extensor carpi radialis while a servomotor passively moves the wrist. | Wrist | Tested in “off” phase, and on the most affected side. In controls, half of the tests performed on each side. Tested at rest and with contralateral activation maneuver. Five records in 60° of ROM with rest intervals were collected. Tests were performed at two cycle frequencies (at 1 and 1.5 Hz). A qualitative scale to evaluate rigidity (CRS) was administered. | Examiner tended to use quasi-sinusoidal mobilizations at frequencies between 0.5 and 2 Hz, and with articular amplitudes of ± 40°. Activation produced a significant increase in CRS when compared to rest in both groups, but the magnitude of this increase was greater in PD. Angular impulse scores were significantly higher in PD, both at rest and with activation maneuver. There were no differences between groups for work scores in any case. In PD, high correlation between CRS and angular impulse was confirmed (work score was not well correlated). Better correlations were found at 1.5 Hz. Activation maneuver increased angular impulse scores. |
| Levin, 2009 [22] | Case–control | 13 people with PD 8 healthy controls | Surface EMG on biceps brachii and triceps brachii. | Elbow | Tested in “off” phase (deep brain stimulation -DBS- electrodes turned off) and “on” phase (30 s after DBS electrodes turned on). Tested on the most affected side. Elbow was flexo-extended (90° amplitude) by an examiner from total extension to 90° flexion (it was registered by a goniometer). Cycles were made at 0.5 Hz. All individuals were tested twice while EMG was recorded for 180 s. | People with PD had greater EMG activity only during middle and final stretch phases compared with healthy controls. During the first two phases, there were no significant differences between groups. There were different EMG patterns when comparing people with high scores (UPDRS 3–4) and lower scores (0–1) in clinical rigidity. In both muscles, people with PD with low clinical rigidity exhibited differences compared with healthy controls. |
| Solopova, 2014 [23] | Case–control | 25 people with PD 22 healthy controls | Surface EMG (bilateral) on rectus femoris, biceps femoris, tibialis anterior, and gastrocnemius lateralis muscles. A potentiometer sensor fixed coaxially with lower limb joints. | Lower limb (hip, knee, and ankle) | Two recordings in “off” phase and one in “on” phase. Passive movement at 7°/s of each joint in isolation, in a range of ± 20° (ankle ± 10°). Each movement was examined three times. The resistance force, the shortening reaction (mean value of EMG values eliminating the basal activity in each test), and the latency period of the test were recorded. | Hip muscles stiffness was 1.5 and 1.7 times higher in people with PD than healthy controls for flexors and extensors, respectively (p < 0.05). In the hip, flexors exhibited greater rigidity than extensors (p < 0.05) in both groups, as well as in the knee (1.5 and 1.6 times). In the ankle, dorsiflexors were 1.3 times more rigid in people with PD than controls. Plantar flexors exhibited similar stiffness levels in both groups (p = 0.3). Plantar flexors were significantly more rigid than dorsal flexors in PD patients. Levodopa improved hip and knee stiffness but not ankle stiffness. |
| Hayashi, 2001 [24] | Pre–post design | 11 people with PD | Surface EMG on flexor carpi radialis and extensor carpi radialis. | Wrist | Assessment before (“off” state) and 2–3 months after (“on” state) pallidotomy surgery. A servomotor moved the wrist while EMG is recorded and M1 and M2 and their amplitudes were obtained. The two conditions were passive (participant was instructed not to resist the movement) and active (participant was instructed to suddenly resist). | There were no significant differences between active and passive conditions at either preoperative or postoperative stages for M1 parameter or basal activity. For M2 parameter, significant differences were found after pallidotomy in active condition (M2 amplitude decreases). In passive condition, the tendency was a decrease, but statistical signification was not reached. Pallidotomy had no effects on inherent muscle stiffness. |
| Marusiak, 2018 [25] | Case–control | 8 people with PD 10 healthy controls | Myotonometry (Myoton-3 device) on biceps brachii (short head) and triceps brachii (long head). | Elbow | Tested on the most affected side; in controls, on dominant side. Test at rest (20 consecutive recordings with 1 s interval between each) and with a voluntary contraction of 10% of the MVC. In the second condition, elbow was placed at 15° flexion and the subject had to hold a 2 kg weight for 1 min while 20 recordings were collected. Test–retest reliability was assessed. | Myotonometry had high test–retest reliability (ICC > 0.9), both at rest and at 10% of the MVC. |
| Marusiak, 2011 [26] | Case–control | 12 people with PD 12 healthy controls | Myometry (Myoton-3 device) on biceps brachii and triceps brachii (belly and tendon). | Elbow | Tested in “on” state. Twenty consecutive recordings were collected in the following order (biceps belly, biceps tendon, triceps belly, triceps tendon). EMG and MMG data were also collected, as well as the elbow ROM at rest. | Myometry had excellent reproducibility. There were no differences between groups for EMG and MMG. There were differences between groups in the myotonometric examination of the bicep belly and tendons. In the triceps this was true in the tendons (p < 0.05), but not so in the belly of the triceps. |
| Marusiak, 2010 [27] | Case–control | 8 people with PD 10 healthy controls | Myotonometry (Myoton-3 device) on biceps brachii (short head). | Elbow | Tested in “on” phase, and on the most affected side; in controls, on the dominant side. Twenty records were collected, in addition to EMG and MMG. Reproducibility, group differences, and correlation with clinical scales and EMG/MMG were analysed. | Good reproducibility in both groups (ICC > 0.9). PD group exhibited higher stiffness values than control group (203 ± 22 N/m vs. 192 ± 8 N/m, p = 0.004). There was a positive correlation between myotonometry and clinical rigidity scores. |
| Du, 2016 [28] | Case–control | 46 people with PD 31 healthy controls | Shear wave elastography (AixPlorer) on biceps brachii. | Elbow | The transducer was placed in the muscle belly to collect grayscale images and then over a color code. Three measurements were collected and de Young’s modulus (longitudinal elasticity modulus) was obtained. | Young’s modulus had a moderate positive correlation with UPDRS-III (r = 0.646; p = 0.000). Inter- and intra-examiner reliability was good (ICC = 0.74 and 0.78, respectively). For Young’s modulus, there were differences between PD (54.94 ± 20.91 Kpa on the most affected side and 47.77 ± 24 Kpa on less affected side) and healthy people (24.44 ± 5.09 Kpa). |
| Gao, 2016 [29] | Case–control | 14 people with PD 10 healthy controls | Elastography (Logic E9 ultrasound scanner) on biceps brachii | Elbow | Tested in “off” phase. A 5-s cycle was performed to collect the deformation-relaxation cycle. Three measurements were made on each subject. To exert the same pressure on the skin, a weight was placed on the end of the apparatus. Biceps stress, reference stress, and stress ratio (mean biceps stress divided by mean reference stress) were measured. | Stress ratio was significantly higher in the PD group with respect to control group (3.3 ± 0.27 vs. 2.65 ± 0.6; p = 0.00011). A correlation was found between stress ratio and UPDRS rigidity subscale score. Reliability of measurements was good (ICC intra-observer = 0.88, ICC inter-observer = 0.84). |
| Gao, 2016 [30] | Case–control | 11 people with PD 7 persons with parkinsonism | Elastography (Logic E9 ultrasound scanner) on biceps brachii. | Elbow | Tested in “off” and “on” phases. A 5 s cycle was performed to collect the deformation–relaxation cycle. Three measurements were made on each subject. To exert the same pressure on the skin, a weight was placed on the end of the apparatus. Biceps stress, reference stress, and stress ratio (mean biceps stress divided by mean reference stress) were measured. | PD group exhibited significant differences in stress rate in “off” phase with respect to “on” phase (2.86 ± 0.51 vs. 4.06 ± 0.78; p = 0.02). This was also true for parkinsonism group (2.56 ± 0.23 vs. 2.87 ± 0.37; p = 0.14). There were significant statistically differences between groups for the increase in the stress ratio parameter (p = 0.0007) in favor of PD. |
| Study | Study Design | Sample | Assessment Method | Joint Explored | Evaluation Protocol | Results |
|---|---|---|---|---|---|---|
| Van den Noort, 2017 [31] | Technology reliability | 4 people with PD | Sensor complex (PowerGlove system) with gyroscopes, accelerometers, and magnetometers. | Wrist and hand | Test in “on” and “off” phase. After calibration, the wrist was passively flexo-extended by an examiner while sensors collected data regarding angular velocity, ROM, torque, rigidity, impulse, and work. | There were significant differences between “off” and “on” phases. ROM increased, while torque, rigidity, impulse, and work scores decreased. |
| Caligiuri, 1994 [32] | Case–control | 29 people with PD 25 healthy controls | A portable transducer with gyroscope and potentiometer. | Wrist | Test performed at rest and with contralateral activation maneuver. Examiner passively moved the wrist (flexion and extension) within a 45° ROM over at least 15 cycles. | The group of PD with clinical rigidity presented higher mean instrumental rigidity scores than control group (1.57 vs. 1.09; p < 0.0001), as well as higher scores than PD without clinical rigidity group (1.57 vs. 1.05; p = 0.0001). During “off”- vs. “on” phase examination (n = 4), instrumental rigidity score was reduced by 26% after levodopa/carbidopa administration. |
| Byung Kyu, 2011 [33] | Case–control | 45 people with PD 12 healthy controls | A sensor with potentiometer, load cell, and accelerometer. | Wrist | Tested in “on” phase. Movement was applied by an examiner. Random flexion and extension (1–5 s) and random rest (2–10 s) within a ROM of −35° and +35°. Six flexion and extension cycles were applied, between 2–4 times each, bilaterally. | Damping viscosity constant was well correlated with clinical rigidity measured by UPDRS. Tissue resistance (viscosity) was greater during extension. Velocity dependence of rigidity was more pronounced in subjects with greater clinical rigidity scores. Viscosity measure was useful for detecting muscle stiffness. |
| Kwon, 2014 [34] | Technology reliability | 8 people with PD | A sensor with potentiometer, load cell, and accelerometer. | Wrist | Tested in “off” (before DBS surgery) and “on” states (after surgery). “Off” and “on” data were compared with each other and with data collected by a neurologist. | Damping viscosity constant was well correlated with clinical rigidity (Spearman coefficient = 0.77), and was the variable that improved the most after DBS (2.38 pre vs. 0.39 post; p < 0.001). The other variables showed moderate to low correlation (Spearman < 0.7), although all improved significantly after surgery (p < 0.05) with the exception of mechanical impedance. |
| Costa, 2015 [35] | Technology reliability | 10 people with PD | Sensor with a gyroscope, accelerometer, and magnetometer. | Wrist | ROM and angular velocity were obtained during flexion and extension while DBS surgery was performed. These data were compared with each other and with data collected by a neurologist. | The device was capable of distinguishing between rigidity and normal tone (83.9%), and its measurements were correlated with clinical rigidity with an error margin of 8.24% ± 7.95%. The system was useful for discriminating discrete rigidity levels and for detecting cog wheel rigidity with good sensitivity (0.93). |
| Ghika, 1993 [36] | Case–control | 20 people with PD 15 healthy controls 20 healthy controls (young) | A semimanual device with goniometer and potentiometer. | Elbow | An examiner passively moved the elbow with 0.67 Hz angular velocity (using a metronome) in a ROM of 45°. The sensor recorded data on angular movement and torque. | Obtained values were correlated with clinical rigidity. PD group had significantly greater values compared with the others in the mean torque (right side 0.391 Nm/degree and left side 0.388 Nm/degree vs. 0.054 and 0.044 Nm/degree, respectively, for the age-matched controls; p < 0.05). |
| Prochazka, 1997 [37] | Proof of concept | 14 people with PD | Sensors with potentiometer to measure the force applied by the examiner at the wrist. A sensor | Elbow | Wrist sensors measured the force applied by the examiner to move the forearm. Additionally, a string was placed with its limits adhered to the shoulder and the forearm of the subject. The string measured elbow ROM during the examination. | Rigidity was significantly reduced after medication administration. Clinical evaluation had very low reliability. The results of the evaluation were influenced by factors such as velocity or ROM, and also attention of the subject, reinforcement maneuver, and distractors. Impedance and rigidity profiles that were generated by examiners were similar, but high differences in the qualitative interpretation of rigidity existed. |
| with a string to measure the elbow ROM. | ||||||
| Van Emmerik, 1999 [38] | Case–control | 27 people with PD 11 healthy controls | Optoelectronic system with infrared LEDs on pelvis and thorax. Accelerometers on both tibias. | Trunk | Analysis during gait. Angular rotation of pelvis and trunk and their velocities were obtained in order to record a relative phase measure along with its variability. | PD group exhibited lower relative phase mean duration and lower variability of this factor. in both groups, relative phase increased as velocity increased, without any group differences. There were no differences between groups for stride duration or its variability. |
| Lauk, 1999 [39] | Technology reliability | 18 people with PD | Centre of pressure (CoP) platform (Kistler). | Whole body | Tested in “on” phase. Ten 30 s tests with open eyes were made. A 60 s rest period between tests was allowed. A “postural rigidity measure” (k) was obtained through a mathematical model that used information regarding AP displacements of the CoP. Reliability was assessed by randomly by separating the tests into 2 groups of 5 tests each and comparing them. UPDRS was administered. | Positive correlations with the UPDRS items of rigidity, bradykinesia, posture, lower limb agility, and retropulsion test were found. There was no correlation with rest tremor, postural tremor, or the getting up items. Results showed that the measure proposed had significant correlations with other relevant variables. |
| Study | Study Design | Sample | Assessment Method | Joint Explored | Evaluation Protocol | Results |
|---|---|---|---|---|---|---|
| Teräväinen, 1989 [40] | Case–control | 29 people with PD 12 healthy controls | Servomotor with position feedback. | Wrist | Movements of flexion and extension were applied in a passive way and in contralateral activation, over a range of angular velocities from 12°/s to 240°/s, and over angular displacements from 15° to 30°. | Higher velocities gave more sensitive results than the lower ones in the assessment of rigidity. Wide angular displacements and velocities gave the highest correlations between objective evaluation and the CRS. |
| Zetterberg, 2015 [41] | Case–control | 25 people with PD 14 healthy controls | Servomotor (NeuroFlexor). | Wrist | Flexion and extension movements were applied in a range of 50°, at two velocities of 5°/s and 236°/s, under passive and dynamic conditions. Bilateral exploration: less affected side was dominant side; more affected side was non-dominant side. | PD showed greater resistance and neural component than controls, with no difference in components between sides. Total resistance was greater in the dynamic test in both groups. |
| Xia, 2016 [42] | Case–control | 14 people with PD 14 healthy controls | Servomotor and surface EMG. | Wrist | Tested without antiparkinsonian medication (at least for 12 h) and under medication (45–60 min before the test). Flexion and extension movements in minimum displacements. | There were significant differences in neural components between “on” and “off” states. |
| Xia, 2009 [43] | Case series | 12 people with PD | Servomotor with EMG. | Wrist | Tested with and without antiparkinsonian medication. Flexion and extension movements were applied in a range of motion of ±30° at two velocities: 50°/s and 280°/s. | There was a correlation between the torque resistance and the ration values of EMG. There was a correlation between the activation of the stretched and shortened muscles. Direction and speed of the passive movement had a great influence on rigidity. |
| Zito, 2018 [44] | Case–control | 4 people with PD 18 healthy controls | Servomotor (exoskeleton wrist resistance robot (WRR)). | Wrist | Flexion and extension movements in a range of motion from −60° to +30° were applied, at 10°/s and 50°/s. The most affected wrist was evaluated in PD group and non-dominant wrist was evaluated in controls. | Significative differences were found in position, speed. and torque for both groups, at every speed. |
| Perera, 2019 [45] | Case–control | 8 people with PD 8 age-matched healthy controls 8 young healthy controls | Servomotor (Bionics Institute Rigidity Device (BiRD)). | Wrist | A cycle per second of flexion/extension movement was executed. Each evaluation consisted of 15 cycles. | Force rate was significantly greater in PD vs. controls, both at rest and during activity. This allowed for detection of “on” and “off” states. A moderate congruence (R = 0.68) was shown with Movement Disorders Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS). |
| Powell, 2012 [46] | Case series | 18 people with PD | Servomotor and EMG. | Wrist | Tested with and without antiparkinsonian medication. Flexion and extension movements in a range of 60° and 90°, at two speeds of 50°/s and 280°/s. | Higher displacements (90 vs. 60) were associated with greater rigidity. There were no significant differences between “on” and “off” states. |
| Rothwell, 1983 [47] | Case–control | 47 people with PD 12 healthy controls | Servomotor and EMG. | Elbow and thumb | Thumb: movement of the interphalangeal joint at about 10° of flexion. Elbow: resting position at 90° flexion. Forces of 8, 16, and 24 N were applied. | Stretch reflexes were augmented in patients with moderate PD. The was a positive correlation between the severity of the disease and the magnitude of the stretch reflex. The saturation of the reflexes occurred at velocities greater than 300°/s in both patients and controls. |
| Kirollos, 1996 [48] | Case–control | 2 young healthy controls 2 elderly healthy controls 2 elderly with activation phenomenon but not hypertonia at rest or other parkinsonian signs 2 untreated PD people and mild–moderate rigidity | Servomotor. | Elbow | Contralateral activation by squeezing a sphygmomanometer cuff. Test was started with an acclimatization phase for 2 min to the passive arm movement. Then, 6 tests were executed at 10 s intervals in both resting and contralateral grasping states. | PD required higher work values by unit of displacement. |
| Lee, 2002 [49] | Case–control | 16 people with PD 12 hemiparetic spastic people 12 healthy controls | Servomotor with sensors and electromyography of biceps and triceps. | Elbow | Flexion and extension movements in a range of motion of 75°. Stretch velocities were set at 40, 80, 120, and 160°/s, were applied randomly for | Hemiparesis and PD patients reported significantly greater torque values at higher speeds. Healthy controls revealed low values. |
| PD patients, and a single speed of 175°/s was selected for healthy controls. | ||||||
| Relja, 1996 [50] | Case–control | 24 people with PD 103 healthy controls | Tonometer with a transductor device. | Elbow | Basal and activated rigidity were tested with and without antiparkinsonian medication. Movements of flexion and extension were applied through a 53° angle, at a constant frequency of 0.5Hz. Torque and angular displacements were measured by sensor. Twenty cycles of flexion and extension were executed. | Significant differences in rigidity were found in: (a) basal activation between PD patients and healthy controls; (b) in “on” state vs. “off” states; (c) in contralateral activation vs. passive assessment. |
| Sepehri, 2007 [51] | Case–control | 52 people with PD | Servomotor with a transducer system and a potentiometer. | Elbow | An examiner was trained to execute flexion and extension movements at a constant speed (1 cycle/sec). In some cases, EMG was used to check the absence of voluntary muscle activity. | There was no correlation between age and severity of rigidity. Normalized total hysteresis had the greatest correlation for rigidity. This means that the measure of viscous properties may better score the level of the disease than the elastic ones. |
| Huang, 2016 [52] | Case–control | 21 people with PD 14 people with stroke 22 healthy controls | Servomotor with electric goniometer. | Elbow | Two initial positions were measured, at 50° and 130° of flexion. Six pendulum tests were executed to register the number of oscillations, number of peaks and troughs during the swing, and | Significant differences in the number of oscillations were found between PD vs. controls. There were no differences between PD and stroke patients. Differences in relaxation index were found between controls vs. PD and |
| relaxation index. | stroke. Differences in stiffness coefficient and damping coefficient were found between the two initial positions (greater in extension). | |||||
| Nuyens, 2000 [53] | Case–control | 10 people with PD 10 healthy controls | Isokinetic device with servomotor and electromyography. | Knee | Clinical evaluation was carried out by AS. Knee movements were executed at 60°, 180°, and 300°/s. Torque and EMG activity of quadriceps femoris, hamstrings, and gastrocnemius medialis muscles were recorded. | The greatest torque took place during flexion movements and high speeds. Rigidity depended on speed and direction of the movement, as well as coactivation of stretched and shortened muscles, especially during extension movements. |
| Anastasopoulos, 2009 [54] | Case–control | 14 people with PD 23 healthy controls | Bárány chair with a cervical stabilizer. | Neck | Rotation of head and trunk. Stimuli were applied in darkness and without any auditory cues for the subject. | PD patients had more difficulty relaxing neck muscles than controls. PD showed greater peak torque than control. |
| Cano de la Cuerda, 2014 [55] | Case series | 36 people with PD | Servomotor (Biodex System isokinetic dynamometer). | Trunk | A passive flexion movement was applied in a range of 80°, and an extension movement of 30° was applied at three velocities: 30°/s, 45°/s, and 60°/s. | The greatest resistance was obtained at the end of the range for both movements at every speed. There was a correlation between extensor rigidity and clinical scales (H&Y and UPDRS) for all speeds. There was a correlation between Schwab and England scale and flexor and extensor rigidity. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira-Sánchez, M.d.R.; Moreno-Verdú, M.; Cano-de-la-Cuerda, R. Quantitative Measurement of Rigidity in Parkinson’s Disease: A Systematic Review. Sensors 2020, 20, 880. https://doi.org/10.3390/s20030880
Ferreira-Sánchez MdR, Moreno-Verdú M, Cano-de-la-Cuerda R. Quantitative Measurement of Rigidity in Parkinson’s Disease: A Systematic Review. Sensors. 2020; 20(3):880. https://doi.org/10.3390/s20030880
Chicago/Turabian StyleFerreira-Sánchez, María del Rosario, Marcos Moreno-Verdú, and Roberto Cano-de-la-Cuerda. 2020. "Quantitative Measurement of Rigidity in Parkinson’s Disease: A Systematic Review" Sensors 20, no. 3: 880. https://doi.org/10.3390/s20030880
APA StyleFerreira-Sánchez, M. d. R., Moreno-Verdú, M., & Cano-de-la-Cuerda, R. (2020). Quantitative Measurement of Rigidity in Parkinson’s Disease: A Systematic Review. Sensors, 20(3), 880. https://doi.org/10.3390/s20030880






