Photothermal Effect in Plasmonic Nanotip for LSPR Sensing
Abstract
1. Introduction
2. Materials and Methods
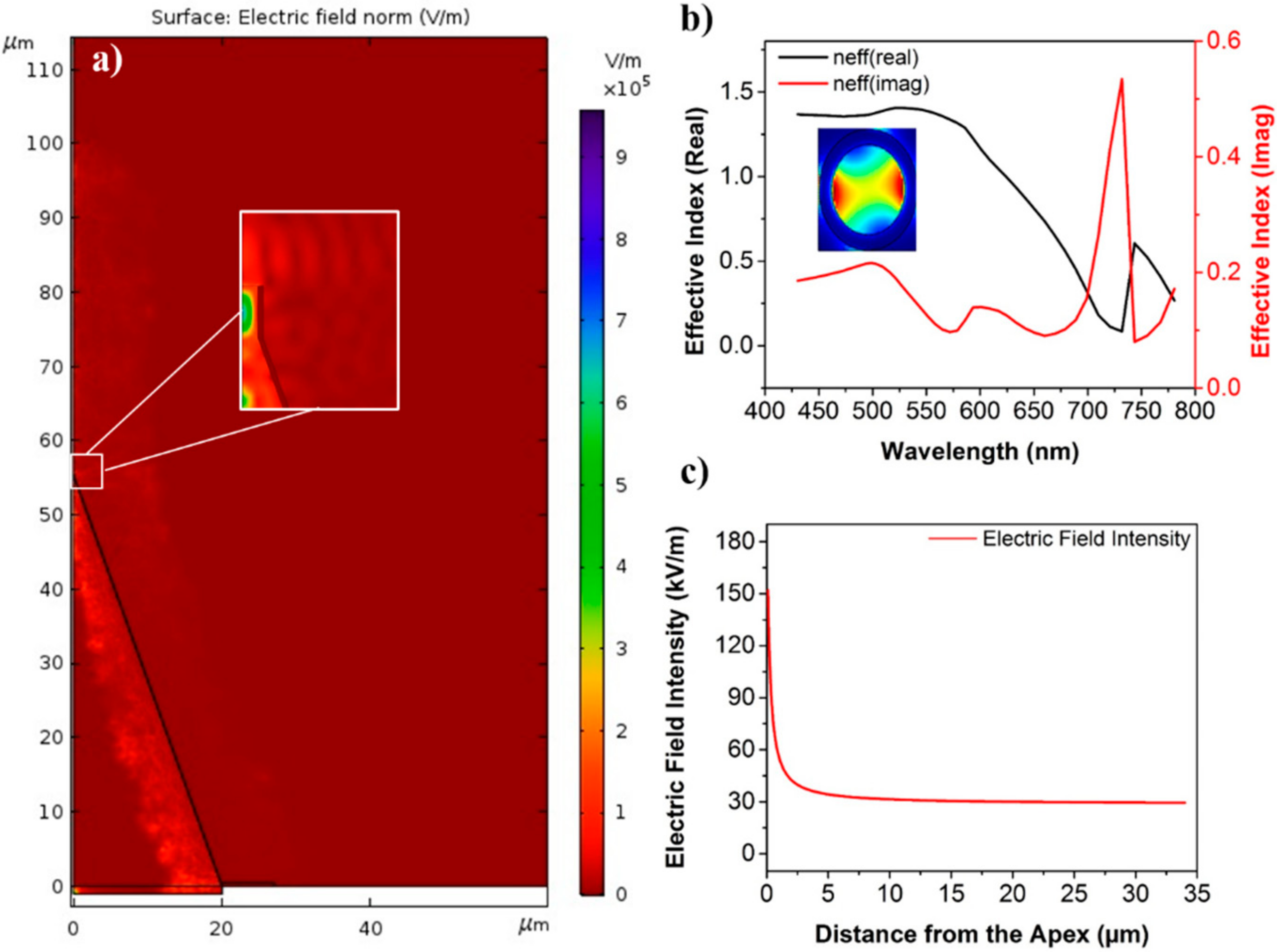
2.1. Simulation Design
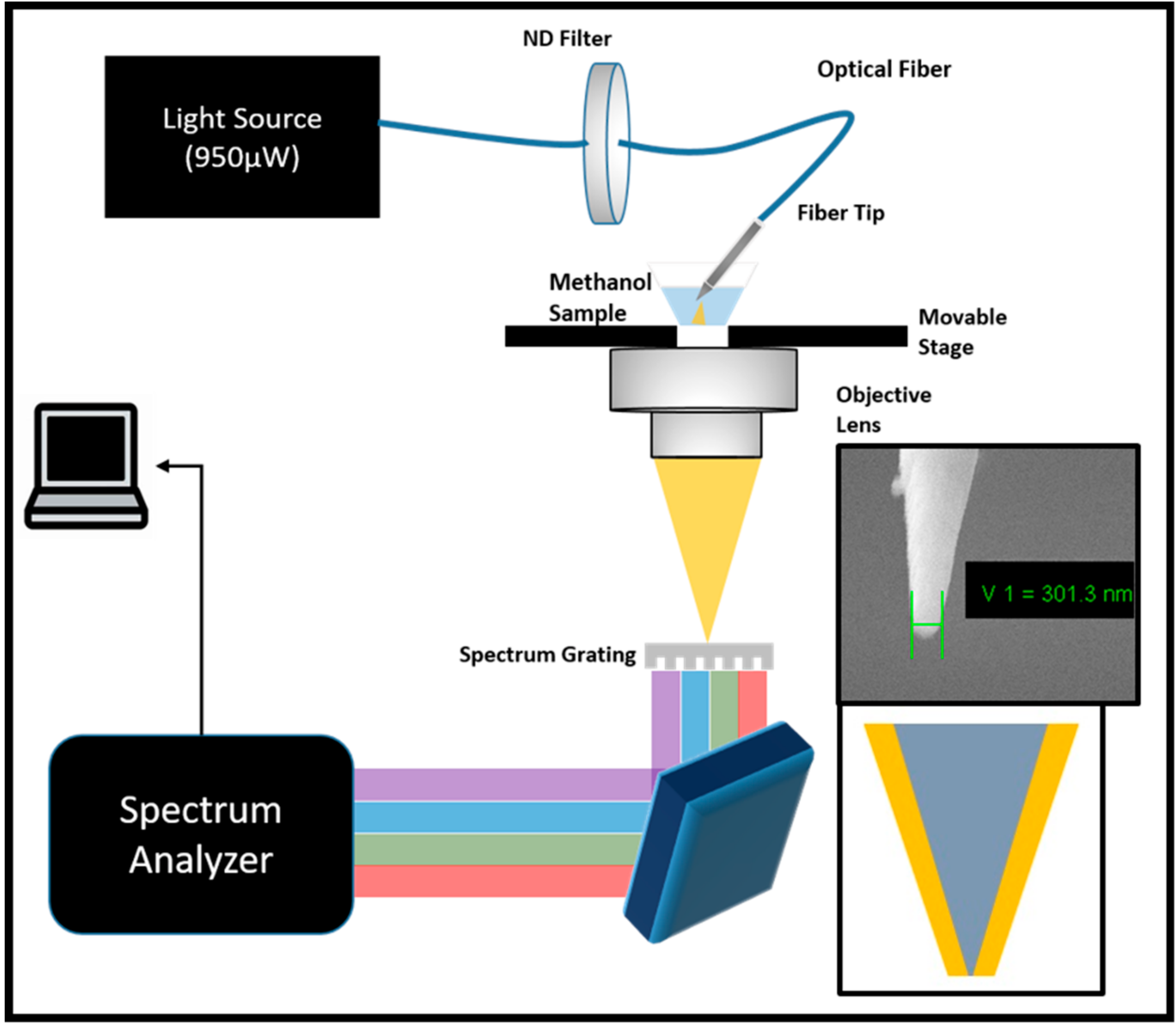
2.2. Experimental Design
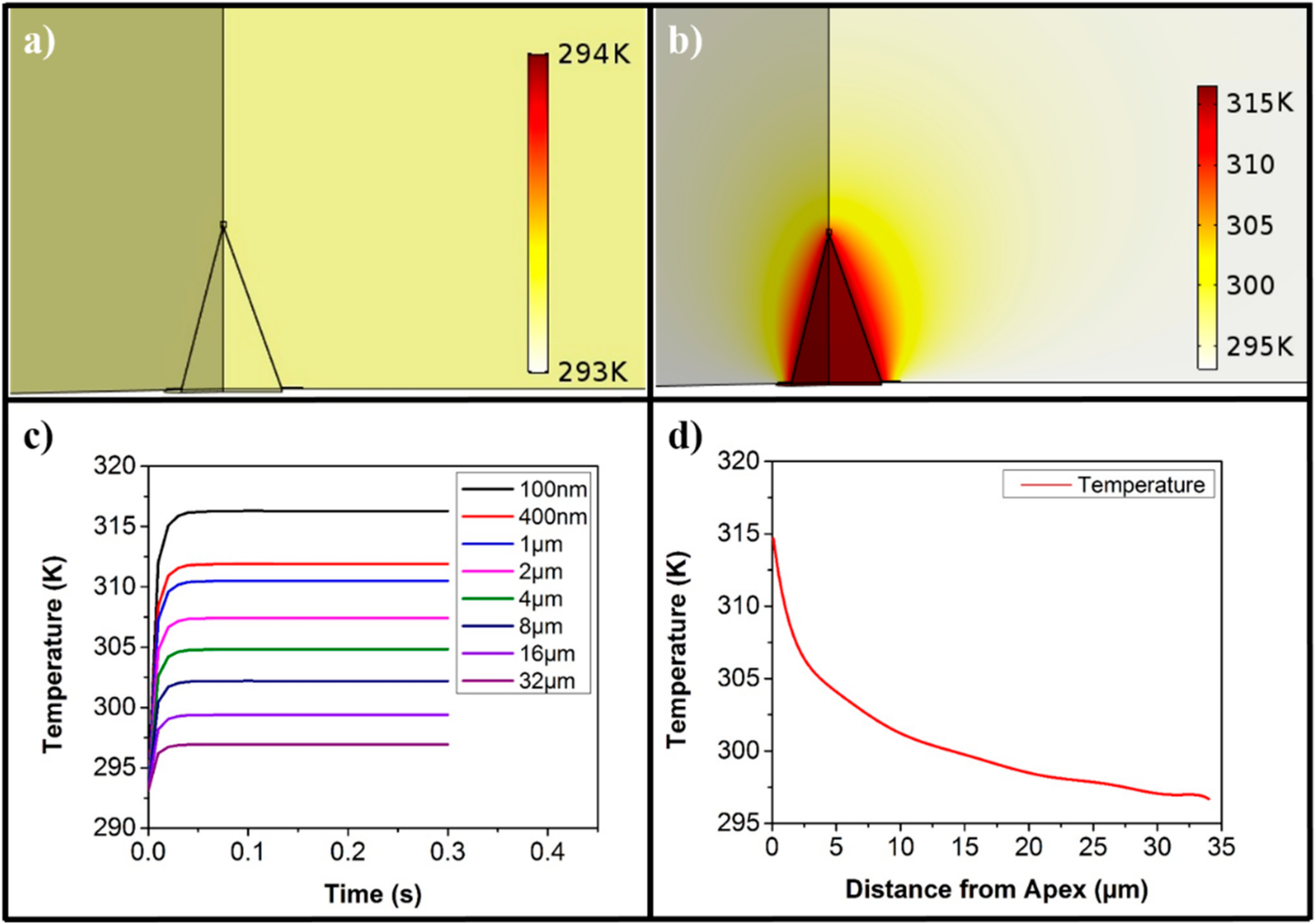
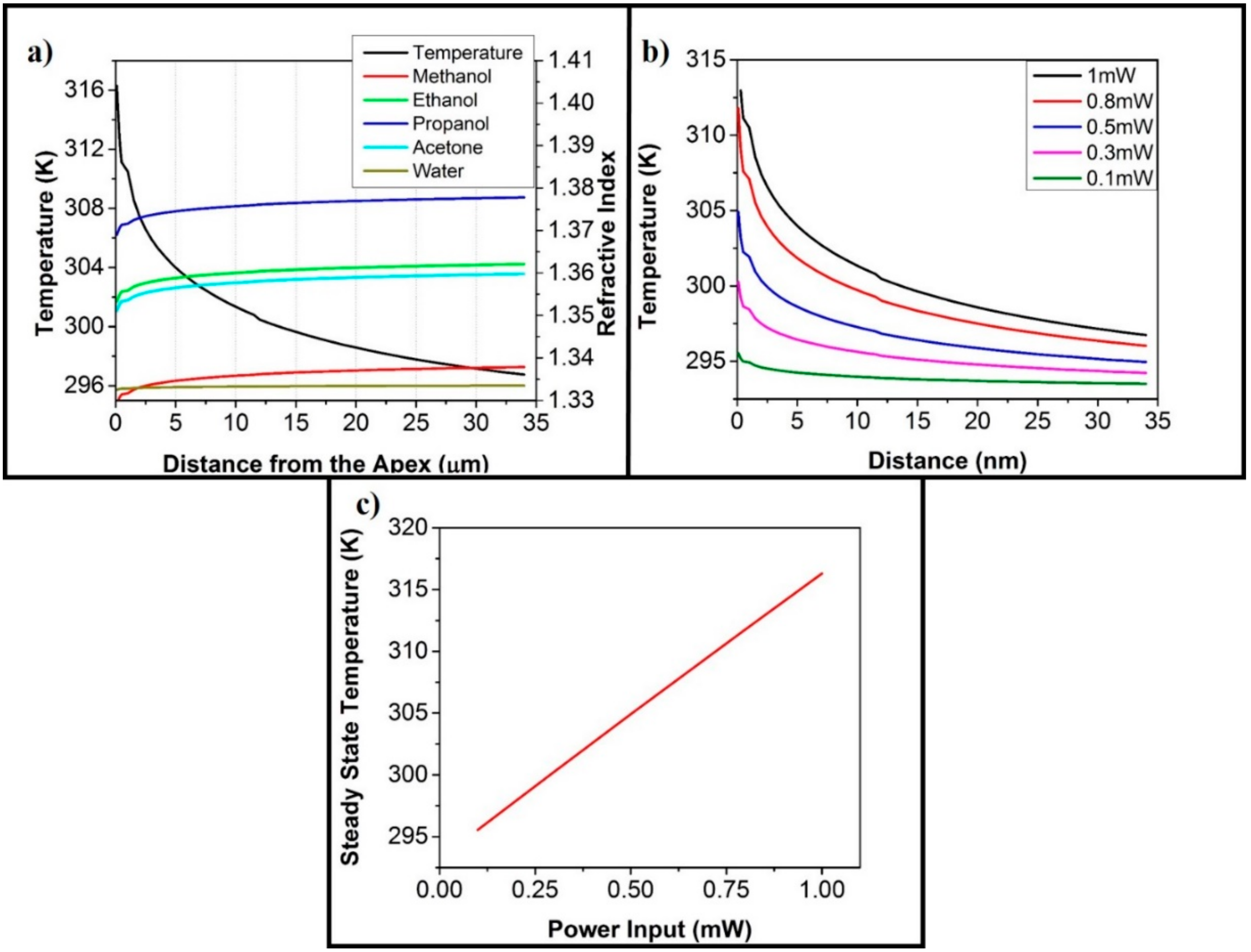
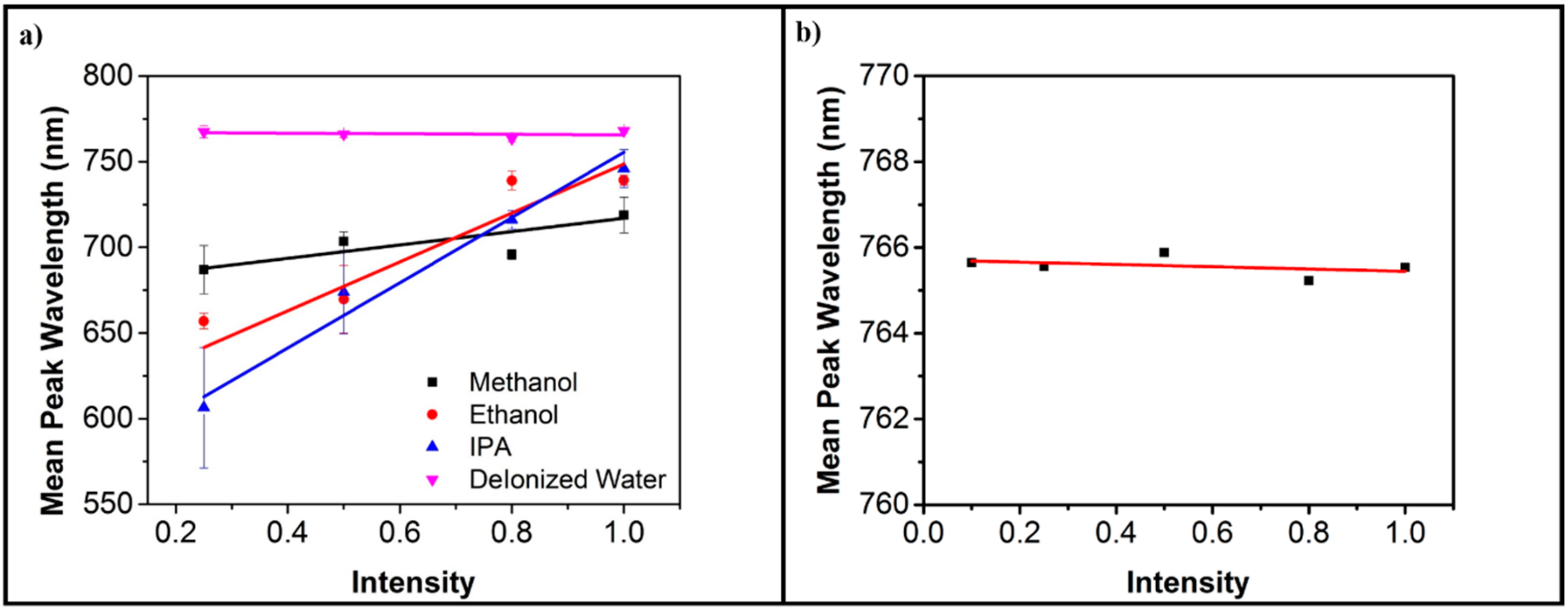
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Stewart, M.E.; Anderton, C.R.; Thompson, L.B.; Maria, J.; Gray, S.K.; Rogers, J.A.; Nuzzo, R.G. Nanostructured Plasmonic Sensors. Chem. Rev. 2008, 108, 494–521. [Google Scholar] [CrossRef] [PubMed]
- Spackova, B.; Wrobel, P.; Bockova, M.; Homola, J. Optical Biosensors Based on Plasmonic Nanostructures: A Review. Proc. IEEE 2016, 104, 2380–2408. [Google Scholar] [CrossRef]
- Ali, M.R.K.; Chen, K.; Wu, Y.; El-Sayed, M.A.; Fang, N. Gold nanoparticles in biological optical imaging. Nano Today 2019, 24, 120–140. [Google Scholar]
- Yeo, B.S.; Zhang, W.; Vannier, C.; Zenobi, R. Enhancement of Raman signals with silver-coated tips. Appl. Spectrosc. 2006, 60, 1142–1147. [Google Scholar] [CrossRef]
- Yu, J.; Guo, Y.; Wang, H.; Su, S.; Zhang, C.; Man, B.; Lei, F. Quasi Optical Cavity of Hierarchical ZnO Nanosheets@Ag Nanoravines with Synergy of Near- And Far-Field Effects for in Situ Raman Detection. J. Phys. Chem. Lett. 2019, 10, 3676–3680. [Google Scholar] [CrossRef]
- Huang, T.X.; Huang, S.C.; Li, M.H.; Zeng, Z.C.; Wang, X.; Ren, B. Tip-enhanced Raman spectroscopy: Tip-related issues. Anal. Bioanal. Chem. 2015, 407, 8177–8195. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Z.; Zhuang, M.; De Zhang, H.M.; Wang, X.; Xie, Z.X.; Wu, D.Y.; Ren, B.; Tian, Z.Q. Tip-enhanced Raman spectroscopy for investigating adsorbed species on a single-crystal surface using electrochemically prepared Au tips. Appl. Phys. Lett. 2007, 91, 101105. [Google Scholar] [CrossRef]
- Freedman, K.J.; Crick, C.R.; Albella, P.; Barik, A.; Ivanov, A.P.; Maier, S.A.; Oh, S.H.; Edel, J.B. On-Demand Surface-And Tip-Enhanced Raman Spectroscopy Using Dielectrophoretic Trapping and Nanopore Sensing. ACS Photonics 2016, 3, 1036–1044. [Google Scholar] [CrossRef]
- Smith, D.R.; Pendry, J.B.; Wiltshire, M.C.K. Metamaterials and negative refractive index. Science 2004, 305, 788–792. [Google Scholar] [CrossRef]
- Pendry, J.B. Negative Refraction Makes a Perfect Lens. Phys. Rev. Lett. 2000, 85, 3966–3969. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liu, M.; Liu, W.; Liu, Y.; Li, Z.; Wang, X.; Yang, F. Absorption enhancement in thin film solar cells with bilayer silver nanoparticle arrays. J. Phys. Commun. 2018, 2, 055032. [Google Scholar] [CrossRef]
- Singh, P. LSPR Biosensing: Recent Advances and Approaches. In Reviews in Plasmonics 2016; Springer: Cham, Switzerland, 2017; pp. 211–238. ISBN 978-3-319-48080-0. [Google Scholar]
- Jeon, H.B.; Tsalu, P.V.; Ha, J.W. Shape Effect on the Refractive Index Sensitivity at Localized Surface Plasmon Resonance Inflection Points of Single Gold Nanocubes with Vertices. Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; He, S.; Guo, W.; Hu, Y.; Huang, J.; Mulcahy, J.R.; Wei, W.D. Surface-Plasmon-Driven Hot Electron Photochemistry. Chem. Rev. 2018, 118, 2927–2954. [Google Scholar] [CrossRef]
- Hasheminejad, M.; Fang, Y.; Li, M.; Jiang, Y.; Wang, W.; Chen, H.Y. Plasmonic Imaging of the Interfacial Potential Distribution on Bipolar Electrodes. Angew. Chem. Int. Ed. 2017, 56, 1629–1633. [Google Scholar] [CrossRef] [PubMed]
- Yin, A.; He, Q.; Lin, Z.; Luo, L.; Liu, Y.; Yang, S.; Wu, H.; Ding, M.; Huang, Y.; Duan, X. Optical Voltage Sensors Plasmonic/Nonlinear Optical Material Core/Shell Nanorods as Nanoscale Plasmon Modulators and Optical Voltage Sensors Communications Angewandte. Angew. Chem. Int. Ed. 2016, 55, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.F.; Chau, L.K. Colloidal gold-modified optical fiber for chemical and biochemical sensing. Anal. Chem. 2003, 75, 16–21. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Yi, H.; Long, J.; Zhou, X.; Yang, J.; Yang, T. Plasmonic crystal cavity on single-mode optical fiber end facet for label-free biosensing. Appl. Phys. Lett. 2016, 108, 231105. [Google Scholar] [CrossRef]
- Alam, M.S.; Karim, F.; Zhao, C. Single-molecule detection at high concentrations with optical aperture nanoantennas. Nanoscale 2016, 8, 9480–9487. [Google Scholar] [CrossRef]
- Baaske, M.D.; Vollmer, F. Optical observation of single atomic ions interacting with plasmonic nanorods in aqueous solution. Nat. Photonics 2016, 10, 733–739. [Google Scholar] [CrossRef]
- Jung, I.; Kim, M.; Kwak, M.; Kim, G.; Jang, M.; Kim, S.M.; Park, D.J.; Park, S. Surface plasmon resonance extension through two-block metal-conducting polymer nanorods. Nat. Commun. 2018, 9, 1010. [Google Scholar] [CrossRef]
- Yu, J.; Wei, Y.; Wang, H.; Zhang, C.; Wei, Y.; Wang, M.; Man, B.; Lei, F. In situ detection of trace pollutants: A cost-effective SERS substrate of blackberry-like silver/graphene oxide nanoparticle cluster based on quick self-assembly technology. Opt. Express 2019, 27, 9879–9894. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wong, M.M.; Chiu, S.; Pang, S.W. Biosensors and Bioelectronics Effects of three-layered nanodisk size on cell detection sensitivity of plasmon resonance biosensors. Biosens. Bioelectron. 2015, 74, 799–807. [Google Scholar] [CrossRef]
- Wang, W.; Foley, K.; Shan, X.; Wang, S.; Eaton, S.; Nagaraj, V.J.; Wiktor, P.; Patel, U.; Tao, N. Single cells and intracellular processes studied by a plasmonic-based electrochemical impedance microscopy. Nat. Chem. 2011, 3, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, R.S.; Alarcon-Llado, E.; Krogstrup, P.; Bojarskaite, L.; Buch-Månson, N.; Bolinsson, J.; Nygård, J.; Fontcuberta i Morral, A.; Martinez, K.L. Nanowire-Aperture Probe: Local Enhanced Fluorescence Detection for the Investigation of Live Cells at the Nanoscale. ACS Photonics 2016, 3, 1208–1216. [Google Scholar] [CrossRef]
- Kim, E.; Baaske, M.D.; Schuldes, I.; Wilsch, P.S.; Vollmer, F. Label-free optical detection of single enzyme-reactant reactions and associated conformational changes. Sci. Adv. 2017, 3, e1603044. [Google Scholar] [CrossRef]
- Wu, F.C.; Wu, Y.; Niu, Z.; Vollmer, F. Ratiometric detection of oligonucleotide stoichiometry on multifunctional gold nanoparticles by whispering gallery mode biosensing. Analyst 2015, 140, 2969–2972. [Google Scholar] [CrossRef]
- Yuan, L.; Tao, N.; Wang, W. Plasmonic Imaging of Electrochemical Impedance. Annu. Rev. Anal. Chem. 2017, 10, 183–200. [Google Scholar] [CrossRef]
- Wang, W. Imaging the chemical activity of single nanoparticles with optical microscopy. Chem. Soc. Rev. 2018, 47, 2485–2508. [Google Scholar] [CrossRef]
- Luo, Z.; Zhang, J.; Wang, Y.; Chen, J.; Li, Y.; Duan, Y. Sensors and Actuators B: Chemical An aptamer based method for small molecules detection through monitoring salt-induced AuNPs aggregation and surface plasmon resonance (SPR) detection. Sens. Actuators B Chem. 2016, 236, 474–479. [Google Scholar] [CrossRef]
- Spasopoulos, D.; Kaziannis, S.; Danakas, S.; Ikiades, A.; Kosmidis, C. Sensors and Actuators B: Chemical LSPR based optical fiber sensors treated with nanosecond laser irradiation for refractive index sensing. Sens. Actuators B Chem. 2018, 256, 359–366. [Google Scholar] [CrossRef]
- Caucheteur, C.; Guo, T.; Albert, J. Review of plasmonic fiber optic biochemical sensors: Improving the limit of detection. Anal. Bioanal. Chem. 2015, 407, 3883–3897. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.; Zhou, X.; Yang, J.; He, X.; Wang, Y.; Yang, T. Second-order distributed-feedback surface plasmon resonator for single-mode fiber end-facet biosensing. Appl. Phys. Lett. 2017, 110, 171107. [Google Scholar] [CrossRef]
- Muri, H.I.; Hjelme, D.R. LSPR coupling and distribution of interparticle distances between nanoparticles in hydrogel on optical fiber end face. Sensors 2017, 17, 2723. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; He, X.; Zhou, X.; Lei, Z.; Wang, Y.; Yang, J.; Cai, D.; Chen, S.L.; Wang, X. Surface plasmon cavities on optical fiber end-facets for biomolecule and ultrasound detection. Opt. Laser Technol. 2018, 101, 468–478. [Google Scholar] [CrossRef]
- Sanders, M.; Lin, Y.; Wei, J.; Bono, T.; Lindquist, R.G. An enhanced LSPR fiber-optic nanoprobe for ultrasensitive detection of protein biomarkers. Biosens. Bioelectron. 2014, 61, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Paiva, J.S.; Jorge, P.A.S.; Rosa, C.C.; Cunha, J.P.S. BBA-General Subjects Optical fi ber tips for biological applications: From light con fi nement, biosensing to bioparticles manipulation. BBA Gen. Subj. 2018, 1862, 1209–1246. [Google Scholar] [CrossRef]
- Berweger, S.; Atkin, J.M.; Olmon, R.L.; Raschke, M.B. Adiabatic tip-plasmon focusing for nano-raman spectroscopy. J. Phys. Chem. Lett. 2010, 1, 3427–3432. [Google Scholar] [CrossRef]
- Kumar, S.; Kaushik, B.K.; Singh, R.; Chen, N.-K.; Yang, Q.S.; Zhang, X.; Wang, W.; Zhang, B. LSPR-based cholesterol biosensor using a tapered optical fiber structure. Biomed. Opt. Express 2019, 10, 2150. [Google Scholar] [CrossRef]
- Ortega, J. Densities and refractive indices of pure alcohols as a function of temperature. J. Chem. Eng. Data 1982, 27, 312–317. [Google Scholar] [CrossRef]
- Bashkatov, A.N.; Genina, E.A. Water Refractive Index in Dependence on Temperature and Wavelength: A Simple Approximation. In Proceedings of the Proc. SPIE 5068, Saratov Fall Meeting 2002: Optical Technologies in Biophysics and Medicine IV, Saratov, Russia, 13th October 2003; Tuchin, V.V., Ed.; pp. 393–395. [Google Scholar]
- Atad-Ettedgui, E.; Antebi, J.; Lemke, D. (Eds.) Optomechanical Technologies for Astronomy. In Proceedings of the Proc. SPIE 6273, Orlando, FL, USA, 6th July 2006; p. 62732J. [Google Scholar]
- Christopher, P.; Xin, H.; Linic, S. Visible-light-enhanced catalytic oxidation reactions on plasmonic silver nanostructures. Nat. Chem. 2011, 3, 467. [Google Scholar] [CrossRef]
- Govorov, A.O.; Richardson, H.H. Generating heat with metal nanoparticles. Nano Today 2007, 2, 30–38. [Google Scholar] [CrossRef]
- Adleman, J.R.; Boyd, D.A.; Goodwin, D.G.; Psaltis, D. Heterogenous catalysis mediated by plasmon heating. Nano Lett. 2009, 9, 4417–4423. [Google Scholar] [CrossRef] [PubMed]
- Lalisse, A.; Tessier, G.; Plain, J.; Baffou, G. Quantifying the Efficiency of Plasmonic Materials for Near-Field Enhancement and Photothermal Conversion. J. Phys. Chem. C 2015, 119, 25518–25528. [Google Scholar] [CrossRef]
- Ren, Y.; Chen, Q.; Qi, H.; Ruan, L. Experimental comparison of photothermal conversion efficiency of gold nanotriangle and nanorod in laser induced thermal therapy. Nanomaterials 2017, 7, 416. [Google Scholar]
- Elkalsh, A.; Vukovic, A.; Sewell, P.D.; Benson, T.M. Electro-thermal modelling for plasmonic structures in the TLM method. Opt. Quantum Electron. 2016, 48, 263. [Google Scholar] [CrossRef][Green Version]
- Wassel, H.M.G.; Dai, D.; Tiwari, M.; Valamehr, J.K.; Theogarajan, L.; Dionne, J.; Chong, F.T.; Sherwood, T. Opportunities and challenges of using plasmonic components in nanophotonic architectures. IEEE J. Emerg. Sel. Top. Circuits Syst. 2012, 2, 154–168. [Google Scholar] [CrossRef]
- Baffou, G.; Quidant, R. Thermo-plasmonics: Using metallic nanostructures as nano-sources of heat. Laser Photonics Rev. 2013, 7, 171–187. [Google Scholar] [CrossRef]
- Atwater, H.A.; Polman, A. Plasmonics for improved photovoltaic devices. Nat. Mater. 2010, 9, 205–213. [Google Scholar] [CrossRef]
- Paci, B.; Kakavelakis, G.; Generosi, A.; Wright, J.; Ferrero, C.; Stratakis, E.; Kymakis, E. Improving stability of organic devices: A time/space resolved structural monitoring approach applied to plasmonic photovoltaics. Sol. Energy Mater. Sol. Cells 2017, 159, 617–624. [Google Scholar] [CrossRef]
- Liu, G.L.; Kim, J.; Lu, Y.U.; Lee, L.P. Optofluidic control using photothermal nanoparticles. Nat. Mater. 2006, 5, 27–32. [Google Scholar] [CrossRef]
- Fang, Z.; Zhen, Y.-R.; Neumann, O.; Polman, A.; García de Abajo, F.J.; Nordlander, P.; Halas, N.J. Evolution of light-induced vapor generation at a liquid-immersed metallic nanoparticle. Nano Lett. 2013, 13, 1736–1742. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, P.; Han, G.; De, M.; Kim, C.K.; Rotello, V.M. Gold nanoparticles in delivery applications. Adv. Drug Deliv. Rev. 2008, 60, 1307–1315. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Baffou, G.; Meyerbröker, N.; Polleux, J. Micropatterning thermoplasmonic gold nanoarrays to manipulate cell adhesion. ACS Nano 2012, 6, 7227–7233. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Ai, K.; Jiang, C.; Li, Y.; Song, X.; Lu, L. Plasmonic titanium nitride nanoparticles for in vivo photoacoustic tomography imaging and photothermal cancer therapy. Biomaterials 2017, 132, 37–47. [Google Scholar] [CrossRef]
- Shao, J.; Xie, H.; Huang, H.; Li, Z.; Sun, Z.; Xu, Y.; Xiao, Q.; Yu, X.F.; Zhao, Y.; Zhang, H.; et al. Biodegradable black phosphorus-based nanospheres for in vivo photothermal cancer therapy. Nat. Commun. 2016, 7, 1–13. [Google Scholar] [CrossRef]
- Lal, S.; Clare, S.E.; Halas, N.J. Nanoshell-enabled photothermal cancer therapy: Impending clinical impact. Acc. Chem. Res. 2008, 41, 1842–1851. [Google Scholar] [CrossRef]
- Jin, X.; He, J.; Ye, J. Nanotriangle-based gap-enhanced Raman tags for bioimaging and photothermal therapy. J. Appl. Phys. 2019, 125, 073102. [Google Scholar] [CrossRef]
- Boyer, D.; Tamarat, P.; Maali, A.; Lounis, B.; Orrit, M. Photothermal imaging of nanometer-sized metal particles among scatterers. Science 2002, 5584, 1160–1163. [Google Scholar] [CrossRef]
- Kim, C.; Cho, E.C.; Chen, J.; Song, K.H.; Au, L.; Favazza, C.; Zhang, Q.; Cobley, C.M.; Gao, F.; Xia, Y.; et al. In vivo molecular photoacoustic tomography of melanomas targeted by bioconjugated gold nanocages. ACS Nano 2010, 4, 4559–4564. [Google Scholar] [CrossRef]
- Kim, J. Joining plasmonics with microfluidics: From convenience to inevitability. Lab Chip 2012, 12, 3611–3623. [Google Scholar] [CrossRef]
- Donner, J.S.; Baffou, G.; McCloskey, D.; Quidant, R. Plasmon-assisted optofluidics. Proc. ACS Nano 2011, 5, 5457–5462. [Google Scholar] [CrossRef]
- Joshi, G.K.; Smith, K.A.; Johnson, M.A.; Sardar, R. Temperature-controlled reversible localized surface plasmon resonance response of polymer-functionalized gold nanoprisms in the solid state. J. Phys. Chem. C 2013, 117, 26228–26237. [Google Scholar] [CrossRef]
- Jackman, J.A.; Ferhan, A.R.; Yoon, B.K.; Park, J.H.; Zhdanov, V.P.; Cho, N.J. Indirect Nanoplasmonic Sensing Platform for Monitoring Temperature-Dependent Protein Adsorption. Anal. Chem. 2017, 89, 12976–12983. [Google Scholar] [CrossRef]
- Baffou, G.; Quidant, R.; García De Abajo, F.J. Nanoscale control of optical heating in complex plasmonic systems. Proc. ACS Nano 2010, 4, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Chou, H.T.; Huang, W.H.; Wu, T.M.; Yu, Y.K.; Hsu, H.C. LSPR effects of Au nanoparticles/ZnO nano-composite films. Sens. Bio Sens. Res. 2017, 14, 17–20. [Google Scholar] [CrossRef]
- El-Kashef, H. The necessary requirements imposed on polar dielectric laser dye solvents—II. Phys. B Condens. Matter 2002, 311, 376–379. [Google Scholar] [CrossRef]
- Hale, G.M.; Querry, M.R. Optical Constants of Water in the 200-nm to 200-μm Wavelength Region. Appl. Opt. 1973, 12, 555. [Google Scholar] [CrossRef]
- Rheims, J.; Köser, J.; Wriedt, T. Refractive-index measurements in the near-IR using an Abbe refractometer. Meas. Sci. Technol. 1997, 8, 601–605. [Google Scholar] [CrossRef]
- Kozma, I.Z.; Krok, P.; Riedle, E. Direct measurement of the group-velocity mismatch and derivation of the refractive-index dispersion for a variety of solvents in the ultraviolet. J. Opt. Soc. Am. B 2005, 22, 1479. [Google Scholar] [CrossRef]
- Laluet, J.-Y.; Devaux, E.; Genet, C.; Ebbesen, T.W.; Weeber, J.-C.; Dereux, A. Optimization of surface plasmons launching from subwavelength hole arrays: Modelling and experiments. Opt. Express 2007, 15, 3488. [Google Scholar] [CrossRef]
- Zayats, A.V.; Smolyaninov, I.I.; Maradudin, A.A. Nano-optics of surface plasmon polaritons. Phys. Rep. 2005, 408, 131–314. [Google Scholar] [CrossRef]
- Teperik, T.V.; Archambault, A.; Marquier, F.; Greffet, J.J. Huygens-Fresnel principle for surface plasmons. Opt. Express 2009, 17, 17483. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nisar, M.S.; Kang, S.; Zhao, X. Photothermal Effect in Plasmonic Nanotip for LSPR Sensing. Sensors 2020, 20, 671. https://doi.org/10.3390/s20030671
Nisar MS, Kang S, Zhao X. Photothermal Effect in Plasmonic Nanotip for LSPR Sensing. Sensors. 2020; 20(3):671. https://doi.org/10.3390/s20030671
Chicago/Turabian StyleNisar, Muhammad Shemyal, Siyu Kang, and Xiangwei Zhao. 2020. "Photothermal Effect in Plasmonic Nanotip for LSPR Sensing" Sensors 20, no. 3: 671. https://doi.org/10.3390/s20030671
APA StyleNisar, M. S., Kang, S., & Zhao, X. (2020). Photothermal Effect in Plasmonic Nanotip for LSPR Sensing. Sensors, 20(3), 671. https://doi.org/10.3390/s20030671






