The Coefficient of Variation of Step Time Can Overestimate Gait Abnormality: Test-Retest Reliability of Gait-Related Parameters Obtained with a Tri-Axial Accelerometer in Healthy Subjects
Abstract
1. Introduction
2. Materials and Methods
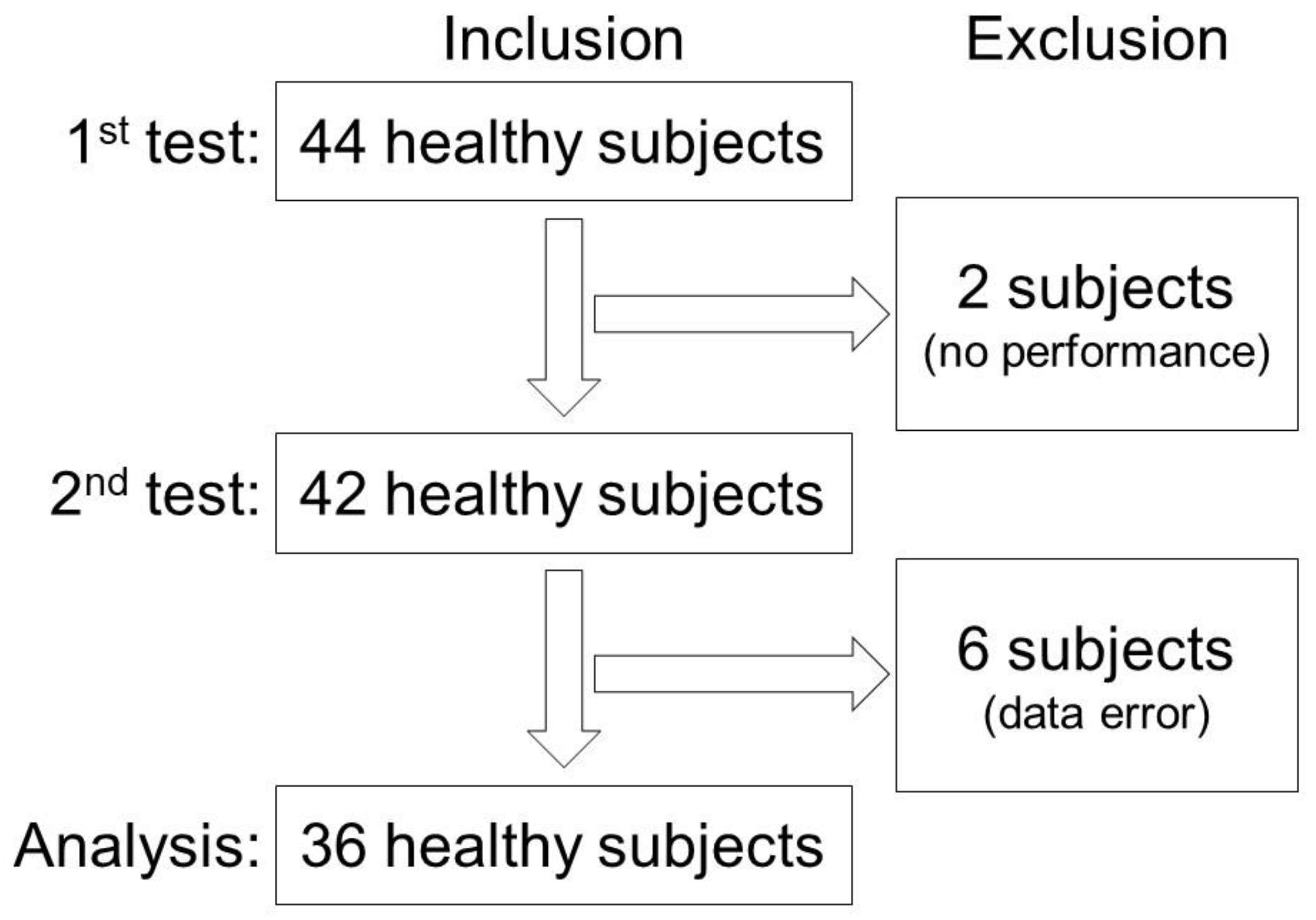
2.1. Subjects
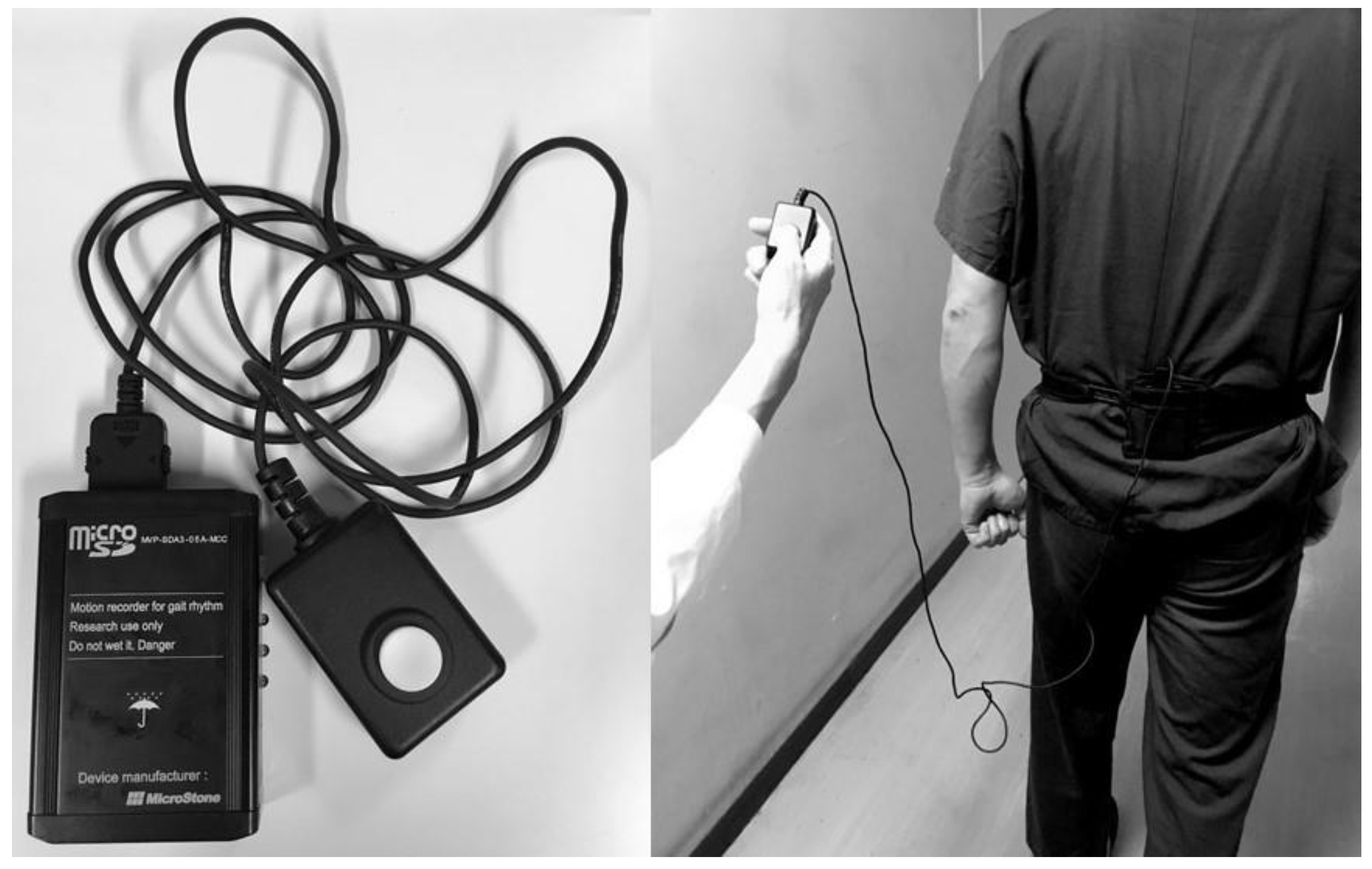
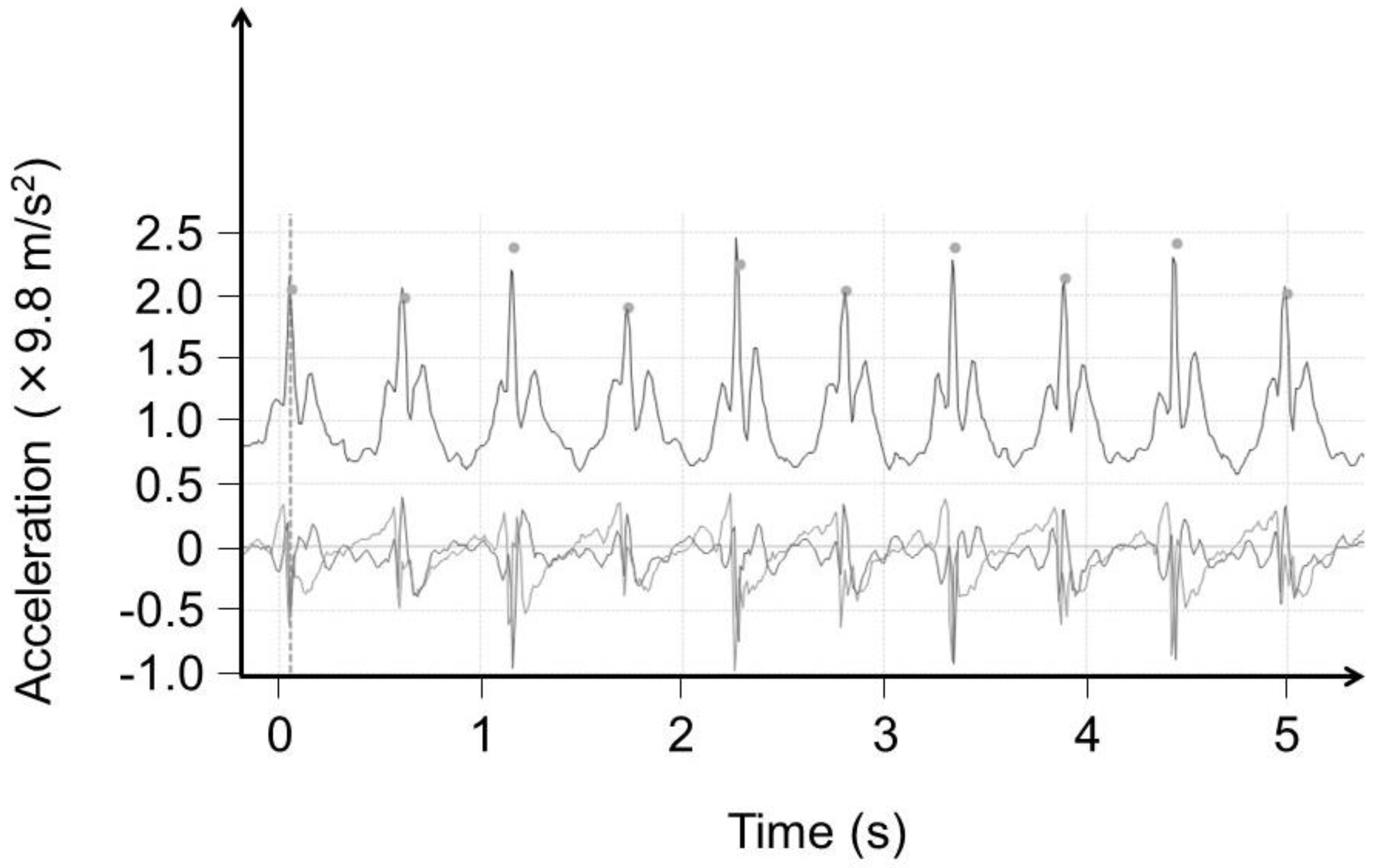
2.2. Data Analysis
2.3. Statistical Analysis
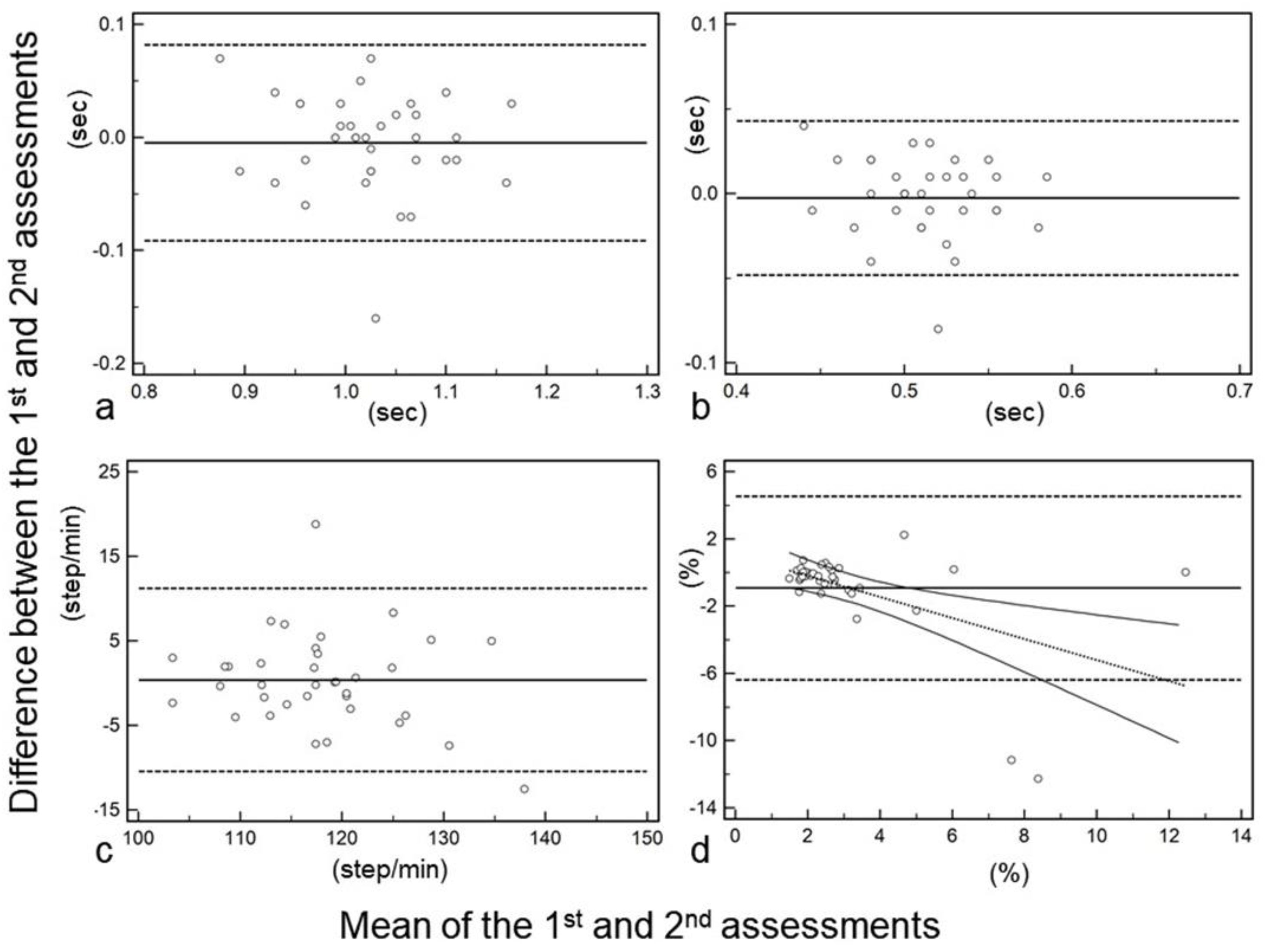
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nooijen, C.F.; Ter Hoeve, N.; Field-Fote, E.C. Gait quality is improved by locomotor training in individuals with SCI regardless of training approach. J. Neuroeng. Rehabil. 2009, 6, 36. [Google Scholar] [CrossRef] [PubMed]
- von Schroeder, H.P.; Coutts, R.D.; Lyden, P.D.; Billings, E., Jr.; Nickel, V.L. Gait parameters following stroke: A practical assessment. J. Rehabil. Res. Dev. 1995, 32, 25–31. [Google Scholar] [PubMed]
- Imms, F.J.; Edholm, O.G. The assessment of gait and mobility in the elderly. Age Ageing 1979, 8, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, S.; Iwakura, H. Foot-force measuring device for clinical assessment of pathological gait. Med. Biol. Eng. Comput. 1978, 16, 429–436. [Google Scholar] [CrossRef]
- Grieve, D.W. The assessment of gait. Physiotherapy 1969, 55, 452–460. [Google Scholar]
- Kennedy, P.E. Locomotion and gait: The physiotherapists assessment. Ir. Nurs. Hosp. World 1966, 32, 11–12. [Google Scholar]
- Bohnen, N.I.; Kanel, P.; Zhou, Z.; Koeppe, R.A.; Frey, K.A.; Dauer, W.T.; Albin, R.L.; Muller, M. Cholinergic system changes of falls and freezing of gait in Parkinson disease. Ann. Neurol. 2019. [Google Scholar] [CrossRef]
- Bohnen, N.I.; Frey, K.A.; Studenski, S.; Kotagal, V.; Koeppe, R.A.; Constantine, G.M.; Scott, P.J.; Albin, R.L.; Muller, M.L. Extra-nigral pathological conditions are common in Parkinson’s disease with freezing of gait: An in vivo positron emission tomography study. Mov. Disord. Off. J. Mov. Disord. Soc. 2014, 29, 1118–1124. [Google Scholar] [CrossRef]
- Hausdorff, J.M.; Cudkowicz, M.E.; Firtion, R.; Wei, J.Y.; Goldberger, A.L. Gait variability and basal ganglia disorders: Stride-to-stride variations of gait cycle timing in Parkinson’s disease and Huntington’s disease. Mov. Disord. Off. J. Mov. Disord. Soc. 1998, 13, 428–437. [Google Scholar] [CrossRef]
- Bazner, H.; Oster, M.; Daffertshofer, M.; Hennerici, M. Assessment of gait in subcortical vascular encephalopathy by computerized analysis: A cross-sectional and longitudinal study. J. Neurol. 2000, 247, 841–849. [Google Scholar] [CrossRef]
- Wajda, D.A.; Sandroff, B.M.; Pula, J.H.; Motl, R.W.; Sosnoff, J.J. Effects of walking direction and cognitive challenges on gait in persons with multiple sclerosis. Mult. Scler. Int. 2013, 2013, 859323. [Google Scholar] [CrossRef] [PubMed]
- Motl, R.W.; Pilutti, L.; Sandroff, B.M.; Dlugonski, D.; Sosnoff, J.J.; Pula, J.H. Accelerometry as a measure of walking behavior in multiple sclerosis. Acta Neurol. Scand. 2013, 127, 384–390. [Google Scholar] [CrossRef]
- Grobelny, A.; Behrens, J.R.; Mertens, S.; Otte, K.; Mansow-Model, S.; Kruger, T.; Gusho, E.; Bellmann-Strobl, J.; Paul, F.; Brandt, A.U.; et al. Maximum walking speed in multiple sclerosis assessed with visual perceptive computing. PLoS ONE 2017, 12, e0189281. [Google Scholar] [CrossRef]
- Sehle, A.; Mundermann, A.; Starrost, K.; Sailer, S.; Becher, I.; Dettmers, C.; Vieten, M. Objective assessment of motor fatigue in Multiple Sclerosis using kinematic gait analysis: A pilot study. J. Neuroeng. Rehabil. 2011, 8, 59. [Google Scholar] [CrossRef] [PubMed]
- Al-Amri, M.; Al Balushi, H.; Mashabi, A. Intra-rater repeatability of gait parameters in healthy adults during self-paced treadmill-based virtual reality walking. Comput. Methods Biomech. Biomed. Eng. 2017, 20, 1669–1677. [Google Scholar] [CrossRef] [PubMed]
- Sosnoff, J.J.; Socie, M.J.; Sandroff, B.M.; Balantrapu, S.; Suh, Y.; Pula, J.H.; Motl, R.W. Mobility and cognitive correlates of dual task cost of walking in persons with multiple sclerosis. Disabil. Rehabil. 2014, 36, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Phan-Ba, R.; Pace, A.; Calay, P.; Grodent, P.; Douchamps, F.; Hyde, R.; Hotermans, C.; Delvaux, V.; Hansen, I.; Moonen, G.; et al. Comparison of the timed 25-foot and the 100-m walk as performance measures in multiple sclerosis. Neurorehabil. Neural Repair 2011, 25, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Terui, Y.; Suto, E.; Konno, Y.; Kubota, K.; Iwakura, M.; Satou, M.; Nitta, S.; Hasegawa, K.; Satake, M.; Shioya, T. Evaluation of gait symmetry using a tri-axial accelerometer in stroke patients. NeuroRehabilitation 2018, 42, 173–180. [Google Scholar] [CrossRef]
- Supratak, A.; Datta, G.; Gafson, A.R.; Nicholas, R.; Guo, Y.; Matthews, P.M. Remote Monitoring in the Home Validates Clinical Gait Measures for Multiple Sclerosis. Front. Neurol. 2018, 9, 561. [Google Scholar] [CrossRef]
- Donath, L.; Faude, O.; Lichtenstein, E.; Nuesch, C.; Mundermann, A. Validity and reliability of a portable gait analysis system for measuring spatiotemporal gait characteristics: Comparison to an instrumented treadmill. J. Neuroeng. Rehabil. 2016, 13, 6. [Google Scholar] [CrossRef]
- Manor, B.; Yu, W.; Zhu, H.; Harrison, R.; Lo, O.Y.; Lipsitz, L.; Travison, T.; Pascual-Leone, A.; Zhou, J. Smartphone App-Based Assessment of Gait During Normal and Dual-Task Walking: Demonstration of Validity and Reliability. JMIR Mhealth Uhealth 2018, 6, e36. [Google Scholar] [CrossRef] [PubMed]
- Silsupadol, P.; Teja, K.; Lugade, V. Reliability and validity of a smartphone-based assessment of gait parameters across walking speed and smartphone locations: Body, bag, belt, hand, and pocket. Gait Posture 2017, 58, 516–522. [Google Scholar] [CrossRef]
- Ojeda, L.V.; Rebula, J.R.; Kuo, A.D.; Adamczyk, P.G. Influence of contextual task constraints on preferred stride parameters and their variabilities during human walking. Med. Eng. Phys. 2015, 37, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Yentes, J.M.; Rennard, S.I.; Schmid, K.K.; Blanke, D.; Stergiou, N. Patients with Chronic Obstructive Pulmonary Disease Walk with Altered Step Time and Step Width Variability as Compared with Healthy Control Subjects. Ann. Am. Thorac. Soc. 2017, 14, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.Y.; Spruit, M.A.; Delbressine, J.M.; Willems, P.J.; Franssen, F.M.E.; Wouters, E.F.M.; Meijer, K. Spatiotemporal gait characteristics in patients with COPD during the Gait Real-time Analysis Interactive Lab-based 6-minute walk test. PLoS ONE 2017, 12, e0190099. [Google Scholar] [CrossRef] [PubMed]
- Heredia-Jimenez, J.; Orantes-Gonzalez, E.; Soto-Hermoso, V.M. Variability of gait, bilateral coordination, and asymmetry in women with fibromyalgia. Gait Posture 2016, 45, 41–44. [Google Scholar] [CrossRef]
- Clermont, C.A.; Barden, J.M. Accelerometer-based determination of gait variability in older adults with knee osteoarthritis. Gait Posture 2016, 50, 126–130. [Google Scholar] [CrossRef]
- Neely, J.G.; Karni, R.J.; Engel, S.H.; Fraley, P.L.; Nussenbaum, B.; Paniello, R.C. Practical guides to understanding sample size and minimal clinically important difference (MCID). Otolaryngol. Head Neck Surg. 2007, 136, 14–18. [Google Scholar] [CrossRef]
- Pau, M.; Caggiari, S.; Mura, A.; Corona, F.; Leban, B.; Coghe, G.; Lorefice, L.; Marrosu, M.G.; Cocco, E. Clinical assessment of gait in individuals with multiple sclerosis using wearable inertial sensors: Comparison with patient-based measure. Mult. Scler. Relat. Disord. 2016, 10, 187–191. [Google Scholar] [CrossRef]
- Mutoh, T.; Mutoh, T.; Takada, M.; Doumura, M.; Ihara, M.; Taki, Y.; Tsubone, H.; Ihara, M. Application of a tri-axial accelerometry-based portable motion recorder for the quantitative assessment of hippotherapy in children and adolescents with cerebral palsy. J. Phys. Ther. Sci. 2016, 28, 2970–2974. [Google Scholar] [CrossRef]
- Hsu, C.Y.; Tsai, Y.S.; Yau, C.S.; Shie, H.H.; Wu, C.M. Test-Retest Reliability of an Automated Infrared-Assisted Trunk Accelerometer-Based Gait Analysis System. Sensors 2016, 16, 1156. [Google Scholar] [CrossRef] [PubMed]
- Schmid, A.; Duncan, P.W.; Studenski, S.; Lai, S.M.; Richards, L.; Perera, S.; Wu, S.S. Improvements in speed-based gait classifications are meaningful. Stroke J. Cereb. Circ. 2007, 38, 2096–2100. [Google Scholar] [CrossRef] [PubMed]
| 1st | 95% CI | 2nd | 95% CI | p Value * | ICC | 95% CI | |
|---|---|---|---|---|---|---|---|
| Stride time [sec] | 1.02 | 1.01–1.05 | 1.03 | 0.99–1.05 | 0.689 | 0.803 | 0.647–0.894 |
| Cadence [step/min] | 119 | 115–120 | 117 | 114–121 | 0.765 | 0.784 | 0.616–0.884 |
| Step time [sec] | 0.505 | 0.500–0.523 | 0.515 | 0.500–0.523 | 0.697 | 0.788 | 0.624–0.886 |
| Number of steps [step] | 13.8 | 13.5–14.2 | 13.9 | 13.5–14.2 | 0.765 | 0.685 | 0.462–0.827 |
| Step length [cm] | 72.3 | 69.7–73.8 | 72.0 | 70.7–73.6 | 0.981 | 0.663 | 0.429–0.813 |
| Ground reaction force [×9.8 m/s2] | 0.360 | 0.330–0.383 | 0.355 | 0.327–0.373 | 0.980 | 0.615 | 0.361–0.784 |
| Velocity [m/min] | 85.3 | 82.1–87.1 | 84.3 | 82.3–86.3 | 0.753 | 0.598 | 0.339–0.773 |
| Assessment time [s] | 7.04 | 6.89–7.33 | 7.13 | 6.96–7.30 | 0.831 | 0.565 | 0.293–0.752 |
| Coefficient of variance [%] | 2.16 | 1.98–2.57 | 2.50 | 2.15–2.95 | 0.0188 | 0.425 | 0.129–0.655 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujiwara, S.; Sato, S.; Sugawara, A.; Nishikawa, Y.; Koji, T.; Nishimura, Y.; Ogasawara, K. The Coefficient of Variation of Step Time Can Overestimate Gait Abnormality: Test-Retest Reliability of Gait-Related Parameters Obtained with a Tri-Axial Accelerometer in Healthy Subjects. Sensors 2020, 20, 577. https://doi.org/10.3390/s20030577
Fujiwara S, Sato S, Sugawara A, Nishikawa Y, Koji T, Nishimura Y, Ogasawara K. The Coefficient of Variation of Step Time Can Overestimate Gait Abnormality: Test-Retest Reliability of Gait-Related Parameters Obtained with a Tri-Axial Accelerometer in Healthy Subjects. Sensors. 2020; 20(3):577. https://doi.org/10.3390/s20030577
Chicago/Turabian StyleFujiwara, Shunrou, Shinpei Sato, Atsushi Sugawara, Yasumasa Nishikawa, Takahiro Koji, Yukihide Nishimura, and Kuniaki Ogasawara. 2020. "The Coefficient of Variation of Step Time Can Overestimate Gait Abnormality: Test-Retest Reliability of Gait-Related Parameters Obtained with a Tri-Axial Accelerometer in Healthy Subjects" Sensors 20, no. 3: 577. https://doi.org/10.3390/s20030577
APA StyleFujiwara, S., Sato, S., Sugawara, A., Nishikawa, Y., Koji, T., Nishimura, Y., & Ogasawara, K. (2020). The Coefficient of Variation of Step Time Can Overestimate Gait Abnormality: Test-Retest Reliability of Gait-Related Parameters Obtained with a Tri-Axial Accelerometer in Healthy Subjects. Sensors, 20(3), 577. https://doi.org/10.3390/s20030577





