A COVID-19-Based Modified Epidemiological Model and Technological Approaches to Help Vulnerable Individuals Emerge from the Lockdown in the UK
Abstract
1. Introduction
- The use of wearable devices (henceforth, wearables) to enable vulnerable people to take part in contact tracing,
- The development of effective incentive mechanisms to motivate people to engage in contact tracing,
- The use of digital tools to maintain physical distancing and monitor health symptoms,
- The use of personal protective equipment.
- Reduced rates of transmission post-lockdown in vulnerable populations;
- Fewer restrictions on the vulnerable post-lockdown with noticeable improvement in their well-being (many may already be suffering from loneliness and mental health problems due to the lockdown);
- Ensuring that the vulnerable people and the hard-to-reach are connected and closely monitored.
2. Infectious Diseases Spread Modelling
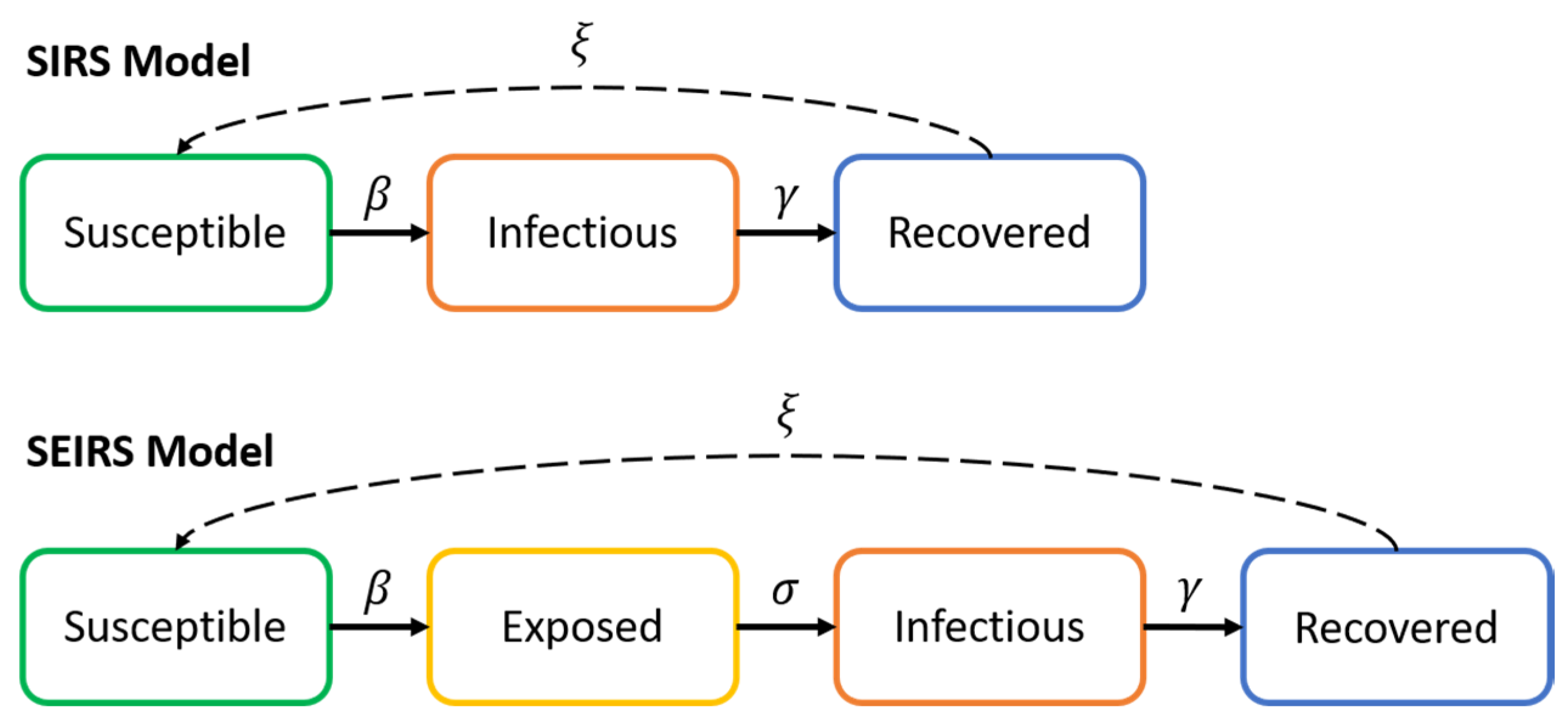
2.1. SIRS Model
- Infectious rate (β): is the rate of spread of the virus given by the probability of transmitting the disease between an infectious individual and a susceptible individual. This is subject to the disease transmission probability and the chance of contact.
- Recovery rate is determined by the average duration of the infectious period of the disease (Tlat).
- Re-susceptibility rate (ξ) is the rate at which recovered individuals return to the susceptible state due to loss of immunity (normally ignored due to long-term immunity).
2.2. SEIRS Model
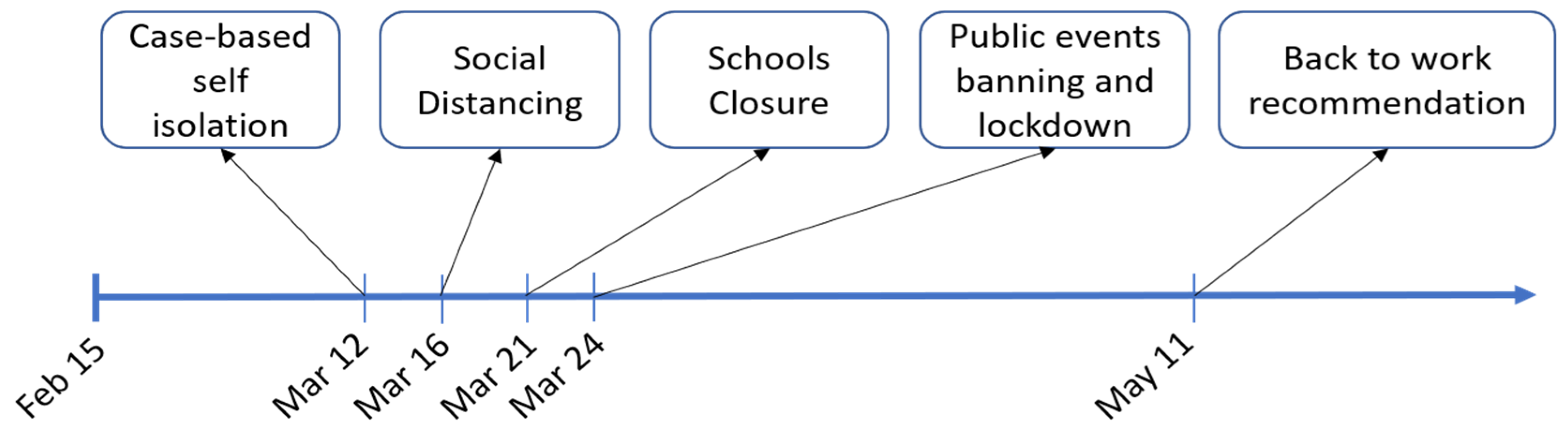
2.3. The Role of Non-Pharmaceutical Interventions and Herd Immunity
3. Methods
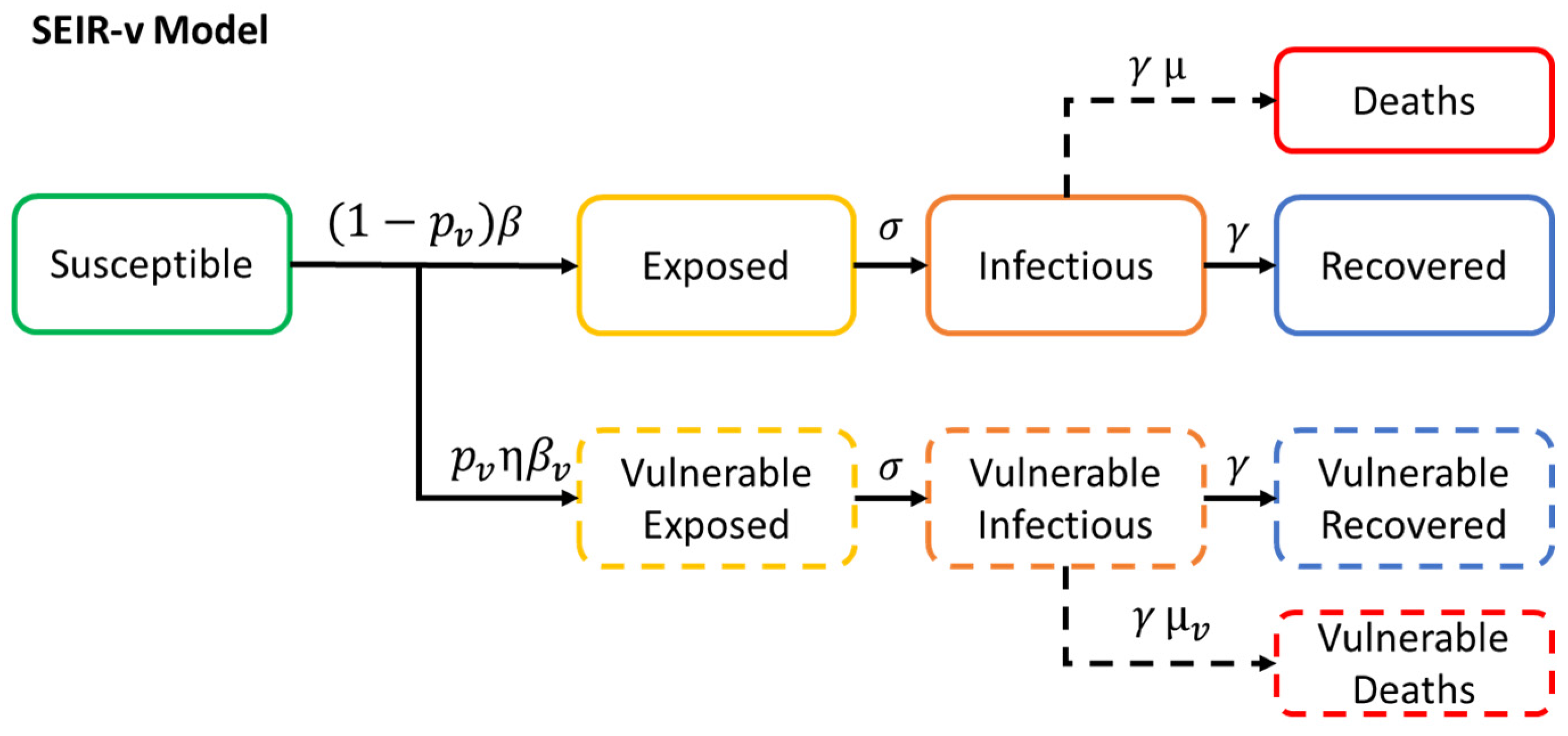
3.1. Proposed Model: SEIR-v
3.2. Model Parametrization
3.3. Virus Transmissibility Study
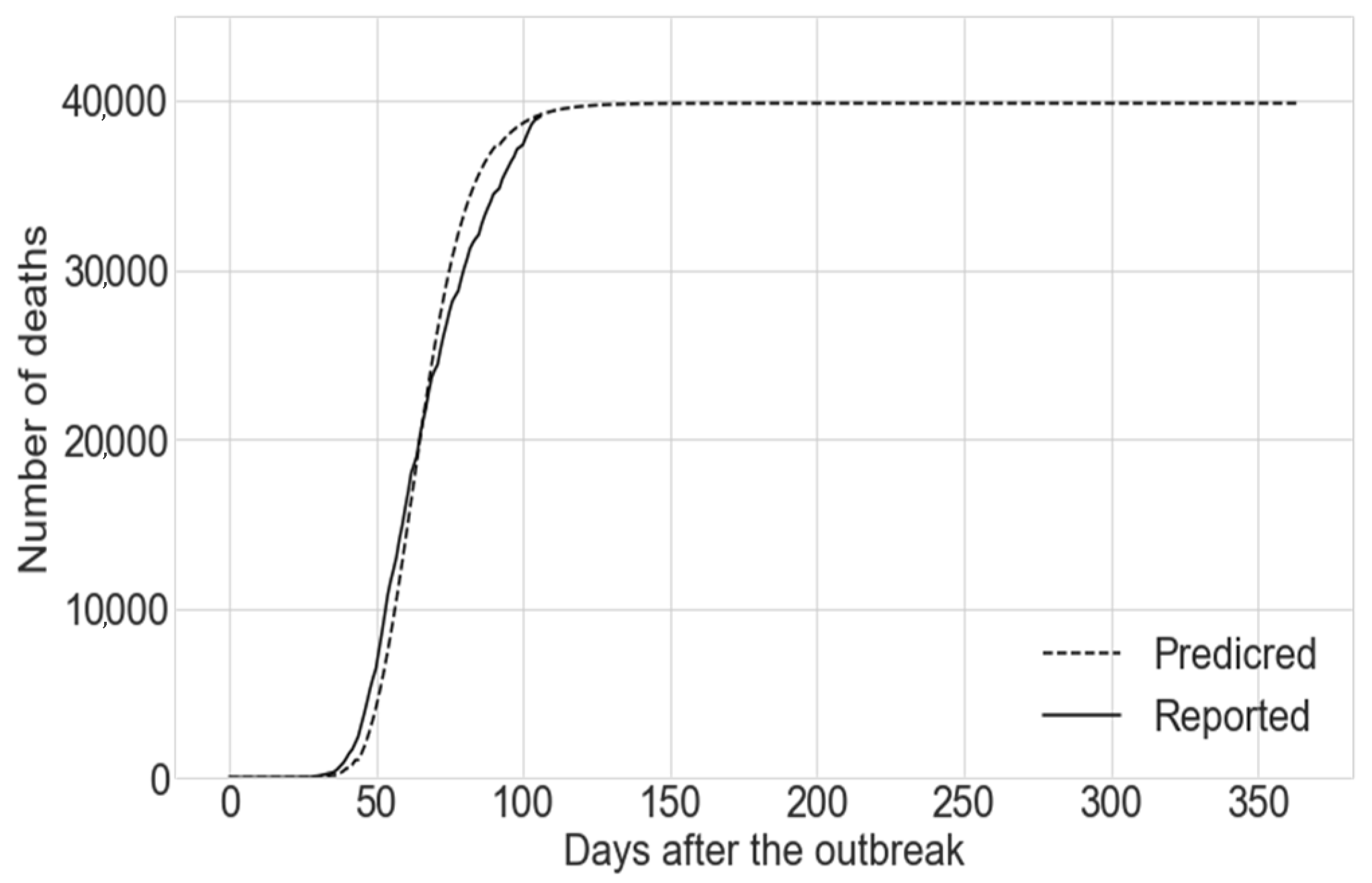
4. Results
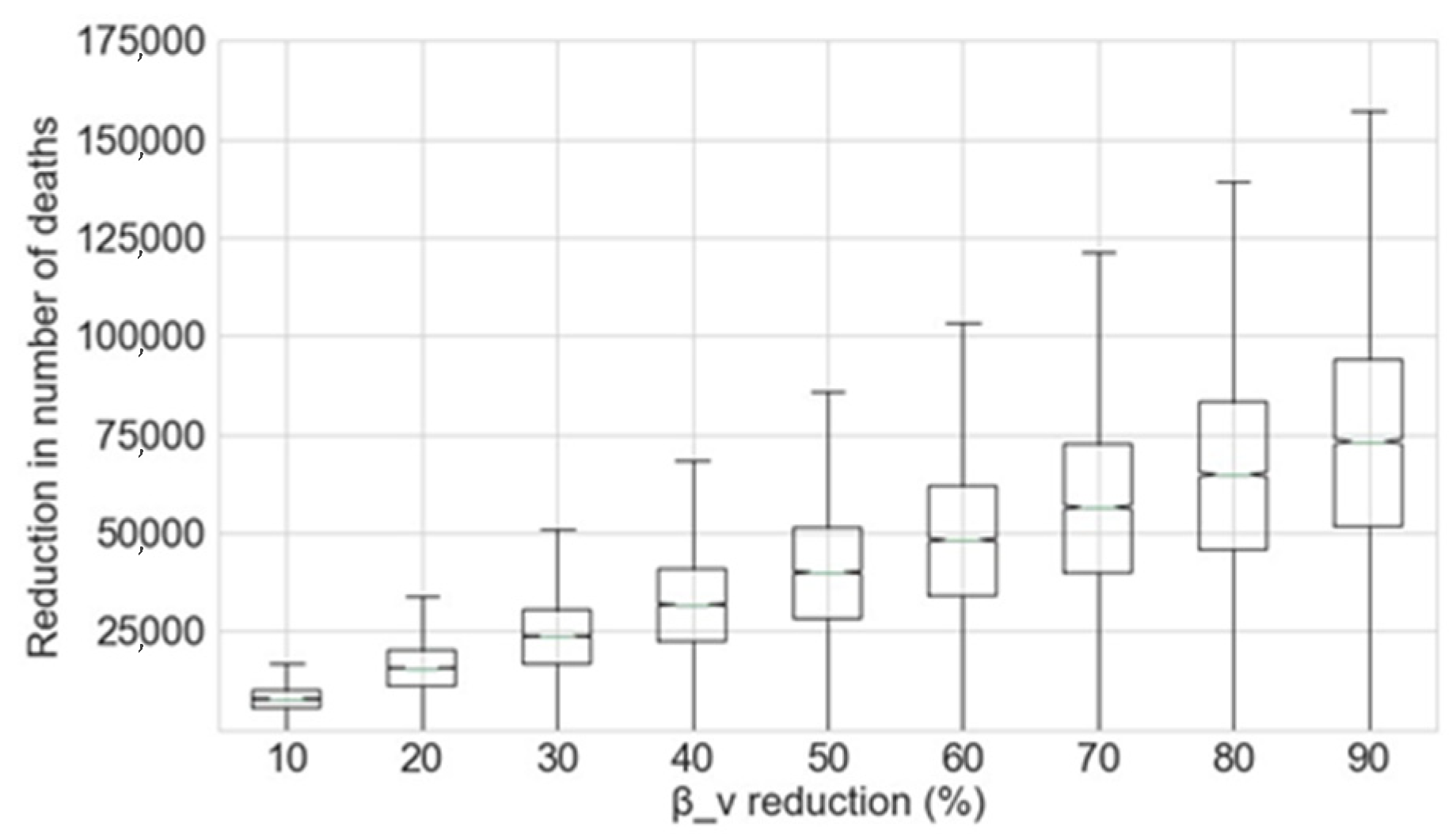
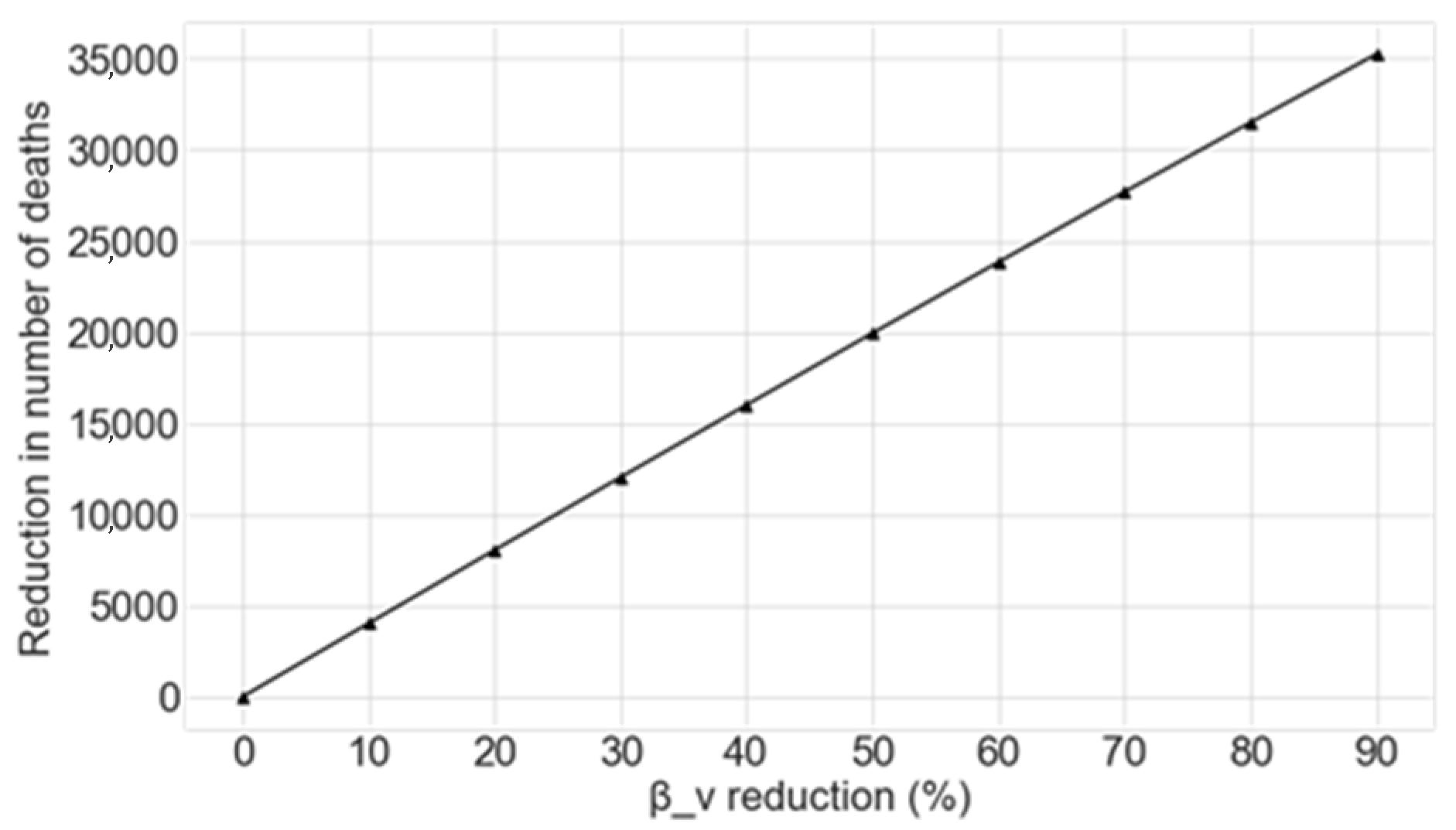
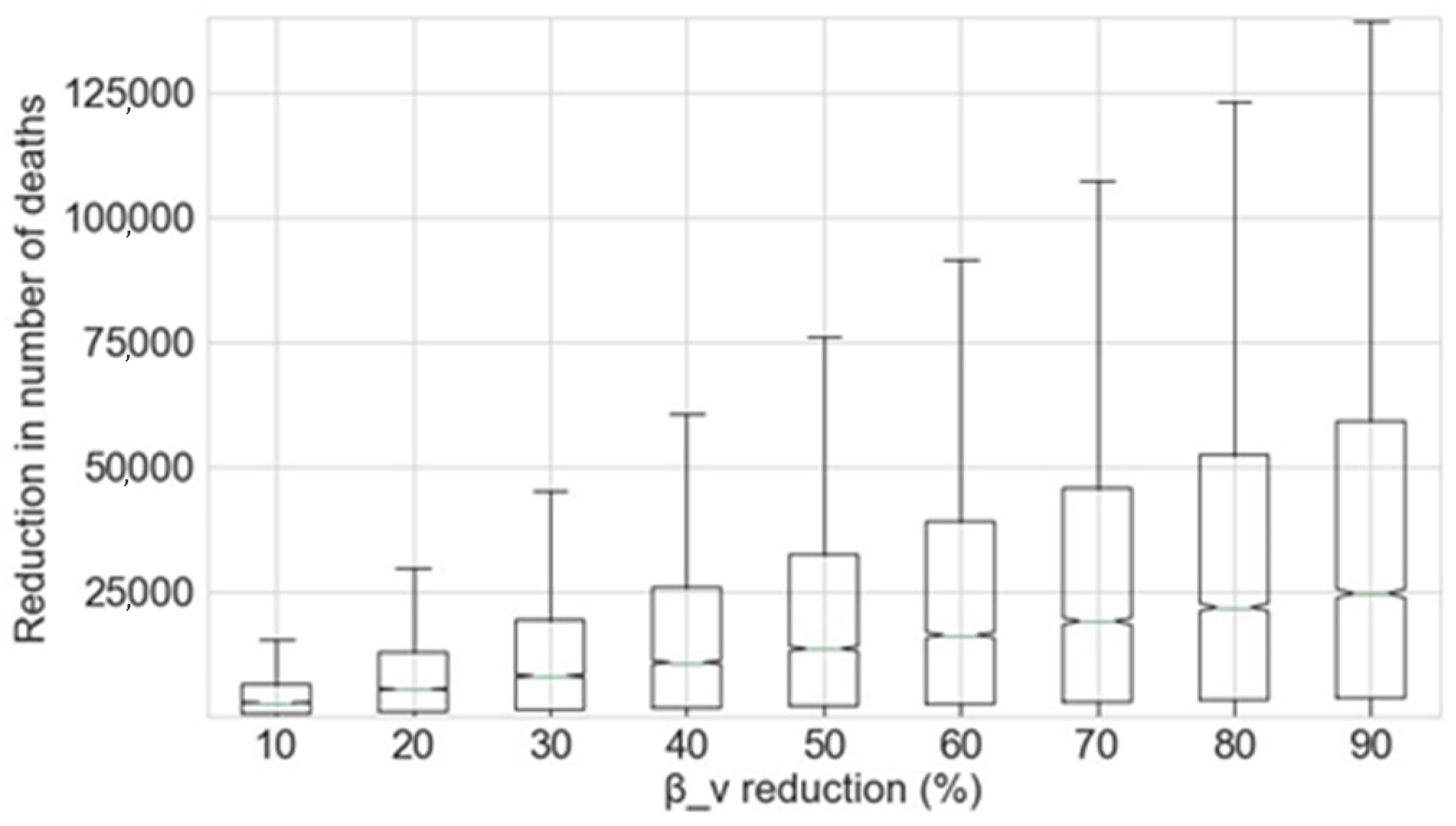
- The vulnerable group contact rate, βv, is decreased from the beginning of the outbreak. For this scenario, the potential reduction in the number of deaths if more protective measures for vulnerable groups had been applied from the beginning of the outbreak was studied.
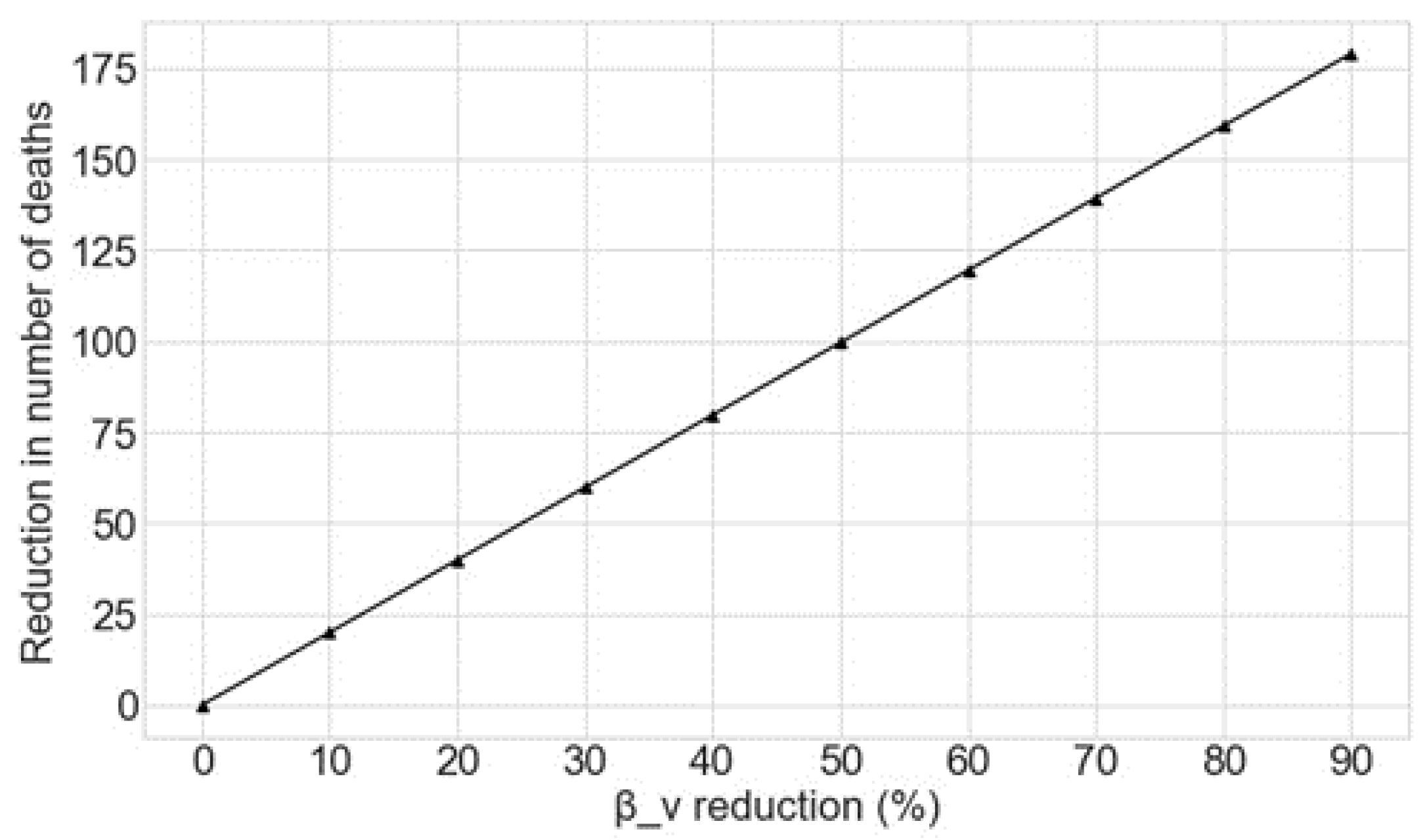
- The vulnerable group contact rate, βv, is decreased from June 2020. In this case. the potential reduction in the number of deaths resulting from the implementation of measures from June 2020 was studied.
4.1. Reduction of the Contact Rate of Vulnerable Individuals from the Beginning of the Outbreak
4.2. Reduction of the Contact Rate of Vulnerable Individuals from June 2020
4.3. Discussion
5. Protecting the Vulnerable People
5.1. Wearables for Contact Tracing
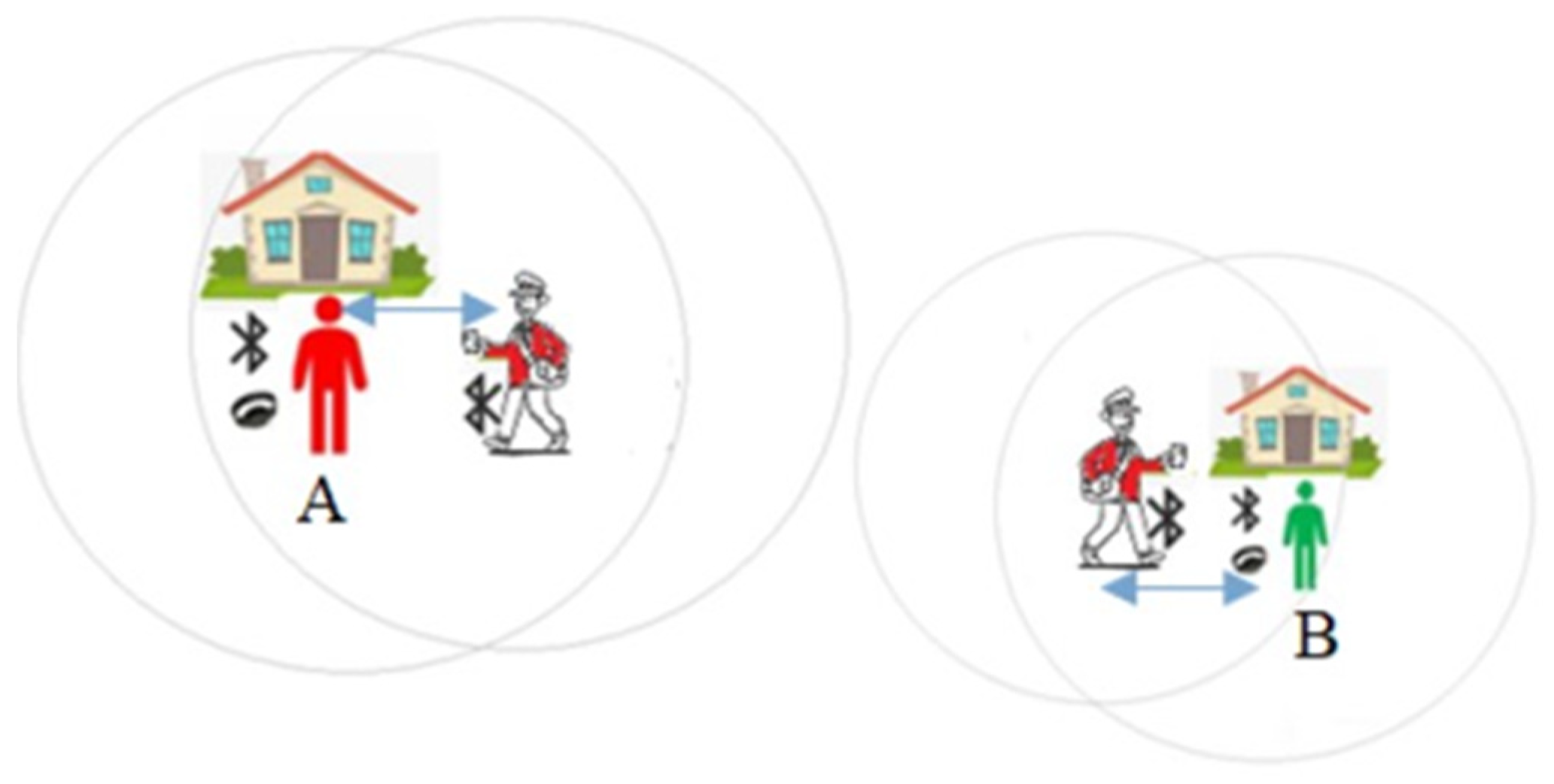
- Mobile phones might not be always with users. Instead, they might be left at home, in the car or at work, which means their social encounters don’t always correspond to actual contact.
- A wristband solution will only have radio technology with a small memory and a battery with no access to users’ data which could help to preserve privacy. As it is low power it can always be on. It does not require set-up up or installation by the wearer.
- Phone apps need to be installed and activated by users, and Bluetooth needs to be switched on. These requirements represent real barriers to the use of apps by vulnerable people who may have difficulties in remembering instructions, digital literacy, vision or motor control.
- Contact Tracing apps can consume more energy as they are often kept active with battery optimisation features disabled.
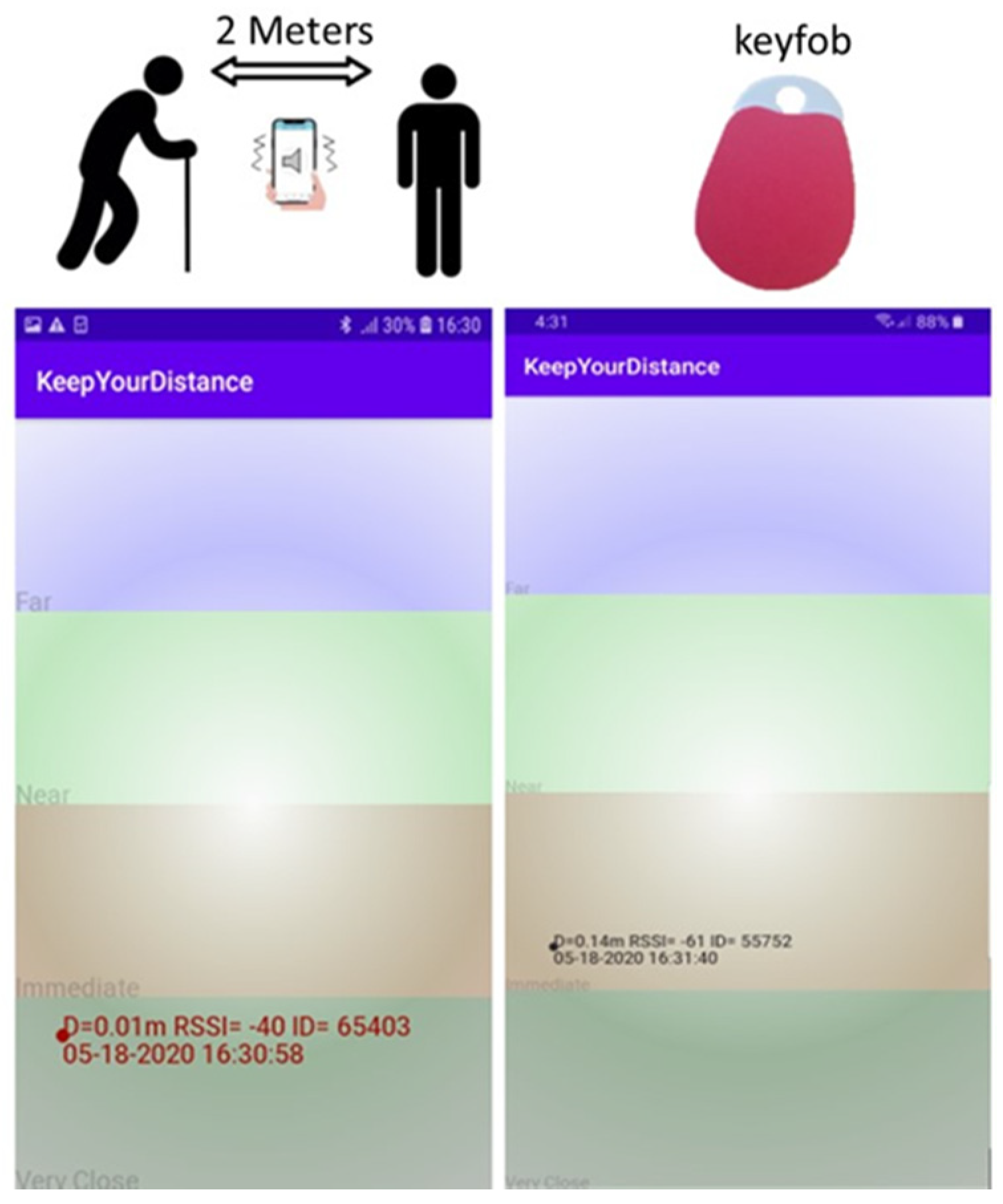
- Wearables are more likely to be worn at the front side of the body (e.g., wristbands, necklace or a keyfob), which could potentially improve the accuracy of the proximity detection in the case of face of face contact.
- Smart phones come with different operating systems and settings, which means each model might require individual calibration and configuration.
5.2. Digital Tools to Maintain Social Distancing
5.3. Wearable to Monitor Symptoms
5.4. Disease Transmission and the Use of Personal Protective Equipment
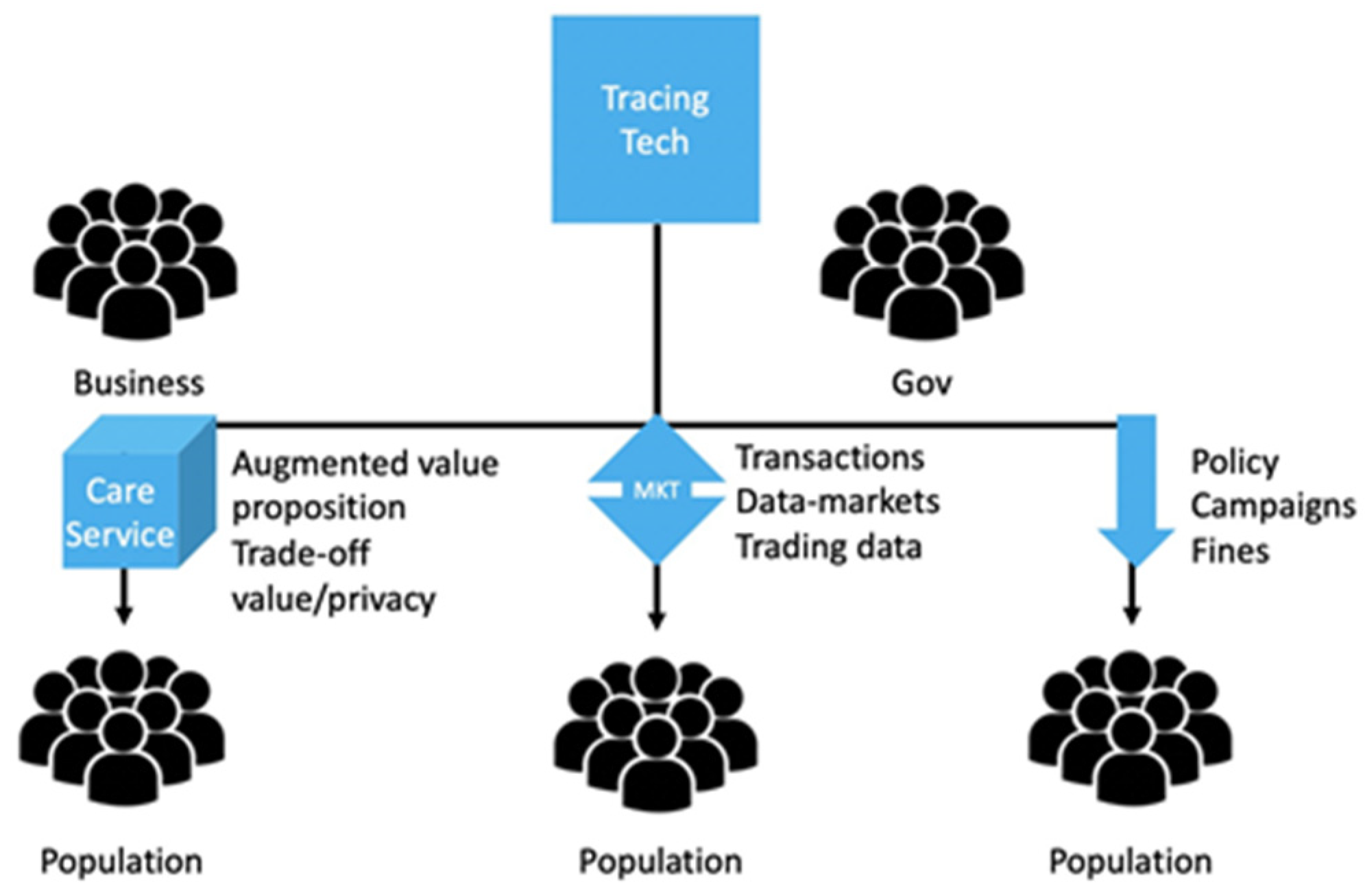
5.5. Adoption and Incentive Mechanisms for Behavioural Change
6. Conclusions and Future Work
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Channappanavar, R.; Perlman, S. Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. In Seminars in Immunopathology; Springer: Berlin/Heidelberg, Germany, 2017; Volume 39, pp. 529–539. [Google Scholar]
- Su, S.; Wong, G.; Shi, W.; Liu, J.; Lai, A.C.; Zhou, J.; Liu, W.; Bi, Y.; Gao, G.F. Epidemiology, genetic recombination, and pathogenesis of coronaviruses. Trends Microbiol. 2016, 24, 490–502. [Google Scholar] [CrossRef]
- Worldometers.Info. Coronavirus Update (Live): Worldometer. Available online: https://www.worldometers.info/coronavirus (accessed on 27 June 2020).
- Coates, S.; Tanna, P.; Scott-Allen, E. Overview of the UK Population: August 2019; Office for National Statistics: Newport, UK, 2019. [Google Scholar]
- Kermack, W.O.; McKendrick, A.G. A contribution to the mathematical theory of epidemics. Proc. R. Soc. Lond. Ser. A Contain. Pap. Math. Phys. Character 1927, 115, 700–721. [Google Scholar]
- Aron, J.L.; Schwartz, I.B. Seasonality and period-doubling bifurcations in an epidemic model. J. Theor. Biol. 1984, 110, 665–679. [Google Scholar] [CrossRef]
- Ferretti, L.; Wymant, C.; Kendall, M.; Zhao, L.; Nurtay, A.; Abeler-Dörner, L.; Parker, M.; Bonsall, D.; Fraser, C. Quantifying SARS-CoV-2 transmission suggests epidemic control with digital contact tracing. Science 2020, 368, 1–9. [Google Scholar] [CrossRef]
- Armitage, R.; Nellums, L.B. COVID-19 and the consequences of isolating the elderly. Lancet Public Health 2020, 5, e256. [Google Scholar] [CrossRef]
- Yang, Z.; Zeng, Z.; Wang, K.; Wong, S.S.; Liang, W.W.; Zanin, M.M.; Liu, P.P.; Cao, X.X.; Gao, Z.Z.; Mai, Z.; et al. Modified SEIR and AI prediction of the epidemics trend of COVID-19 in China under public health interventions. J. Thorac. Dis. 2020, 12, 165. [Google Scholar] [CrossRef]
- López, L.; Rodo, X. A modified SEIR model to predict the COVID-19 outbreak in Spain and Italy: Simulating control scenarios and multi-scale epidemics. Lancet 2020. [Google Scholar] [CrossRef]
- Bongolan, V.P.; Minoza, J.M.A.; de Castro, R.; Sevilleja, J.E. Age-stratified Infection Probabilities Combined with Quarantine-Modified SEIR Model in the Needs Assessments for COVID-19. medRxiv 2020. [Google Scholar] [CrossRef]
- Röst, G.; Bartha, F.A.; Bogya, N.; Boldog, P.; Dénes, A.; Tamás, F.; Horváth, K.J.; Juhász, A.; Nagy, C.; Tekeli, T.; et al. Early phase of the COVID-19 outbreak in Hungary and post-lockdown scenarios. medRxiv 2020. [Google Scholar] [CrossRef]
- Moghadas, S.M.; Shoukat, A.; Fitzpatrick, M.C.; Wells, C.R.; Sah, P.; Pandey, A.; Sachs, J.D.; Wang, Z.; Meyers, L.A.; Singer, B.H.; et al. Projecting hospital utilization during the COVID-19 outbreaks in the United States. Proc. Natl. Acad. Sci. USA 2020, 117, 9122–9126. [Google Scholar] [CrossRef]
- Aleta, A.; Moreno, Y. Evaluation of the potential incidence of COVID-19 and effectiveness of contention measures in Spain: A data-driven approach. medRxiv 2020. [Google Scholar] [CrossRef]
- Mo, H.; Zeng, G.; Ren, X.; Li, H.; Ke, C.; Tan, Y.; Cai, C.; Lai, K.; Chen, R.; Chan-Yeung, M.; et al. Longitudinal profile of antibodies against SARS-coronavirus in SARS patients and their clinical significance. Respirology 2006, 11, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.P.; Wang, N.C.; Chang, Y.H.; Tian, X.Y.; Na, D.Y.; Zhang, L.Y.; Zheng, L.; Lan, T.; Wang, L.F.; Liang, G.D. Duration of antibody responses after severe acute respiratory syndrome. Emerg. Infect. Dis. 2007, 13, 1562. [Google Scholar] [CrossRef] [PubMed]
- Alshukairi, A.N.; Khalid, I.; Ahmed, W.A.; Dada, A.M.; Bayumi, D.T.; Malic, L.S.; Althawadi, S.; Ignacio, K.; Alsalmi, H.S.; Al-Abdely, H.M.; et al. Antibody response and disease severity in healthcare worker MERS survivors. Emerg. Infect. Dis. 2016, 22, 1113. [Google Scholar] [CrossRef]
- Payne, D.C.; Iblan, I.; Rha, B.; Alqasrawi, S.; Haddadin, A.; Al Nsour, M.; Alsanouri, T.; Ali, S.S.; Harcourt, J.; Miao, C.; et al. Persistence of antibodies against Middle East respiratory syndrome coronavirus. Emerg. Infect. Dis. 2016, 22, 1824. [Google Scholar] [CrossRef]
- Ferguson, N.; Laydon, D.; Nedjati Gilani, G.; Imai, N.; Ainslie, K.; Baguelin, M.; Bhatia, S.; Boonyasiri, A.; Cucunuba, Z.; Dighe, A.; et al. Report 9: Impact of non-pharmaceutical interventions (NPIs) to reduce COVID19 mortality and healthcare demand. Imp. Coll. Lond. 2020, 10, 77482. [Google Scholar]
- Worldometers.Info. United Kingdom Coronavirus: Worldometer. Available online: https://www.worldometers.info/coronavirus/country/uk (accessed on 2 June 2020).
- Linton, N.M.; Kobayashi, T.; Yang, Y.; Hayashi, K.; Akhmetzhanov, A.R.; Jung, S.M.; Yuan, B.; Kinoshita, R.; Nishiura, H. Incubation period and other epidemiological characteristics of 2019 novel coronavirus infections with right truncation: A statistical analysis of publicly available case data. J. Clin. Med. 2020, 9, 538. [Google Scholar] [CrossRef]
- Who.Int. WHO|Contact Tracing. Available online: https://www.who.int/csr/disease/ebola/training/contact-tracing/en (accessed on 1 June 2020).
- Kanjo Eiman, K.C. Viral message passing framework for crowd and sensor applications. In Networked Digital Technologies; Springer: Berlin/Heidelberg, Germany, 2012; pp. 24–33. [Google Scholar]
- Eiman, K. Tools and architectural support for mobile phone based crowd control systems. Netw. Protoc. Algorithms 2012, 4, 4–14. [Google Scholar]
- Apple Newsroom. Apple and Google Partner on COVID-19 Contact Tracing Technology. Available online: https://www.apple.com/uk/newsroom/2020/04/apple-and-google-partner-on-covid-19-contact-tracing-technology (accessed on 12 May 2020).
- Ofcom.Org.UK. Adult’s Media Use and Attitudes Report 2019. Available online: https://www.ofcom.org.uk/__data/assets/pdf_file/0021/149124/adults-media-use-and-attitudes-report.pdf (accessed on 23 May 2020).
- Statista. Internet Usage on Mobile Phone by Age GB 2010–2019. Available online: https://www.statista.com/statistics/320974/internet-usage-on-mobile-phones-gb-age (accessed on 16 May 2020).
- O’brien, T.; Troutman-Jordan, M.; Hathaway, D.; Armstrong, S.; Moore, M. Acceptability of wristband activity trackers among community dwelling older adults. Geriatr. Nurs. 2015, 36, S21–S25. [Google Scholar] [CrossRef]
- BBC News. People-Tracking Wristbands Put to the Test. Available online: https://www.bbc.co.uk/news/technology-52409893 (accessed on 8 May 2020).
- Kwok, Y.L.A.; Gralton, J.; McLaws, M.L. Face touching: A frequent habit that has implications for hand hygiene. Am. J. Infect. Control. 2015, 43, 112–114. [Google Scholar] [CrossRef]
- Anderez, D.O.; Lotfi, A.; Pourabdollah, A. Temporal convolution neural network for food and drink intake recognition. In Proceedings of the 12th ACM International Conference on PErvasive Technologies Related to Assistive Environments, Rhodes, Greece, 6–8 June 2019; pp. 580–586. [Google Scholar]
- Ortega Anderez, D.; Lotfi, A.; Pourabdollah, A. Eating and drinking gesture spotting and recognition using a novel adaptive segmentation technique and a gesture discrepancy measure. Expert Syst. Appl. 2020, 140, 112888. [Google Scholar] [CrossRef]
- Who.Int. Modes of Transmission of Virus Causing COVID-19: Implications for IPC Precaution Recommendations. Available online: https://www.who.int/news-room/commentaries/detail/modes-of-transmission-of-virus-causing-covid-19-implications-for-ipc-precaution-recommendations (accessed on 2 May 2020).
- Leder, K.; Newman, D. Respiratory infections during air travel. Intern. Med. J. 2005, 35, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Stahlhofen, W.; Rudolf, G.; James, A. Inter comparison of experimental regional aerosol deposition data. J. Aerosol Med. 1989, 2, 285–308. [Google Scholar] [CrossRef]
- Booth, T.F.; Kournikakis, B.; Bastien, N.; Ho, J.; Kobasa, D.; Stadnyk, L.; Li, Y.; Spence, M.; Paton, S.; Henry, B.; et al. Detection of airborne severe acute respiratory syndrome (SARS) coronavirus and environmental contamination in SARS outbreak units. J. Infect. Dis. 2005, 191, 1472–1477. [Google Scholar] [CrossRef] [PubMed]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef]
- Bake, B.; Larsson, P.; Ljungkvist, G.; Ljungström, E.; Olin, A. Exhaled particles and small airways. Respir. Res. 2019, 20, 8. [Google Scholar] [CrossRef]
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Di Napoli, R. Features, evaluation and treatment coronavirus (COVID-19). In Statpearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Leung, N.H.; Chu, D.K.; Shiu, E.Y.; Chan, K.H.; McDevitt, J.J.; Hau, B.J.; Yen, H.L.; Li, Y.; Ip, D.K.M.; Peiris, J.S.M.; et al. Respiratory virus shedding in exhaled breath and efficacy of face masks. Nat. Med. 2020, 26, 676–680. [Google Scholar] [CrossRef]
- Feng, S.; Shen, C.; Xia, N.; Song, W.; Fan, M.; Cowling, B.J. Rational use of face masks in theCOVID-19 pandemic. Lancet Respir. Med. 2020, 8, 434–436. [Google Scholar] [CrossRef]
- Musicant, O.; Lotan, T. Can novice drivers be motivated to use a smartphone based app that monitors their behavior? Transp. Res. Part. F Traffic Psychol. Behav. 2016, 42, 544–557. [Google Scholar] [CrossRef]
- Marteau, T.M.; Ashcroft, R.E.; Oliver, A. Using financial incentives to achieve healthy behaviour. BMJ 2009, 338, b1415. [Google Scholar] [CrossRef]
- O’Donoghue, T.; Rabin, M. Doing it now or later. Am. Econ. Rev. 1999, 89, 103–124. [Google Scholar] [CrossRef]
- Drobetz, R.; Hänggi, J.; Maercker, A.; Kaufmann, K.; Jäncke, L.; Forstmeier, S. Structural brain correlates of delay of gratification in the elderly. Behav. Neurosci. 2014, 128, 134. [Google Scholar] [CrossRef]
- Wang, Y.; Sloan, F.A. Present bias and health. J. Risk Uncertain. 2018, 57, 177–198. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, K.; Volz, K.G.; Sutter, M.; von Cramon, D.Y. What do I want and when do I want it: Brain correlates of decisions made for self and other. PLoS ONE 2013, 8, e73531. [Google Scholar] [CrossRef]
- Delaney, L.; Lades, L.K. Present bias and everyday self-control failures: A day reconstruction study. J. Behav. Decis. Mak. 2017, 30, 1157–1167. [Google Scholar] [CrossRef]
- Wu, Y.; Li, L.; Yuan, B.; Tian, X. Individual differences in resting-state functional connectivity predict procrastination. Personal. Individ. Differ. 2016, 95, 62–67. [Google Scholar] [CrossRef]
- Shivayogi, P. Vulnerable population and methods for their safeguard. Perspect. Clin. Res. 2013, 4, 53. [Google Scholar] [CrossRef]
- Vegesna, A.; Tran, M.; Angelaccio, M.; Arcona, S. Remote patient monitoring via non-invasivedigital technologies: A systematic review. Telemed. E Health 2017, 23, 3–17. [Google Scholar] [CrossRef]
- Roupa, Z.; Nikas, M.; Gerasimou, E.; Zafeiri, V.; Giasyrani, L.; Kazitori, E.; Sotiropoulou, P. The use of technology by the elderly. Health Sci. J. 2010, 4, 118. [Google Scholar]
- Norberg, P.A.; Horne, D.R.; Horne, D.A. The privacy paradox: Personal information disclosure intentions versus behaviors. J. Consum. Aff. 2007, 41, 100–126. [Google Scholar] [CrossRef]
- Xu, H.; Luo, X.R.; Carroll, J.M.; Rosson, M.B. The personalization privacy paradox: An exploratory study of decision making process for location-aware marketing. Decis. Support. Syst. 2011, 51, 42–52. [Google Scholar] [CrossRef]
- Schwartz, P.M. Property, privacy, and personal data. Harv. L. Rev. 2003, 117, 2056. [Google Scholar] [CrossRef]
- Bizon, W.; Poszewiecki, A. The willingness to trade privacy in the context of WTA and WTP. Int. J. Trade Econ. Financ. 2016, 7, 121–124. [Google Scholar] [CrossRef]
- Milberg, S.J.; Burke, S.J.; Smith, H.J.; Kallman, E.A. Values, personal information privacy, and regulatory approaches. Commun. ACM 1995, 38, 65–74. [Google Scholar] [CrossRef]
- Ullah, F.; Abdullah, A.H.; Kaiwartya, O.; Cao, Y. TraPy-MAC: Traffic priority aware medium access control protocol for wireless body area network. J. Med. Syst. 2017, 41, 93. [Google Scholar] [CrossRef]
- Prasad, M.; Liu, Y.T.; Li, D.L.; Lin, C.T.; Shah, R.R.; Kaiwartya, O.P. A new mechanism for data visualization with TSK-type preprocessed collaborative fuzzy rule based system. J. Artif. Intell. Soft Comput. Res. 2017, 7, 33–46. [Google Scholar] [CrossRef]

| Parameter | Unit | Description | Value | Comments |
|---|---|---|---|---|
| N | N People | Population | 67,838,235 [15] | Total population in the UK as of 2020 |
| Ev0 | N People | Vulnerable Exposed | 2 | Vulnerable individuals exposed to the disease at the beginning of the outbreak |
| E0 | N People | Exposed | 4 | Non-vulnerable individuals exposed to the disease at the beginning of the outbreak |
| Iv0 | N People | Infected | 0 | Vulnerable infected individuals at the beginning of the outbreak |
| I0 | N People | Infected | 1 | Non-vulnerable infected individuals at the beginning of the outbreak |
| Tinc | Days | Incubation period | 5.6 [16] | σ = 1/Tinc, where Tinc is the time it takes for an exposed individual to become infectious |
| Tlat | Days | Latent period | 7.5 [16] | γ = 1/Tlat, where Tlat is the time it takes for an infectious individual to recover |
| µv | Vulnerable deaths/Vulnerable Infected | Vulnerable Case Fatality Rate | [0.005–0.037, 95% CI]% | Case fatality rate of COVID-19 on vulnerable individuals |
| µ | Non-vulnerable deaths/non-vulnerable Infected | Non-vulnerable Case Fatality Rate | [0.000007–0.000011, 95% CI]% | Case fatality rate of COVID-19 on non-vulnerable individuals |
| pv | *- | Vulnerable probability | 0.2 | Probability of an individual being vulnerable to the disease |
| η | *- | Fear Factor | 0.33 | Fear factor caused by the recommendation made by the UK government for vulnerable individuals to stay at home for at least 12 weeks at the beginning of the outbreak and the widespread severity of the disease within this group |
| β0 | 1/(person*day) | Initial Contact Rate | [0.5–2.1, 95% CI] | Contact rate at the beginning of the outbreak |
| β1 | 1/(person*day) | Contact Rate 1 | [0.9–0.95, 95% CI] * β0 | Contact rate after the mandate of case-based self-isolation |
| β2 | 1/(person*day) | Contact Rate 2 | [0.9–0.95, 95% CI] * β1 | Contact rate after government encouragement for social distancing |
| β3 | 1/(person*day) | Contact Rate 3 | [0.75–0.85, 95% CI] * β2 | Contact rate after schools closure |
| β4 | 1/(person*day) | Contact Rate 4 | [0.40–0.60, 95% CI] * β3 | Contact rate after lockdown order and banning of public events |
| β5 | 1/(person*day) | Contact Rate 5 | [1.1–1.9, 95% CI] * β4 | Contact rate after recommendation for people to go back to work |
| βvi | 1/(person*day) | Vulnerable Contact Rate | η * βi | Contact rate of vulnerable individuals. |
| Decrease in βv | 10% | 20% | 30% | 40% | 50% | 60% | 70% | 80% | 90% |
| Decrease in Number of Deaths | 7699 | 15,512 | 23,428 | 31,434 | 39,519 | 47,671 | 55,876 | 64,122 | 72,395 |
| Decrease in βv | 10% | 20% | 30% | 40% | 50% | 60% | 70% | 80% | 90% |
| Decrease in Number of Deaths | 3681 | 7406 | 11,172 | 14,975 | 18,810 | 22,673 | 26,559 | 30,464 | 34,383 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anderez, D.O.; Kanjo, E.; Pogrebna, G.; Kaiwartya, O.; Johnson, S.D.; Hunt, J.A. A COVID-19-Based Modified Epidemiological Model and Technological Approaches to Help Vulnerable Individuals Emerge from the Lockdown in the UK. Sensors 2020, 20, 4967. https://doi.org/10.3390/s20174967
Anderez DO, Kanjo E, Pogrebna G, Kaiwartya O, Johnson SD, Hunt JA. A COVID-19-Based Modified Epidemiological Model and Technological Approaches to Help Vulnerable Individuals Emerge from the Lockdown in the UK. Sensors. 2020; 20(17):4967. https://doi.org/10.3390/s20174967
Chicago/Turabian StyleAnderez, Dario Ortega, Eiman Kanjo, Ganna Pogrebna, Omprakash Kaiwartya, Shane D. Johnson, and John Alan Hunt. 2020. "A COVID-19-Based Modified Epidemiological Model and Technological Approaches to Help Vulnerable Individuals Emerge from the Lockdown in the UK" Sensors 20, no. 17: 4967. https://doi.org/10.3390/s20174967
APA StyleAnderez, D. O., Kanjo, E., Pogrebna, G., Kaiwartya, O., Johnson, S. D., & Hunt, J. A. (2020). A COVID-19-Based Modified Epidemiological Model and Technological Approaches to Help Vulnerable Individuals Emerge from the Lockdown in the UK. Sensors, 20(17), 4967. https://doi.org/10.3390/s20174967







