3D Printable Dry EEG Electrodes with Coiled-Spring Prongs
Abstract
1. Introduction
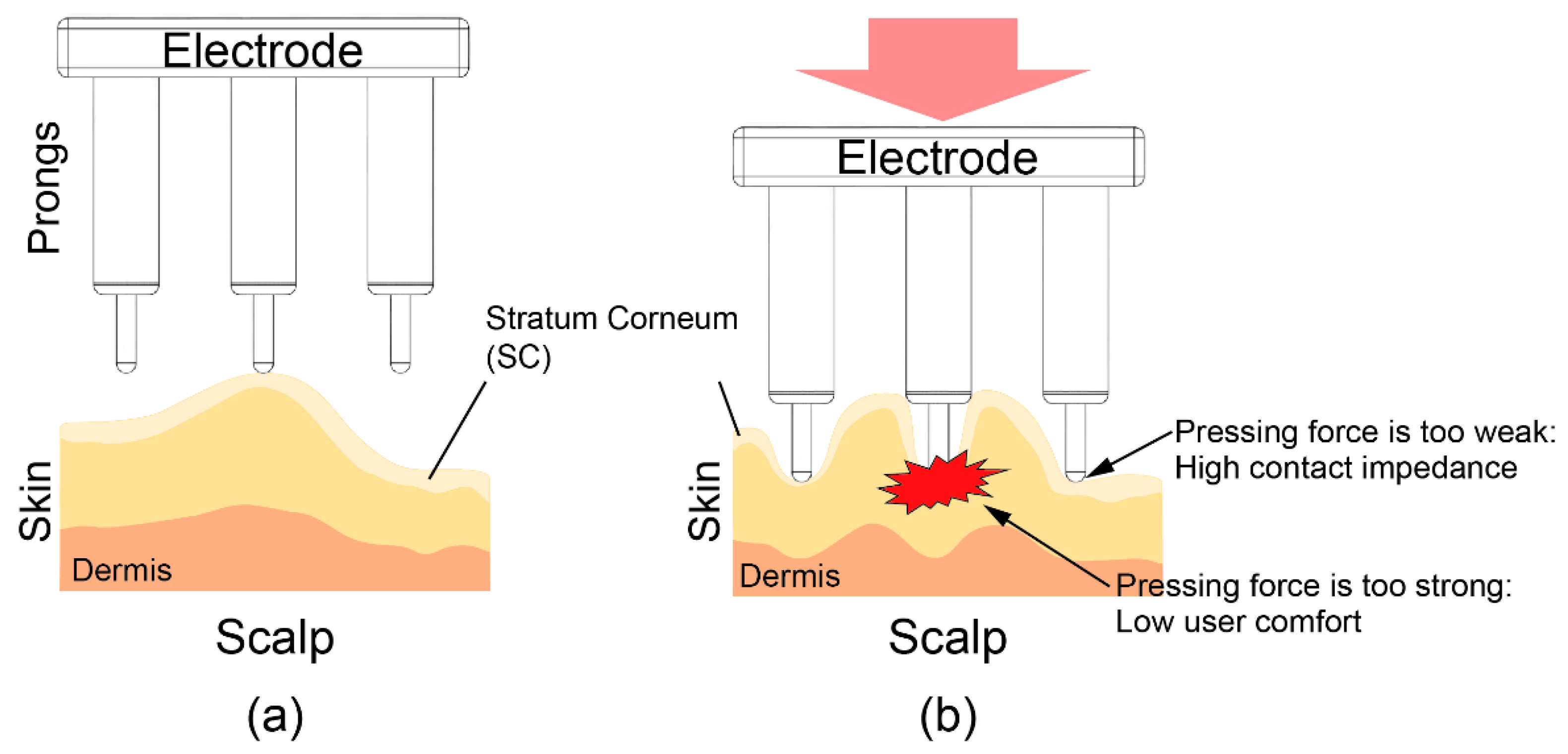
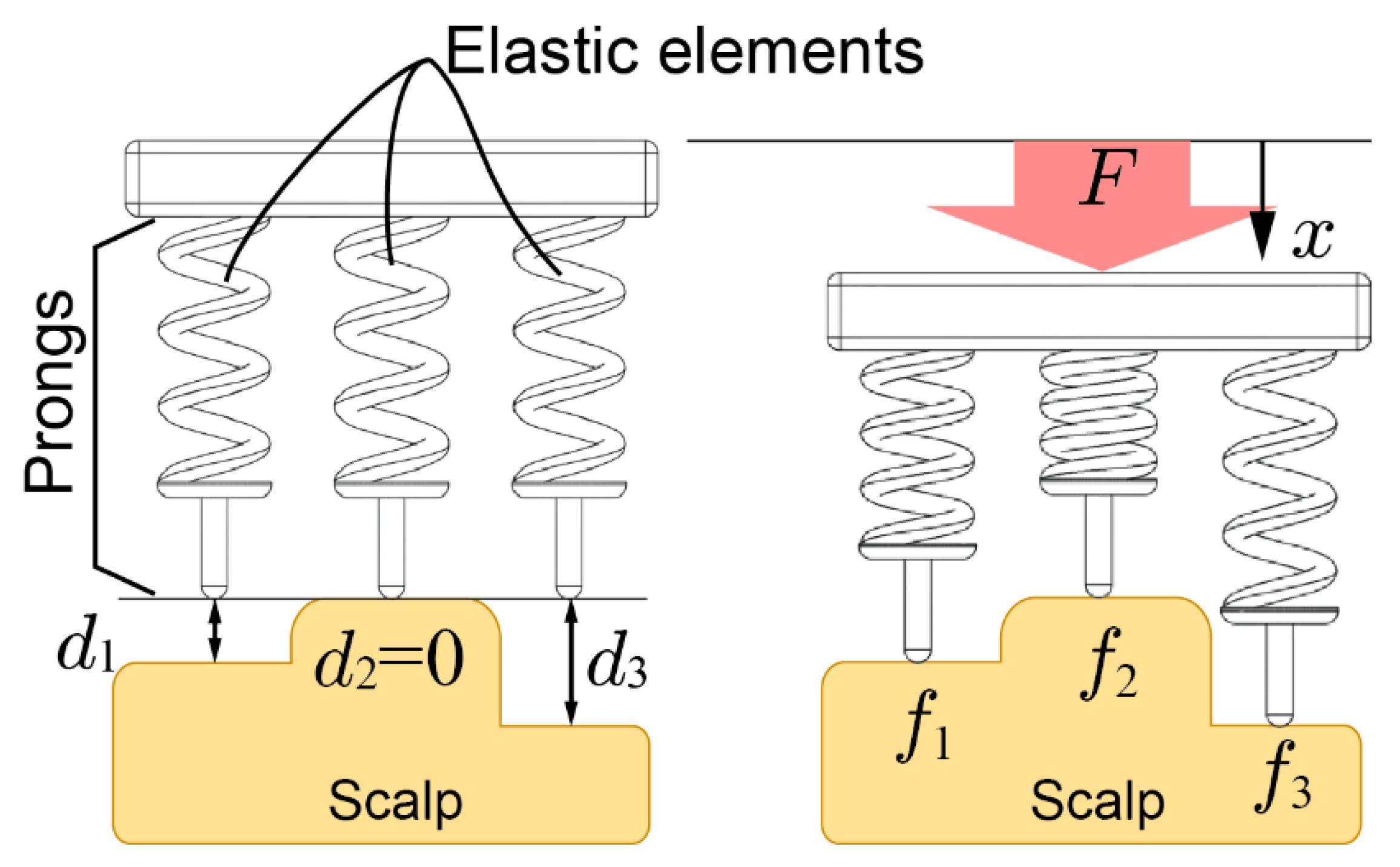
2. Contact Model of Electrode
3. Design and Development of 3D Printed Electrodes
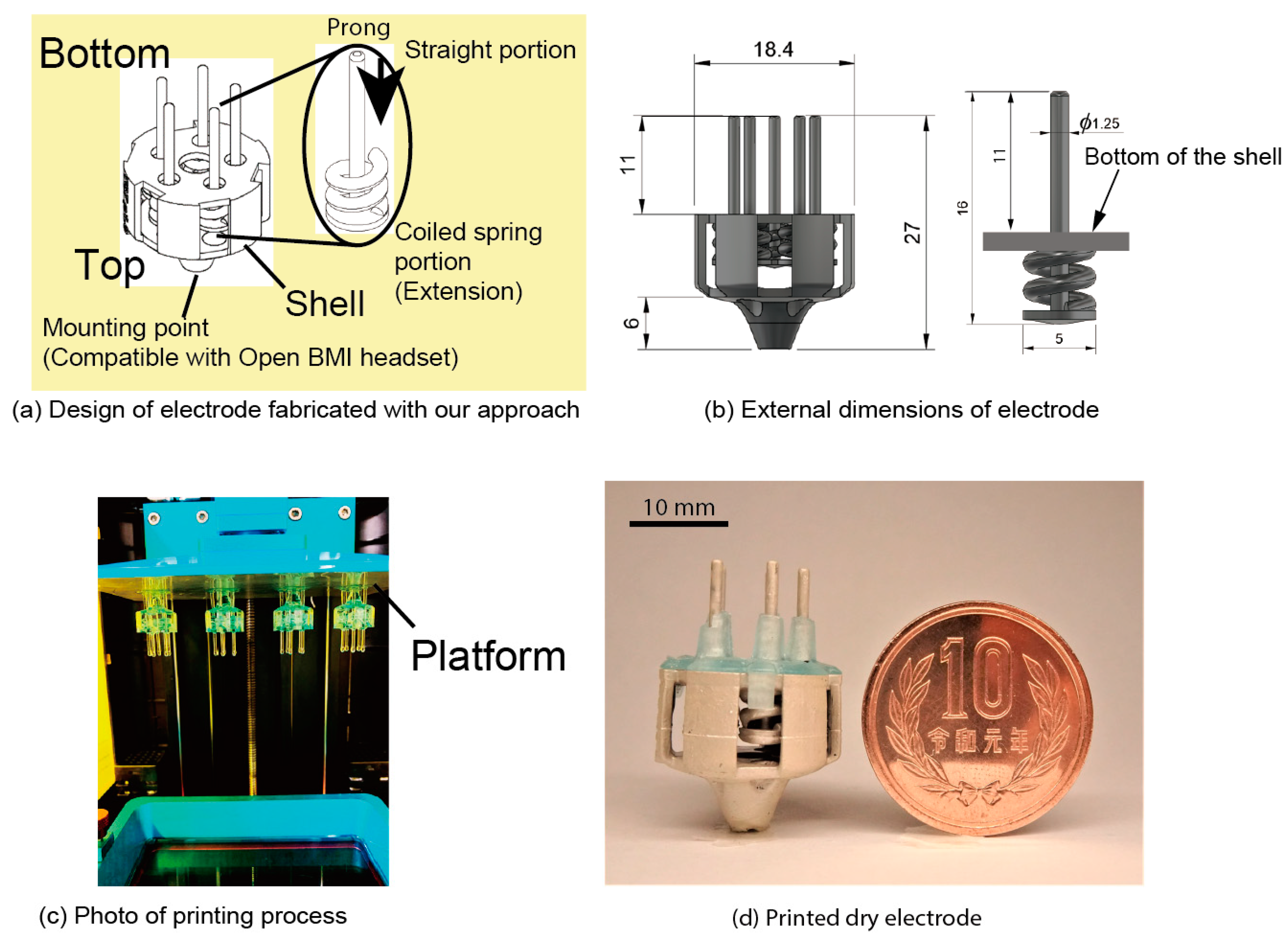
3.1. Electrode Design
3.2. 3D Printing
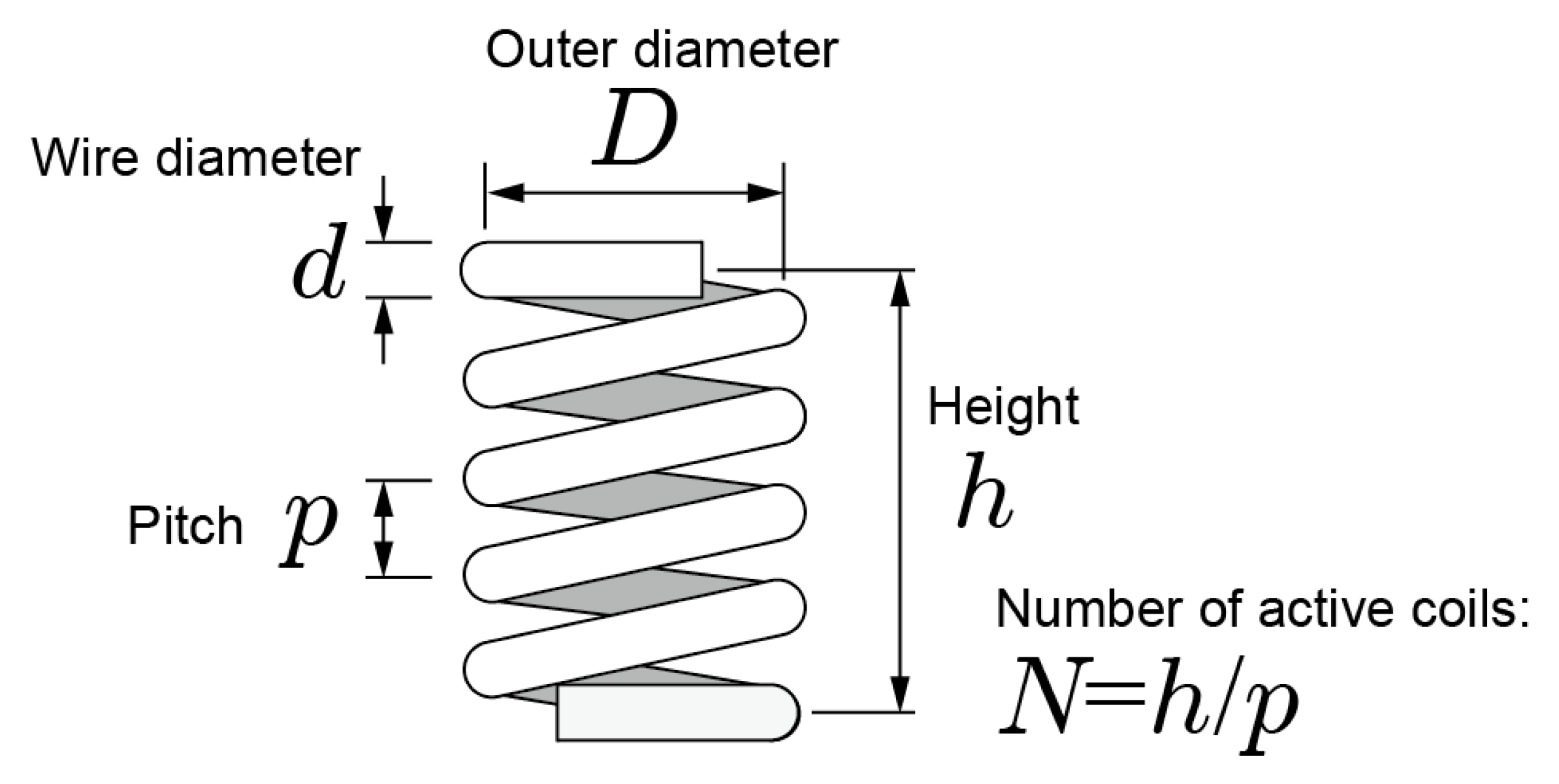
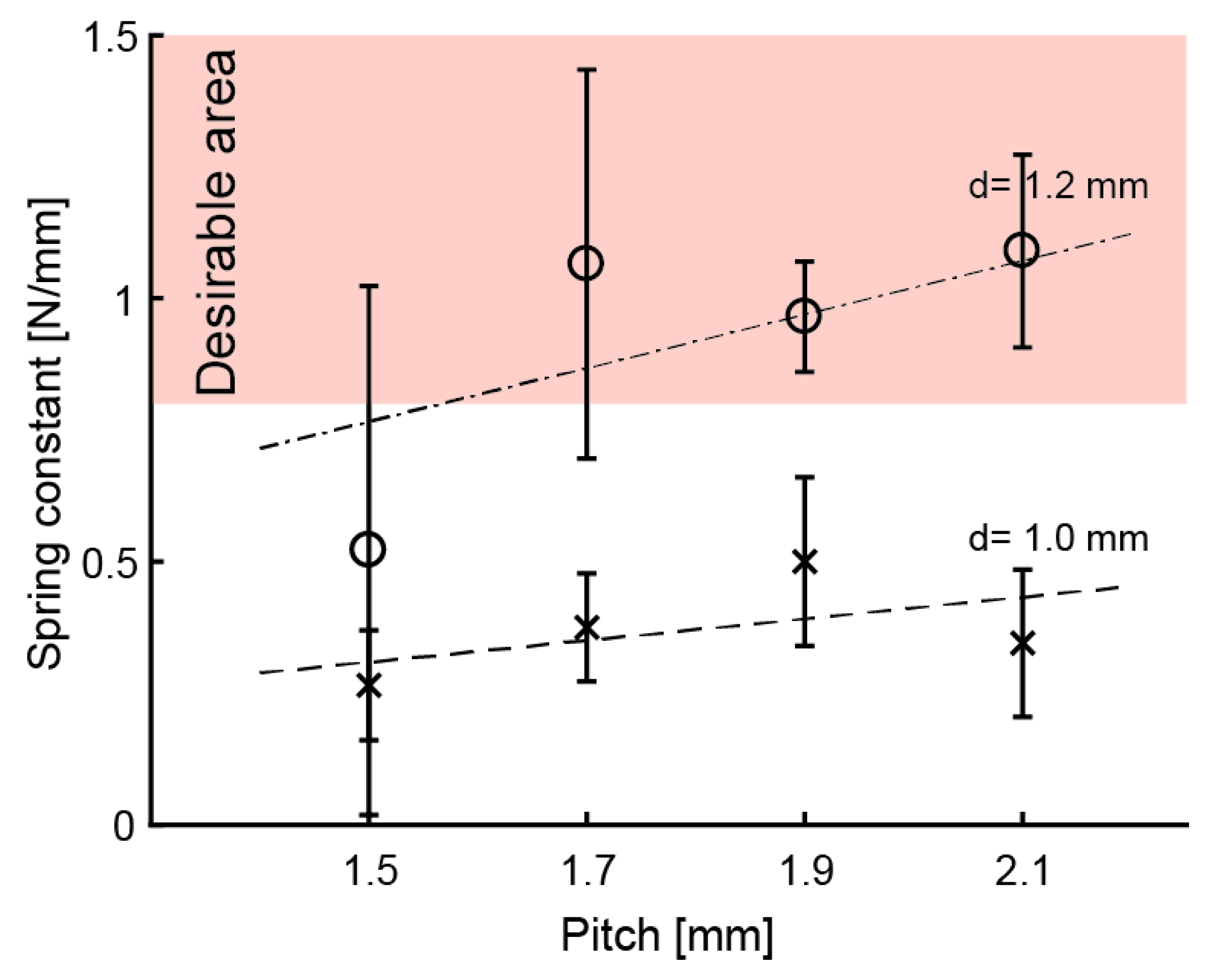
3.3. Coiled-Spring Design
3.4. Mechanical Evaluation
3.5. Elasticity Evaluation of Electrode
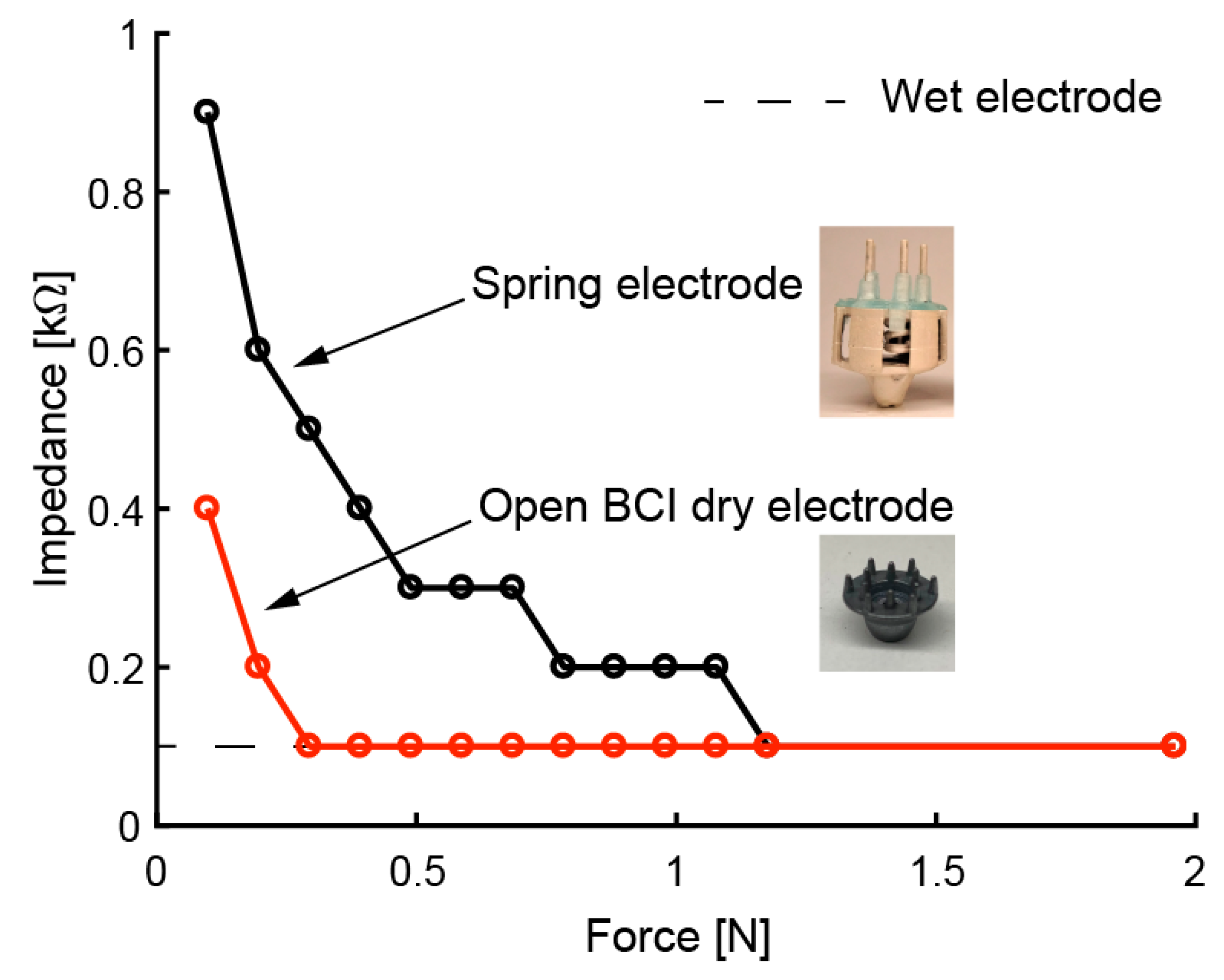
4. Electrical Evaluation
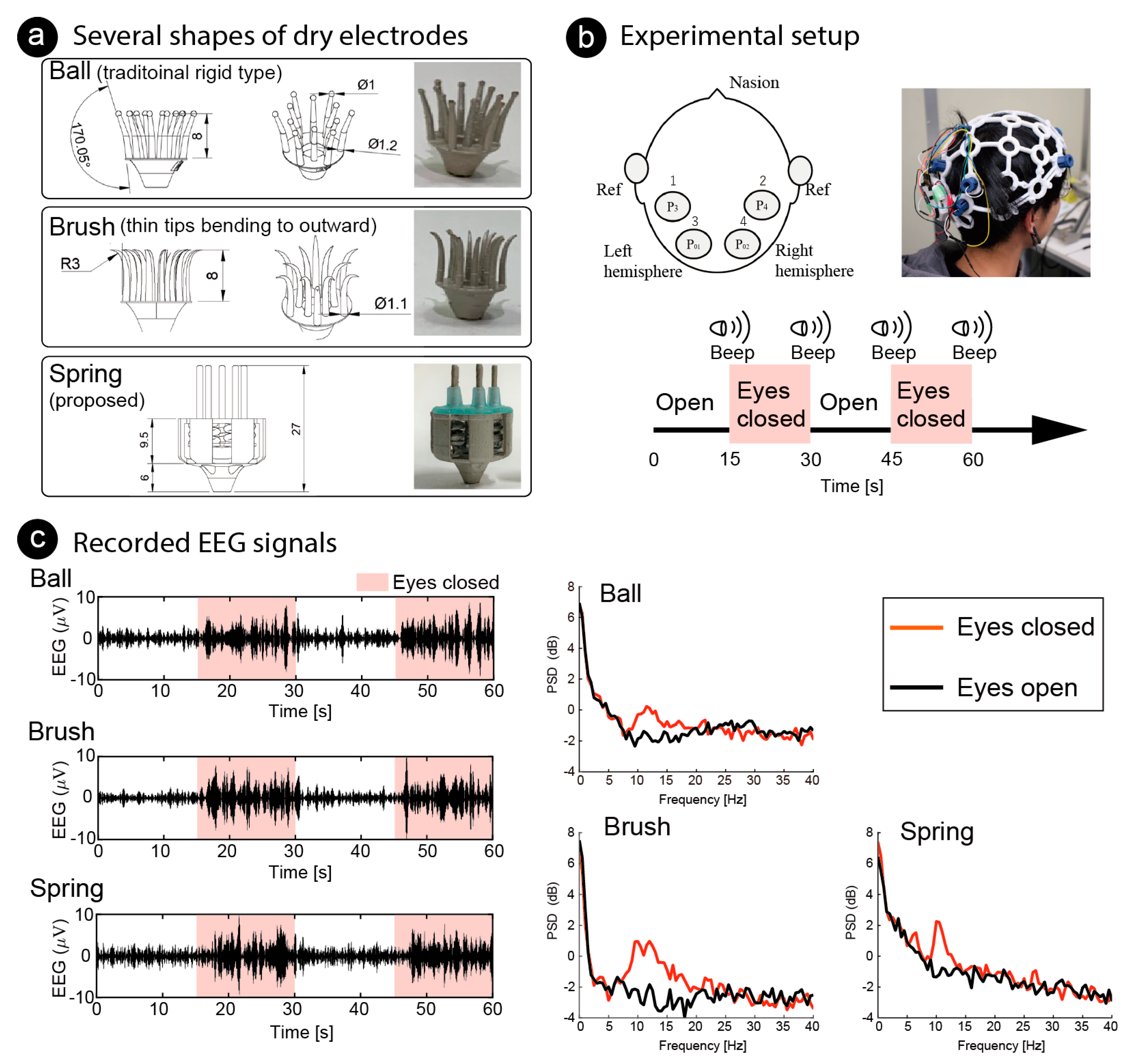
5. Functional Testing
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lebedev, M.A.; Nicolelis, M.A.L. Brain-machine interfaces: Past, present and future. Trends Neurosci. 2006, 29, 536–546. [Google Scholar] [CrossRef]
- Wolpaw, J.R.; Birbaumer, N.; McFarland, D.J.; Pfurtscheller, G.; Vaughan, T.M. Brain-computer interfaces for communication and control. Clin. Neurophysiol. 2002, 113, 767–791. [Google Scholar] [CrossRef]
- Dornhege, G.; Millán, J.d.R.; Hinterberger, T.; McFarland, D.; Müller, K.-R. Non-Invasive Brain-Computer Interface Research at the Wadsworth Center. In Towards Brain-Computer Interfacing; The MIT Press: Cambridge, MA, USA, 2007; pp. 31–42. [Google Scholar]
- Katona, J.; Ujbanyi, T.; Sziladi, G.; Kovari, A. Electroencephalogram-Based Brain-Computer Interface for Internet of Robotic Things. In Intelligent Technologies and Robotics; Springer Nature: Cham, Switzerland, 2019; pp. 253–275. [Google Scholar]
- Katona, J.; Ujbanyi, T.; Sziladi, G.; Kovari, A. Speed control of Festo Robotino mobile robot using NeuroSky MindWave EEG headset based brain-computer interface. In Proceedings of the 2016 7th IEEE International Conference on Cognitive Infocommunications (CogInfoCom), Wroclaw, Poland, 16–18 October 2016; pp. 000251–000256. [Google Scholar]
- Townsend, G.; LaPallo, B.K.; Boulay, C.B.; Krusienski, D.J.; Frye, G.E.; Hauser, C.K.; Schwartz, N.E.; Vaughan, T.M.; Wolpaw, J.R.; Sellers, E.W. A novel P300-based brain-computer interface stimulus presentation paradigm: Moving beyond rows and columns. Clin. Neurophysiol. 2010, 121, 1109–1120. [Google Scholar] [CrossRef] [PubMed]
- Leeb, R.; Lee, F.; Keinrath, C.; Scherer, R.; Bischof, H.; Pfurtscheller, G. Brain–Computer Communication: Motivation, Aim, and Impact of Exploring a Virtual Apartment. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Selfslagh, A.; Shokur, S.; Campos, D.S.F.; Donati, A.R.C.; Almeida, S.; Yamauti, S.Y.; Coelho, D.B.; Bouri, M.; Nicolelis, M.A.L. Non-invasive, Brain-controlled Functional Electrical Stimulation for Locomotion Rehabilitation in Individuals with Paraplegia. Sci. Rep. 2019, 9, 1–17. [Google Scholar] [CrossRef]
- Lopez-Gordo, M.A.; Sanchez Morillo, D.; Pelayo Valle, F. Dry EEG electrodes. Sensors 2014, 14, 12847–12870. [Google Scholar] [CrossRef]
- Lin, C.T.; Liao, L.D.; Liu, Y.H.; Wang, I.J.; Lin, B.S.; Chang, J.Y.; Lin, C.T.; Liao, L.D.; Liu, Y.H.; Wang, I.J.; et al. Novel dry polymer foam electrodes for long-term EEG measurement. IEEE Trans. Biomed. Eng. 2011, 58, 1200–1207. [Google Scholar] [CrossRef]
- Gargiulo, G.; Calvo, R.A.; Bifulco, P.; Cesarelli, M.; Jin, C.; Mohamed, A.; van Schaik, A. A new EEG recording system for passive dry electrodes. Clin. Neurophysiol. 2010, 121, 686–693. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Yamamoto, T.; Ozawa, T. Characteristics of skin admittance for dry electrodes and the measurement of skin moisturisation. Med. Biol. Eng. Comput. 1986, 24, 71–77. [Google Scholar] [CrossRef]
- Mathewson, K.E.; Harrison, T.J.L.; Kizuk, S.A.D. High and dry? Comparing active dry EEG electrodes to active and passive wet electrodes. Psychophysiology 2017, 54, 74–82. [Google Scholar] [CrossRef]
- Grozea, C.; Voinescu, C.D.; Fazli, S. Bristle-sensors—Low-cost flexible passive dry EEG electrodes for neurofeedback and BCI applications. J. Neural Eng. 2011, 8. [Google Scholar] [CrossRef] [PubMed]
- Ng, W.C.; Seet, H.L.; Lee, K.S.; Ning, N.; Tai, W.X.; Sutedja, M.; Fuh, J.Y.H.; Li, X.P. Micro-spike EEG electrode and the vacuum-casting technology for mass production. J. Mater. Process. Technol. 2009, 209, 4434–4438. [Google Scholar] [CrossRef]
- Vanlerberghe, F.; De Volder, M.; De Beeck, M.O.; Penders, J.; Reynaerts, D.; Puers, R.; Van Hoof, C. 2-Scale topography dry electrode for biopotential measurements. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, EMBS 2011, Boston, MA, USA, 30 August–3 September 2011; pp. 1892–1895. [Google Scholar] [CrossRef]
- Arai, M.; Nishinaka, Y.; Miki, N. Long-term electroencephalogram measurement using polymer-based dry microneedle electrode. In Proceedings of the 18th International Conference on Solid-State Sensors, Actuators Microsystems, TRANSDUCERS 2015, Anchorage, Alaska, 21–25 June 2015; pp. 81–84. [Google Scholar] [CrossRef]
- Ruffini, G.; Dunne, S.; Fuentemilla, L.; Grau, C.; Farrés, E.; Marco-Pallarés, J.; Watts, P.C.P.; Silva, S.R.P. First human trials of a dry electrophysiology sensor using a carbon nanotube array interface. Sens. Actuators A Phys. 2008, 144, 275–279. [Google Scholar] [CrossRef]
- Chen, Y.H.; Op de Beeck, M.; Vanderheyden, L.; Carrette, E.; Mihajlović, V.; Vanstreels, K.; Grundlehner, B.; Gadeyne, S.; Boon, P.; van Hoof, C. Soft, comfortable polymer dry electrodes for high quality ECG and EEG recording. Sensors 2014, 14, 23758–23780. [Google Scholar] [CrossRef]
- Gao, K.P.; Yang, H.J.; Wang, X.L.; Yang, B.; Liu, J.Q. Soft pin-shaped dry electrode with bristles for EEG signal measurements. Sens. Actuators A Phys. 2018, 283, 348–361. [Google Scholar] [CrossRef]
- Naruse, Y. Development of mobile wireless EEG system with dry electrode. In Proceedings of the SICE Life Engineering Symposium 2014, Kanazawa, Japan, 17–19 September 2014; pp. 130–132. [Google Scholar]
- De Liao, L.; Wang, I.J.; Chen, S.F.; Chang, J.Y.; Lin, C.T. Design, fabrication and experimental validation of a novel dry-contact sensor for measuring electroencephalography signals without skin preparation. Sensors 2011, 11, 5819–5834. [Google Scholar] [CrossRef]
- Nathan, V.; Jafari, R. Reducing the noise level of EEG signal acquisition through reconfiguration of dry contact electrodes. In Proceedings of the IEEE Biomedical Circuits and Systems Conference (BioCAS) 2014, Lausanne, Switzerland, 22–24 October 2014; pp. 572–575. [Google Scholar] [CrossRef]
- Mota, A.R.; Duarte, L.; Rodrigues, D.; Martins, A.C.; Machado, A.V.; Vaz, F.; Fiedler, P.; Haueisen, J.; Nóbrega, J.M.; Fonseca, C. Development of a quasi-dry electrode for EEG recording. Sens. Actuators A Phys. 2013, 199, 310–317. [Google Scholar] [CrossRef]
- Karyappa, R.; Hashimoto, M. Chocolate-based Ink Three-dimensional Printing (Ci3DP). Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Wang, J.; Goyanes, A.; Gaisford, S.; Basit, A.W. Stereolithographic (SLA) 3D printing of oral modified-release dosage forms. Int. J. Pharm. 2016, 503, 207–212. [Google Scholar] [CrossRef]
- Martinez, P.R.; Goyanes, A.; Basit, A.W.; Gaisford, S. Influence of Geometry on the Drug Release Profiles of Stereolithographic (SLA) 3D-Printed Tablets. AAPS PharmSciTech 2018, 19, 3355–3361. [Google Scholar] [CrossRef]
- Goyanes, A.; Det-Amornrat, U.; Wang, J.; Basit, A.W.; Gaisford, S. 3D scanning and 3D printing as innovative technologies for fabricating personalized topical drug delivery systems. J. Control. Release 2016, 234, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Salvo, P.; Raedt, R.; Carrette, E.; Schaubroeck, D.; Vanfleteren, J.; Cardon, L. A 3D printed dry electrode for ECG/EEG recording. Sens. Actuators A Phys. 2012, 174, 96–102. [Google Scholar] [CrossRef]
- Krachunov, S.; Casson, A. 3D Printed Dry EEG Electrodes. Sensors 2016, 16, 1635. [Google Scholar] [CrossRef] [PubMed]
- Velcescu, A.; Lindley, A.; Cursio, C.; Krachunov, S.; Beach, C.; Brown, C.A.; Jones, A.K.P.; Casson, A.J. Flexible 3D-printed EEG electrodes. Sensors 2019, 19, 1650. [Google Scholar] [CrossRef]
- Peele, B.N.; Wallin, T.J.; Zhao, H.; Shepherd, R.F. 3D printing antagonistic systems of artificial muscle using projection stereolithography. Bioinspiration Biomim. 2015, 10. [Google Scholar] [CrossRef]
- Collins, J.A.; Busby, H.R.; Staab, G.H. Mechanical Design of Machine Elements and Machines: A Failure Prevention Perspective, 2nd ed.; Wiley: New York, NY, USA, 2009; ISBN 0470413034. [Google Scholar]
- Owda, A.Y.; Casson, A.J. Electrical properties, accuracy, and multi-day performance of gelatine phantoms for electrophysiology. BioRxiv 2020. [Google Scholar] [CrossRef]
- Hairston, W.D.; Slipher, G.A.; Yu, A.B. Ballistic gelatin as a putative substrate for EEG phantom devices. arXiv 2016, arXiv:1609.07691. [Google Scholar]
- Farrer, A.I.; Odéen, H.; de Bever, J.; Coats, B.; Parker, D.L.; Payne, A.; Christensen, D.A. Characterization and evaluation of tissue-mimicking gelatin phantoms for use with MRgFUS. J. Ther. Ultrasound 2015, 3, 9. [Google Scholar] [CrossRef]
- Craig, A.; Moses, P.; Tran, Y.; McIsaac, P.; Kirkup, L. The effectiveness of a hands-free environmental control system for the profoundly disabled. Arch. Phys. Med. Rehabil. 2002, 83, 1455–1458. [Google Scholar] [CrossRef]
- Craig, A.; McIsaac, P.; Tran, Y.; Kirkup, L.; Searle, A. Alpha wave reactivity following eye closure: A potential method of remote hands free control for the disabled. Technol. Disabil. 1999, 10, 187–194. [Google Scholar] [CrossRef]
- Binnie, C.D.; Cooper, R.; Mauguiere, F.; Osselton, J.W.; Prior, P.F.; Tedman, B.M. Clinical Neurophysiology, Vol. 2 EEG, Paediatric Neurophysiology, Special Techniques and Applications, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2003; ISBN 0444512578. [Google Scholar]
- Brown, T.; Johnson, R.; Milavetz, G. Identifying periods of drowsy driving using EEG. In Proceedings of the Annals of Advances in Automotive Medicine, Quebec City, QC, Canada, 23–25 September 2013; Volume 57, pp. 99–108. [Google Scholar]
- Yeo, M.V.M.; Li, X.; Shen, K.; Wilder-Smith, E.P.V. Can SVM be used for automatic EEG detection of drowsiness during car driving? Saf. Sci. 2009, 47, 115–124. [Google Scholar] [CrossRef]
- Li, G.; Chung, W.Y. A context-aware EEG headset system for early detection of driver drowsiness. Sensors 2015, 15, 20873–20893. [Google Scholar] [CrossRef] [PubMed]
- Katona, J.; Kovari, A. Examining the Learning Efficiency by a Brain-Computer Interface System. Acta Polytech. Hung. 2018, 15. [Google Scholar] [CrossRef]
- Katona, J.; Kovari, A. A Brain–Computer Interface Project Applied in Computer Engineering. IEEE Trans. Educ. 2016, 59, 319–326. [Google Scholar] [CrossRef]
- Available online: https://github.com/1nakatan/EEG_electrodes_with_coiled-spring_prongs (accessed on 20 August 2020).



| Printing Parameters | Value |
|---|---|
| Layer thickness | 50 μm |
| Normal exposure time | 10 s |
| Bottom exposure time | 75 s |
| Resin temperature | 20–25 °C |
| Post-exposure time | 180 min. |
| Sample | Prong 1 | Prong 2 | Prong 3 | Prong 4 | Prong 5 | Mean ± S.D. |
|---|---|---|---|---|---|---|
| ID 1 | 1.07 | 0.90 | 1.15 | 1.17 | 0.72 | 1.00 ± 0.19 |
| ID 2 | 1.42 | 1.08 | 1.25 | 1.39 | 1.22 | 1.27 ± 0.14 |
| ID 3 | 0.90 | 0.73 | 0.82 | 0.77 | 1.27 | 0.90 ± 0.22 |
| ID 4 | 0.78 | 1.03 | 0.61 | 0.68 | 0.73 | 0.76 ± 0.16 |
| Band with a Significant Difference (Hz) | |||
|---|---|---|---|
| Participant | Ball | Brush | Spring |
| PID1 | 9.0–14.0, 15.0–17.5 | 8.0–19.5 | 9.5–13.0 |
| PID2 | 10.5–12.5 | 10.5–12.5 | 9.5–10.5, 11.5–13.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kimura, M.; Nakatani, S.; Nishida, S.-I.; Taketoshi, D.; Araki, N. 3D Printable Dry EEG Electrodes with Coiled-Spring Prongs. Sensors 2020, 20, 4733. https://doi.org/10.3390/s20174733
Kimura M, Nakatani S, Nishida S-I, Taketoshi D, Araki N. 3D Printable Dry EEG Electrodes with Coiled-Spring Prongs. Sensors. 2020; 20(17):4733. https://doi.org/10.3390/s20174733
Chicago/Turabian StyleKimura, Masaya, Shintaro Nakatani, Shin-Ichiro Nishida, Daiju Taketoshi, and Nozomu Araki. 2020. "3D Printable Dry EEG Electrodes with Coiled-Spring Prongs" Sensors 20, no. 17: 4733. https://doi.org/10.3390/s20174733
APA StyleKimura, M., Nakatani, S., Nishida, S.-I., Taketoshi, D., & Araki, N. (2020). 3D Printable Dry EEG Electrodes with Coiled-Spring Prongs. Sensors, 20(17), 4733. https://doi.org/10.3390/s20174733





