Experimental Assessment of Sleep-Related Parameters by Passive Infrared Sensors: Measurement Setup, Feature Extraction, and Uncertainty Analysis
Abstract
1. Introduction
1.1. Sleep Stages
1.2. Sleep Monitoring Sensors
2. Materials and Methods
2.1. Measurement Set-Up
- Operative range: 5 m;
- Field of view: 140° (horizontal);
- Time delay: 2 s;
- Measurement range: −20–40 °C;
- Trigger mode: Continuous sensing;
- Working Voltage: 3 V.
Preliminary Investigation
2.2. Data Acquisition
2.2.1. Data Analysis and Processing
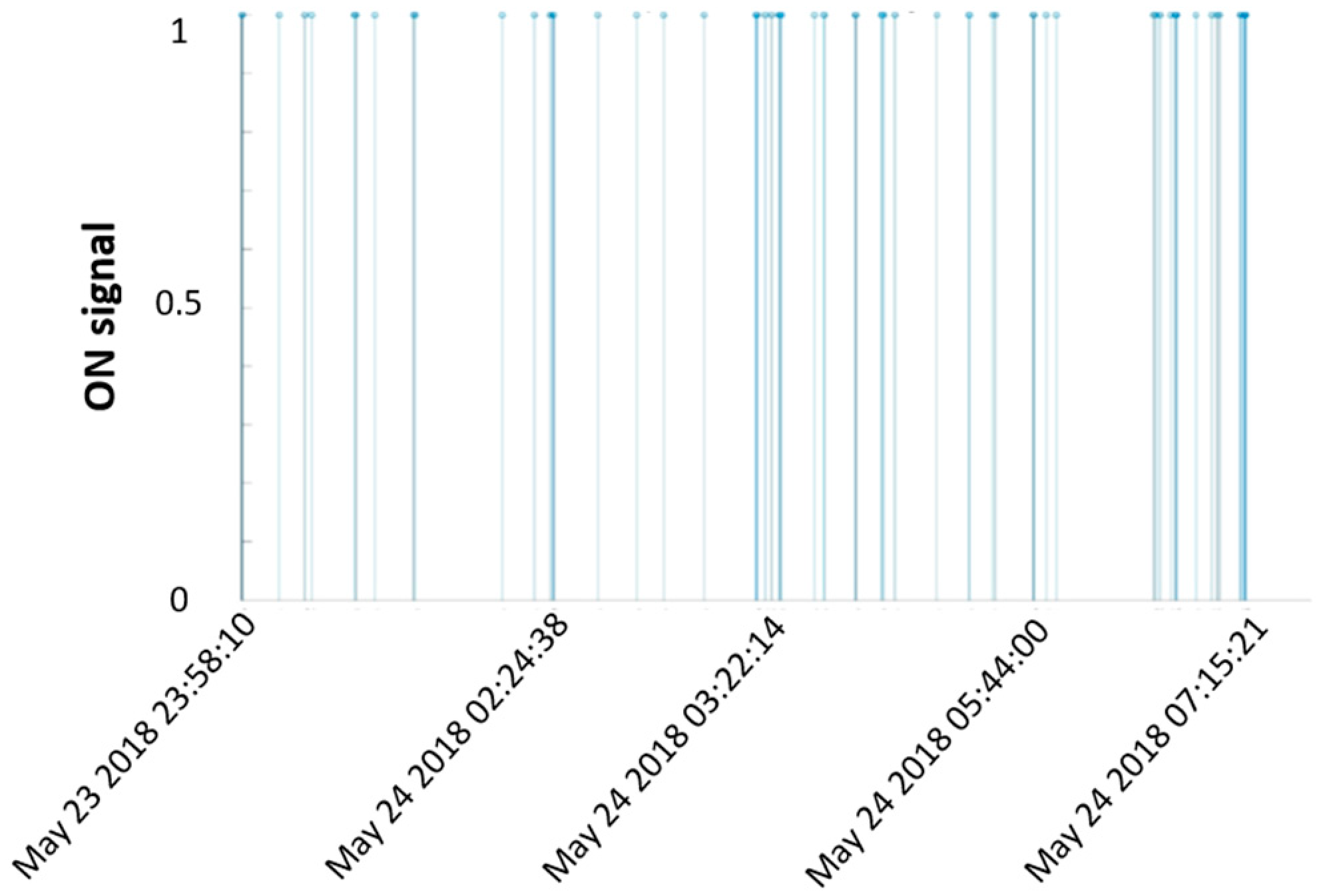
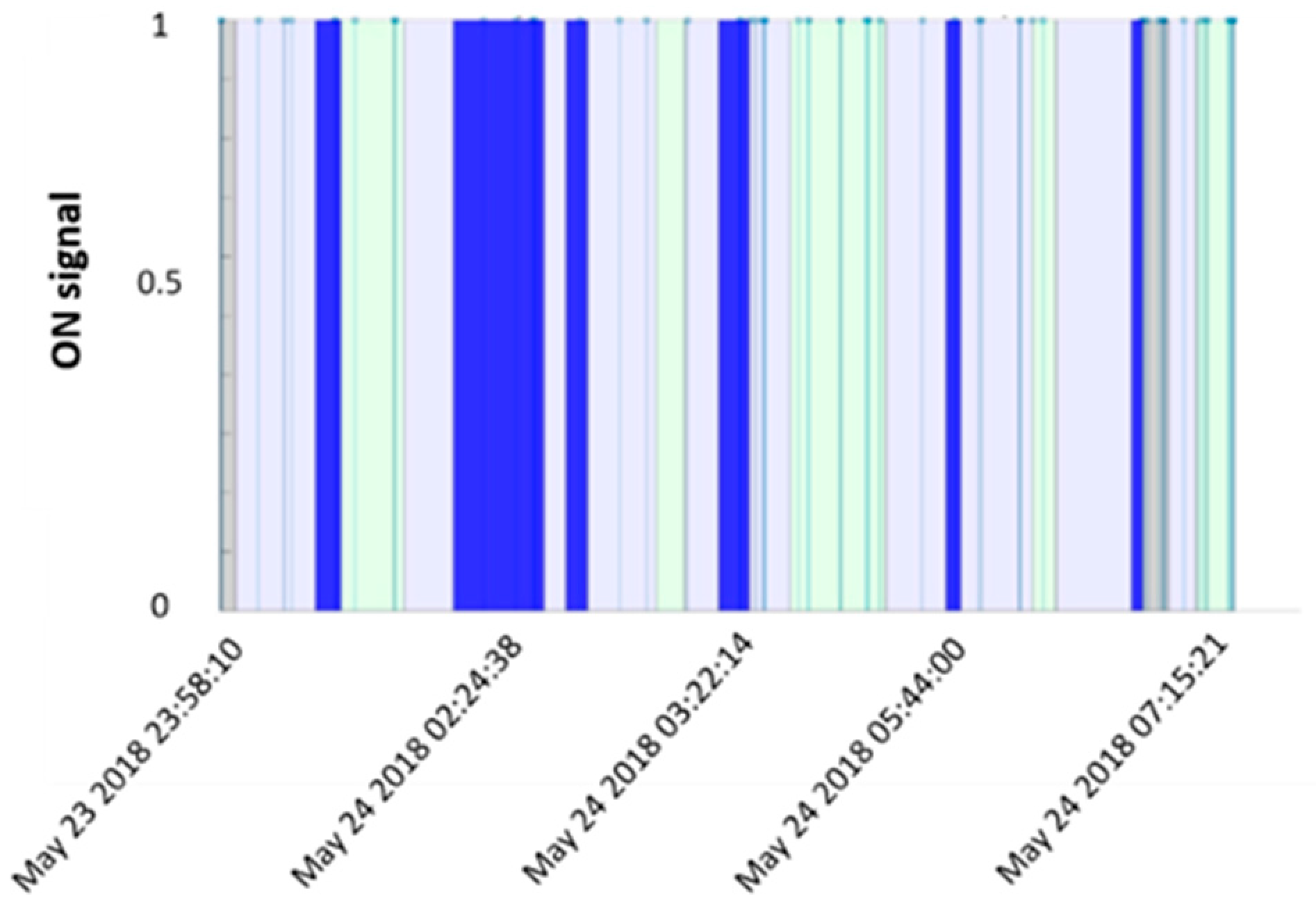
2.2.2. Algorithm
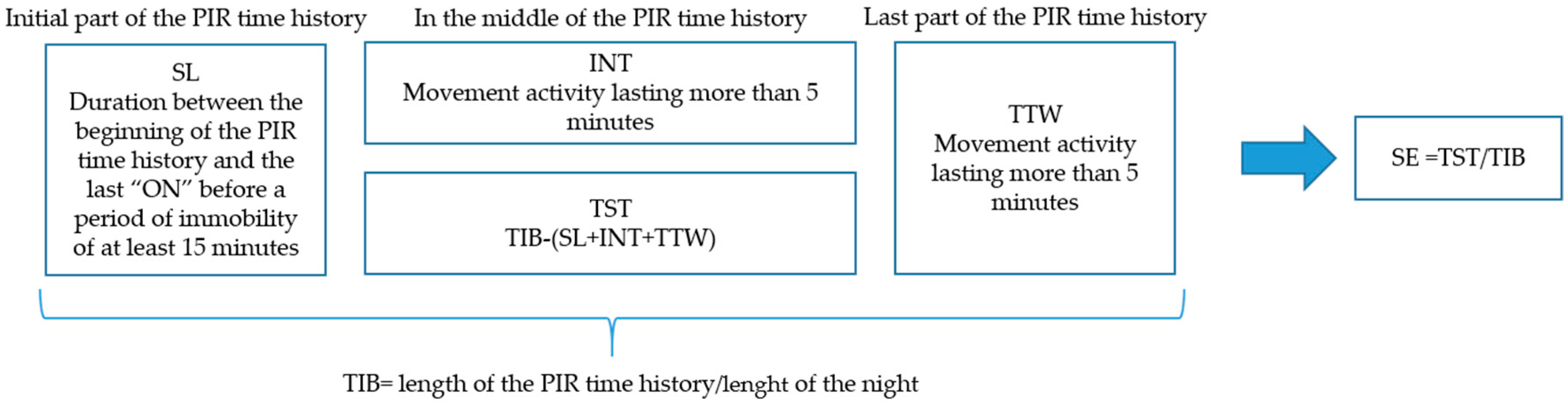
2.2.3. Feature Extraction
2.2.4. Statistical Analysis
2.2.5. Uncertainty Analysis
3. Results
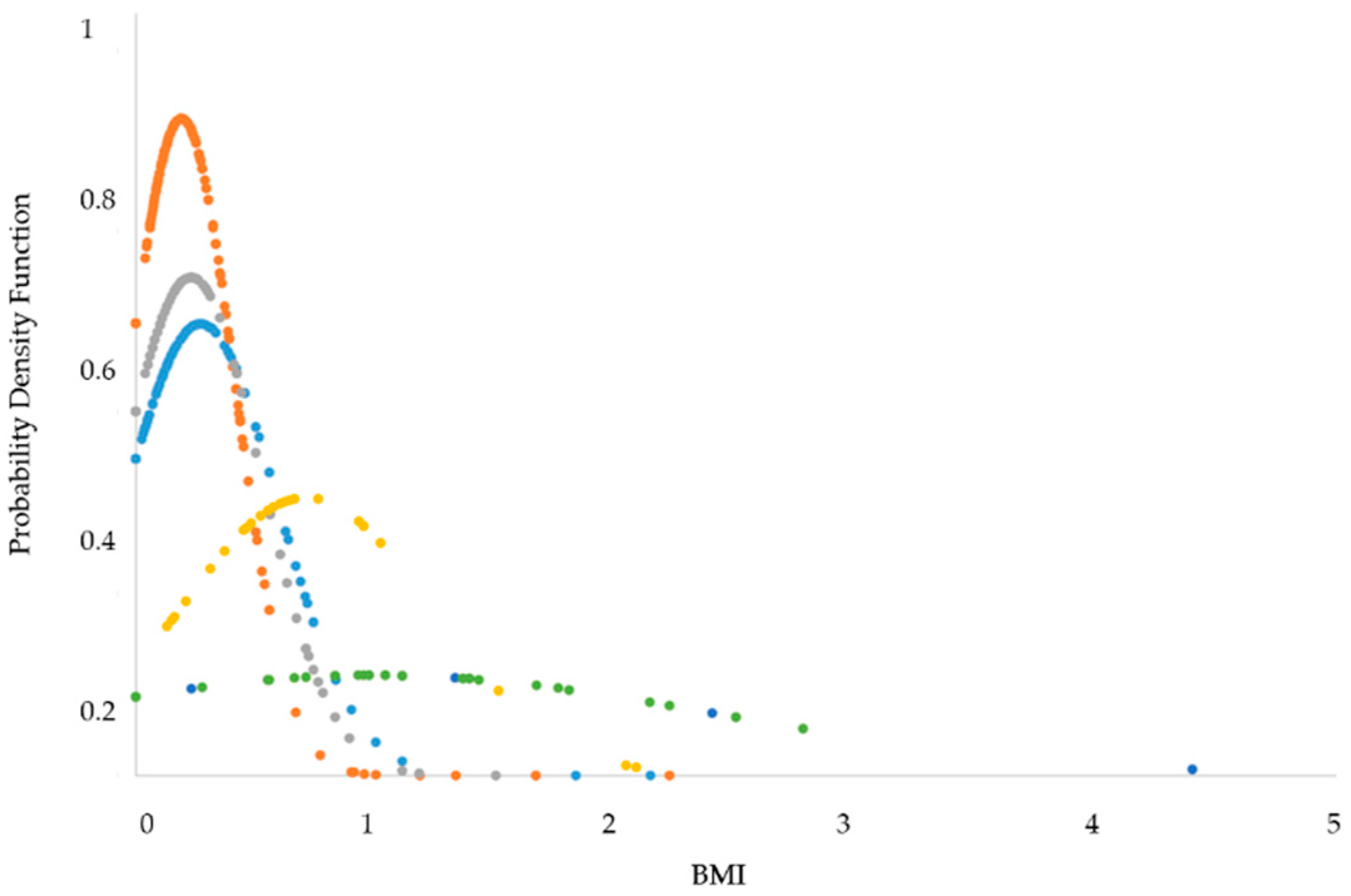
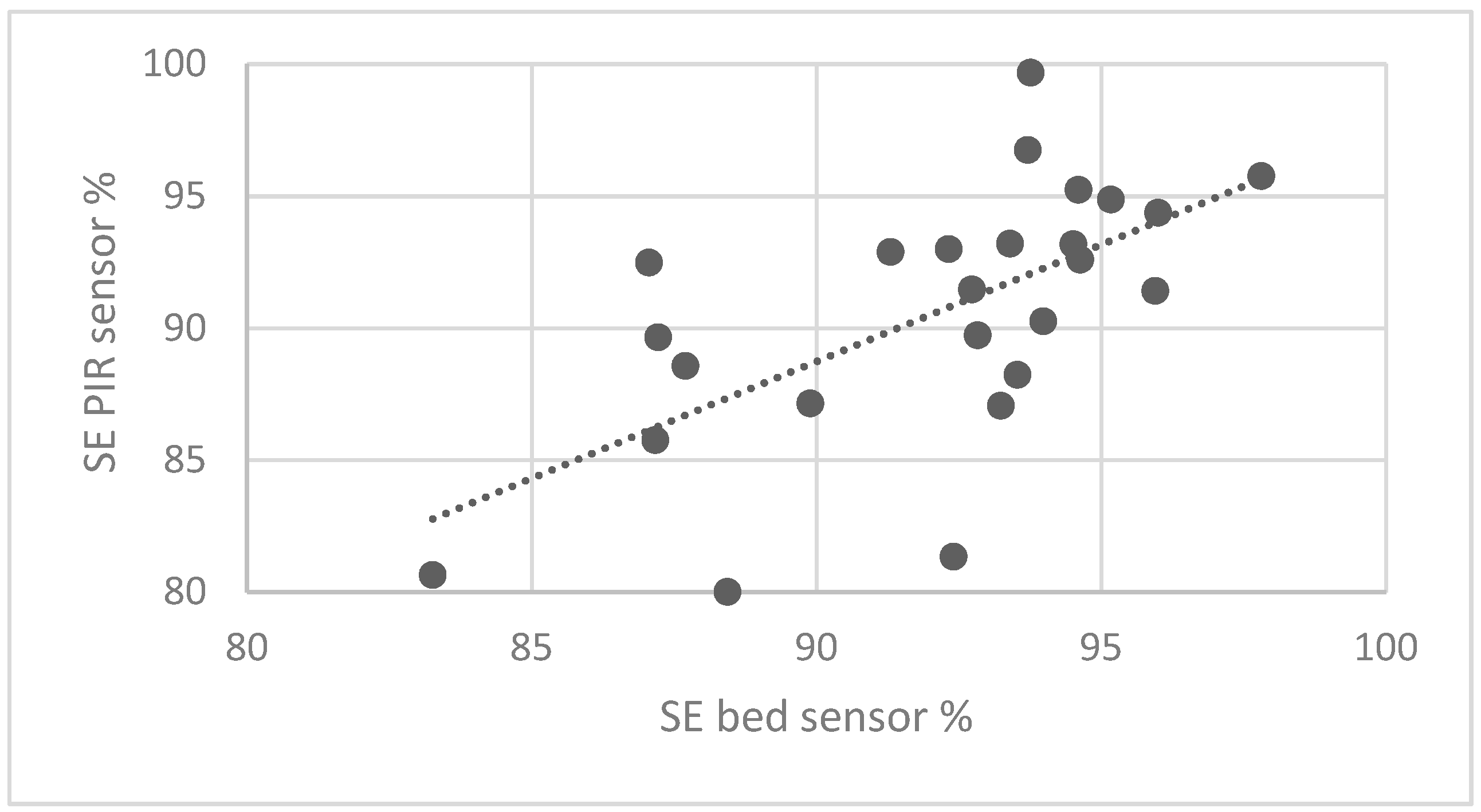
3.1. Statistical Analysis
3.2. Uncertainty Analysis
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yaffe, K.; Falvey, C.M.; Hoang, T. Connections between sleep and cognition in older adults. Lancet Neurol. 2014, 13, 1017–1028. [Google Scholar] [CrossRef]
- Driscoll, H.C.; Serody, L.; Patrick, S.; Maurer, J.; Bensasi, S.; Houck, P.R.; Mazumdar, S.; Nofzinger, E.A.; Bell, B.; Nebes, R.D.; et al. Sleeping Well, Aging Well: A Descriptive and Cross-Sectional Study of Sleep in “Successful Agers” 75 and Older. Am. J. Geriatr. Psychiatry 2008, 16, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Thorpy, M.J. Classification of Sleep Disorders. Neurotherapeutics 2012, 9, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Casaccia, S.; Pietroni, F.; Scalise, L.; Revel, G.M.; Monteriù, A.; Prist, M.R.; Frontoni, E.; Longhi, S. Health@Home: Pilot cases and preliminary results: Integrated residential sensor network to promote the active aging of real users. In Proceedings of the 2018 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Rome, Italy, 11–13 June 2018; pp. 1–6. [Google Scholar]
- Monteriù, A.; Prist, M.R.; Frontoni, E.; Longhi, S.; Pietroni, F.; Casaccia, S.; Scalise, L.; Cenci, A.; Romeo, L.; Berta, R.; et al. A Smart Sensing Architecture for Domestic Monitoring: Methodological Approach and Experimental Validation. Sensors 2018, 18, 2310. [Google Scholar] [CrossRef] [PubMed]
- Pietroni, F.; Casaccia, S.; Revel, G.M.; Prist, M.; Monteriù, A.; Longhi, S.; Scalise, L. Smart Monitoring of User and Home Environment: The Health@Home Acquisition Framework. In Ambient Assisted Living; Casiddu, N., Porfirione, C., Monteriù, A., Cavallo, F., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2019; pp. 23–37. [Google Scholar]
- Paalasmaa, J.; Waris, M.; Toivonen, H.; Leppäkorpi, L.; Partinen, M. Unobtrusive online monitoring of sleep at home. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2012, 2012, 3784–3788. [Google Scholar] [PubMed]
- Rashwan, H.A.; Solanas, A.; Puig, D.; Martínez-Ballesté, A. Understanding trust in privacy-aware video surveillance systems. Int. J. Inf. Secur. 2016, 15, 225–234. [Google Scholar] [CrossRef]
- Migliorini, M.; Bianchi, A.M.; Nisticò, D.; Kortelainen, J.; Arce-Santana, E.; Cerutti, S.; Mendez, M.O. Automatic sleep staging based on ballistocardiographic signals recorded through bed sensors. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2010, 2010, 3273–3276. [Google Scholar]
- Alivar, A.; Carlson, C.; Suliman, A.; Warren, S.; Prakash, P.; Thompson, D.; Natarajan, B. Motion Detection in Bed-Based Ballistocardiogram to Quantify Sleep Quality. In Proceedings of the GLOBECOM 2017 IEEE Global Communications Conference, Singapore, 4–8 December 2017; pp. 1–6. [Google Scholar]
- Stiller, J.W.; Postolache, T.T. Sleep-wake and Other Biological Rhythms: Functional Neuroanatomy. Clin. Sports Med. 2005, 24, 205–235. [Google Scholar] [CrossRef]
- III. SLEEP ASSESSMENT METHODS - Sadeh - 2015 - Monographs of the Society for Research in Child Development Wiley Online Library. Available online: https://onlinelibrary.wiley.com/doi/full/10.1111/mono.12143 (accessed on 21 February 2019).
- Åkerstedt, T.; Billiard, M.; Bonnet, M.; Ficca, G.; Garma, L.; Mariotti, M.; Salzarulo, P.; Schulz, H. Awakening from sleep. Sleep Med. Rev. 2002, 6, 267–286. [Google Scholar] [CrossRef]
- Kelly, J.M.; Strecker, R.E.; Bianchi, M.T. Recent Developments in Home Sleep-Monitoring Devices. ISRN Neurol. 2012, 2012, 10. [Google Scholar] [CrossRef]
- Park, K.S.; Yoon, H. Ballistocardiography. In Seamless Healthcare Monitoring: Advancements in Wearable, Attachable, and Invisible Devices; Tamura, T., Chen, W., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 127–155. ISBN 978-3-319-69362-0. [Google Scholar]
- Hayes, T.L.; Riley, T.; Pavel, M.; Kaye, J.A. Estimation of rest-activity patterns using motion sensors. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2010, 2010, 2147–2150. [Google Scholar] [PubMed]
- Nikamalfard, H.; Zheng, H.; Wang, H.; Mulvenna, M.; McCullagh, P.; Jeffers, P. A sleep pattern analysis and visualization system to support people with early dementia. In Proceedings of the 2011 5th International Conference on Pervasive Computing Technologies for Healthcare (PervasiveHealth) and Workshops, Dublin, Ireland, 23–26 May 2011; pp. 510–513. [Google Scholar]
- Peake, J.M.; Kerr, G.; Sullivan, J.P. A Critical Review of Consumer Wearables, Mobile Applications, and Equipment for Providing Biofeedback, Monitoring Stress, and Sleep in Physically Active Populations. Front. Physiol. 2018, 9, 743. [Google Scholar] [CrossRef] [PubMed]
- Moghavvemi, M.; Lu, C.S. Pyroelectric infrared sensor for intruder detection. In Proceedings of the 2004 IEEE Region 10 Conference TENCON 2004, Chiang Mai, Thailand, 24 November 2004; pp. 656–659. [Google Scholar]
- Pinheiro, E.; Postolache, O.; Girão, P.S. Ballistocardiogram: Model and sensing systems. In Proceedings of the 2016 10th International Conference on Sensing Technology (ICST), Nanjing, China, 11–13 November 2016; pp. 1–6. [Google Scholar]
- Wu, L.; Wang, Y.; Liu, H. Occupancy Detection and Localization by Monitoring Nonlinear Energy Flow of a Shuttered Passive Infrared Sensor. IEEE Sens. J. 2018, 18, 8656–8666. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, Y.; Liu, H. Unobtrusive Sensor-Based Occupancy Facing Direction Detection and Tracking Using Advanced Machine Learning Algorithms. IEEE Sens. J. 2018, 18, 6360–6368. [Google Scholar] [CrossRef]
- Brown, L.A.; Hasan, S.; Foster, R.G.; Peirson, S.N. COMPASS: Continuous Open Mouse Phenotyping of Activity and Sleep Status. Wellcome Open Res. 2017, 1, 2. [Google Scholar] [CrossRef] [PubMed]
- Bresler, M.; Sheffy, K.; Pillar, G.; Preiszler, M.; Herscovici, S. Differentiating between light and deep sleep stages using an ambulatory device based on peripheral arterial tonometry. Physiol. Meas. 2008, 29, 571–584. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.A.; Lee, H.J.; Moon, J.H.; Lee, T.; Kim, M.G.; In, H.; Cho, C.H.; Kim, L. Comparison of Wearable Activity Tracker with Actigraphy for Sleep Evaluation and Circadian Rest-Activity Rhythm Measurement in Healthy Young Adults. Psychiatry Investig. 2017, 14, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Kasuya, E. On the use of r and r squared in correlation and regression. Ecol. Res. 2019, 34, 235–236. [Google Scholar] [CrossRef]
- Lorenz, K.; Freddolino, P.P.; Comas-Herrera, A.; Knapp, M.; Damant, J. Technology-based tools and services for people with dementia and carers: Mapping technology onto the dementia care pathway. Dementia 2019, 18, 725–741. [Google Scholar] [CrossRef]
- Gochoo, M.; Tan, T.; Huang, S.; Batjargal, T.; Hsieh, J.; Alnajjar, F.S.; Chen, Y. Novel IoT-Based Privacy-Preserving Yoga Posture Recognition System Using Low-Resolution Infrared Sensors and Deep Learning. IEEE Internet Things J. 2019, 6, 7192–7200. [Google Scholar] [CrossRef]
- Klemets, J.; Määttälä, J.; Hakala, I. Integration of an in-home monitoring system into home care nurses’ workflow: A case study. Int. J. Med. Inform. 2019, 123, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Rantz, M.J.; Scott, S.D.; Miller, S.J.; Skubic, M.; Phillips, L.; Alexander, G.; Koopman, R.J.; Musterman, K.; Back, J. Evaluation of Health Alerts from an Early Illness Warning System in Independent Living. Comput. Inf. Nurs. 2013, 31, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Wolkove, N.; Elkholy, O.; Baltzan, M.; Palayew, M. Sleep and aging: 1. Sleep disorders commonly found in older people. CMAJ 2007, 176, 1299–1304. [Google Scholar] [CrossRef] [PubMed]
- Sculthorpe, L.D.; Douglass, A.B. Sleep Pathologies in Depression and the Clinical Utility of Polysomnography. Can J. Psychiatry 2010, 55, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, S.; Lapierre, S.; Hudon, C.; Desgagné, A. Factors involved in sleep efficiency: A population-based study of community-dwelling elderly persons. Sleep 2019, 42. [Google Scholar] [CrossRef] [PubMed]

| Sleep Phase | R Value (Pearson) | R2 Value |
|---|---|---|
| DEEP | −0.03 | 0.00 |
| LIGHT | 0.61 | 0.37 |
| REM | 0.40 | 0.16 |
| SL | 0.67 | 0.45 |
| INT | 0.71 | 0.55 |
| TTW | 0.86 | 0.69 |
| Sleep Phase | BMI (AVG) | BMI (SD) |
|---|---|---|
| DEEP | 0.27 | 0.32 |
| LIGHT | 0.19 | 0.27 |
| REM | 0.23 | 0.29 |
| SL | 0.71 | 0.52 |
| INT | 1 | 1.44 |
| TTW | 1.14 | 0.73 |
| Sleep Phases | Null Hypothesis | p-Value |
|---|---|---|
| DEEP-LIGHT | 1 | 0.001 |
| DEEP-REM | 0 | 0.175 |
| DEEP-SL | 1 | 0.000 |
| DEEP-INT | 1 | 0.037 |
| DEEP-TTW | 1 | 0.000 |
| LIGHT-REM | 0 | 0.223 |
| LIGHT-SL | 1 | 0.000 |
| LIGHT-INT | 1 | 0.008 |
| LIGHT-TTW | 1 | 0.000 |
| REM-SL | 1 | 0.000 |
| REM-INT | 1 | 0.023 |
| REM-TTW | 1 | 0.000 |
| SL-INT | 0 | 0.296 |
| SL-TTW | 1 | 0.008 |
| INT-TTW | 0 | 0.676 |
| Sleep Phase | Precision (%) |
|---|---|
| SL algorithm detection | 100 |
| INT algorithm detection | 17 |
| TTW algorithm detection | 100 |
| Sleep Phase | Uncertainty (SD) |
|---|---|
| SL | 23.9% |
| INT | 56.1% |
| TTW | 49.5% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casaccia, S.; Braccili, E.; Scalise, L.; Revel, G.M. Experimental Assessment of Sleep-Related Parameters by Passive Infrared Sensors: Measurement Setup, Feature Extraction, and Uncertainty Analysis. Sensors 2019, 19, 3773. https://doi.org/10.3390/s19173773
Casaccia S, Braccili E, Scalise L, Revel GM. Experimental Assessment of Sleep-Related Parameters by Passive Infrared Sensors: Measurement Setup, Feature Extraction, and Uncertainty Analysis. Sensors. 2019; 19(17):3773. https://doi.org/10.3390/s19173773
Chicago/Turabian StyleCasaccia, Sara, Eleonora Braccili, Lorenzo Scalise, and Gian Marco Revel. 2019. "Experimental Assessment of Sleep-Related Parameters by Passive Infrared Sensors: Measurement Setup, Feature Extraction, and Uncertainty Analysis" Sensors 19, no. 17: 3773. https://doi.org/10.3390/s19173773
APA StyleCasaccia, S., Braccili, E., Scalise, L., & Revel, G. M. (2019). Experimental Assessment of Sleep-Related Parameters by Passive Infrared Sensors: Measurement Setup, Feature Extraction, and Uncertainty Analysis. Sensors, 19(17), 3773. https://doi.org/10.3390/s19173773








