Detection of Cardiopulmonary Activity and Related Abnormal Events Using Microsoft Kinect Sensor
Abstract
:1. Introduction
2. Cardiopulmonary Activity and Related Abnormal Events
3. Methods & Procedures
3.1. Participants
3.2. Experimental Setup & Validation Methods
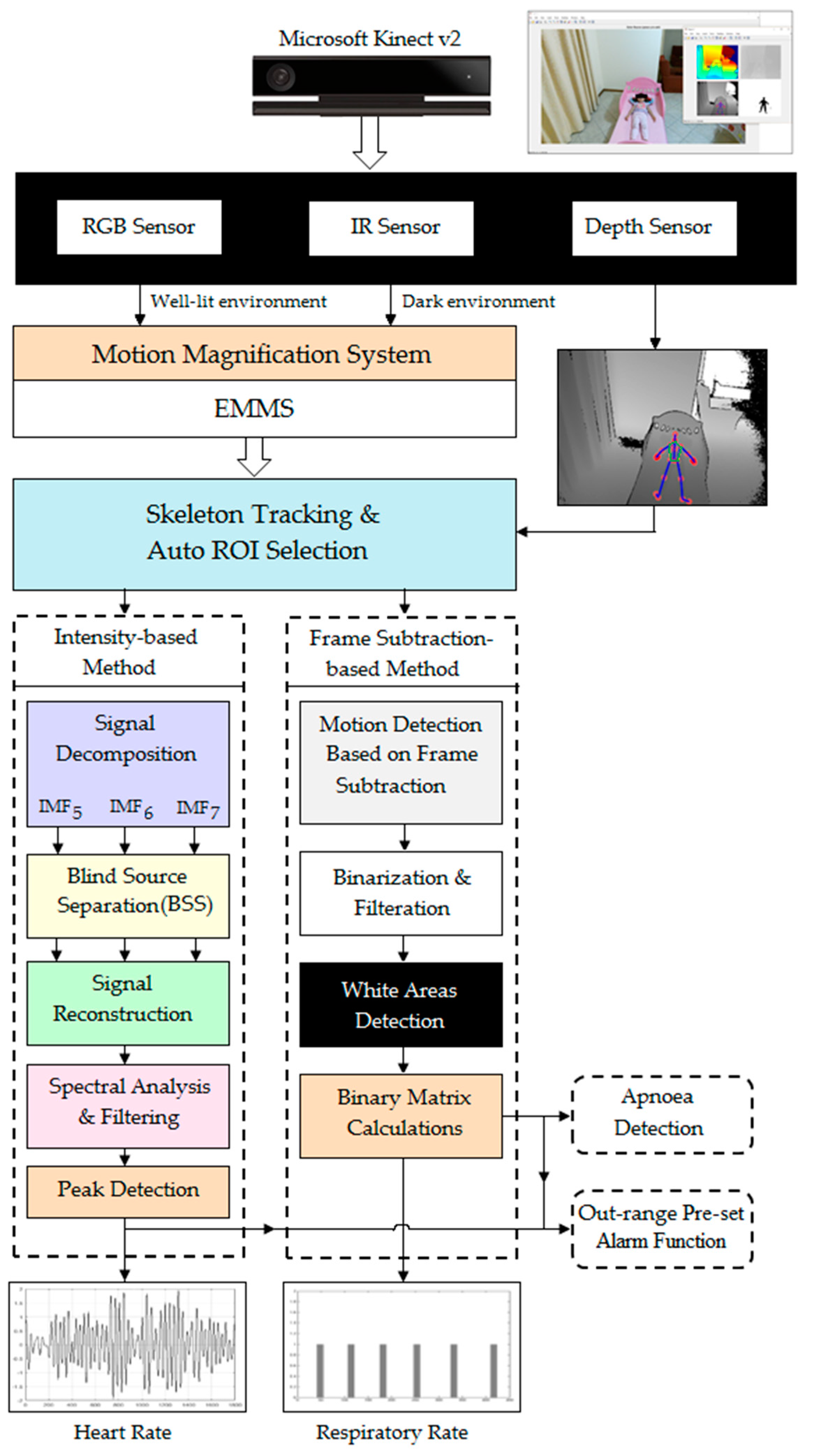
3.3. System Framework & Data Analysis
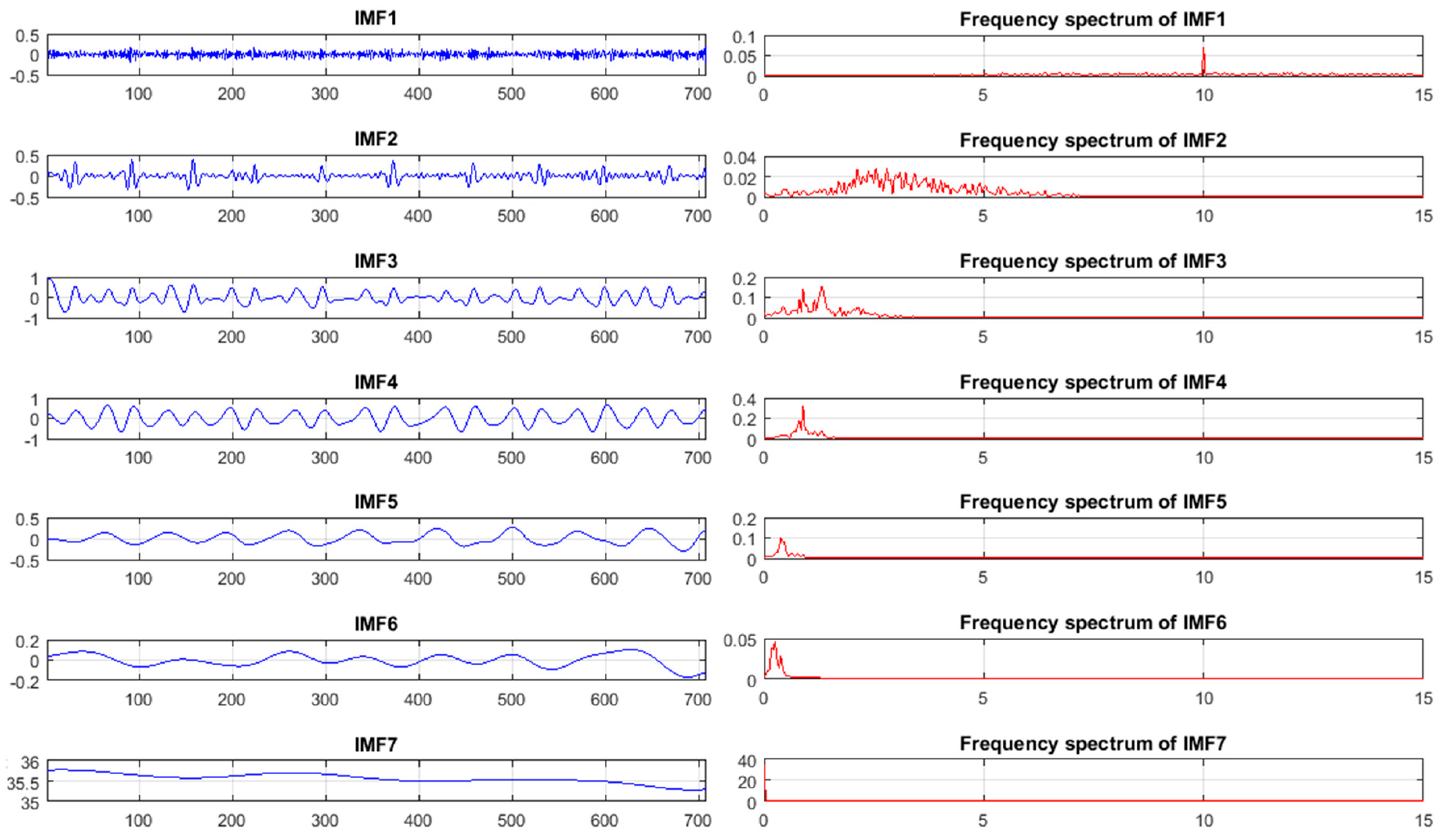
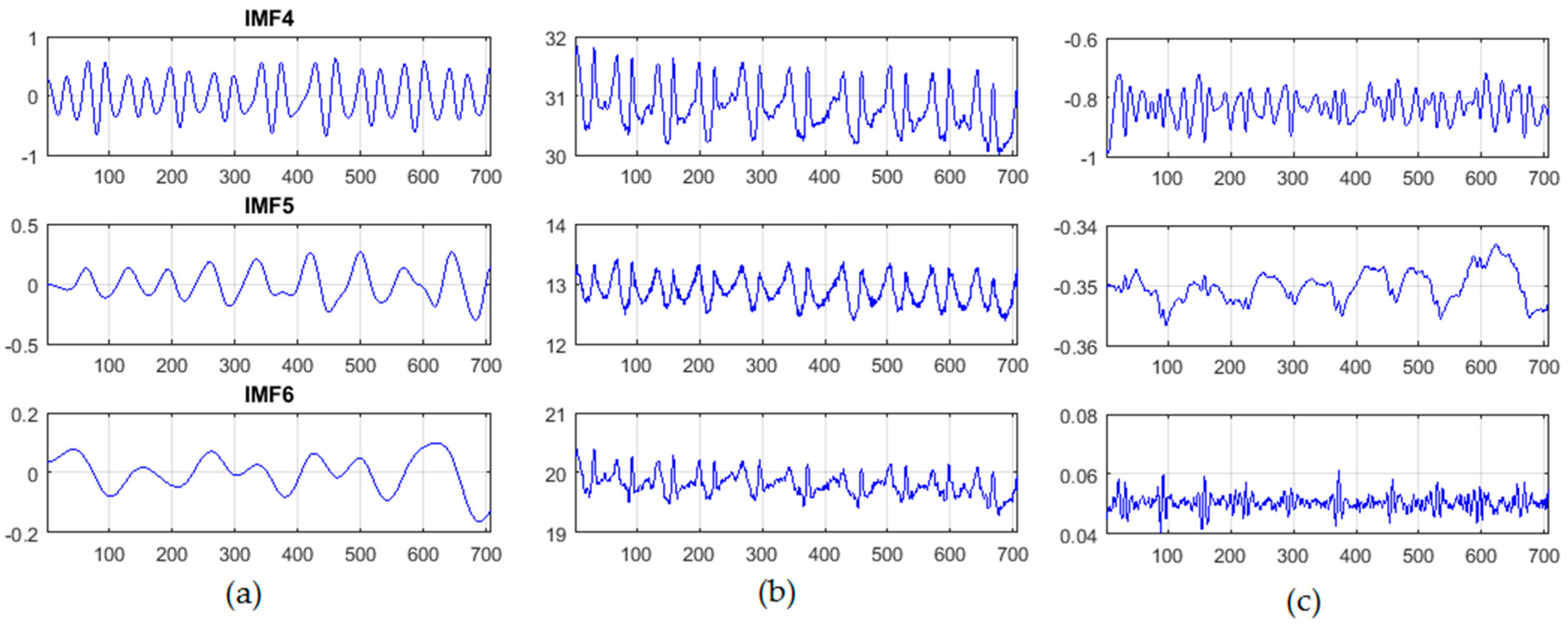
3.3.1. Extraction of Cardiac Signal Using Intensity-Based Method
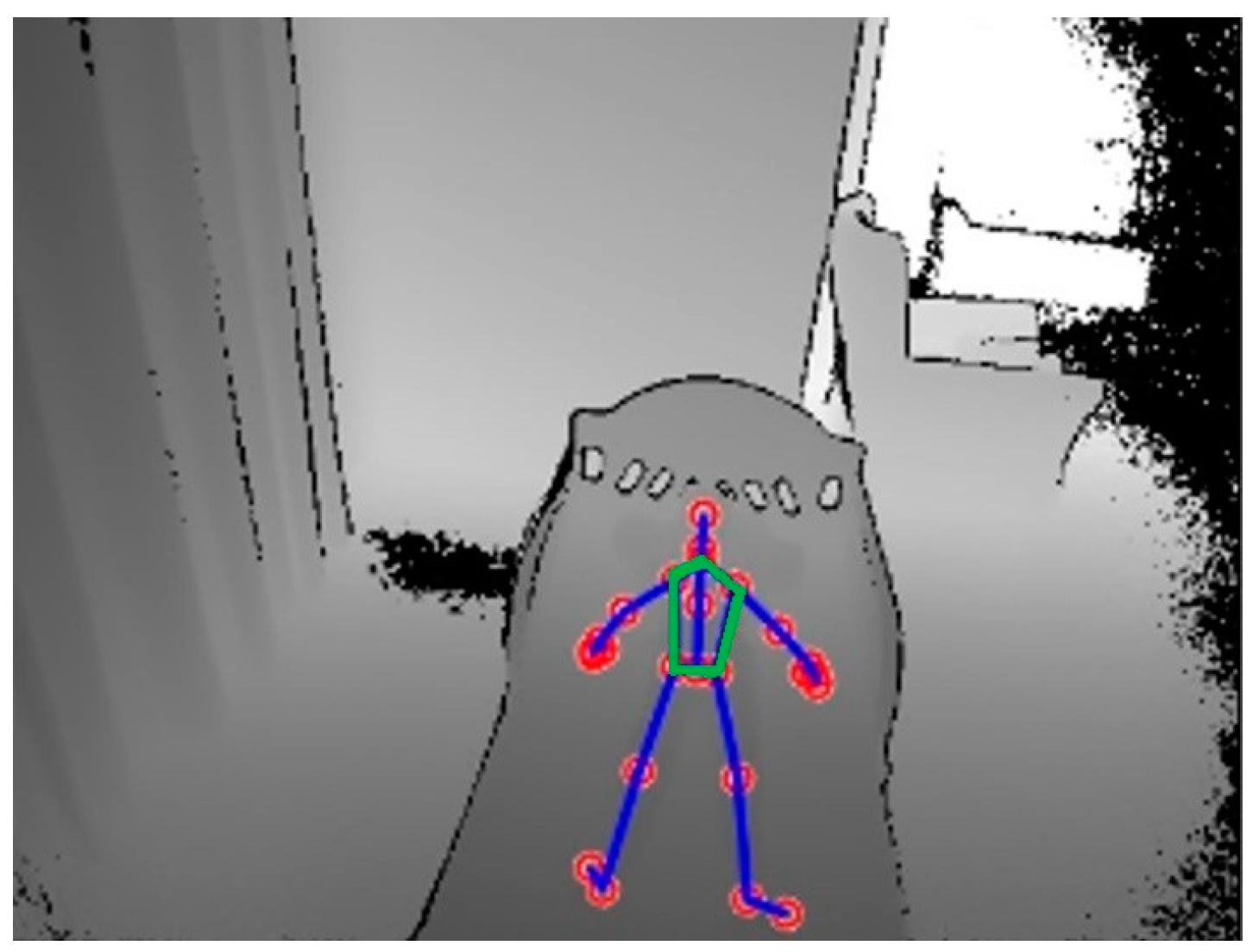
3.3.2. Extraction of Respiratory Signal Using a Frame-Subtraction-Based Method
3.3.3. Sleep Apnoea Detection
4. Experimental Results
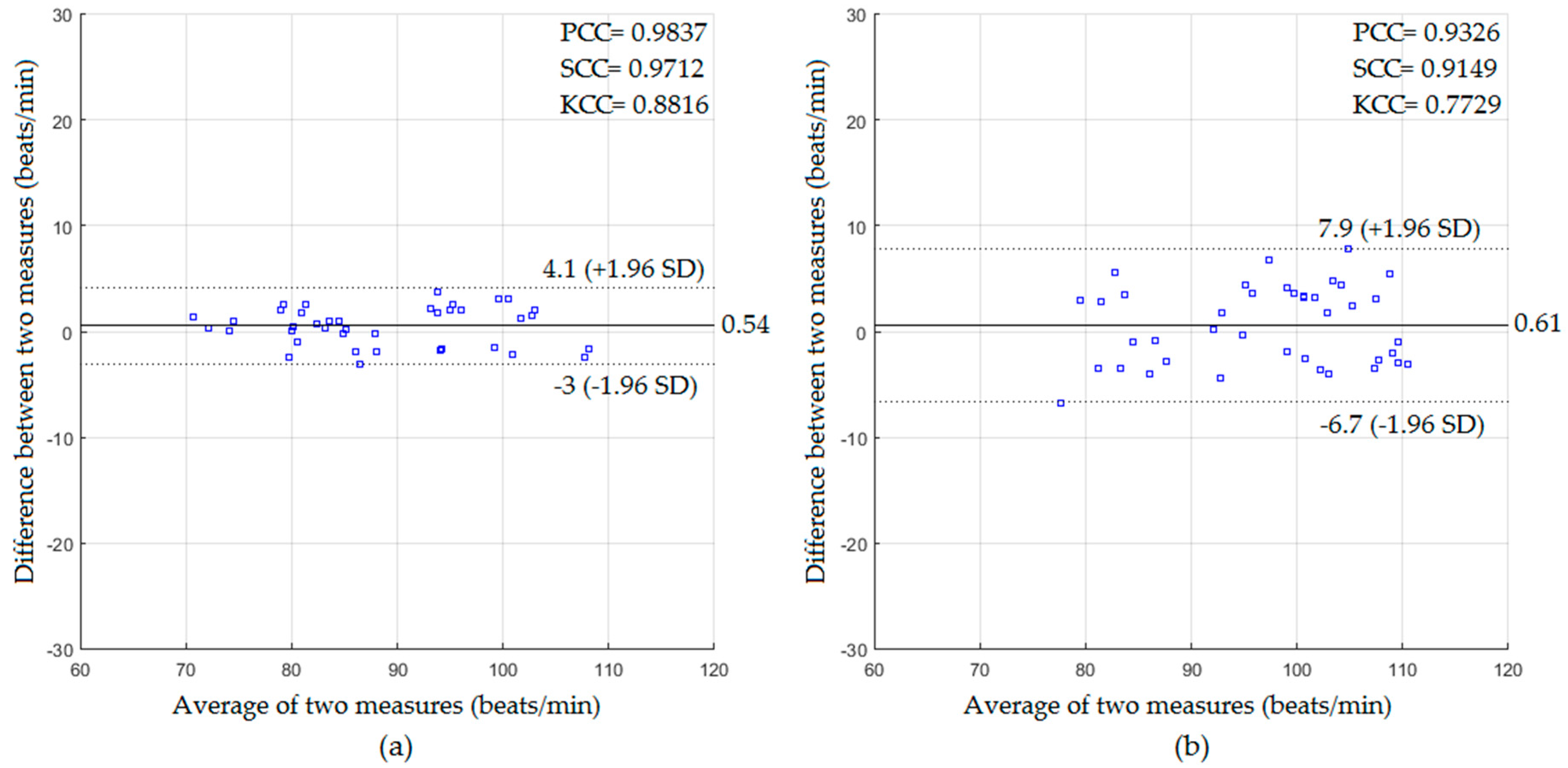
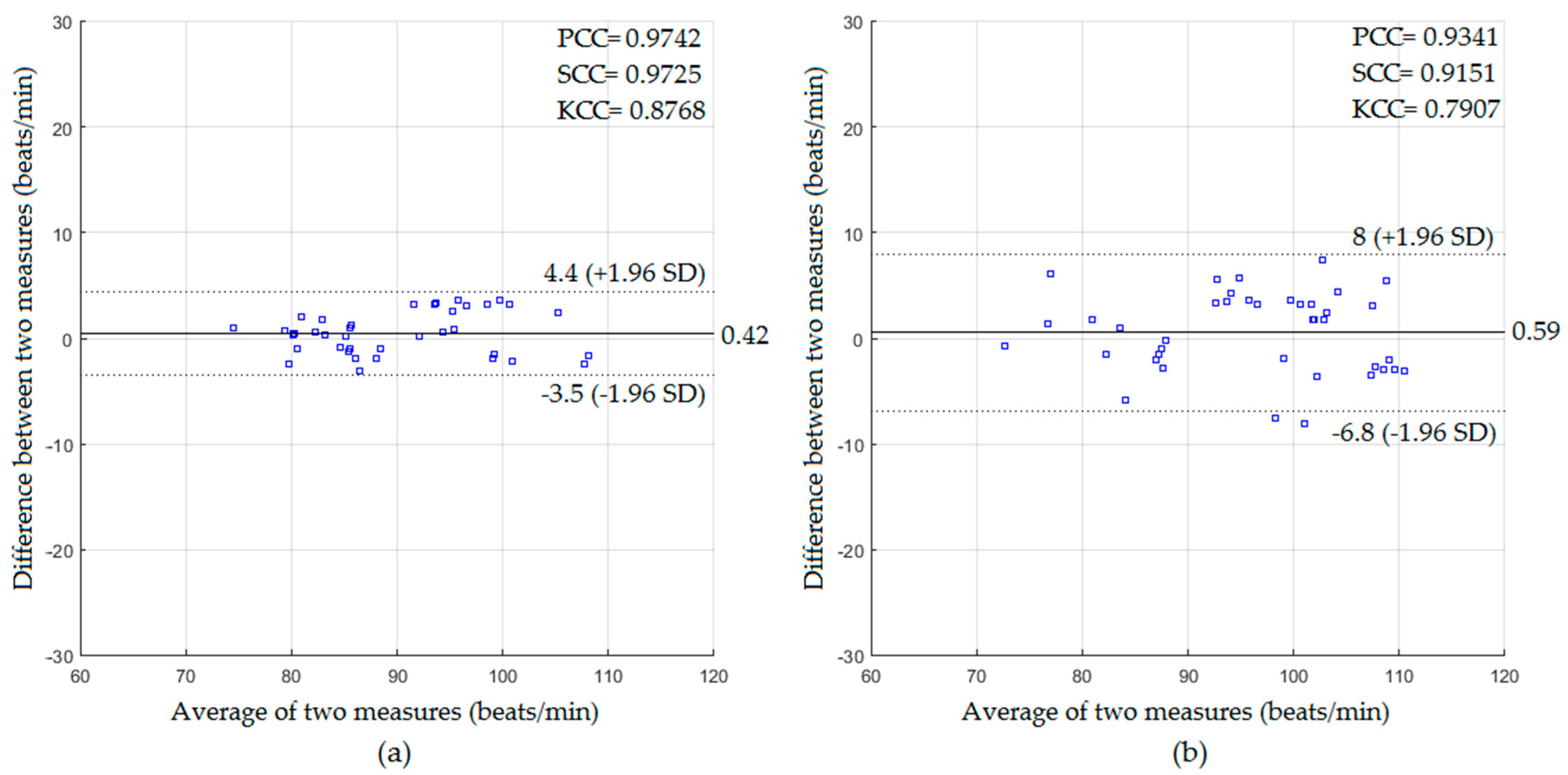
4.1. Measurements of Cardiac Activity
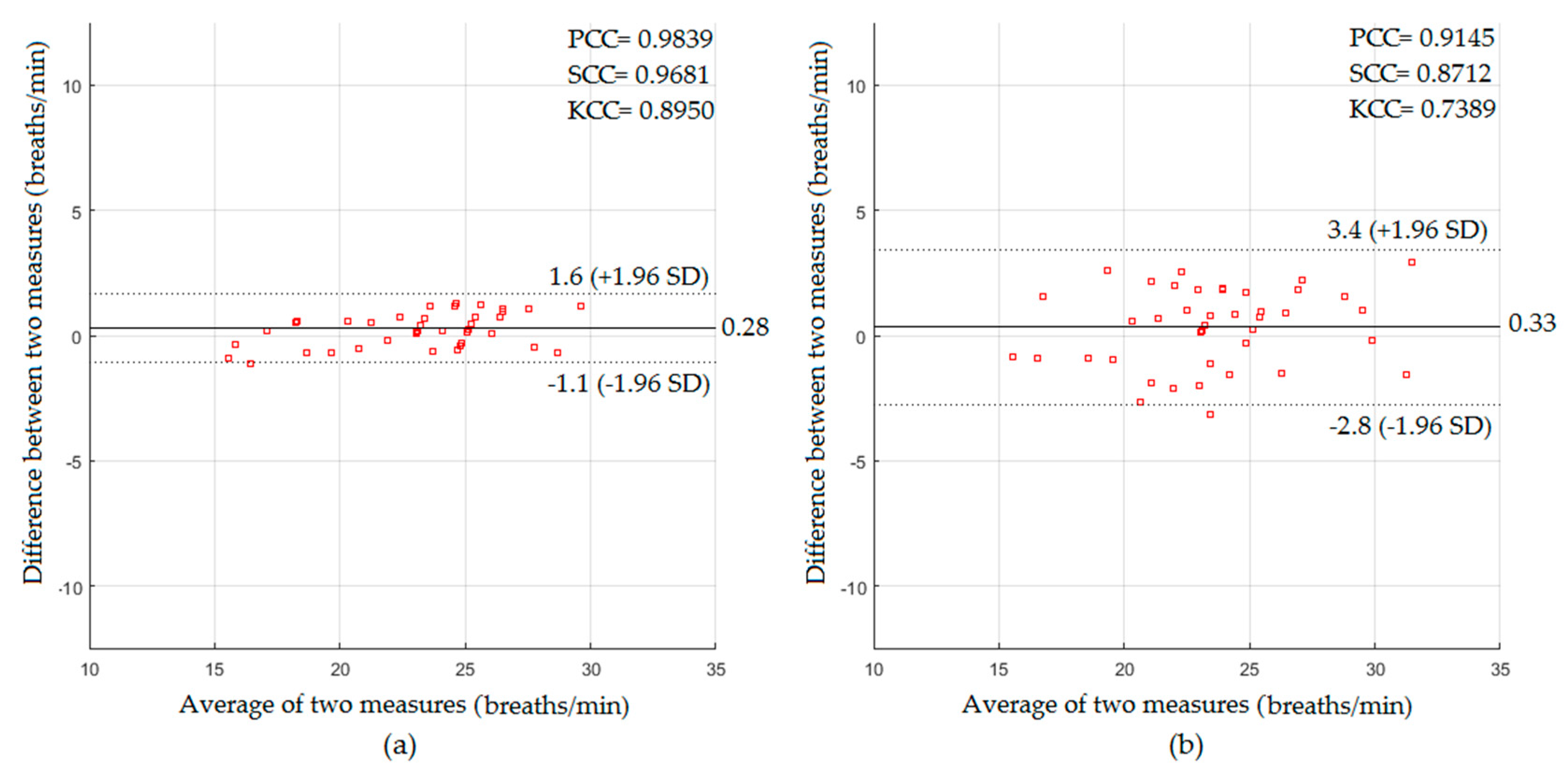
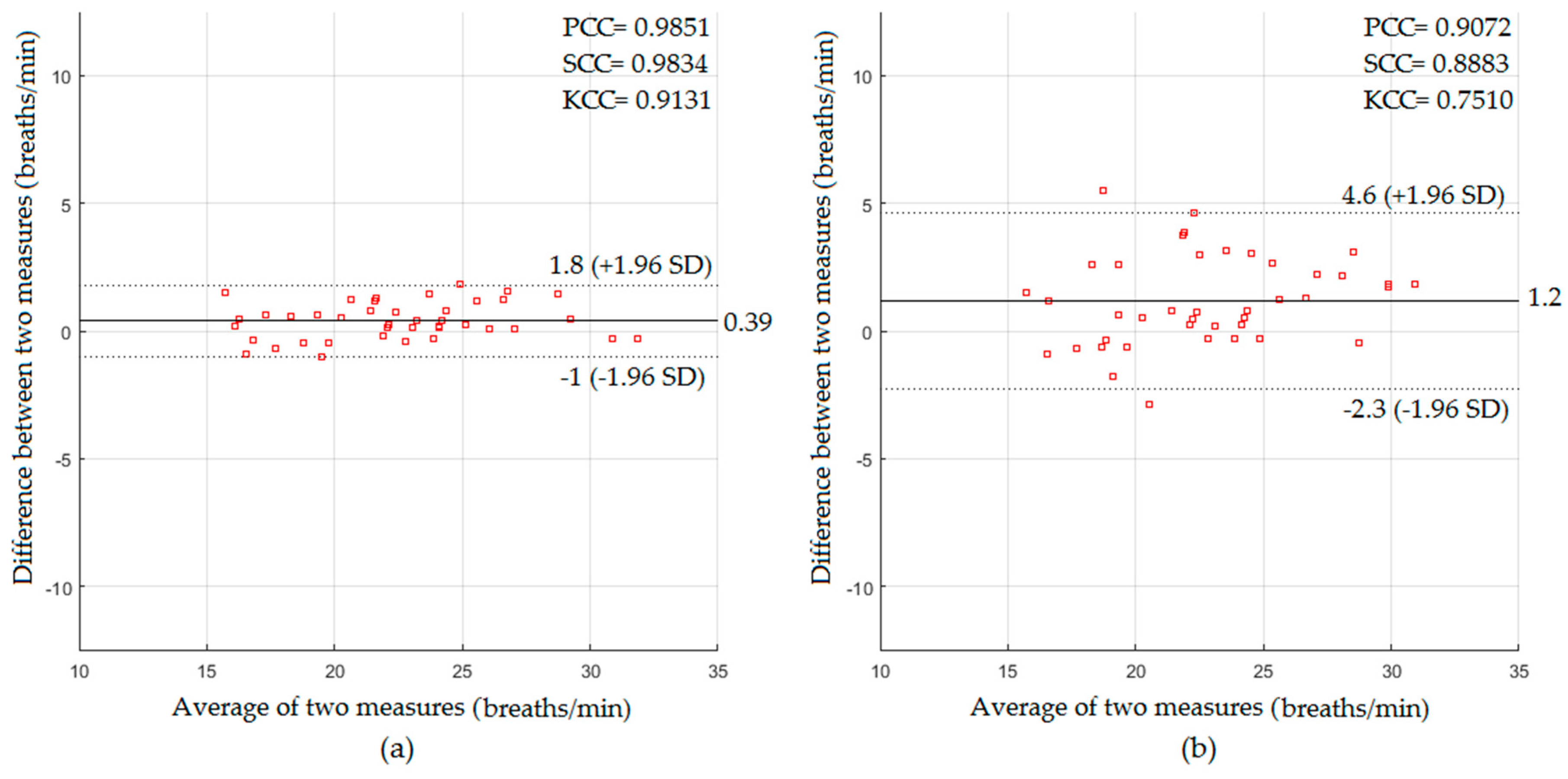
4.2. Measurements of Respiratory Activity
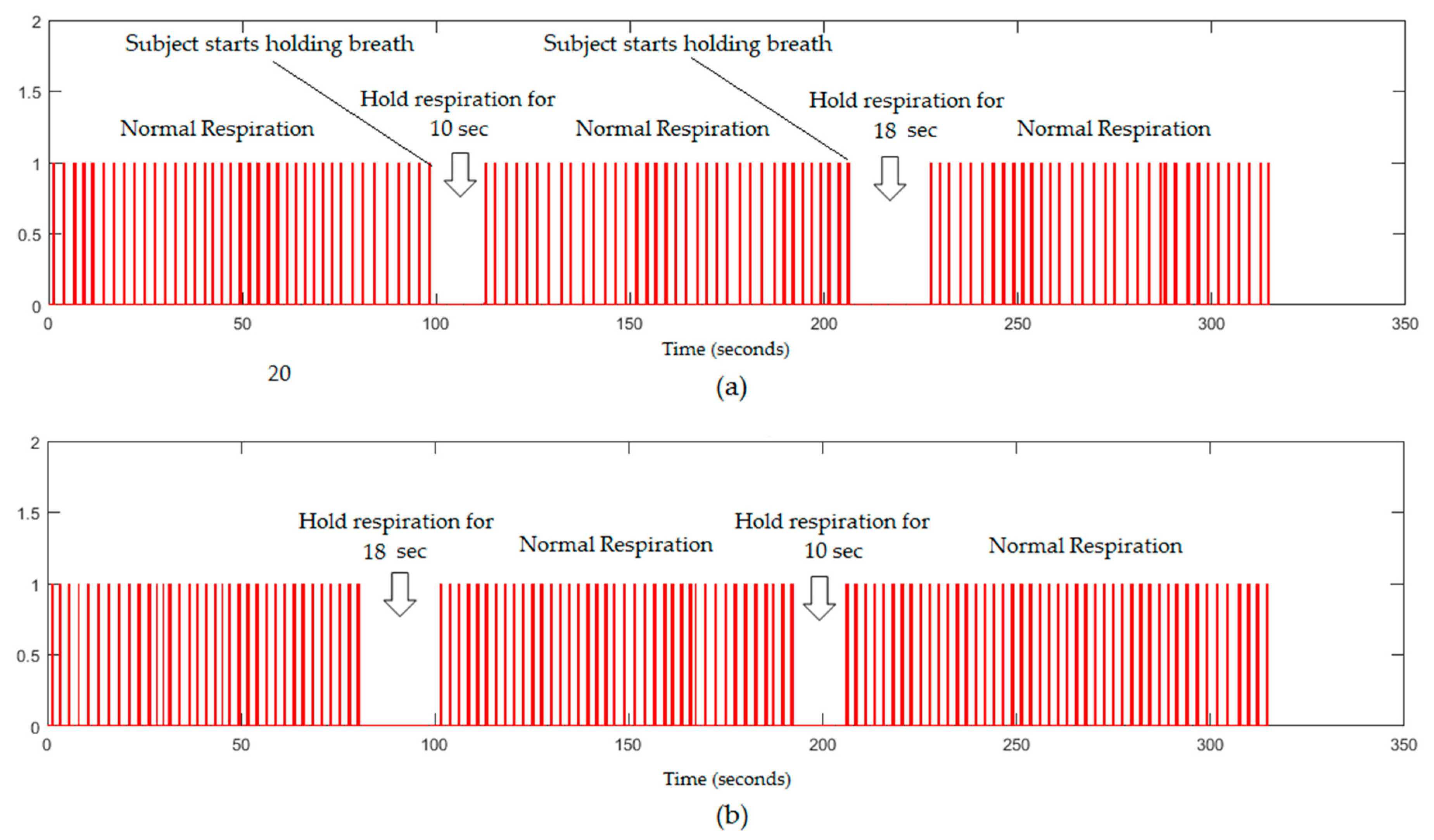
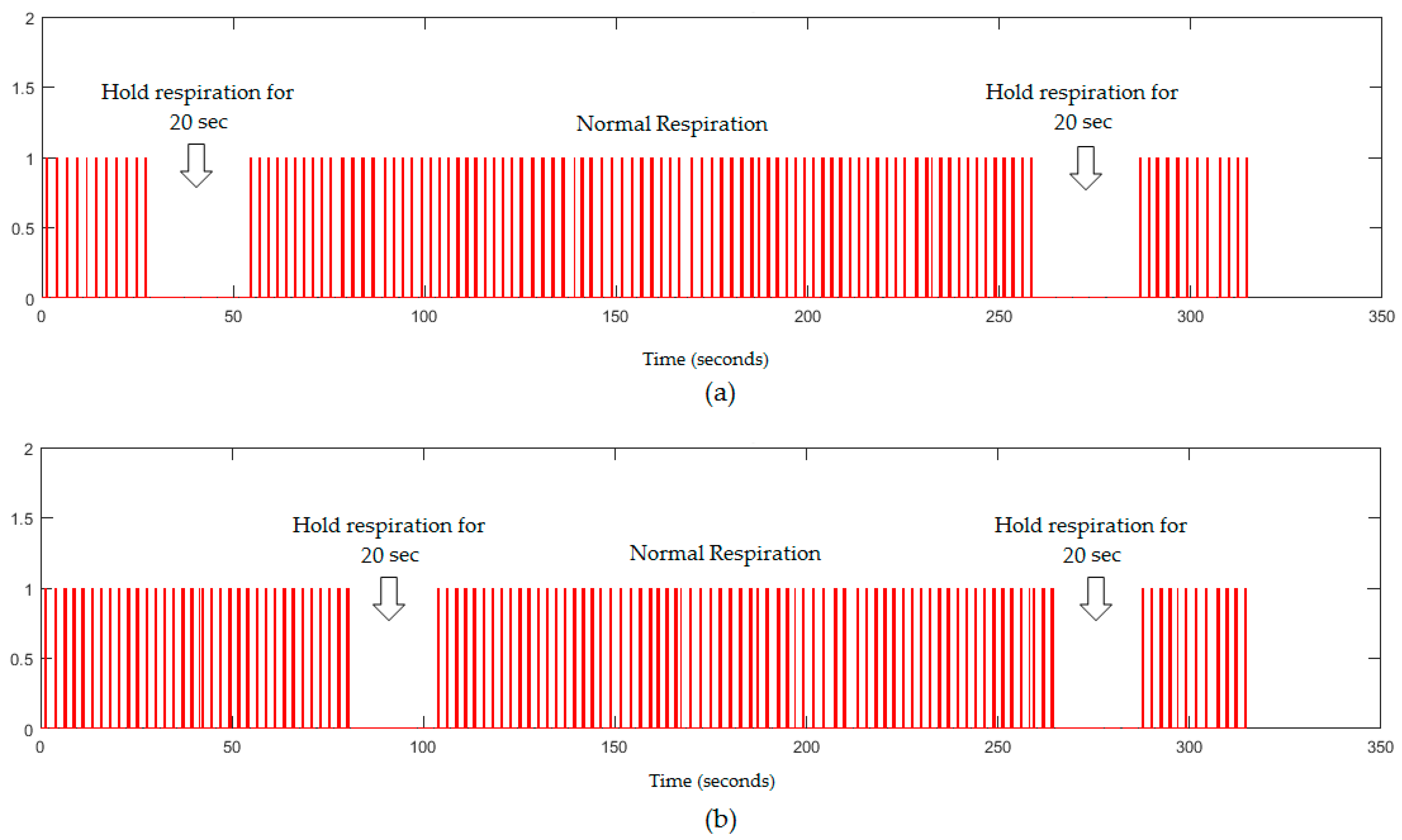
4.3. Apnoea Detection
5. Discussion
6. Conclusions
Author Contributions
Conflicts of Interest
References
- Geng, J. Structured-light 3D surface imaging: A tutorial. Adv. Opt. Photonics 2011, 3, 128–160. [Google Scholar] [CrossRef]
- Mutto, C.D.; Zanuttigh, P.; Cortelazzo, G.M. Time-of-Flight Cameras and Microsoft Kinect (TM); Springer Publishing Company Incorporated: Berlin, Germany, 2012. [Google Scholar]
- Yang, L.; Zhang, L.; Dong, H.; Alelaiwi, A.; El Saddik, A. Evaluating and improving the depth accuracy of Kinect for Windows v2. Sens. J. IEEE 2015, 15, 4275–4285. [Google Scholar] [CrossRef]
- Sarbolandi, H.; Lefloch, D.; Kolb, A. Kinect range sensing: Structured-light versus Time-of-Flight Kinect. Comput. Vis. Image Underst. 2015, 139, 1–20. [Google Scholar] [CrossRef]
- Samir, M.; Golkar, E.; Rahni, A.A.A. Comparison between the KinectTM V1 and KinectTM V2 for respiratory motion tracking. In Proceedings of the 2015 IEEE International Conference on Signal and Image Processing Applications (ICSIPA), Kuala Lumpur, Malaysia, 19–21 October 2015; pp. 150–155. [Google Scholar]
- Kim, C.; Yun, S.; Jung, S.-W.; Won, C.S. Color and depth image correspondence for Kinect v2. In Advanced Multimedia and Ubiquitous Engineering; Springer: Berlin, Germany, 2015; pp. 111–116. [Google Scholar]
- Wasenmüller, O.; Stricker, D. Comparison of kinect v1 and v2 depth images in terms of accuracy and precision. In Computer Vision–ACCV 2016 Workshops: ACCV 2016; Lecture Notes in Computer Science; Springer: Cham, Switzerland, 2017; Volume 10117, pp. 34–45. [Google Scholar]
- Xia, J.; Siochi, R.A. A real-time respiratory motion monitoring system using KINECT: Proof of concept. Med. Phys. 2012, 39, 2682–2685. [Google Scholar] [CrossRef] [PubMed]
- Smilkstein, T.; Buenrostro, M.; Kenyon, A.; Lienemann, M.; Larson, G. Heart rate monitoring using Kinect and color amplification. In Proceedings of the Healthcare Innovation Conference (HIC), Seattle, WA, USA, 8–10 October 2014; pp. 60–62. [Google Scholar]
- Wu, H.-Y.; Rubinstein, M.; Shih, E.; Guttag, J.V.; Durand, F.; Freeman, W.T. Eulerian video magnification for revealing subtle changes in the world. ACM Trans. Graph. 2012, 31, 65. [Google Scholar] [CrossRef]
- Bernacchia, N.; Scalise, L.; Casacanditella, L.; Ercoli, I.; Marchionni, P.; Tomasini, E.P. Non contact measurement of heart and respiration rates based on Kinect™. In Proceedings of the 2014 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Lisboa, Portugal, 11–12 June 2014; pp. 1–5. [Google Scholar]
- Yu, M.-C.; Wu, H.; Liou, J.-L.; Lee, M.-S.; Hung, Y.-P. Breath and position monitoring during sleeping with a depth camera. In Proceedings of the International Conference on Health Informatics (HEALTHINF-2012), Algarve, Portugal, 1–4 February 2012; pp. 12–22. [Google Scholar]
- Yang, C.; Cheung, G.; Chan, K.L.; Stankovic, V. Sleep monitoring via depth video compression & analysis. In Proceedings of the 2014 IEEE International Conference on Multimedia and Expo Workshops (ICMEW), Chengdu, China, 14–18 July 2014; pp. 1–6. [Google Scholar]
- Tanaka, M. Application of depth sensor for breathing rate counting. In Proceedings of the 2015 10th Asian Control Conference (ASCC), Kota Kinabalu, Malaysia, 31 May–3 June 2015; pp. 1–5. [Google Scholar]
- Ortmüller, J.; Gauer, T.; Wilms, M.; Handels, H.; Werner, R. Respiratory surface motion measurement by Microsoft Kinect. Curr. Dir. Biomed. Eng. 2015, 1, 270–273. [Google Scholar] [CrossRef]
- Aoki, H.; Nakamura, H.; Fumoto, K.; Nakahara, K.; Teraoka, M. Basic study on non-contact respiration measurement during exercise tolerance test by using kinect sensor. In Proceedings of the 2015 IEEE/SICE International Symposium on System Integration (SII), Nagoya, Japan, 11–13 December 2015; pp. 217–222. [Google Scholar]
- Centonze, F.; Schatz, M.; Prochazka, A.; Kuchynka, J.; Vysata, O.; Cejnar, P.; Valis, M. Feature extraction using MS Kinect and data fusion in analysis of sleep disorders. In Proceedings of the 2015 International Workshop on Computational Intelligence for Multimedia Understanding (IWCIM), Prague, Czech Republic, 29–30 October 2015; pp. 1–5. [Google Scholar]
- Yang, C.; Cheung, G.; Stankovic, V. Estimating heart rate via depth video motion tracking. In Proceedings of the 2015 IEEE International Conference on Multimedia and Expo (ICME), Turin, Italy, 29 June–3 July 2015; pp. 1–6. [Google Scholar]
- Harte, J.M.; Golby, C.K.; Acosta, J.; Nash, E.F.; Kiraci, E.; Williams, M.A.; Arvanitis, T.N.; Naidu, B. Chest wall motion analysis in healthy volunteers and adults with cystic fibrosis using a novel Kinect-based motion tracking system. Med. Boil. Eng. Comput. 2016, 54, 1631–1640. [Google Scholar] [CrossRef] [PubMed]
- Tahavori, F.; Adams, E.; Dabbs, M.; Aldridge, L.; Liversidge, N.; Donovan, E.; Jordan, T.; Evans, P.; Wells, K. Combining marker-less patient setup and respiratory motion monitoring using low cost 3D camera technology. In Proceedings of the SPIE Medical Imaging. International Society for Optics and Photonics, Orlando, FL, USA, 21 February 2015; pp. 94152I-1–94152I-7. [Google Scholar]
- Kumagai, S.; Uemura, R.; Ishibashi, T.; Nakabayashi, S.; Arai, N.; Kobayashi, T.; Kotoku, J.I. Markerless Respiratory Motion Tracking Using Single Depth Camera. Open J. Med. Imaging 2016, 6, 20–31. [Google Scholar] [CrossRef]
- Lee, J.; Hong, M.; Ryu, S. Sleep monitoring system using kinect sensor. Int. J. Distrib. Sens. Netw. 2015, 2015, 1–9. [Google Scholar] [CrossRef]
- Gambi, E.; Agostinelli, A.; Belli, A.; Burattini, L.; Cippitelli, E.; Fioretti, S.; Pierleoni, P.; Ricciuti, M.; Sbrollini, A.; Spinsante, S. Heart Rate Detection Using Microsoft Kinect: Validation and Comparison to Wearable Devices. Sensors 2017, 17, 1776. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Cheung, G.; Stankovic, V. Estimating heart rate and rhythm via 3D motion tracking in depth video. IEEE Trans. Multimed. 2017, 19, 1625–1636. [Google Scholar] [CrossRef]
- Al-Naji, A.; Gibson, K.; Lee, S.-H.; Chahl, J. Real time apnoea monitoring of children using the microsoft kinect sensor: A pilot study. Sensors 2017, 17, 286. [Google Scholar] [CrossRef] [PubMed]
- Kranjec, J.; Beguš, S.; Geršak, G.; Drnovšek, J. Non-contact heart rate and heart rate variability measurements: A review. Biomed. Signal Process. Control 2014, 13, 102–112. [Google Scholar] [CrossRef]
- Alnaji, A.; Gibson, K.; Lee, S.-H.; Chahl, J. Monitoring of cardiorespiratory signal: Principles of remote measurements and review of methods. IEEE Access 2017, 5, 15776–15790. [Google Scholar] [CrossRef]
- Papon, T.I.; Ahmad, I.; Saquib, N.; Rahman, A. Non-invasive heart rate measuring smartphone applications using on-board cameras: A short survey. In Proceedings of the 2015 International Conference on Networking Systems and Security (NSysS), Dhaka, Bangladesh, 5–7 January 2015; pp. 1–6. [Google Scholar]
- Alonso, A.; Sasin, J.; Bottini, N. Signs of respiratory disease. Apoptosis 2006, 85, 721–726. [Google Scholar]
- Alnaji, A.; Lee, S.-H.; Chahl, J. Quality index evaluation of videos based on fuzzy interface system. IET Image Proc. 2017, 11, 292–300. [Google Scholar] [CrossRef]
- Alnaji, A.; Perera, A.G.; Chahl, J. Remote monitoring of cardiorespiratory signals from a hovering unmanned aerial vehicle. Biomed. Eng. Online 2017, 16, 101. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Javaan, C. Non-contact heart activity measurement system based on video imaging analysis. Int. J. Pattern Recognit. Artif. Intell. 2017, 31, 1–21. [Google Scholar]
- Colominas, M.A.; Schlotthauer, G.; Torres, M.E. Improved complete ensemble EMD: A suitable tool for biomedical signal processing. Biomed. Signal Proc. Control 2014, 14, 19–29. [Google Scholar] [CrossRef]
- Huang, N.E.; Shen, Z.; Long, S.R.; Wu, M.C.; Shih, H.H.; Zheng, Q.; Yen, N.-C.; Tung, C.C.; Liu, H.H. The empirical mode decomposition and the Hilbert spectrum for nonlinear and non-stationary time series analysis. Proc. Math Phys. Eng. Sci. 1998, 454, 903–995. [Google Scholar] [CrossRef]
- Wu, Z.; Huang, N.E. Ensemble empirical mode decomposition: A noise-assisted data analysis method. Adv. Adapt. Data Anal. 2009, 1, 1–41. [Google Scholar] [CrossRef]
- Torres, M.E.; Colominas, M.A.; Schlotthauer, G.; Flandrin, P. A complete ensemble empirical mode decomposition with adaptive noise. In Proceedings of the 2011 IEEE international conference on Acoustics, Speech and Signal Processing (ICASSP), Prague, Czech Republic, 22–27 May 2011; pp. 4144–4147. [Google Scholar]
- Alnaji, A.; Chahl, J. Simultaneous tracking of cardiorespiratory signals for multiple persons using a machine vision system with noise artifact removal. IEEE J. Transl. Eng. Health Med. 2017, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Mandic, D.P.; Cichocki, A. Analysis and online realization of the CCA approach for blind source separation. IEEE Trans. Neural Netw. 2007, 18, 1505–1510. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-O.; Adali, T.; Wang, W.; Calhoun, V.D. Joint blind source separation by multiset canonical correlation analysis. IEEE Trans. Signal Proc. 2009, 57, 3918–3929. [Google Scholar] [CrossRef] [PubMed]
- Safieddine, D.; Kachenoura, A.; Albera, L.; Birot, G.; Karfoul, A.; Pasnicu, A.; Biraben, A.; Wendling, F.; Senhadji, L.; Merlet, I. Removal of muscle artifact from EEG data: Comparison between stochastic (ICA and CCA) and deterministic (EMD and wavelet-based) approaches. EURASIP J. Adv. Signal Proc. 2012, 2012, 127. [Google Scholar] [CrossRef]
- Lin, Y.-P.; Wang, Y.; Jung, T.-P. A mobile SSVEP-based brain-computer interface for freely moving humans: The robustness of canonical correlation analysis to motion artifacts. In Proceedings of the 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 1350–1353. [Google Scholar]
- Gao, J.; Zheng, C.; Wang, P. Online removal of muscle artifact from electroencephalogram signals based on canonical correlation analysis. Clin. EEG Neurosci. 2010, 41, 53–59. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, W.; Vergult, A.; Vanrumste, B.; Van Paesschen, W.; Van Huffel, S. Canonical correlation analysis applied to remove muscle artifacts from the electroencephalogram. IEEE Trans. Biomed. Eng. 2006, 53, 2583–2587. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Chen, X.; Servati, A.; Soltanian, S.; Servati, P.; Wang, Z.J. A Blind Source Separation Framework for Monitoring Heart Beat Rate Using Nanofiber-Based Strain Sensors. IEEE Sens. J. 2016, 16, 762–772. [Google Scholar] [CrossRef]
- Sweeney, K.T.; McLoone, S.F.; Ward, T.E. The use of ensemble empirical mode decomposition with canonical correlation analysis as a novel artifact removal technique. IEEE Trans. Biomed. Eng. 2013, 60, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Liu, A.; Chiang, J.; Wang, Z.J.; McKeown, M.J.; Ward, R.K. Removing Muscle Artifacts From EEG Data: Multichannel or Single-Channel Techniques? IEEE Sens. J. 2016, 16, 1986–1997. [Google Scholar] [CrossRef]
- Martínez-Martín, E.; del Pobil, Á.P. Motion Detection in Static Backgrounds. In Robust Motion Detection in Real-Life Scenarios; Springer: Berlin, Germany, 2012; pp. 5–42. [Google Scholar]
- Zuiderveld, K. Contrast Limited Adaptive Histogram Equalization; Graphics Gems IV; Academic Press Professional, Inc.: San Diego, CA, USA, 1994; pp. 474–485. [Google Scholar]
- Pediatrics, A.A.O. Apnea, sudden infant death syndrome, and home monitoring. Pediatrics 2003, 111, 914–917. [Google Scholar]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Int. J. Nurs. Stud. 2010, 47, 931–936. [Google Scholar] [CrossRef]

| Features | Kinect v1 | Kinect v2 |
|---|---|---|
| Technology used | Structured light coding technology | ToF technology |
| RGB sensor resolution | 640 × 480, 30 fps | 1920 × 1080, 30 fps |
| IR sensor resolution | 320 × 240, 30 fps | 512 × 424, 30 fps |
| RGB sensor Field of view | 62° × 48.6° | 84.1° × 53.8° |
| Depth sensor Field of view | 57° × 43° | 70° × 60° |
| Operative distance | 0.8 m–4 m (Default) 0.4 m–3.5 m (Near) | 0.5 m–4.5 m |
| Skeleton joints tracking | 20 joints | 25 joints |
| Number of detected subjects | 2 | 6 |
| Age | HR (Beats/Min) | RR (Breaths/Min) |
|---|---|---|
| Infant < 1 year | 110–160 | 22–55 |
| 1–3 years | 80–150 | 22–30 |
| 3–6 years | 70–120 | 16–24 |
| 6–13 years | 60–110 | 16–22 |
| Adults | 60–100 | 12–20 |
| States | Environmental Settings | Measured HR (Beats/Min) | Predicted HR without a Blanket | Predicted HR with a Blanket | Events |
|---|---|---|---|---|---|
| S1 | Well-lit | 66 | 68.48 | 69.41 | Normal |
| S2 | 124 | 126.01 | 127.68 | Tachycardia | |
| S3 | 56 | 58.82 | 59.07 | Bradycardia | |
| S1 | Dark | 65 | 67.94 | 68.93 | Normal |
| S2 | 130 | 133.05 | 135.14 | Tachycardia | |
| S3 | 56 | 58.13 | 59.62 | Bradycardia |
| States | Environmental Settings | Measured RR (Breaths/min) | Predicted RR without a Blanket | Predicted RR with a Blanket | Events |
|---|---|---|---|---|---|
| S1 | Well-lit | 14 | 14.88 | 15.69 | Normal |
| S2 | 34 | 34.91 | 35.78 | Tachypnea | |
| S3 | 8 | 8.82 | 9.54 | Bradypnea | |
| S1 | Dark | 14 | 14.95 | 16.01 | Normal |
| S2 | 33 | 34.05 | 35.14 | Tachypnea | |
| S3 | 9 | 10.04 | 10.83 | Bradypnea |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Naji, A.; Chahl, J. Detection of Cardiopulmonary Activity and Related Abnormal Events Using Microsoft Kinect Sensor. Sensors 2018, 18, 920. https://doi.org/10.3390/s18030920
Al-Naji A, Chahl J. Detection of Cardiopulmonary Activity and Related Abnormal Events Using Microsoft Kinect Sensor. Sensors. 2018; 18(3):920. https://doi.org/10.3390/s18030920
Chicago/Turabian StyleAl-Naji, Ali, and Javaan Chahl. 2018. "Detection of Cardiopulmonary Activity and Related Abnormal Events Using Microsoft Kinect Sensor" Sensors 18, no. 3: 920. https://doi.org/10.3390/s18030920
APA StyleAl-Naji, A., & Chahl, J. (2018). Detection of Cardiopulmonary Activity and Related Abnormal Events Using Microsoft Kinect Sensor. Sensors, 18(3), 920. https://doi.org/10.3390/s18030920







