A New Method for Sensing Soil Water Content in Green Roofs Using Plant Microbial Fuel Cells
Abstract
1. Introduction
2. Materials and Methods
Plant Microbial Fuel Cell (PMFC) Construction and Operation
3. Results
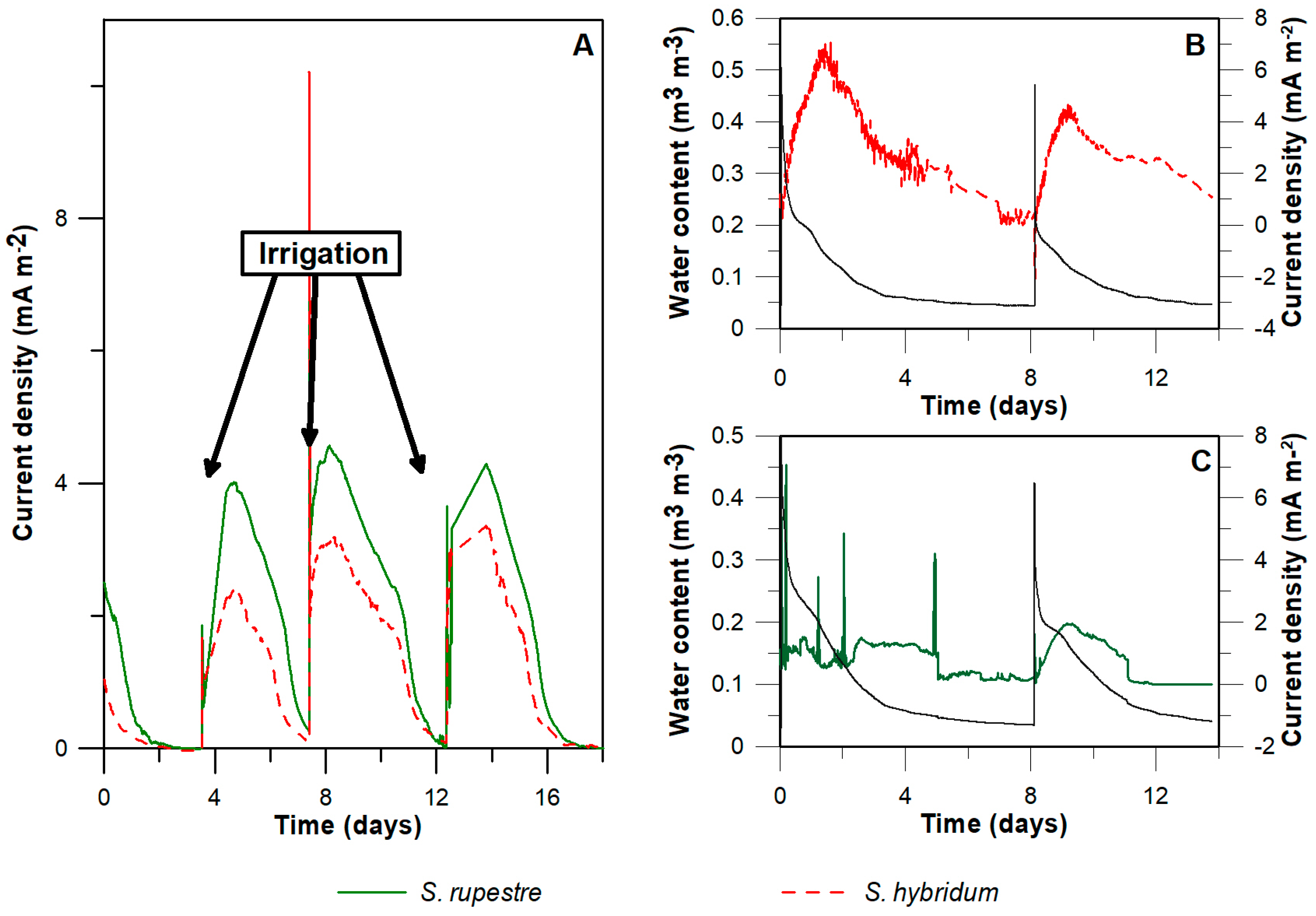
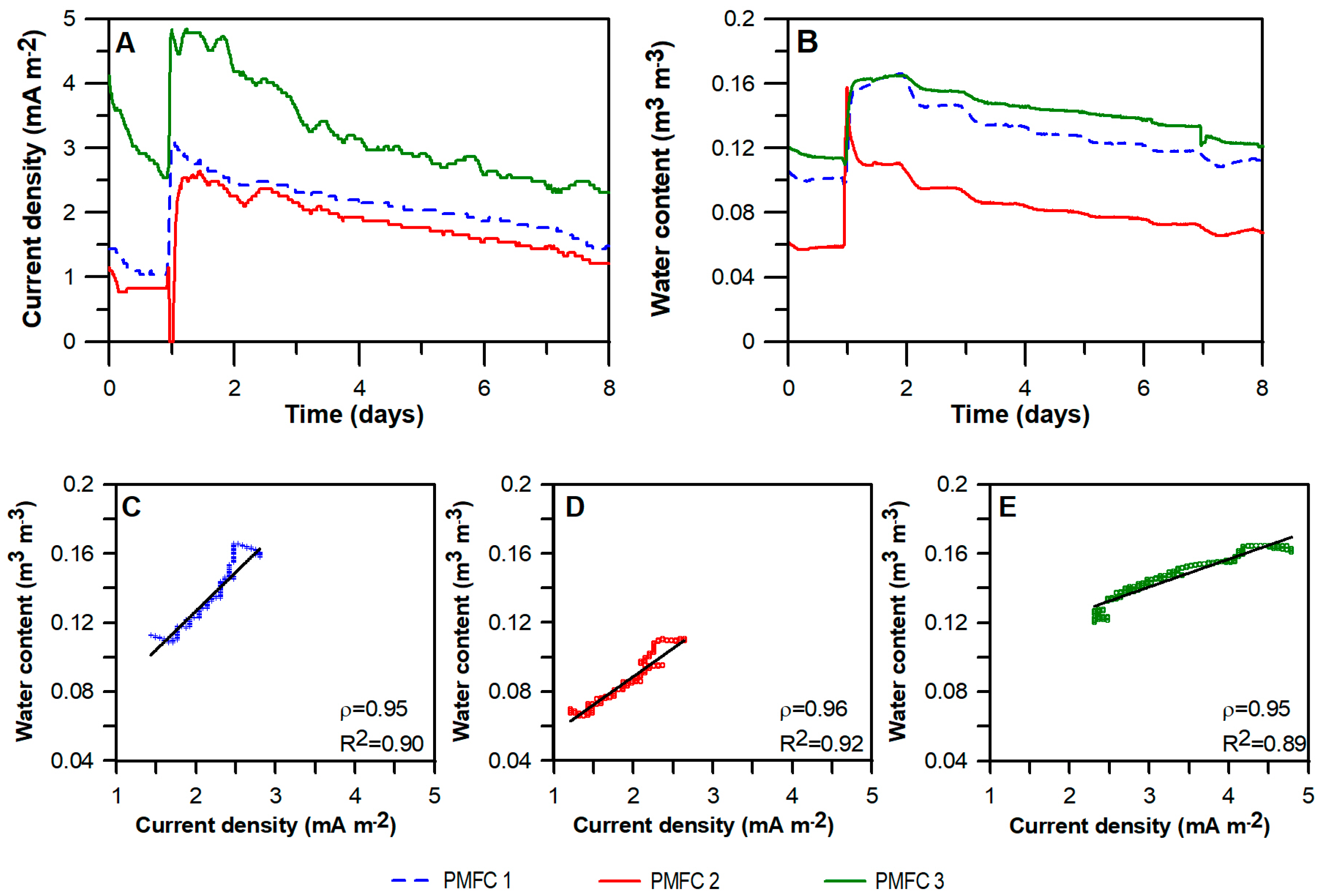
3.1. Water Content and Current Density
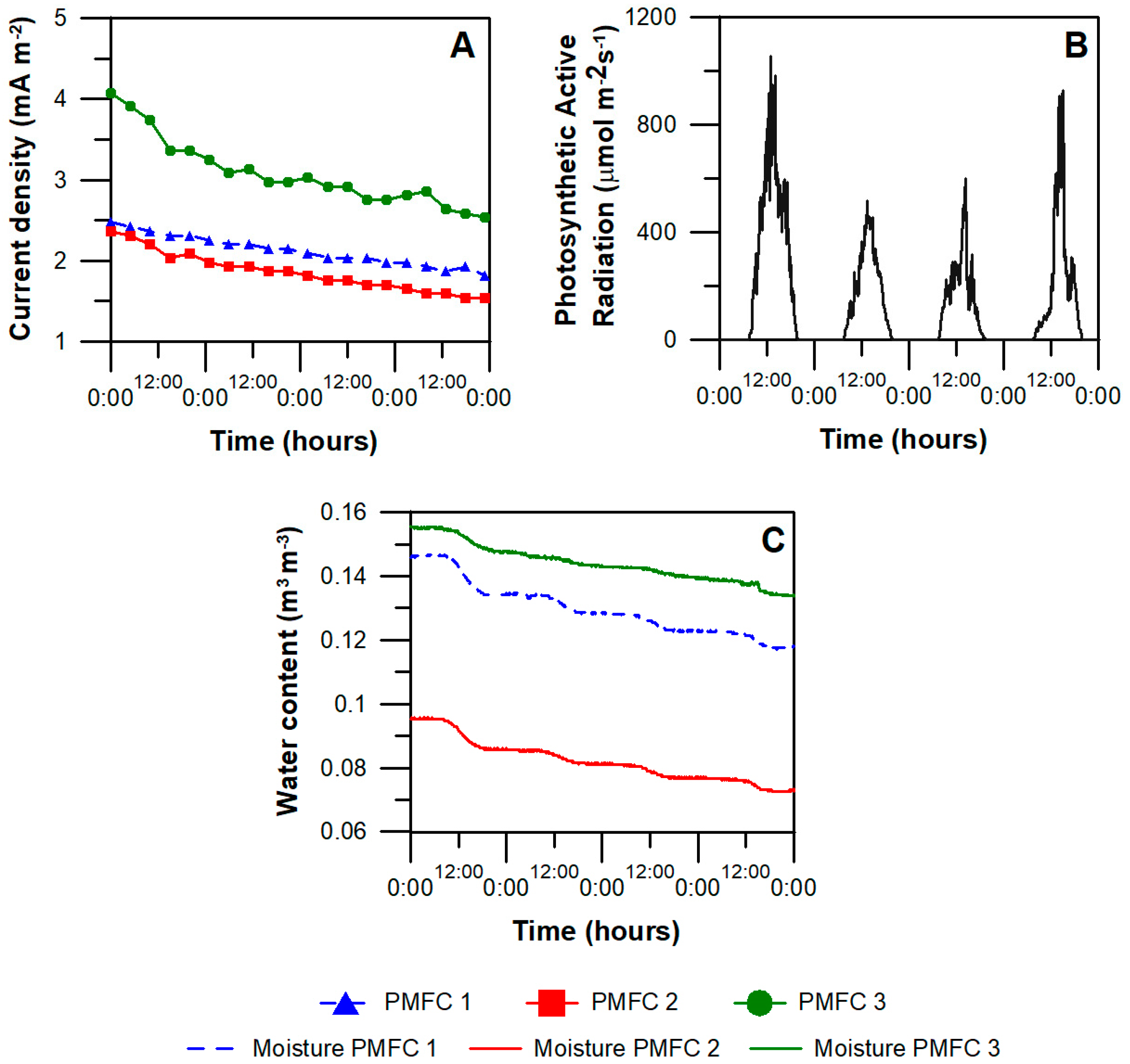
3.2. PMFC Performance
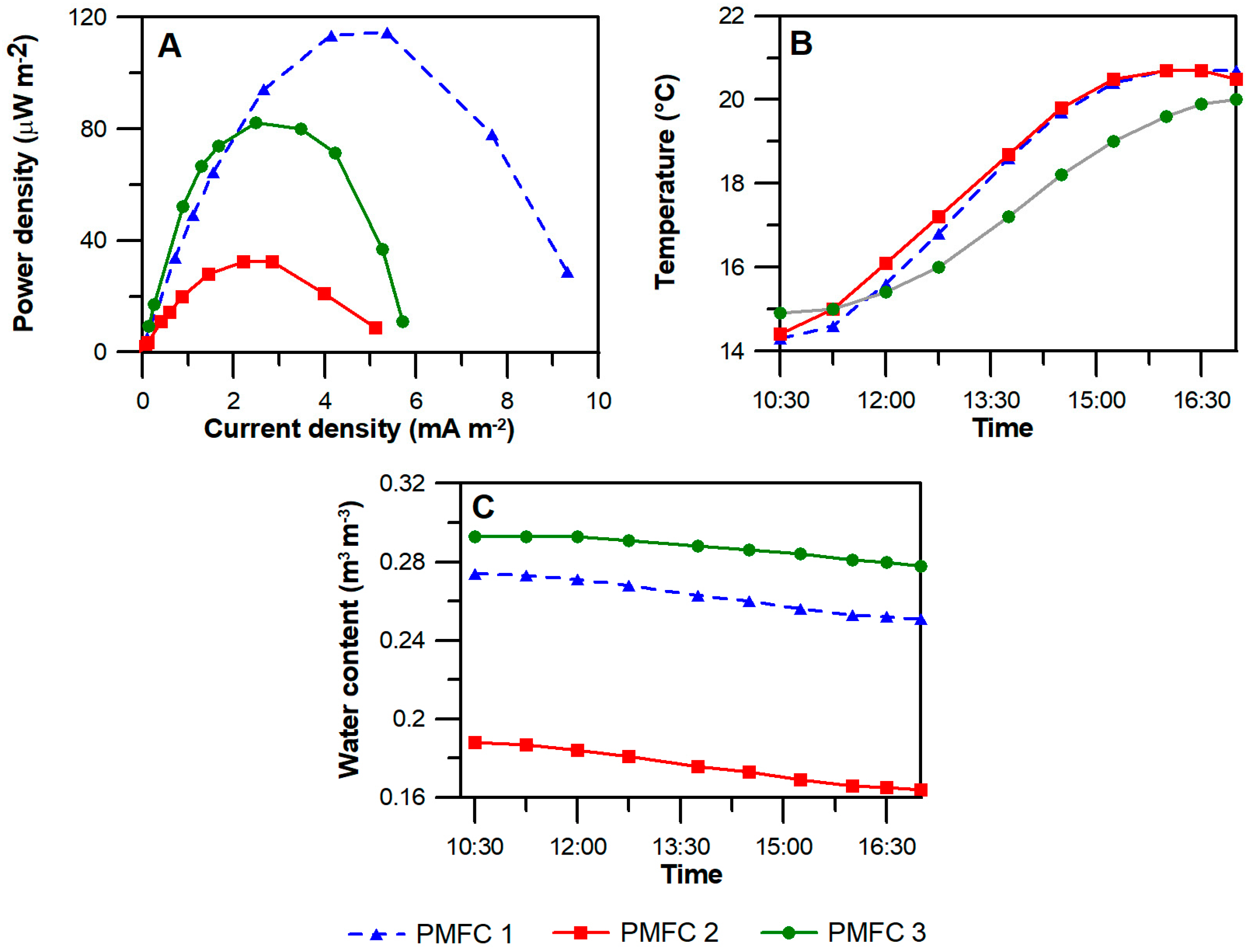
3.3. Power Density and Polarization Curves
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Berndtsson, J. Green roof performance towards management of runoff water quantity and quality: A review. Ecol. Eng. 2010, 36, 351–360. [Google Scholar] [CrossRef]
- Bass, B.; Liu, K.; Baskaran, B. Evaluating Rooftop and Vertical Gardens as an Adaptation Strategy for Urban Areas; National Research Council Canada: Ottawa, ON, Canada, 2003; pp. 1–106. [Google Scholar]
- Berardi, U.; GhaffarianHoseini, A.; GhaffarianHoseini, A. State-of-the-art analysis of the environmental benefits of green roofs. Appl. Energy 2014, 115, 411–428. [Google Scholar] [CrossRef]
- Brenneisen, S. Space for urban wildlife: Designing green roofs as habitats in Switzerland. Urban Habitats 2006, 4, 27–36. [Google Scholar]
- Yang, J.; Yu, Q.; Gong, P. Quantifying air pollution removal by green roofs in Chicago. Atmos. Environ. 2008, 42, 7266–7273. [Google Scholar] [CrossRef]
- Williams, N.S.G.; Rayner, J.P.; Raynor, K.J. Green roofs for a wide brown land: Opportunities and barriers for rooftop greening in Australia. Urban For. Urban Green. 2010, 9, 245–251. [Google Scholar] [CrossRef]
- Olate, E.; Gómez, M.F.; Musalem, M.; Sepúlveda, C.; Ferrer, J.M. Evaluation of native species for use in green roofs in the semiarid Mediterranean region of central Chile. Acta Hortic. 2013, 171–177. [Google Scholar] [CrossRef]
- Reyes, R.; Bustamante, W.; Gironás, J.; Pastén, P.A.; Rojas, V.; Suárez, F.; Vera, S.; Victorero, F.; Bonilla, C.A. Effect of substrate depth and roof layers on green roof temperature and water requirements in a semi-arid climate. Ecol. Eng. 2016, 97, 624–632. [Google Scholar] [CrossRef]
- Tapia, N.F.; Rojas, C.; Bonilla, C.A.; Vargas, I.T. Evaluation of sedum as driver for plant microbial fuel cells in a semi-arid green roof ecosystem. Ecol. Eng. 2017, 108, 203–210. [Google Scholar] [CrossRef]
- Zheng, Q.; Xiong, L.; Mo, B.; Lu, W.; Kim, S.; Wang, Z. Temperature and humidity sensor powered by an individual microbial fuel cell in a power management system. Sensors 2015, 15, 23126–23144. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Song, H.-L.; Yu, C.-Y.; Li, X.-N. Simultaneous degradation of toxic refractory organic pesticide and bioelectricity generation using a soil microbial fuel cell. Bioresour. Technol. 2015, 189, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, M.-C.; Cheng, C.-Y.; Liu, M.-H.; Chung, Y.-C. Effects of operating parameters on measurements of biochemical oxygen demand using a Mediatorless microbial fuel cell biosensor. Sensors 2016, 16, 35. [Google Scholar] [CrossRef] [PubMed]
- Logroño, W.; Guambo, A.; Pérez, M.; Kadier, A.; Recalde, C. A terrestrial single chamber microbial fuel cell-based biosensor for biochemical oxygen demand of synthetic rice washed wastewater. Sensors 2016, 16, 101. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.-H.; Cheng, C.-Y.; Liu, M.-H.; Chen, T.-Y.; Hsieh, M.-C.; Chung, Y.-C. Utility of Ochrobactrum Anthropi yc152 in a microbial fuel cell as an early warning device for hexavalent chromium determination. Sensors 2016, 16, 1272. [Google Scholar] [CrossRef] [PubMed]
- Grattieri, M.; Minteer, S.D. Self-powered biosensors. ACS Sens. 2017. [Google Scholar] [CrossRef] [PubMed]
- Grattieri, M.; Hasan, K.; Minteer, S.D. Bioelectrochemical systems as a multipurpose biosensing tool: Present perspective and future outlook. ChemElectroChem 2017, 4, 834–842. [Google Scholar] [CrossRef]
- Wang, X.; Cai, Z.; Zhou, Q.; Zhang, Z.; Chen, C. Bioelectrochemical stimulation of petroleum hydrocarbon degradation in saline soil using u-tube microbial fuel cells. Biotechnol. Bioeng. 2012, 109, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Chiranjeevi, P.; Mohanakrishna, G.; Mohan, S.V. Rhizosphere mediated electrogenesis with the function of anode placement for harnessing bioenergy through CO2 sequestration. Bioresour. Technol. 2012, 124, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, X.; Zhao, Q.; Wan, L.; Li, Y.; Zhou, Q. Carbon fiber enhanced bioelectricity generation in soil microbial fuel cells. Biosens. Bioelectron. 2016, 85, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Wolińska, A.; Stępniewska, Z.; Bielecka, A.; Ciepielski, J. Bioelectricity production from soil using microbial fuel cells. Appl. Biochem. Biotechnol. 2014, 173, 2287–2296. [Google Scholar] [CrossRef] [PubMed]
- Timmers, R.; Strik, D.B.T.B.; Hamelers, H.M.; Buisman, C.N. Long-term performance of a plant microbial fuel cell with Spartina Anglica. Appl. Microbiol. Biotechnol. 2010, 86, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Helder, M.; Strik, D.P.; Hamelers, H.V.; Kuhn, A.J.; Blok, C.; Buisman, C.J. Concurrent bio-electricity and biomass production in three plant-microbial fuel cells using Spartina Anglica, arundinella anomala and arundo donax. Bioresour. Technol. 2010, 101, 3541–3547. [Google Scholar] [CrossRef] [PubMed]
- Timmers, R.A.; Strik, D.P.B.T.B.; Arampatzoglou, C.; Buisman, C.J.N.; Hamelers, H.V.M. Rhizosphere anode model explains high oxygen levels during operation of a glyceria maxima PMFC. Bioresour. Technol. 2012, 108, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Helder, M.; Strik, D.P.; Timmers, R.A.; Raes, S.M.; Hamelers, H.V.; Buisman, C.J. Resilience of roof-top plant-microbial fuel cells during Dutch winter. Biomass Bioenergy 2013, 51, 1–7. [Google Scholar] [CrossRef]
- Kaku, N.; Yonezawa, N.; Kodama, Y.; Watanabe, K. Plant/microbe cooperation for electricity generation in a rice paddy field. Appl. Microbiol. Biotechnol. 2008, 79, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.R.; Mele, P.M. Subtle changes in rhizosphere microbial community structure in response to increased boron and sodium chloride concentrations. Soil Biol. Biochem. 2007, 39, 340–351. [Google Scholar] [CrossRef]
- Vranova, V.; Rejsek, K.; Skene, K.R.; Janous, D.; Formanek, P. Methods of collection of plant root exudates in relation to plant metabolism and purpose: A review. J. Plant Nutr. Soil Sci. 2013, 176, 175–199. [Google Scholar] [CrossRef]
- Strik, D.P.B.T.B.; Timmers, R.A.; Helder, M.; Steinbusch, K.J.J.; Hamelers, H.V.M.; Buisman, C.J.N. Microbial solar cells: Applying photosynthetic and electrochemically active organisms. Trends Biotechnol. 2011, 29, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Dunaj, S.J.; Vallino, J.J.; Hines, M.E.; Gay, M.; Kobyljanec, C.; Rooney-Varga, J.N. Relationships between soil organic matter, nutrients, bacterial community structure, and the performance of microbial fuel cells. Environ. Sci. Technol. 2012, 46, 1914–1922. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.A.; Campbell, C.S.; Hopmans, J.W.; Hornbuckle, B.K.; Jones, S.B.; Knight, R.; Ogden, F.; Selker, J.; Wendroth, O. Soil moisture measurement for ecological and hydrological watershed-scale observatories: A review. Vadose Zone J. 2008, 7, 358–389. [Google Scholar] [CrossRef]
- Quails, R.J.; Scott, J.M.; DeOreo, W.B. Soil moisture sensors for urban landscape irrigation: Effectweness and reliability1. JAWRA J. Am. Water Resour. Assoc. 2001, 37, 547–559. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tapia, N.F.; Rojas, C.; Bonilla, C.A.; Vargas, I.T. A New Method for Sensing Soil Water Content in Green Roofs Using Plant Microbial Fuel Cells. Sensors 2018, 18, 71. https://doi.org/10.3390/s18010071
Tapia NF, Rojas C, Bonilla CA, Vargas IT. A New Method for Sensing Soil Water Content in Green Roofs Using Plant Microbial Fuel Cells. Sensors. 2018; 18(1):71. https://doi.org/10.3390/s18010071
Chicago/Turabian StyleTapia, Natalia F., Claudia Rojas, Carlos A. Bonilla, and Ignacio T. Vargas. 2018. "A New Method for Sensing Soil Water Content in Green Roofs Using Plant Microbial Fuel Cells" Sensors 18, no. 1: 71. https://doi.org/10.3390/s18010071
APA StyleTapia, N. F., Rojas, C., Bonilla, C. A., & Vargas, I. T. (2018). A New Method for Sensing Soil Water Content in Green Roofs Using Plant Microbial Fuel Cells. Sensors, 18(1), 71. https://doi.org/10.3390/s18010071




