Supplemental Blue LED Lighting Array to Improve the Signal Quality in Hyperspectral Imaging of Plants
Abstract
:1. Introduction
2. Experimental Section
2.1. Hyperspectral Measuring Setup

2.2. Supplemental Blue LED Lighting Array
3. Results and Discussion
3.1. Noise Profile
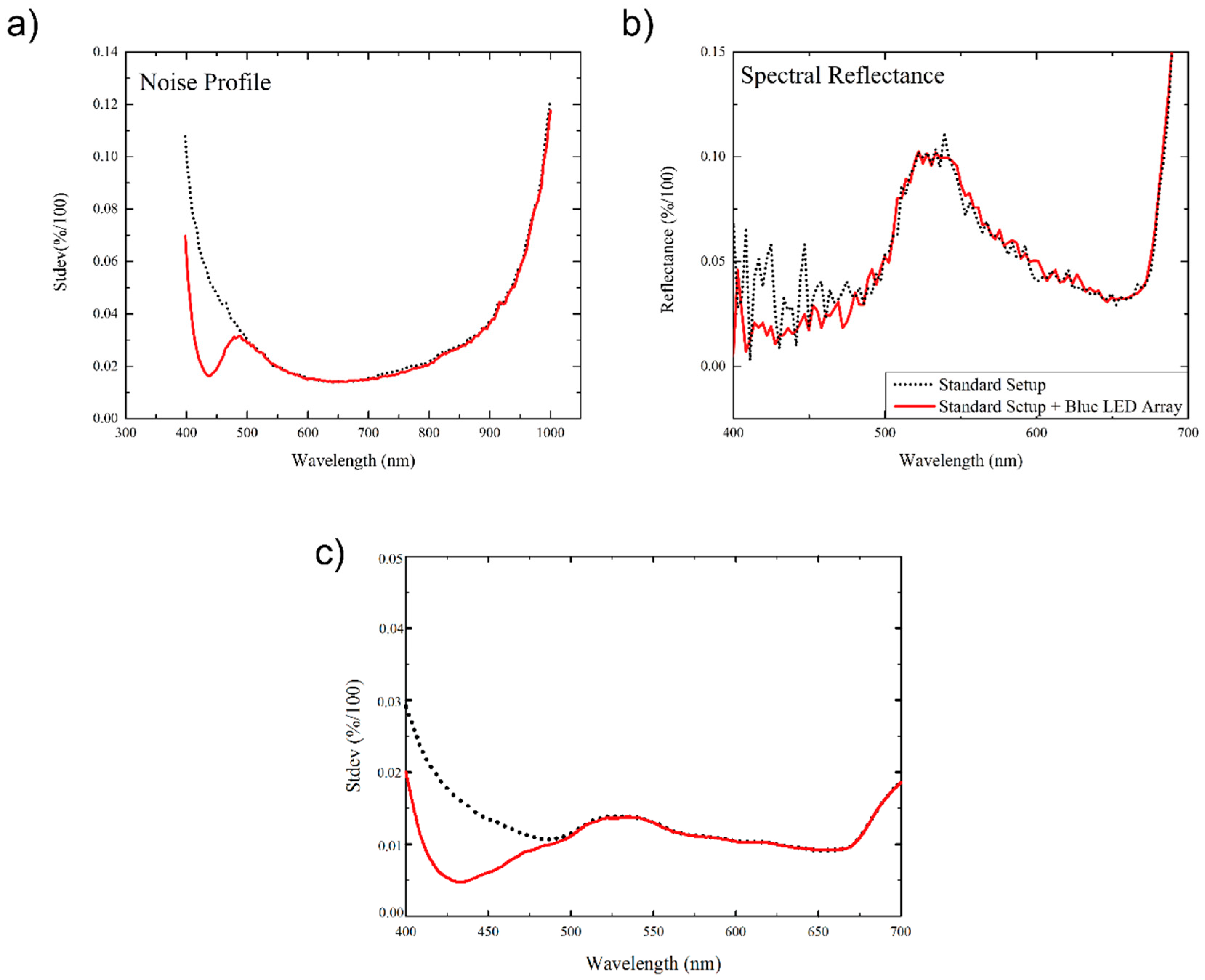
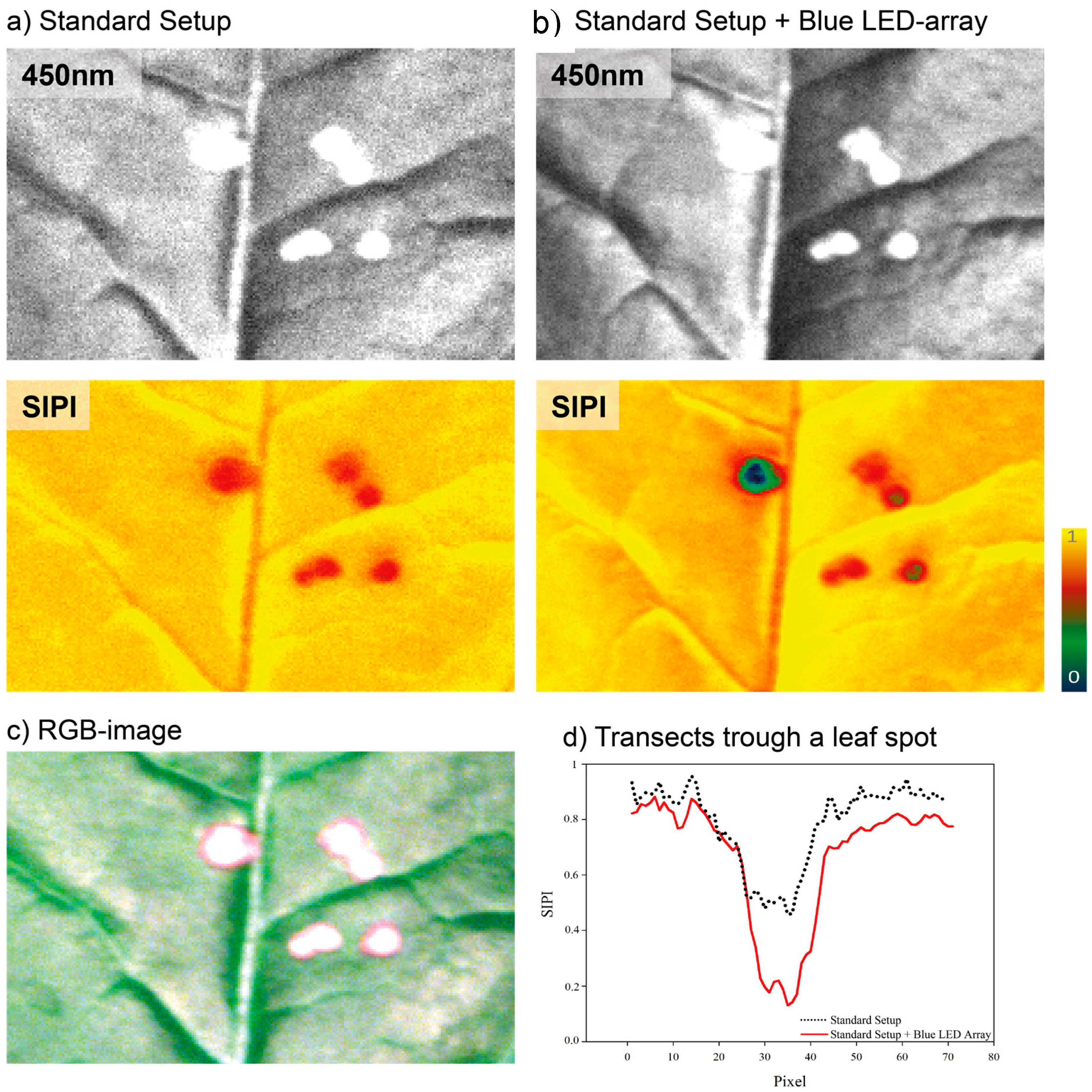
3.2. Improved Sensitivity of Spectral Vegetation Indices
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Carter, G.A.; Knapp, A.K. Leaf optical properties in higher plants: Linking spectral characteristics to stress and chlorophyll concentration. Am. J. Bot. 2001, 88, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Mahlein, A.-K.; Steiner, U.; Oerke, E.-C.; Dehne, H.-W. Recent advances in sensing plant diseases for precision crop protection. Eur. J. Plant Pathol. 2012, 133, 197–209. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Merzlyak, M.N. Signature analysis of leaf reflectance spectra: Algorithm development for remote sensing of chlorophyll. J. Plant Physiol. 1996, 148, 494–500. [Google Scholar] [CrossRef]
- Sims, D.A.; Gamon, J.A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Penuelas, J.; Filella, I. Visible and near-infrared reflectance techniques for diagnosing plant physiological status. Trends Plant Sci. 1998, 3, 151–156. [Google Scholar] [CrossRef]
- Penuelas, J.; Filella, I.; Lloret, P.; Munoz, F.; Vilajeliu, M. Reflectance assessment of mite effects on apple trees. Int. J. Remote Sens. 1995, 16, 2727–2733. [Google Scholar] [CrossRef]
- Mahlein, A.-K.; Rumpf, T.; Welke, P.; Dehne, H.-W.; Plümer, L.; Steiner, U.; Oerke, E.-C. Development of spectral indices for detecting and identifying plant diseases. Remote Sens. Environ. 2013, 128, 21–30. [Google Scholar] [CrossRef]
- Bousquet, L.; Lachérade, S.; Jacquemoud, S.; Moya, I. Leaf BRDF measurements and model for specular and diffuse components differentiation. Remote Sens. Environ. 2005, 98, 201–211. [Google Scholar] [CrossRef]
- Mahlein, A.-K.; Steiner, U.; Hillnhütter, C.; Dehne, H.-W.; Oerke, E.-C. Hyperspectral imaging for small-scale analysis of symptoms caused by different sugar beet diseases. Plant Methods 2012, 8, 3. [Google Scholar] [CrossRef] [PubMed]
- Feilhauer, H.; Oerke, E.C.; Schmidtlein, S. Quantifying empirical relations between planted species mixtures and canopy reflectance with PROTEST. Remote Sens. Environ. 2010, 114, 1513–1521. [Google Scholar] [CrossRef]
- Carvalho, S.; Macel, M.; Schlerf, M.; Skidmore, A.K.; van der Putten, W.H. Soil biotic impact on plant species shoot chemistry and hyperspectral reflectance patterns. New Phytol. 2012, 196, 1133–1144. [Google Scholar] [CrossRef] [PubMed]
- Delalieux, S.; Somers, B.; Verstaeten, W.W.; van Aardt, J.A.N.; Keulemans, W.; Coppin, P. Hyperspectral indices to diagnose leaf biotic stress of apple plants, considering leaf phenology. Int. J. Remote Sens. 2009, 30, 1887–1912. [Google Scholar] [CrossRef]
- Savitzky, A.; Golay, J.M.E. Smoothing and differentiation of data by simplified least squares procedures. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- Grieve, B.D; Hammersley, S.; Mahlein, A.K.; Oerke, E.C.; Goldbach, H. Localized multispectral crop imaging sensors: Engineering & validation of a cost effective plant stress and disease sensor. In Proceedings of the IEEE 2015 Sensors Applications Symposium, Zadar, Croatia, 11–15 April 2015; Gurkan, D., Baglio, S., Eds.; IEEE Instrument & Measurement Society: Piscataway, NJ, USA, 2015. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahlein, A.-K.; Hammersley, S.; Oerke, E.-C.; Dehne, H.-W.; Goldbach, H.; Grieve, B. Supplemental Blue LED Lighting Array to Improve the Signal Quality in Hyperspectral Imaging of Plants. Sensors 2015, 15, 12834-12840. https://doi.org/10.3390/s150612834
Mahlein A-K, Hammersley S, Oerke E-C, Dehne H-W, Goldbach H, Grieve B. Supplemental Blue LED Lighting Array to Improve the Signal Quality in Hyperspectral Imaging of Plants. Sensors. 2015; 15(6):12834-12840. https://doi.org/10.3390/s150612834
Chicago/Turabian StyleMahlein, Anne-Katrin, Simon Hammersley, Erich-Christian Oerke, Heinz-Wilhelm Dehne, Heiner Goldbach, and Bruce Grieve. 2015. "Supplemental Blue LED Lighting Array to Improve the Signal Quality in Hyperspectral Imaging of Plants" Sensors 15, no. 6: 12834-12840. https://doi.org/10.3390/s150612834
APA StyleMahlein, A.-K., Hammersley, S., Oerke, E.-C., Dehne, H.-W., Goldbach, H., & Grieve, B. (2015). Supplemental Blue LED Lighting Array to Improve the Signal Quality in Hyperspectral Imaging of Plants. Sensors, 15(6), 12834-12840. https://doi.org/10.3390/s150612834





