Electronic-Nose Applications for Fruit Identification, Ripeness and Quality Grading
Abstract
: Fruits produce a wide range of volatile organic compounds that impart their characteristically distinct aromas and contribute to unique flavor characteristics. Fruit aroma and flavor characteristics are of key importance in determining consumer acceptance in commercial fruit markets based on individual preference. Fruit producers, suppliers and retailers traditionally utilize and rely on human testers or panels to evaluate fruit quality and aroma characters for assessing fruit salability in fresh markets. We explore the current and potential utilization of electronic-nose devices (with specialized sensor arrays), instruments that are very effective in discriminating complex mixtures of fruit volatiles, as new effective tools for more efficient fruit aroma analyses to replace conventional expensive methods used in fruit aroma assessments. We review the chemical nature of fruit volatiles during all stages of the agro-fruit production process, describe some of the more important applications that electronic nose (e-nose) technologies have provided for fruit aroma characterizations, and summarize recent research providing e-nose data on the effectiveness of these specialized gas-sensing instruments for fruit identifications, cultivar discriminations, ripeness assessments and fruit grading for assuring fruit quality in commercial markets.1. Introduction
Fruit quality is judged by consumers primarily from their perception of the acceptability of fruits based on characteristics including visual appeal (lack of blemishes, color, size, and texture), ripeness, aroma and flavor. The quality of fruits (as measured by aroma, flavor, color, and textural characteristics) constantly changes during fruit development from pre-harvest through post-harvest stages as fruits grow and ripen, and during maintenance in storage [1]. Personal consumer preferences for different types of fruits are reflected in their particular choices of fruit varieties or cultivars selected for purchase. Fruit varieties vary widely in aroma characteristics due to differences in the composition of aromatic volatiles present in fruit aromas which are ultimately determined by plant genetics [2,3]. Previously, professional human graders and panelists have been used to judge fruit quality based on visual and aroma characteristics for selecting and evaluating fruits for ripeness at harvest and salability in commercial fruit markets [4]. The advent of electronic-nose (e-nose) devices has offered new alternative tools for grading fruits and other perishable foods using more consistent qualitative and quantitative measures of aroma characteristics that avoid the highly variable subjective opinions of human graders [5,6]. These instruments provide new means for characterizing fruit aromas for numerous applications ranging from the development of new fruit varieties by geneticists or fruit breeders to the timing of fruit harvests, transportation, storage operations (handling), and final selection by commercial dealers and retailers in fresh produce markets.
Fruit aroma is often the most valued characteristic determining fruit quality and consumer choice because aroma is usually the best indicator of fruit flavor. Electronic-noses are ideal digital, electronic devices for identifying, characterizing and grading fruit aromas from different fruits and fruit varieties because these instruments are capable of rapidly and consistently evaluating complex volatile gaseous mixtures without having to identify all of the chemical constituents present in the bouquet of fruit aromas [5,7]. E-noses contain a sensor array that evaluates all of the chemical constituents present in an aroma mixture (as a whole sample) and coverts the electronic output signals (via a transducer) from all of the sensors in the array and collectively assembles them to form a distinct digital pattern, sometimes referred to as an Electronic Aroma Signature Pattern (EASP) that is highly unique and specific to the particular gas mixture being analyzed [8,9]. In this way, the instrument output generates an aroma signature or smell-print that can be used to identify the particular type and variety of fruit being analyzed.
Fruits produce and release a wide variety of Volatile Organic Compounds (VOCs) that make up their characteristic aromas with esters, terpenoids, lactones and derivatives of amino acids, fatty-acids and phenolic compounds being the dominant classes of organic volatiles represented in fruit aromas [3]. Even though different fruits share some aromatic characteristics, each fruit has a distinctive aroma that depends upon the specific combination of VOCs present in the aroma mixture [10]. Whereas some specific volatiles are common to different fruit types, other fruit volatiles are specific to only one or only a few related fruits. Production and emission of volatiles from fruits is markedly influenced by numerous factors that interact in complex ways to determine fruit volatile composition. Multiple biochemical pathways are responsible for determining the final composition of volatile compounds released from different fruit types.
The purpose of this review is to summarize some of main chemical characteristics of fruit volatile gaseous mixtures which are conducive to characterization and analysis by electronic-nose technologies, to describe the diverse potential applications of e-nose technologies in the agro-fruit production sector of the agricultural production industry, and to provide examples of research that have demonstrated many ways in which e-nose devices have been utilized to distinguish between the fruit volatiles of different plant species and varieties for the purpose of analyzing and grading fruit quality and aroma characteristics.
2. Chemical Characteristics of Fruit Volatiles
Fruit aromas consist of a complex mixture of VOCs whose composition is specific to plant species and fruit variety [2,3]. Although different fruits often share many aromatic characteristics, each fruit has a distinctive aroma that depends upon the combination of volatiles, the concentration and the perception threshold of individual volatile compounds [10]. The most important aroma compounds include amino acid-derived compounds, lipid-derived compounds, phenolic derivatives, and mono- and sesquiterpenes [3]. Although fruit aromas are generally complex mixtures of a wide range of compounds, volatile esters often represent the major components of aroma volatiles present in rosaceous fruits such as apple (Malaus domestica Borkh.) and peach (Prunus persica L.) [11,12].
Fruit volatiles are mainly composed of VOCs in relatively few chemical classes, including primarily aliphatic esters, alcohols, aldehydes, ketones, lactones, terpenoids (monoterpenes, sesquitepenes) and apocarotenoids [13]. However, there are many thousands of volatile compounds represented by these chemical classes that make up the complex aroma mixtures of the numerous fruit types cultivated for agronomic markets of the world. Some of the predominant VOC principal components, comprising the distinctive aroma mixtures of selective fruit types which are representatives of the most common chemical classes found in fruit volatiles, are presented in Table 1. Volatile compounds in the aliphatic ester chemical class are the most abundant types of organic compounds found in many fruit volatiles; and esters are most responsible for the sweet smell of flowers and fruits of most angiosperms or seed plants [14]. In aromatic melon varieties for example, volatile esters predominate in fruit aromas that also contain aldehydes, short-chain alcohols, sesquiterpenes, norisoprenes, and aromatic sulfur-containing compounds [15]. Non-aromatic fruit varieties often have much lower levels of total volatiles and lack volatile esters [16]. Esters are a particularly important component of strawberry fruit aroma, accounting for 90% of the total number of volatiles in ripe strawberry fruit [17]. Esters also are the key volatiles responsible for the flavor characteristics of citrus [18].
Fruit volatiles, in addition to chemical class categorizations, may be classified as primary compounds (present in intact fruit tissue) or secondary compounds (produced as a result of tissue disruption of fruit tissue) [13]. Consequently, the condition of the fruit tissue being analyzed, either intact or disrupted, will influence the characteristics (chemical composition) and output patterns of the resulting aroma profiles. Some aroma compounds may be released only from cell disruption due to physical damage or injury to the fruit. Other fruit volatiles are more bound internally in the fruit, perhaps due to lower volatility or lack of direct exposure to the air as a result of fruit tissue compartmentalization.
Numerous factors affect fruit volatile chemical composition during all phases of the agronomic production process, including plant genetics, harvest time, fruit maturity, and agronomic environmental conditions, as well as postharvest handling, transportation and storage. Plant genetics, plant hormones, and environmental factors strongly influence the biosynthesis pathways responsible for the release of volatile aroma compounds from fruits under various conditions over time. The availability of primary precursor substrates for biosynthetic pathways producing aroma compounds is highly regulated both in amount and composition during fruit development [34]. All of these factors have varying effects on the aroma volatiles released at different stages of fruit development and after fruit harvest when the detached fruit is no longer influenced by the biochemical processes of the plant.
Fatty acids are the major primary precursor substrates of many aroma volatiles in most fruit types [2]. Aliphatic alcohols, aldehydes, ketones, organic acids, esters and lactones, ranging from C1 to C20, are all derived from fatty acid precursors through three key biosynthetic processes: α-oxidation, β-oxidation and the lipoxygenase pathway [35]. Volatiles derived from fatty acid precursors are important character-impact aroma compounds responsible for fresh fruit flavors at high concentrations.
The terpenoids comprise the largest class of plant secondary metabolites (about 20,000 identified), derived from the universal C5 precursor isopentenyl diphosphate (IPP) and its allylic isomer dimethylallyl diphosphate (DMAPP) from two independent pathways, the mevalonic acid (MVA) and methylerythritol phosphate (MEP) biochemical pathways, with many volatile products represented [36,37]. Terpenes are classified into monoterpenes, diterpenes and sesquiterpenes, depending on the number of repeating units of a 5-carbon molecule (isoprene), the structural unit of all terpenoids, present in the molecule. Hemiterpenes (C5), monoterpenes (C10), sesquiterpenes (C15), homoterpenes (C11 and C16), and some diterpenes (C20) are quite volatile VOCs because they have a high vapor pressure, allowing their rapid release into the atmosphere [13].
The complex gaseous mixtures of VOCs released from various fruit types (plant species), detectable by e-nose instruments, depend on the extent to which different metabolic pathways predominate in the generation of fruit volatiles as determined by genetic and environmental factors. Certain types of VOCs are more frequently associated with specific fruits as a result of unique combinations of metabolic pathways that control primary and secondary metabolite production in fruit tissues. The major chemical classes and representative VOCs found in fruit volatile mixtures, most associated as principal components derived from specific fruit types, are presented in Table 2. These associations do not preclude the occurrence of VOCs from many other chemical classes in volatile mixtures from each fruit type, but occur in different relative proportions (molar ratios) as lesser components in VOC mixtures derived from various fruit types.
The total number of aromatic compounds that contribute to fruit aromas vary considerably in different fruit types, but the complex mixture of VOCs found in individual fruit aromas usually is an extensive list of volatiles from different classes of organic compounds. Fruit aromas from fresh apples (Malus domestica Borkr) have been reported to contain at least 300 volatile compounds [51]. The total number and concentration of VOCs emitted by ripening apples are cultivar specific [52].
The epidermal tissue (peel) of apples produces a greater quantity of volatile compounds than internal fleshy (pericarp) tissues [53]. This higher capacity for aroma production by peel tissue is due to either the abundance of fatty acid substrates or higher metabolic activity in the peel [54,55]. Peach (Prunus persica (L.) Batsch) fruit aromas consist of about 100 volatile compounds, among them, C6 aldehydes and alcohols provide the green-note aroma, while lactones and esters are responsible for fruity aromas [56–58]. Esters, including hexyl acetate and (Z)-3-hexenyl acetate, are key odorants influencing the flavor characteristics of peach fruit [59]. Changes in these volatiles occur during fruit development and postharvest ripening [60]. Aldehydes tend to decline, while esters increase in the fruit during development. Postharvest treatments, low temperature and controlled atmosphere, can influence changes in peach aroma quality [12,61]. More than 300 VOCs have been identified in pear fruit (Pyrus pyrifolia Nak.) [62]. Methyl and hexyl esters of decadienoate are the main character-imparting compounds of European pear [62,63].
Apricot (Prunus armeniaca L.) fruit aromas have more than 200 different volatile compounds [64]. The most abundant volatile compounds by concentration were aldehydes, primarily hexanal and (E)-2-hexenal, that decreased in concentration during ripening [21,22].
Banana (Musa spp.) fruit aroma has about 250 VOCs [40], although the characteristic banana fruity top notes are from volatile esters, such as isoamyl acetate and isobutyl acetate that tend to increase in concentration during ripening [23,40]. Volatile compounds in citrus fruits accumulate in oil glands of flavedo and in the oil bodies of the juice sacs from which 100 VOCs have been identified, but varietal differences in the volatile profiles are primarily quantitative and only a few compounds are variety-specific [65–68].
Approximately 42 volatiles has been associated with the fruit aromas of southern highbush blueberry (Vaccinium species) cultivars [43]. Certain varieties contain a large amount of esters and C6 aldehydes, whereas others produce more terpenoids and less esters.
Melon (Cucumis and Citrullus species) fruit aromas have more than 240 VOCs identified in different varieties [69]. Melon fruits release numerous compounds, particularly C9 aliphatic compounds that are the major determinants of fruit quality as perceived by consumers. These compounds are strongly dependent on variety and particular physiological characteristics of the fruit. For example, climacteric melons (cantaloupes) have greater aroma intensity and shorter shelf life than less climacteric melons (honeydew melons) [70]. Volatiles derived from amino acids are major contributors to the aromas of both aromatic and non-aromatic melon varieties [71,72].
Grape (Vitis vinifera L.) fruit aromas contain many VOCs, including monoterpenes, C13 norisoprenoids, alcohols, esters and carbonyl compounds [73]. Grape varieties may be divided into aromatic and nonaromatic categories. Terpenoids are major volatiles in both red and white grapes [44]. At veraison, terpene production (in both Riesling and Cabernet Sauvignon varieties) generally is low, but Riesling grapes produced some terpenes (geraniol and α-muurolene) post-veraison.
Kiwi (Actinidia species) fruit aromas consist of more than 80 compounds with the major volatile components being methyl and ethyl butanoate, (Z)- and (E)-2-hexenal, hexanal, (Z)- and (E)-3-hexenol, and methyl benzoate [27]. Some important variations in fruit aroma volatiles produced by different kiwifruit varieties have been found to be due to the presence or absence of diverse sulfur-containing VOCs.
Mango (Mangifera indica L.) fruit aromas contain more than 270 VOCs in different mango varieties [74]. Monoterpenes are the most important compounds contributing to mango flavor [75]. Generally, terpenes are the major class of compounds in New World and Colombian mangoes whereas alcohols, ketones, and esters are mainly responsible for the characteristic aroma of Old World mangoes [76,77].
At least 280 VOCs have been found in pineapple (Ananas comosus L. Merr.) fruit aromas [30]. Esters and hydrocarbons were found to be the major constituents of fruit aromas, whereas octenoic acid, methyl ester, hexanoic acid, octanoic acid and ethyl ester were minor aromatic components. The relative content of different volatiles in pineapple aroma varied significantly during fruit development [49].
Raspberry (Rubus idaeus x ursinus) fruit aromas are composed of at least 200 volatile compounds that vary in concentrations for different cultivars. Many alcohols, aldehydes and ketones (including raspberry ketone, α-ionone, β-ionone, linalool, (Z)-3-hexenol, geraniol, nerol, α-terpineol, furaneol, hexanal, β-ocimene, 1-octanol, β-pinene, β-damascenone, ethyl 2-methylpropanoate, (E)-2-hexenal, heptanal, and benzaldehyde have been identified in raspberry aroma [13]. Among them, α-ionone, β-ionone, geraniol, nerol, linalool, and raspberry ketone probably contribute most to red raspberry aroma [31].
The complex fruit aroma of strawberry (Fragaria x ananassa Duch.) contains approximately 350 volatile compounds [3,78]. The furanone compound (furaneol), 2,5-dimethyl-4-hydroxy-3(2H)-furanone, and its methyl derivative (mesifurane) are considered the dominating compounds that contribute the typical caramel-like, sweet, floral and fruity aroma [17]. Aldehydes and alcohols (such as hexanal, trans-2-hexenal and cis-3-hexen-1-ol) contribute the unripe notes to green strawberry aroma in which the concentrations of these components are cultivar and ripeness dependent [17].
Notice from comparisons of key volatiles (principal components) that distinguish between different fruit types that certain variations in fruit volatiles from specific chemical classes often are most useful for fruit aroma discriminations. For example, apple and pineapple varieties may be distinguished primarily by differences in aroma ester composition, i.e., variations in ester volatiles present in fruit aromas, whereas grapes and mango varieties are discriminated mostly by terpene volatiles that are detected in the aroma. By contrast, banana varieties are distinguished mainly by aldehydes and aliphatic alcohol volatiles, but ketones, furaneols, and alcohols are more important for distinguishing between blackberry and raspberry varieties. Discriminations between blueberry varieties are determined predominantly by the presence of esters, aldehydes, and terpenoids in the fruit aromas.
Despite the fact that a very large number of VOCs have been detected in various fresh fruit types, only a small fraction of these compounds have been identified as contributing significantly to the impact components (top and middle notes) of fruit aroma based on quantitative abundance and human olfactory thresholds [79]. The human olfactory threshold (for a particular aroma) is defined as the lowest concentration of aromatic compounds present in an aroma in which human subjects (usually 50 percent of a human panel) can smell the presence of the aroma [80].
Differences in the aroma characteristics of different fruit varieties are attributed to variations in their chemical profiles, based on the types of VOCs present and the relative concentrations of individual volatiles found in the aroma mixture. The principal VOCs, found in the aromas of specific fruit cultivars, may be used to distinguish between different fruit varieties. These differences in aroma composition and relative abundance of fruit volatiles in different fruit varieties are the means by which e-nose devices are capable of recognizing differences in fruit aromas and discriminating between fruit cultivars based on their distinct aroma signature patterns resulting from variations in e-nose response (sensor-array outputs) to different fruit aromas. Thus, e-nose discriminations of fruit aroma are determined both by the volatiles present and molar ratios of individual components found in each aroma (gaseous mixture)s.
3. Electronic-Nose Applications for Fruit Aroma Characterizations
Initial interest in the use of electronic noses as a non-destructive method to study the characteristics of fruits was shown by Benady et al. [81] who developed a sensing machine with a single semiconductor gas sensor in 1995, located within a small cup, placed on the surface of fruits of three different muskmelon cultivars. The instrument could discriminate between ripe and unripe fruits with an accuracy of 90.2%, and sort fruits into three ripeness categories (unripe, half-ripe and fully ripe) with an accuracy of 83%. The same research group worked on blueberries (the same year) to determine the variability in e-nose response among blueberry cultivars and to assess ripening stage and fruit quality (Simon et al.) [82].
Since these pioneering works, researchers have focused on developing and testing non-destructive sensorial techniques for the evaluation of the many and diverse characteristics and qualities of various fruits, particularly fruit maturity stages, shelf life and genotypic effects on aroma or bouquet characteristics. A comprehensive list of e-nose applications for characterizations and chemical discrimination of fruit volatiles for many fruit types is given in Table 3.
3.1. Apples and Pears
The fruit species receiving the most interest among e-nose researchers is apple (Malus domestica Borkr). Bai et al. [83] examined changes in the aroma profile on freshly-cut Gala apple slices treated with ethanol vapor, heat and 1-methylcyclopropene to prolong visual shelf-life. The FOX 4000 e-nose system (Alpha MOS, Toulouse, France) was utilized to assess aroma quality and the results demonstrated that pretreatments with ethanol and heat are effective in prolonging visual shelf life, but at the expense of aroma quality.
One of the main needs of fruits producers is to determine optimal picking date for different fruit types in order to assure the presence of preferred traits expected by consumers for maximum salability. Harvesting fruits at the optimal physiological condition results in fruits with the highest quality characteristics (aroma, firmness, color, flavor), and an extended shelf life. Fruit picked too early will not ripen sufficiently after storage and may suffer from physiological disorders, whereas those picked too late will be mealy and too soft following storage. Traditional techniques for assessing apple quality are destructive, requiring that only random samples are tested. For these reasons, electronic noses have been preferred for assessing the maturity stage of apples (at harvest) since 1999 to determine the optimal picking date. Hines et al. [88] first tested a prototype e-nose equipped with four commercial tin- oxide gas sensors on “Golden Delicious” apples to accurately classify fruit ripeness. The same year, Young et al. [98] worked on Gala apples, discriminating the ripening stage at harvest in four different classes. Saevels et al. [95] used the Technobiochip (Elba Island, Italy) Lybra Nose for predicting the optimal harvest date of apples as well as the cultivar effect on aroma of “Jonagold” and “Braeburn” varieties. The e-nose used in this study contained a sensor array based on Quartz Crystal Microbalance (QCM) sensors coated with metalloporphyrins and related compounds. Data were collected for two years and the results yielded a good predictive model developed for each cultivar based on one year of data, but a similar model based on two years of data was less effective at predicting optimal harvest dates.
Another commercial electronic nose (Cyranose 320, Cyrano Sciences Inc., Pasadena, CA, USA) was employed by Pathange et al. [93] to assess apple maturity for the apple cultivar “Gala”. This instrument could positively classify the fruits into three groups according to their maturity stage (immature, mature and over mature) with an accuracy of 83%. Brezmes et al. [85–87] published several works on the assessment of apple ripeness. They initially worked on “Pink Lady” apples using a prototype e-nose equipped with 21 commercial tin oxide or Metal Oxide Semiconducting (MOS) sensors. The accuracy of e-nose classification of fruit maturity stage depended on the statistical classification technique used on e-nose data. PCA analysis did not show any clustering behavior that could be attributed to ripening, whereas a neural network classification algorithm provided good results. Brezmes et al. [85] could not determine the correct maturity stage of some apple cultivars, but had very good results on peaches and pears. More recently, Baumgartner et al. [84] showed very good results in discriminating the ripening stage of “Golden Delicious” apples, using a lesser-known Swiss e-nose (SMart Nose, SMart Nose SA, Marin-Epanier, Switzerland).
Of all commercial fruit species, apples rank highest with the largest number of experimental and commercial cultivars (including ancient, traditional and modern varieties) available in world markets. Apples have the greatest diversity of flesh and pericarp colors, flesh firmness, shelf life, and especially flavors and aromas. The cultivar effect, or variation in aroma characteristics or bouquet of apples, was studied by Marrazzo et al. [92] using a Cyrano Science prototype e-nose equipped with an array of 31 chemical sensors (Pasadena, CA, USA) to discriminate between “McIntosh”, “Delicious” and “Gala” apple varieties. This electronic-nose prototype discriminated between fruit-aroma classes (varieties) based on data from the first day after harvest when the cultivar effect on aroma was more intense for freshly-harvested fruits. A more difficult test was recently performed by Pruteanu et al. [94] who successfully employed a FOX 4000 e-nose to discriminate between seven different varieties of Romanian apples.
One of the most important quality features of any fruit species or variety is the duration or longevity that optimal fruit characteristics can be maintained, referred to as shelf life, prior to decline to an unsalable state. The ultimate goal is to develop fruit cultivars with a very low after-harvest perishability coefficient, indicating a low cull-rate following commercial display to consumers. The merchantability of fresh products is largely influenced by shelf life. Traditionally, shelf life is measured and assessed through an evaluation of the chemical and physical properties (factors) that most determine ripening, maturation and post-harvest deterioration. Some of the key parameters measured included color, soluble solids content (SSC), percent of sugar (brix), and titratable acidity (TA). All of these parameters are commonly used by researchers and industry fruit graders, but none of these parameters are utilized by or matched with criteria used in fruit selection from the consumer perspective. Thus, measuring and evaluating post-harvest perishability through one single parameter, changes in volatile compounds directly associated with aroma, is market-oriented. Herrmann et al. [89] probably was the first researcher to become interested in monitoring aroma of apples during shelf life. It was well known at that time that the ratio of aldehydes within apple headspace volatiles could be used as indicators of ripeness for many apple varieties. Consequently, a QCM prototype electronic nose containing sensors coated with aldehyde-sensitive materials was used to monitor the increase in trans-2-hexenal concentration as an indicator of post-harvest development over time. Saevels et al. [96] and Baumgartner et al. [84] monitored the post-harvest shelf life as well, obtaining very interesting good results, as did Guohua et al. [90] with “Fuji” apples, employing an electronic nose prototype equipped with an array of eight tin oxide MOS sensors.
Li et al. [91] examined the aroma bouquet of deteriorating apples during the shelf life period. Physical damage to apples dramatically reduced economic value due to changes in color, shape, flavor and aroma, as well as increased susceptibility to attack by various post-harvest pathogens. Results of this work demonstrate that differences in numbers of physical cuts to the fruits had effects on volatile compound emissions. Apples subjected to two and three cuts generated aroma bouquets significantly different from uncut fruits. A similar study was published by Di Natale et al. [129] who used the Technobiochip Lybranose QCM e-nose to successfully discriminate between no cuts, one and two cut fruits. Xiaobo et al. [97] utilized a more comprehensive approach by combining three different sensors (a near-infrared spectrophotometer, a machine vision system and an electronic nose) to classify “Fuji” apples according to several quality parameters.
Other researchers have focused on pears (Pyrus species) to assess the maturity stage at harvest and shelf-life. Oshita et al. [130] successfully classified “La France” pears into three groups according to three storage treatments applied after harvest to prolong the shelf-life by using an Aromascan conducting polymer (CP) e-nose. Zhang et al. [131,132], published similar papers on the prediction of acidity, soluble solids content and firmness of “Xuequing” pears by employing a prototype e-nose equipped with eight different MOS sensors and an artificial neural network to analyze their response. Finally, Li et al. [164] worked on a Chinese species of Pyrus (Pyrus ussuriensis Maxim.) to characterize its volatile organic compounds (VOCs) at different ripening stages by traditional methods compared to an electronic nose.
3.2. Peach and Apricot
Three research groups have worked on apricot (Prunus armeniaca L.), obtaining good results by the use of three different commercial electronic noses [99–101]. The cultivar effect on aroma bouquet was tested using a PEN2 electronic nose, a portable (AlphaMos, Schweirin, Germany) and light-weight sensing machine [100]. It consists of a sensor array composed of 10 different doped semi-conductive MOS sensors positioned in a small chamber. The signal of the electronic nose was statistically analyzed by a trained artificial neural network (ANN) which is a data-processing tool that mimics the structure of the biological neural system, exhibiting brain characteristics of learning. In association with gas chromatography-mass spectroscopy (GC-MS), a FOX 4000 e-nose was used in the second study to characterize and discriminate between eight different apricot cultivars with promising results. Non-destructive cultivar assessment is very important in particular for apricot fruits to classify unknown samples and to prevent adulterations.
Defilippi et al. [99] worked on post-harvest quality of apricots, assessed by changes in VOCs detected with an EOS 835 electronic nose (Sacmi scarl, Imola, Italy). In order to determine differences in aroma profile, apricots were harvested at two maturity stages and stored at 0 °C and 20 °C (shelf-life simulation) for 15 and 30 days, then analyzed by a trained panel test and electronic nose. This e-nose could not classify maturity level of cold samples, but could only be classified after simulated shelf-life and panel test.
Peaches, Prunus persica (L.) Batsch and P. persica var. nucipersica, have been thoroughly studied by researchers interested in finding new ways of characterizing fruits in a non-destructive way. The first report come from Brezmes et al. [85]. The initial approach was aimed at discriminating between cultivars in an attempt to assess the feasibility of a fast, non-destructive, and cheaper way to classify unknown samples and decrease food frauds [133,135–137,139,140].
Peaches, nectarines and most varieties of P. persica are climacteric and particularly perishable at harvest and during storage, requires that these fruits be maintained at 0 °C for only a few days because shelf-life of this species is particularly short. These are only some of the reasons why producers utilize many alternative methods to evaluate the maturity stage of the fruit directly on the tree, traditionally based on personal experience. The same thing is asked by industry and retailers who need non-destructive, fast and systematic methods to evaluate the shelf life of fruits and other perishable products. Most of the previously mentioned researchers, including Brezmes et al. [87] and Zhang et al. [142], have tried to test the feasibility of using an electronic nose to assess shelf life of fruits. Benedetti et al. [133] employed a PEN2 e-nose to successfully classify samples of four different cultivars of peach according to their ripening stage. Performing Principal Components Analysis (PCA) on sensor data, peaches showed a linear data distribution for PC1 (from right to left), with increasing days of shelf-life. They concluded that no more than three sensors had a high influence on the sensor-output pattern for the fruit aroma, but only one sensor was relevant in the discrimination of peaches on the basis of shelf-life. They interpreted this single-sensor response to indicate that the sensor signal was directly linked to ethylene production, responsible for ripeness of peaches. A similar response was published by Rizzolo et al. [138] by using a similar but more advanced electronic nose (PEN3) manufactured by the same Company. In this case, only three sensors seemed to be associated with fruit ripeness and a linear correlation between PCs and quality indices indicated PC1 was related to ethylene production as well. More recently, Guohua and colleagues developed a model for the prediction of peach freshness based on a home-made electronic nose [165].
Monitoring the sensorial qualities of stored (refrigerated) fruits, in particular the loss of flavor and aroma of peaches throughout the period between harvest and the arrival at retail stores and during transportation to far-away markets, is one of the main problems facing fresh-fruit exporting companies and producers. Electronic noses were successfully employed by Infante et al. [136] and Zhang et al. [143] to evaluate the development (or loss) of aroma during transportation to markets. In particular, Infante et al. [137] could discriminate between aroma qualities of four cultivars, showing that “Tardibelle” peaches have the highest quality attributes even after 42 days of cold storage following harvest.
Zhang et al. [141,142,166] attempted to establish a quality index model to describe the effects of different picking dates of peaches by the use of a self-made prototype e-nose equipped with eight commercial tin semiconductor sensors and a commercial acquisition card. Sensor responses were validated by traditional peach quality parameters such as firmness, sugar content and pH at three different picking times. With the aim of predicting fruit quality based on these parameters, Principal Component Regression (PCR) and Partial Least Squares (PLS) regression where applied. The results showed that the two methods allow the determination of firmness, sugar content and pH by use of electronic nose. A similar research was performed by Su et al. [140] who tried to assess harvest season and quality of 39 cultivars of peaches and nectarines. By using a FOX 4000 e-nose and manipulating data by Discriminant Function Analysis (DFA), they successfully linked sensor responses to total soluble solids (TSS) concentration and titratable acidity (TA) in all harvest seasons, but only for those samples with a low or high concentration of TSS and a low or high TSS/TA ratio. Infante et al. [137] employed the Italian EOS 835 (Sacmi, Imola) e-nose to predict the quality of four peach cultivars by applying a multiple linear regression (MLR) to sensors-response data. They were able to describe the quality attributes “acidity”, “sweetness” and the more general “acceptability” by the use of the e-nose, concluding that the instrument could discriminate between peach varieties through descriptors that mainly determine acceptability by the peach consumer. A similar paper was published by Di Natale et al. [134] who applied both electronic nose and panel sensor analysis to determine through an advanced data analysis, some proper sensorial indicators for the classification of peach fruits according to consumer palatability.
3.3. Citrus
Fruits belonging to the complex genus Citrus are commonly called citruses. These fruits are well known since ancient times for their nutraceutical properties, providing medical and health benefits,and the unmistakable pungent notes of their aroma. Hernandez Gomez et al. [119,121] worked on mandarin (C. reticulata Blanco) by using a PEN2 e-nose to associate sensor responses to harvest date (five different dates = aroma classes). No more than three sensors were employed in the Linear Discriminant Analysis (LDA), which gave clearer results than PCA. The later work by Hernandez Gomez et al. [120] was focused on evaluating the change in aroma bouquet emitted by mandarins during different storage treatments (plastic bag, paper box and refrigerator) and time (shelf-life), as well as other quality parameters. In this case, no significant predictive results were shown, while quality indices such as firmness were predicted by e-nose sensor responses.
Di Natale et al. [129] conducted research on oranges using a QCM prototype electronic nose. They distinguished between the different storage days (duration) for oranges, whereas Russo et al. [104] recently were able to discriminate between genuine bergamot (C. bergamia Risso et Poiteau) essential oils from other non-genuine types in order to defend the uniqueness of this very economically-important Southern Italian product. Akakabe et al. [149] worked on some Chinese species and varieties of citruses (including C. nagato-yusukichi Tanaka, C. sudachi Hort. ex Shirai, C. junos Siebold. ex Tanaka, C. sphaerocarpa Tanaka) to evaluate the capability of an electronic nose to discriminate between species and varieties by their aroma profiles.
3.4. Grape
One of the most important and widely-studied applications of electronic gas-sensing machines (electronic noses and tongues) in the food industry concerns analysis of the aroma characteristics of wine, the alcoholic product of fermented grapes [167–173]. However, some researchers have assessed the possibility of employing these gas-sensing machines directly on grapes in the field, or immediately after harvest, to provide important data on fruit quality and physiochemical parameters that are vital to the production success of quality wines and to limit food frauds or adulterations by dilutions with cheaper products. In this regard, Zoecklein et al. [114] studied the effects of ethanol treatments on volatile aromatic compounds emitted by two V. vinifera L. varieties (“Cabernet Franc” and “Merlot”) at the onset of ripening (veraison). Both a conducting polymer (CP) e-nose and a surface acoustic wave-based (SAW) e-nose were capable of successfully discriminating between treated and untreated fruits.
A very interesting paper was published by Devarajan et al. [115] who discovered that the canopy side (north versus south and east versus west) has an effect on the aroma bouquet emitted by fruits (and wine) of Cabernet franc. Data were processed by using two different electronic noses (a Cyranose 320 and a zNose 730) on the basis of two growing seasons. Both sensing systems provided effective discrimination of canopy sides for grapes VOCs using canonical discriminant analysis.
The maturity level at harvest over two seasons was evaluated by Athamneh et al. [110] using a portable Cyranose 320 e-nose on “Cabernet Sauvignon” grape samples picked at three different maturity stages. The instrument proved capable of discriminating between different stages of maturity and fruits from different vine canopy sides.
Post-harvest dehydration is one of the most important steps in the wine-making process. Although the volatile fraction of a wine can be formed by hundreds of chemically-different compounds, the aroma compounds formed during drying have significant effects on wine quality. Currently, winemakers judge optimum drying times in terms of sugar concentration (brix) or water loss rather than based on more precise continuous monitoring of aroma profiles by e-noses until optimum drying conditions are met. Thus, some researchers have tried to determine the optimum drying times for wines using an electronic nose to assess the quality of aroma bouquets derived from dehydrated grapes [111–113]. In all cases, very good results were obtained by the use of a QCM prototype electronic nose developed by the University of Rome Tor Vergata.
3.5. Strawberry and Other Berries
To our knowledge, only three papers have been published on different applications of electronic noses for the evaluation of aroma characteristics in strawberry (Fragaria x ananassa Duch.) fruits. Agulheiro-Santos [151] reported on the capability of an electronic nose to assess the influence of different nitrogen fertilizations on the aroma quality of “Camarosa” strawberry. This represents one of the few examples of scientific research into electronic-nose technologies being applied directly to evaluate and modify growth protocols and agronomic techniques for the improvement of fruit aromatic and flavor qualities.
Strawberry fruit maturity was evaluated using a MOS electronic nose containing 18-metal oxide gas sensors [152]. The instrument discriminated between five stages of fruit maturity (from white to overripe) and three picking dates for two varieties. A PEN2 e-nose was employed by Qiu et al. [153] to characterize the aroma of five strawberry varieties, derived from freshly-squeezed juice produced according to a squeezing-processing technique.
Blackberry (Rubus glaucus Benth), bilberry (Vaccinium meridionale Swartz) and blueberry (Vaccinium spp.), comprise a group of fruits called “berries” with peculiarities of shape and size (always round and small fruits), with colors-ranging from yellow to black-blue-and flavor (sweet or sour). This definition of berry is not linked to the botanical meaning of the term “berry”. Berries are among the most perishable fruits before and after harvesting. Thus, they have to be very carefully harvested and processed at the proper time and in specific ways to maintain quality and shelf life. Quality control in this field is particularly important. Simon et al. [82] tested a very early e-nose prototype, equipped with only two commercial sensors, to assess the maturity level of harvested fruits based on aromatic profile, and to detect damaged fruits in a closed container. The nutraceutical role of blackberries and bilberries was assessed by Bernal et al. [105]. In addition, changes in volatile components during different stages of fruit maturity were evaluated by employing a PEN3 electronic nose [106].
3.6. Mango and Other Tropical Fruits
Among so-called “tropical fruits”, mango (Mangifera indica L.) is the most studied species in the field of gas-sensing e-nose machines. The first report by Lebrun et al. [123] attempted to assess the optimal harvest date. Whole and homogenated mango fruits were sampled and the aroma from each fruit was analyzed using a FOX 4000 e-nose. Although a deep study on the optimal dilution of the sample was carried out, this e-nose was not effective in determining the harvest date. Similar mediocre results were obtained by Kitthawee et al. [122], involving a study of hard green mangoes. The electronic nose could correctly classify only 68% of fruits according to their ripening stage, too low to be introduced into the retail market as a non-destructive method to assess ripening stage in mangoes. Afterwards, several researchers pursued the same goal some years later. Lebrun et al. [124] worked on three different mango cultivars (‘Cogshall’, ‘Kent’ and ‘Keitt’) harvested at different fruit maturities. This e-nose could separate fruits from different picking dates as well as fruits from different varieties. Zakaria et al. [125] published similar good results employing a Cyranose 320 e-nose.
A pioneering work by Llobet et al. [102], utilizing a self-made electronic prototype equipped with a tin-oxide MOS commercial sensor array, was able to discriminate between different stages of fruit maturity in banana (Musa x paradisiaca L.) fruits, and also predict the maturity stage of unknown samples, applying a neural-net classifier.
The Italian research group of Torri et al. [146] worked on pineapple (Ananas comosus L. Merr.) fruits. One experiment was aimed at monitoring the freshness of minimally-processed slices of pineapple during storage by use of a PEN2 electronic nose. Results indicated that this e-nose was successfully employed in the field to determine that pineapple fruit is particularly perishable and that even minimally-processed pineapple fruits lose their aroma characteristics very quickly.
Other researchers have evaluated the maturity stage for harvesting tropical fruits. Supriyadi et al. [148] employed a FOX 4000 e-nose to discriminate between ripe and unripe snake fruits (Salacca edulis Reinw.). Pokhum et al. [109] identified the ripeness stage of durian fruits (Durio spp.) by use of a MOS e-nose. Márquez Cardozo et al. [150] worked on the Columbian exotic and highly perishable soursop fruit (Annona muricata L.) a member of the custard apple tree family (Anonaceae). In this case, a PEN3 e-nose easily classified fruit samples as unripe, half ripe, ripe or overripe. Nugroho et al. [147] recently utilized an array of four commercial tin-semiconductor sensors, their prototype electronic nose, to discriminate between different maturity-classes sapodilla (Manilkara zapota (L.) P. Royen) fruits.
3.7. Other Miscellaneous Fruits
A paper by Lebrun et al. [108] hitherto has been the only research study to report on the application of an electronic nose for the rapid and non-destructive discrimination of date (Phoenix dactylifera L.) varieties. The research group of Alasalvar worked successfully on five raw and eighteen roasted Turkish hazelnuts (Corylus avellana L.) in an attempt at characterizing differences in aroma bouquet according to variety [116,117].
Garcia-Breijo et al. [144] and Li et al. [145] investigated persimmon (Diospyros kaki L.) fruits to discriminate between two different cultivars using a semiconductor commercial e-nose sensor array to assess fruit ripening stage and storage life, applying PCA and LDA statistical methods to a PEN3 e-nose sensor output data. They also attempted to determine the fewest number of sensors that could explain all the variance. Guarrasi et al. [118,174] worked on loquats [Eriobotrya japonica (Thunb.) Lindl.], a plant belonging to the Rosaceae family native to Japan and China and also wide spread in the Mediterranean Regions of Italy. The fruits were characterized chemically (SSC, TA, pH), morphologically, electronically using an e-nose, and olfactorily (via a trained human panel test). Although cross-data from traditional instrumental techniques and e-nose could identify aroma features, neither the panel test nor the electronic nose could discriminate between the four different sample cultivars.
Very recently, a paper by Ghouhua et al. [175] reported about a quality forecasting method using a home-made electronic nose based on an eight MOS sensors array. Samples of Winter jujube (Ziziphus jujuba Mill.) were analyzed each day, for 8 days, for physical and chemical indexes as well as via EN. PCA results indicated that jujubes under different time had an approximate trend, but the samples could not be qualitative or quantitatively discriminated from each other.
3.8. Vegetable Fruits
From a botanic point of view, tomatoes and other vegetables are actually fruits because they are derived from the ripened ovary of a flower [176]. Nevertheless, the general public views these fruits as vegetables especially from a culinary point of view. Quality attributes like the perfect maturity stage at harvest, long shelf-life and attractive visual appearance are critical factors that must be taken into account when evaluating agricultural protocols. Within all vegetable-type fruits, tomato (Solanum lycopersicon L.) is the species receiving the most attention in scientific research efforts in the field of electronic sensing since 1997. Maul et al. [160,161] published some early results on the evaluation of an electronic nose to identify and discriminate tomatoes, exposed to different harvesting and postharvest handling treatments, in order to measure quality diversities (in flavor, aroma and other quality parameters) between physiological (portable) maturity and market maturity, highly influenced by long-distance handling (during transportation) and even more by marketing systems.
Sinesio et al. [162] employed a prototype electronic nose and a panel test to discriminate between fruit samples based on different qualities of tomatoes. Fruits were harvested from two different Italian farms (one of them conducting traditional agriculture, the other organic farming) and classified as “very good”, “good”, “fair” and “poor”, according to visual selection for the presence of injuries and physical damage. A QCM electronic nose was used, containing eight sensors coated with different metalloporphyrines. Their data showed that through the use of a neural-net statistical algorithm, the e-nose could discriminate between classes better than a trained human panel test.
Electronic noses can actually discriminate between different levels of mechanical damage in fruits like tomatoes which apparently release different VOCs according to different degrees of damage [156]. Bruising fruits usually lead to enhanced ripening at a rate proportion to the amount of damage.
Shelf life of tomatoes was assessed using electronic noses by Berna et al. [154,155] and by Hernandez Gomez et al. [158] in 2008. In both cases utilizing three different e-noses (a Technobiochip Lybranose, a University of Rome “Tor Vergata” enQbe, and a AlphaMos PEN2), a clear separation of different aroma classes was not achieved, whereas very good results were obtained by Hernandez Gomez et al. [157] in discriminating between different fruit maturity stages at harvest. Wang and Zhou [163] also obtained good results by crossing PCA sensor data with firmness data.
Hong and Wang [159] recently found that it was relatively simple to identify high quality fruits visually at harvest, but it is very difficult to assess the freshness of squeezed cherry tomatoes (in 100% tomato juices) unless traditional and time-consuming instruments are employed. Unfortunately, their electronic nose system did not give very convincing results.
Abbey et al. [126,127] and Russo et al. [128] worked on bulbs of Allium ssp. which are not named “fruits” either from a botanical or culinary point of view. We report these studies here because these species are somehow used as “fleshy” vegetables in kitchens. The initial work on Allium species was aimed at assessing the ability of an electronic nose to discriminate between different species (A. sativum L., A. ampeloprasum var. porrum, A. cepa var. aggregatum, and A. cepa L.) on the basis of their aroma profile. Based on pyruvic acid and thiosulphinates content (which characterize these species and varieties), the e-nose could successfully discriminate between them. In a secondary experiment, an electronic nose was employed to evaluate the effects of some agronomic factors, such as fertilizations with nitrogen (N) and sulfur (S) as well as the soil effects on plant aroma characteristics in the field and greenhouse. This is an interesting application of an e-nose instrument devoted to classifying different aroma bouquets in agricultural products whose final quality is so influenced by aroma profiles, such as for white pepper (Piper nigrum L.). Mamatha and Prakash [177] could easily discriminate between three cultivars of pepper, whereas a recent work of Liu et al. [143] was devoted to examining flavor quality of five new pepper genotypes. In all cases, the α-Gemini (Alpha MOS SA, Toulouse, France) e-nose, equipped with an array of 6 MOS sensors that can be chosen and customized by users, was useful in identifying known and unknown samples. Another example of this application in which an e-nose was used to identify unknown genotypes of fruit vegetables was published by Zawirska-Wojtasiak et al. [107]. Theyattempted to use the electronic nose to differentiate between transgenic lines of cucumber (Cucumis sativus L.) compared to controls.
The protection of specific geographical labels (as sources of fruits) from frauds and adulterations is one of the main concerns of producers, industry and final users as well as those who want to be extremely sure about the quality and the exact origin of fruits used in their businesses. Italy has the highest number of European protected denominations concerning fresh products [103]. An electronic nose was used to identify the geographical origins of two local varieties of bell pepper (Capsicum annuum L.) and this e-nose was applied in association with other traditional methods to characterize the product according to morphometric, qualitative, spectroscopic and aromatic data.
4. Conclusions
Electronic-nose devices have been utilized in a wide range of diverse applications in the agriculture and forestry industries to improve the effectiveness, efficiency and safety of processes involved in the production of quality food and fiber plant-based products [5]. As summarized in this review, e-nose instruments also offer many new potential applications for the fruit-production industry to facilitate many tasks involving fruit aroma evaluations during all stages of the agro-fruit production process from early cultivation activities, field-applied pest control applications, and timing of fruit harvests to many post-harvest stages including fruit transportation, storage, and finally the maintenance of fruit shelf life during display in commercial markets.
The potential for future developments and new aroma-based applications of electronic-nose devices in fruit production processes include e-nose detection of pesticide residues on harvested fruit surfaces to facilitate enforcement of human health regulations of the Environmental Protection Agency (EPA) [178–180], post-harvest fruit disease detection and management [5,8], and monitoring gases released from fruits in storage to control fruit ripening (maintain fruit shelf life) and fruit quality.
Acknowledgments
The authors would like to thank Gonzalo Pajares Martinsanz (University Complutense of Madrid, Spain), and Pablo Gonzalez-de-Santos (Centre for Automation and Robotics, Arganda del Rey, Madrid, Spain) for the invitation to write this review article on the applications of electronic-nose technologies for fruit identification, discrimination, ripeness and quality grading for the new Sensors special issue “Agriculture and Forestry: Sensors, Technologies and Procedures”. We also appreciate the assistance of Charisse Oberle who helped compile and check the references and provided useful edits for the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ogundiwin, E.A.; Peace, C.P.; Gradziel, T.M.; Parfitt, D.E.; Bliss, F.A.I.; Crisosto, C.H. A. Fruit quality gene map of Prunus. BMC Genomics 2009, 10. [Google Scholar] [CrossRef]
- Sanz, C.; Olias, J.M.; Perez, A.G. Aroma biochemistry of fruits and vegetables. In Phytochemistry of Fruit and Vegetables; Oxford University Press Inc: New York, NY, USA, 1997; pp. 125–155. [Google Scholar]
- Schwab, W.; Davidovich-Rikanati, R.; Lewinsohn, E. Biosynthesis of plant-derived flavor compounds. Plant J. 2008, 54, 712–732. [Google Scholar]
- Eng, T.G.; Tat, M.M. Quality control in fruit processing. JAOCS 1985, 62, 287–291. [Google Scholar]
- Wilson, A.D. Diverse applications of electronic-nose technologies in agriculture and forestry. Sensors 2013, 13, 2295–2348. [Google Scholar]
- Wilson, A.D.; Oberle, C.S.; Oberle, D.F. Detection of off-flavor in catfish using a conducting polymer electronic-nose technology. Sensors 2013, 13, 15968–15984. [Google Scholar]
- Wilson, A.D.; Baietto, M. Applications and advances in electronic-nose technologies. Sensors 2009, 9, 5099–5148. [Google Scholar]
- Wilson, A.D.; Lester, D.G.; Oberle, C.S. Development of conductive polymer analysis for the rapid detection and identification of phytopathogenic microbes. Phytopathology 2004, 94, 419–431. [Google Scholar]
- Wilson, A.D.; Lester, D.G.; Oberle, C.S. Application of conductive polymer analysis for wood and woody plant identifications. For. Ecol. Managem. 2005, 209, 207–224. [Google Scholar]
- Tucker, G.A. Introduction. In Biochemistry of Fruit Ripening; Seymour, G.B., Taylo, R.J.E., Tucker, G.A., Eds.; Chapman & Hall: London, UK, 1993; pp. 1–51. [Google Scholar]
- Ortiz, A.; Graell, J.; Lara, I. Volatile ester-synthesising capacity throughout on-tree maturation of “Golden Reinders” apples. Sci. Hortic. 2011, 131, 6–14. [Google Scholar]
- Ortiz, A.; Graell, J.; López, M.L.; Echeverría, G.; Lara, I. Volatile ester-synthesising capacity in “Tardibelle” peach fruit in response to controlled atmosphere and 1-MCP treatment. Food Chem. 2010, 123, 698–704. [Google Scholar]
- El Hadi, M.A.M.; Zhang, F.-J.; Wu, F.-F.; Zhou, C.-H.; Tao, J. Advances in fruit aroma volatile research. Molecules 2013, 18, 8200–8229. [Google Scholar]
- Dudareva, N.; Pichersky, E.; Gershenzon, J. Biochemistry of plant volatiles. Plant Physiol. 2004, 135, 1893–1902. [Google Scholar]
- Portnoy, V.; Benyamini, Y.; Bar, E. The molecular and biochemical basis for varietal variation in sesquiterpene content in melon (Cucumis melo L.) rinds. Plant Mol. Biol. 2008, 66, 647–661. [Google Scholar]
- Burger, Y.; Sáar, U.; Paris, H.S.; Lewinsohn, E.; Katzir, N.; Tadmor, Y.; Schaffer, A.A. Genetic variability for valuable fruit quality traits in Cucumis melo. Israel J. Plant Sci. 2006, 54, 233–242. [Google Scholar]
- Jetti, R.R.; Yang, E.; Kurnianta, A.; Finn, C.; Qian, M.C. Quantification of selected aroma-active compounds in strawberries by headspace solid-phase microextraction gas chromatography and correlation with sensory descriptive analysis. J. Food Sci. 2007, 72, S487–S496. [Google Scholar]
- Berger, R.G. Flavours and Fragrances-Chemistry, Bioprocessing and Sustainability; Springer-Verlag: Berlin, Germany, 2007. [Google Scholar]
- Holland, D.; Larkov, O.; Bar-Yaákov, I.; Bar, E.; Zax, A.; Brandeis, E. Developmental and varietal differences in volatile ester formation and acetyl-CoA: Alcohol acetyl transferase activities in apple (Malus domestica Borkh.) fruit. J. Agric. Food Chem. 2005, 53, 7198–7203. [Google Scholar]
- Villatoro, C.; Altisent, R.; Echeverria, G.; Graell, J.; Lopez, M.L.; Lara, I. Changes in biosynthesis of aroma volatile compounds during on-tree maturation of “Pink Lady” apples. Postharvest Biol. Technol. 2008, 47, 286–295. [Google Scholar]
- González-Agüero, M.; Troncoso, S.; Gudenschwager, O.; Campos-Vargas, R.; Moya-León, M.A.; Defilippi, B.G. Differential expression levels of aroma-related genes during ripening of apricot (Prunus armeniaca L.). Plant Physiol. Biochem. 2009, 47, 435–440. [Google Scholar]
- Aubert, C.; Chanforan, C. Postharvest changes in physicochemical properties and volatile constituents of apricot (Prunus armeniaca L.). Characterization of 28 cultivars. J. Agric. Food Chem. 2007, 55, 3074–3082. [Google Scholar]
- Wendakoon, S.K.; Ueda, Y.; Imahori, Y.; Ishimaru, M. Effect of short-term anaerobic conditions on the production of volatiles, activity of alcohol acetyltransferase and other quality traits of ripened bananas. J. Sci. Food Agric. 2006, 86, 1475–1480. [Google Scholar]
- Bouwmeester, H.J.; Gershenzon, J.; Konings, M.C.J.M.; Croteau, R. Biosynthesis of the monoterpenes limonene and carvone in the fruit of caraway—I. Demonstration of enzyme activities and their changes with development. Plant Physiol. 1998, 117, 901–912. [Google Scholar]
- Bouwmeester, H.J.; Konings, M.C.J.M.; Gershenzon, J.; Karp, F.; Croteau, R. Cytochrome P-450 dependent (+)-limonene-6-hydroxylation in fruits of caraway (Carum carvi). Phytochemistry 1999, 50, 243–248. [Google Scholar]
- Song, J.; Forney, C.F. Flavour volatile production and regulation in fruit. Can. J. Plant Sci. 2008, 88, 537–550. [Google Scholar]
- Carcia, C.V.; Stevenson, R.J.; Atkinson, R.G.; Winz, R.A.; Yong Quik, S. Changes in the bound aroma profiles of “Hayward” and “Hort16A” kiwifruit (Actinidia spp.) during ripening and GC-olfactometry analysis. Food Chem. 2013, 137, 45–54. [Google Scholar]
- Tressl, R.; Albrecht, W. Biogenesis of aroma compounds through acyl pathways. In Biogeneration of Aromas; Parliament, T.H., Croteau, R., Eds.; ACS: Washington, DC, USA, 1986; pp. 114–133. [Google Scholar]
- Eduardo, I.; Chietera, G.; Bassi, D.; Rossini, L.; Vecchietti, A. Identification of key odor volatile compounds in the essential oil of nine peach accessions. J. Sci. Food Agric. 2010, 90, 1146–1154. [Google Scholar]
- Tokitomo, Y.; Steinhaus, M.; Buttner, A.; Schieberle, P. Odor-active constituents in fresh pineapple (Ananas comosus [L.] Merr.) by quantitative and sensory evaluation. Biosci. Biotechnol. Biochem. 2005, 69, 1323–1330. [Google Scholar]
- Klesk, K.; Qian, M.; Martin, R. Aroma extract dilution analysis of cv. Meeker (Rubus idaeus L.) red raspberries from Oregon and Washington. J. Agric. Food Chem. 2004, 52, 5155–5161. [Google Scholar]
- Malowicki, S.M.; Martin, R.; Qian, M.C. Volatile composition in raspberry cultivars grown in the Pacific Northwest determined by stir bar sorptive extraction-gas chromatography-mass spectrometry. J. Agric. Food Chem. 2008, 56, 4128–4133. [Google Scholar]
- Auldridge, M.E.; McCarty, D.R.; Klee, H.J. Plant carotenoid cleavage oxygenases and their apocarotenoid products. Curr. Opin. Plant Biol. 2006, 9, 315–321. [Google Scholar]
- Song, J.; Bangerth, F. Fatty acids as precursors for aroma volatile biosynthesis in pre-climacteric and climacteric apple fruit. Postharvest Biol. Technol. 2003, 30, 113–121. [Google Scholar]
- Schwab, W.; Schreier, P. Enzymic formation of flavor volatiles from lipids. In Lipid Biotechnology; Kuo, T.M., Gardner, H.W., Eds.; Marcel Dekker: New York, NY, USA, 2002; pp. 293–318. [Google Scholar]
- Newman, J.D.; Chappell, J. Isoprenoid biosynthesis in plants: Carbon partitioning within the cytoplasmic pathway. Crit. Rev. Biochem. Mol. Biol. 1999, 34, 95–106. [Google Scholar]
- Lichtenthaler, H.K. The 1-deoxy-D-xylulose-5-phosphate pathway of isoprenoid biosynthesis in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 47–65. [Google Scholar]
- Boudhrioua, N.; Giampaoli, P.; Bonazzi, C. Changes in aromatic components of banana during ripening and air-drying. Lebensm Wiss Technol. 2003, 36, 633–642. [Google Scholar]
- Aurore, G.; Ginies, C.; Ganou-Parfait, B.; Renard, C.M.G.C.; Fahrasmane, L. Comparative study of free and glycoconjugated volatile compounds of three banana cultivars from French West Indies: Cavendish, Frayssinette and Plantain. Food Chem. 2011, 129, 28–34. [Google Scholar]
- Jayanty, S.; Song, J.; Rubinstein, N.M.; Chong, A.; Beaudry, R.M. Temporal relationship between ester biosynthesis and ripening events in bananas. J. Am. Soc. Hort. Sci. 2002, 127, 998–1005. [Google Scholar]
- Turemis, N.; Kafkas, E.; Kafkas, S.; Kurkcuoglu, M.; Baser, K.H.C. Determination of aroma compounds in blackberry by GC/MS analysis. Chem. Nat. Comp. 2003, 39, 174–176. [Google Scholar]
- Du, X.F.; Finn, C.E.; Qian, M.C. Volatile composition and odour-activity value of thornless “Black Diamond” and “Marion” blackberries. Food Chem. 2010, 119, 1127–1134. [Google Scholar]
- Du, X.F.; Plotto, A.; Song, M.; Olmstead, J.; Rouseff, R. Blueberry volatile composition of four southern highbush cultivars and effect of growing location and harvest date. J. Agric. Food Chem. 2011, 59, 8347–8357. [Google Scholar]
- Rosillo, L.; Salinas, M.R.; Garijo, J.; Alonso, G.L. Study of volatiles in grapes by dynamic headspace analysis Application to the differentiation of some Vitis vinifera varieties. J. Chromatogr. A 1999, 847, 155–159. [Google Scholar]
- Bellincontro, A.; Nicoletti, I.; Valentini, M.; Tomas, A.; de Santis, D.; Corradini, D.; Mencarelli, F. Integration of nondestructive techniques with destructive analyses to study postharvest water stress of winegrapes. Am. J. Enol. Vitic. 2009, 60, 57–65. [Google Scholar]
- Fenoll, J.; Manso, A.; Hellin, P.; Ruiz, L.; Flores, P. Changes in the aromatic composition of the Vitis vinifera grape Muscat Hamburg during ripening. Food Chem. 2009, 114, 420–428. [Google Scholar]
- Narain, N.; Bora, P.S.; Narain, R.; Shaw, P.E. Mango. In Tropical and Subtropical Fruits; Shaw, P.E., Chan, H.T., Nagy, S., Eds.; AgScience: Auburndale, FL, USA, 1997; pp. 1–77. [Google Scholar]
- Chang, B.W.; Sheng, H.L.; Yu, G.L.; Ling, L.L.; Wen, X.Y.; Guang, M.S. Characteristic aroma compounds from different pineapple parts. Molecules 2011, 16, 5104–5112. [Google Scholar]
- Zhang, X.M.; Du, L.Q.; Sun, G.M.; Wei, C.B.; Liu, S.H.; Xie, J.H. Changes of aroma components in Yellow Mauritius pineapple during fruit development (In Chinese). J. Fruit Sci. 2009, 26, 245–249. [Google Scholar]
- Liang, Y.Z.; Guang, M.S.; Yu, G.L.; Ling, L.L.; Wen, X.Y.; Wei, F.Z.; Chang, B.W. Aroma volatile compounds from two fresh pineapple varieties in China. Int. J. Mol. Sci. 2012, 13, 7383–7392. [Google Scholar]
- Nijssen, L.M.; van Ingen-Visscher, C.A.; Donders, J.J.H. VCF Volatile Compounds in Food: Database (Version 13.1.); TNO Triskelion Recuperato da: Zeist, The Netherlands, 2011. [Google Scholar]
- Dixon, J.; Hewett, E.W. Factors affecting apple aroma/flavour volatile concentration: A review. N. Z. J. Crop Hortic. Sci. 2000, 28, 155–173. [Google Scholar]
- Rudell, D.R.; Mattinson, D.S.; Mattheis, J.P.; Wyllie, S.G.; Fellman, J.K. Investigations of aroma volatile biosynthesis under anoxic conditions and in different tissues of “Redchief Dilicious” apple fruit (Malus domestica Borkh.). J. Agric. Food Chem. 2002, 50, 2627–2632. [Google Scholar]
- Guadagni, D.G.; Bomben, J.L.; Hudson, J.S. Factors influencing the development of aroma in apple peel. J. Sci. Food Agric. 1971, 22, 110–115. [Google Scholar]
- Defilippi, B.G.; Dandekar, A.M.; Kader, A.A. Relationship of ethylene biosynthesis to volatile production, related enzymes and precursor availability in apple peel and flesh tissues. J. Agric. Food Chem. 2005, 53, 3133–3141. [Google Scholar]
- Aubert, C.; Milhet, C. Distribution of the volatile compounds in the different parts of a white-fleshed peach (Prunus persica L. Batsch). Food Chem. 2007, 102, 375–384. [Google Scholar]
- Wang, Y.J.; Yang, C.X.; Li, S.H.; Yang, L.; Wang, Y.N.; Zhao, J.B.; Jiang, Q. Volatile characteristics of 50 peaches and nectarines evaluated by HP-SPME with GC-MS. Food Chem. 2009, 116, 356–364. [Google Scholar]
- Aubert, C.; Gunata, Z.; Ambid, C.; Baumes, R. Changes in physicochemical characteristics and volatile constituents of yellow- and white-fleshed nectarines during maturation and artificial ripening. J. Agric. Food Chem. 2003, 51, 3083–3091. [Google Scholar]
- Cheng, L.; Xiao, L.; Chen, W.; Wang, J.; Che, F.; Wu, B. Identification of compounds characterizing the aroma of oblate-peach fruit during storage by GC-MS. J. Stored Prod. Postharv. Res. 2012, 3, 54–62. [Google Scholar]
- Zhang, B.; Shen, J.Y.; Wei, W.W.; Xi, W.P.; Xu, C.J.; Ferguson, I.; Chen, K.S. Expression of genes associated with aroma formation derived from the fatty acid pathway during peach fruit ripening. J. Agric. Food Chem. 2010, 58, 6157–6165. [Google Scholar]
- Zhang, B.; Xi, W.P.; Wei, W.W.; Shen, J.Y.; Ferguson, I.; Chen, K.S. Changes in aroma-related volatiles and gene expression during low temperature storage and subsequent shelf-life of peach fruit. Postharvest Biol. Technol. 2011, 60, 7–16. [Google Scholar]
- Rapparini, F.; Predieri, S. Pear fruit volatiles. In Horticultural Reviews; Janick, J., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2003; pp. 237–324. [Google Scholar]
- Kahle, K.; Preston, C.; Richling, E.; Heckel, F.; Schreier, P. On-line gas chromatography combustion/pyrolysis isotope ratio mass spectrometry (HRGC-C/P-IRMS) of major volatiles from pear fruit (Pyrus communis) and pear products. Food Chem. 2005, 91, 449–455. [Google Scholar]
- Nijssen, L.M.; Visscher, C.A.; Maarse, H.; Willemsens, L.C.; Boelens, M.H. Volatile Compounds in Foods Qualitative and Quantitative Data; TNO Nutrition and Food Research Institute: Zeist, The Netherlands, 2007; pp. 15–16. [Google Scholar]
- Gonza'lez-Mas, M.C.; Rambla, J.L.; Alamar, M.C.; Antonio, A.G. Comparative analysis of the volatile fraction of fruit juice from different Citrus species. PLoS One, 2011, 6, p. e22016. Available online: http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0022016 (accessed on 15 November 2014). [Google Scholar]
- Kelebek, H.; Selli, S. Determination of volatile, phenolic, organic acid and sugar components in a Turkish cv. Dortyol (Citrus sinensis L. Osbeck) orange juice. J. Sci. Food Agric. 2011, 91, 1855–1862. [Google Scholar]
- Tomiyama, K.; Aoki, H.; Oikawa, T.; Sakurai, K.; Kashara, Y.; Kawakami, Y. Characteristic volatile components of Japanese sour citrus fruits: Yuzu, Sudachi and Kabosu. Flavour Fragr. J. 2012, 27, 341–355. [Google Scholar]
- Phi, N.T.L.; Nishiyama, C.; Choi, H.S.; Sawamura, M. Evaluation of characteristic aroma compounds of Citrus natsudaidai Hayata (Natsudaidai) cold-pressed peel oil. Biosci. Biotechnol. Biochem. 2006, 70, 1832–1838. [Google Scholar]
- Obando-Ulloa, J.M.; Moreno, E.; Garc ıa-Mas, J.; Nicolai, B.; Lammertync, J.; Monforte, J.A.; Fernandez-Trujillo, J.P. Climacteric or non-climacteric behavior in melon fruit 1. Aroma volatiles. Postharvest Biol. Technol. 2008, 49, 27–37. [Google Scholar]
- Perry, P.L.; Wang, Y.; Lin, J.M. Analysis of honeydew melon (Cucumis melo var. Inodorus) flavor and GC/MS identification of (E, Z)-2,6-nonadienyl acetate. Flav. Frag. J. 2009, 24, 341–347. [Google Scholar]
- Beaulieu, J.C.; Grimm, C.C. Identification of volatile compounds in cantaloupe at various developmental stages using solid phase microextraction. J. Agric. Food Chem. 2001, 49, 1345–1352. [Google Scholar]
- Jordán, M.J.; Shaw, P.E.; Goodner, K.L. Volatile components in aqueous essence and fresh fruit of Cucumis melo cv. Athena (muskmelon) by GC-MS and GC-O. J. Agric. Food Chem. 2001, 49, 5929–5933. [Google Scholar]
- Dieguez, S.C.; Lois, L.C.; Gomez, E.F.; de Ia Pena, M.L.G. Aromatic composition of the Vitis vinifera grape Albariño. LWT-Food Sci. Technol. 2003, 36, 585–590. [Google Scholar]
- Shibamoto, T.; Tang, C.S. “Minor” tropical fruit mango, papaya, passion fruit, and guava. In Food Flavours: Part C: The Flavour of Fruit; Morton, I.D., MacLeod, A.J., Eds.; Elsevier: Amsterdam, The Netherlands, 1990; pp. 221–234. [Google Scholar]
- Pino, J.A.; Mesa, J. Contribution of volatile compounds to mango (Mangifera indica L.) aroma. Flav. Frag. J. 2006, 21, 207–213. [Google Scholar]
- Quijano, C.E.; Salamanca, G.; Jorge, A.; Pino, J.A. Aroma volatile constituents of Colombian varieties of mango (Mangifera indica L.). Flav. Frag. J. 2007, 22, 401–406. [Google Scholar]
- Goswami, T.K.; Rekha, R.M. Determination of aroma volatiles in mango fruit cv. Amrapali as affected during storage under rapid control atmosphere conditions using liquid nitrogen. Int. J. Agric. Environm. Biotech. 2013, 6, 471–478. [Google Scholar]
- Bood, K.G.; Zabetakis, I. The biosynthesis of strawberry flavor (II): Biosynthetic and molecular biology studies. J. Food Sci. 2002, 67, 2–8. [Google Scholar]
- Wyllie, S.G.; Leach, D.N.; Wang, Y.; Shewfelt, R.L. Key aroma compounds in melons: Their development and cultivar dependence. In Fruit Flavors: Biogenesis, Characterization, and Authentication; Rouseff, R.L., Leahy, M.M., Eds.; American Chemical Society: Washington, DC, USA, 1995; pp. 248–257. [Google Scholar]
- Yoshii, F.; Yamada, Y.; Hoshi, T.; Hagiwara, H. The creation of a database of odorous compounds focused on molecular rigidity and analysis of the molecular features of the compounds in the database. Chem. Sens. 2002, 27, 399–405. [Google Scholar]
- Benady, M.; Simon, J.E.; Charles, D.J.; Miles, G.E. Fruit ripeness determination by electronic sensing or aromatic volatiles. Trans. ASAE 1995, 38, 251–257. [Google Scholar]
- Simon, J.E.; Herzroni, A.; Bordelon, B.; Miles, G.E.; Charles, D.J. Electronic sensing of aromatic volatiles for quality sorting of blueberries. J. Food Sci. 1996, 61, 967–970. [Google Scholar]
- Bai, J.; Baldwin, E.A.; Fortuny, R.C.S.; Mattheis, J.P.; Stanley, R.; Perera, C.; Brecht, J.K. Effect of pretreatment of intact ‘Gala’ apple with ethanol vapor, hear, or 1-methylcyclopropene on quality and shelf life of fresh-cut slices. J. Am. Soc. Hortic. Sci. 2004, 129, 583–593. [Google Scholar]
- Baumgartner, D.; Gabioud, S.; Hohn, E.; Gasser, F.; Nising, A.B. Messung der Aromaentwicklung wahrend der Reifung von Golden Delicious. Schweiz. Z. Für Obst- Weinbau. 2009, 145, 8–11. [Google Scholar]
- Brezmes, J.; Llobet, E.; Vilanova, X.; Saiz, G.; Correig, X. Fruit ripeness monitoring using and electronic nose. Sens. Actuators B Chem. 2000, 69, 223–229. [Google Scholar]
- Brezmes, J.; Llobet, E.; Vilanova, X.; Orts, J.; Saiz, G.; Correig, X. Correlation between electronic nose signals and fruit quality indicators of shelf-life measurements with pink lady apples. Sens. Actuators B Chem. 2001, 80, 41–50. [Google Scholar]
- Brezmes, J.; Fructuoso, L.; Llobet, E.; Vilanova, X.; Recasens, I.; Orts, J.; Saiz, G.; Correig, X. Evaluation of an electronic nose to assess fruit ripeness. IEEE Sens. J. 2005, 5, 97–108. [Google Scholar]
- Hines, E.L.; Llobet, E.; Gardner, J.W. Neural network based electronic nose for apple ripeness determination. Electron. Lett. 1999, 35, 821–823. [Google Scholar]
- Herrmann, U.; Jonischkeit, T.; Bargon, J.; Hahn, U.; Li, Q.; Schalley, C.A.; Vogel, E.; Vogtle, F. Monitoring apple flavor by use of quartz microbalances. Anal. Bioanal. Chem. 2002, 372, 611–614. [Google Scholar]
- Hui, G.H.; Wu, Y.L.; Ye, D.D.; Ding, W.W. Fuji apple storage time predictive method using electronic nose. Food Anal. Method. 2013, 6, 82–88. [Google Scholar]
- Li, C.; Heinemann, P.H.; Irudayaraj, J. Detection of apple deterioration using an electronic nose and zNose™. Trans. ASABE 2007, 50, 1417–1425. [Google Scholar]
- Marrazzo, W.N.; Heinemann, P.H.; Crassweller, R.E.; Leblanc, E. Electronic nose chemical sensor feasibility study for the differentiation of apple cultivars. Trans. ASAE 2005, 48, 1995–2002. [Google Scholar]
- Pathange, L.P.; Mallikarjunan, P.; Marini, R.P.; O'Keefe, S.; Vaughan, D. Non-destructive evaluation of apple maturity using an electronic nose system. J. Food Eng. 2006, 77, 1018–1023. [Google Scholar]
- Pruteanu, E.M.; Duta, D.E.; Hincu, F.A.; Calu, M. Electronic nose for discrimination of Romanian apples. Lucr. Stiintifice 2009, SV, 398–404. [Google Scholar]
- Saevels, S.; Lammertyn, J.; Berna, A.Z.; Veraverbeke, E.A.; Di Natale, C.; Nicolai, B.M. Electronic nose as a non-destructive tool to evaluate the optimal harvest date of apples. Postharvest Biol. Technol. 2003, 30, 3–14. [Google Scholar]
- Saevels, S.; Lammertyn, J.; Berna, A.Z.; Veraverbeke, E.A.; di Natale, C.; Nicolai, B.M. An electronic nose and a mass spectrometry-based electronic nose for assessing apple quality during shelf life. Postharvest Biol. Technol. 2004, 31, 9–19. [Google Scholar]
- Zou, X.B.; Zhao, J.W.; Li, Y.X. Objective quality assessment of apples using machine vision, NIR spectrophotometer, and electronic nose. Trans. ASABE 2010, 53, 1351–1358. [Google Scholar]
- Young, H.; Rossiter, K.; Wang, M.; Miller, M. Characterization of Royal Gala apple aroma using electronic nose technology-potential maturity indicator. J. Agr. Food Chem. 1999, 47, 5173–5177. [Google Scholar]
- Defilippi, B.G.; San Juan, W.; Valdes, H.; Moya-Leon, M.A.; Infante, R.; Campos-Vargas, R. The aroma development during storage of Castlebrite apricots as evaluated by gas chromatography, electronic nose, and sensory analysis. Postharvest Biol. Technol. 2009, 51, 212–219. [Google Scholar]
- Parpinello, G.P.; Fabbri, A.; Domenichelli, S.; Mesisca, V.; Cavicchi, L.; Versari, A. Discrimination of apricot cultivars by gas multisensor array using an artificial neural network. Biosys. Eng. 2007, 97, 371–378. [Google Scholar]
- Solis-Solis, H.M.; Calderon-Santoyo, M.; Gutierrez-Martinez, P.; Schorr-Galindo, S.; Ragazzo-Sanchez, J.A. Discrimination of eight varieties of apricot (Prunus armeniaca) by electronic nose, LLE and SPME using GC-MS and multivariate analysis. Sens. Actuators B Chem. 2007, 125, 415–421. [Google Scholar]
- Llobet, E.; Hines, E.L.; Gardner, J.W.; Franco, S. Non-destructive banana ripeness determination using a neural network-based electronic nose. Meas. Sci. Technol. 1999, 10, 538–548. [Google Scholar]
- Rosso, F.; Zoppellari, F.; Sala, G.; Malusa, E.; Bardi, L.; Bergesio, B. A study to characterize quality and to identify geographical origin of local varieties of sweet pepper from Piedmont (Italy). Acta Hortic. 2012, 936, 401–409. [Google Scholar]
- Russo, M.; Serra, D.; Suraci, F.; Postorino, S. Effectiveness of electronic nose systems to detect bergamot (Citrus bergamia Risso et Poiteau) essential oil quality and genuiness. J. Essent. Oil Res. 2012, 24, 137–151. [Google Scholar]
- Bernal, L.J.; Melo, L.A.; Diaz-Moreno, C. Evaluation of the antioxidant properties and aromatic profile during maturation of the blackberry (Rubus glaucus Benth) and the bilberry (Vaccinium meridional e Swartz). Rev. Fac. Nac. Agron. Medellin 2014, 67, 7209–7218. [Google Scholar]
- Bernal-Roa, L.J.; Melo, L.A.; Diaz-Moreno, C. Aromatic profile and antioxidant properties during blackberry (Rubus glaucus Benth.) fruit ripening. Acta Hortic. 2014, 1016, 39–45. [Google Scholar]
- Zawirska-Wojtasiak, R.; Goslinski, M.; Szwacka, M.; Gajc-Wolska, J.; Mildner-Szkudlarz, S. Aroma evaluation of transgenic, Thaumatin II-producing cucumber fruit. J. Food Sci. 2009, 74, C204–C210. [Google Scholar]
- Lebrun, M.; Billot, C.; Harrak, H.; Self, G. The electronic nose: A fast and efficient tool for characterizing dates. Fruits 2007, 62, 377–382. [Google Scholar]
- Pokhum, C.; Chawengkijwanich, C.; Maolanon, R. Application of electronic-nose for identification of ripeness stage of durian. Acta Hortic. 2010, 875, 319–328. [Google Scholar]
- Athamneh, A.I.; Zoecklein, B.W.; Mallikarjunan, K. Electronic nose evaluation of cabernet sauvignon fruit maturity. J. Wine Res. 2008, 19, 69–80. [Google Scholar]
- Lopez de Lerma, N.; Bellincontro, A.; Mencarelli, F.; Moreno, J.; Peinado, R.A. Use of electronic nose, validated by GC-MS, to establish the optimum off-vine dehydration time of wine grapes. Food Chem. 2012, 130, 447–452. [Google Scholar]
- Lopez de Lerma, N.; Moreno, J.; Peinado, R.A. Determination of the optimum sun-drying time for Vitis vinifera L. cv. Tempranillo grapes by e-nose analysis and characterization of their volatile composition. Food Bioprocess Biotech. 2014, 7, 732–740. [Google Scholar]
- Santonico, M.; Bellincontro, A.; de Santis, D.; di Natale, C.; Mencarelli, F. Electronic nose to study postharvest dehydration of wine grapes. Food Chem. 2010, 121, 789–796. [Google Scholar]
- Zoecklein, B.W.; Devarajan, Y.S.; Mallikarjunan, K.; Gardner, D.M. Monitoring effects of ethanol spray on Cabernet franc and Merlot grapes and wine volatiles using electronic nose systems. Am. J. Enol. Vitic. 2011, 62, 351–358. [Google Scholar]
- Devarajan, Y.S.; Zoecklein, B.W.; Mallikarjunan, M.; Gardner, D.M. Electronic nose evaluation of the effects of canopy side on Cabernet franc (Vitis vinifera L.) grape and wine volatiles. Am. J. Enol. Vitic. 2011, 62, 73–80. [Google Scholar]
- Alasalvar, C.; Odabasi, A.Z.; Demir, N.; Balaban, M.O.; Shahidi, F.; Cadwallader, K.R. Volatiles and flavor of five Turkish hazelnut varieties as evaluated by descriptive sensory analysis, electronic nose, and dynamic headspace analysis/gas chromatography-mass spectrometry. J. Food Sci. 2004, 69, SNQ99–SNQ106. [Google Scholar]
- Alasalvar, C.; Pelvan, E.; Bahar, B.; Korel, F.; Olmez, H. Flavour of natural and roasted Turkish hazelnut varieties (Corylus avellana L.) by descriptive sensory analysis, electronic nose and chemometrics. Int. J. Food Sci. Technol. 2012, 47, 122–131. [Google Scholar]
- Guarrasi, V.; Farina, V.; Germana, M.A.; San Biagio, P.L.; Mazzaglia, A. Fruit quality evaluation of four loquat cultivars grown in Sicily. Acta Hortic. 2011, 887, 299–304. [Google Scholar]
- Hernandez Gomez, A.; Wang, J.; Hu, G.; Garcia Pereira, A. Electronic nose technique potential monitoring mandarin maturity. Sens. Actuators B Chem. 2006, 113, 347–353. [Google Scholar]
- Hernandez Gomez, A.; Wang, J.; Hu, G.; Garcia Pereira, A. Discrimination of storage shelf-life for mandarin by electronic nose technique. LWT 2007, 40, 681–689. [Google Scholar]
- Hernandez Gomez, A.; Wang, J.; Hu, G.; Garcia Pereira, A. Mandarin ripeness monitoring and quality attribute evaluation using an electronic nose technique. Trans. ASABE 2007, 50, 2137–2142. [Google Scholar]
- Kitthawee, U.; Pathaveerat, S.; Slaughter, D.C.; Mitcham, E.J. Nondestructive sensing of maturity and ripeness in mango. Acta Hortic. 2012, 943, 287–296. [Google Scholar]
- Lebrun, M.; Ducamp, M.N.; Plotto, A.; Goodner, D.; Baldwin, E. Development of electronic nose measurements for mango (Mangifera indica) homogenate and whole fruit. Proc. Fla. State Hort. Soc. 2004, 117, 421–425. [Google Scholar]
- Lebrun, M.; Plotto, A.; Goodner, K.; Ducamp, M.; Baldwin, E. Discrimination of mango fruit maturity by volatiles using the electronic nose and gas chromatography. Postharvest Biol. Technol. 2008, 48, 122–131. [Google Scholar]
- Zakaria, A.; Shakaff, A.Y.M.; Masnan, M.J.; Saad, F.S.A.; Adom, A.H.; Ahmad, M.N.; Jaafar, M.N.; Abdullah, A.H.; Kamarudin, L.M. Improved maturity and ripeness classifications of Magnifera indica cv. Harumanis mangoes through sensor fusion of an electronic nose and acoustic sensor. Sensors 2012, 12, 6023–6048. [Google Scholar]
- Abbey, L.; Aked, J.; Joyce, D.C. Discrimination amongst Alliums using an electronic nose. Ann. Appl. Biol. 2001, 139, 337–342. [Google Scholar]
- Abbey, L.; Joyce, D.C.; Aked, J.; Smith, B.; Marshall, C. Electronic nose evaluation of onion headspace volatiles and bulb quality as affected by nitrogen, sulphur and soil type. Ann. Appl. Biol. 2004, 145, 41–50. [Google Scholar]
- Russo, M.; di Sanzo, R.; Cefaly, V.; Carabetta, S.; Serra, D.; Fuda, S. Non-destructive flavour evaluation of red onion (Allium cepa L.) ecotypes: An electronic-nose-based approach. Food Chem. 2013, 141, 896–899. [Google Scholar]
- Di Natale, C.; Macagnano, A.; Martinelli, E.; Paolesse, R.; Proietti, E.; D'Amico, A. The evaluation of quality of post-harvest oranges and apples by means of an electronic nose. Sens. Actuators B Chem. 2001, 78, 26–31. [Google Scholar]
- Oshita, S.; Shima, K.; Haruta, T.; Seo, Y.; Kawagoe, Y.; Nakayama, S.; Takahara, H. Discrimination of odors emanating from ‘La France’ pear by semi-conducting polymer sensors. Comput. Electron. Agric. 2000, 26, 209–216. [Google Scholar]
- Zhang, H.; Wang, J.; Ye, S. Prediction of acidity, soluble solids and firmness of pear using electronic nose technique. J. Food Eng. 2008, 86, 370–378. [Google Scholar]
- Zhang, H.; Wang, J.; Ye, S. Prediction of soluble solids content, firmness and pH of pear by signals of electronic nose sensors. Anal. Chim. Acta 2008, 606, 112–118. [Google Scholar]
- Benedetti, S.; Buratti, S.; Spinardi, A.; Mannino, S.; Mignani, I. Electronic nose as a non-destructive tool to characterize peach cultivars and to monitor their ripening stage during shelf life. Postharvest Biol. Technol. 2008, 47, 181–188. [Google Scholar]
- Di Natale, C.; Macagnano, A.; Martinelli, E.; Proietti, E.; Paolesse, R.; Castellari, L.; Campani, S.; D'Amico, A. Electronic nose based investigation of the sensorial properties of peaches and nectarines. Sens. Actuators B Chem. 2001, 77, 561–566. [Google Scholar]
- Di Natale, C.; Zude-Sasse, M.; Macagnano, A.; Paolesse, R.; Herold, B.; D'Amico, A. Outer product analysis of electronic nose and visible spectra: Application to the measurement of peach fruit characteristics. Anal. Chim. Acta 2002, 459, 107–117. [Google Scholar]
- Infante, R.; Farcuh, M.; Meneses, C. Monitoring the sensorial quality and aroma through an electronic nose in peaches during cold storage. J. Sci. Food Agric. 2008, 88, 2073–2078. [Google Scholar]
- Infante, R.; Rubio, P.; Meneses, C.; Contador, L. Ripe nectarines segregated through sensory quality evaluation and electronic nose assessment. Fruits 2011, 66, 109–119. [Google Scholar]
- Rizzolo, A.; Bianchi, G.; Vanoli, M.; Lurie, S.; Spinelli, L.; Torricelli, A. Electronic nose to detect volatile compound profile and quality changes in “Spring Belle” peach (Prunus persica L.) during cold storage in relation to fruit optical properties measured by time-resolved reflectance spectroscopy. J. Agric. Food Chem. 2013, 61, 1671–1685. [Google Scholar]
- Su, M.; Zhang, B.; Ye, Z.; Shen, J.; Li, H.; Chen, K. Non-destructive detection of peach (Prunus persica) fruit volatiles using an electronic nose. J. Fruit Sci. 2012, 29, 809–813. [Google Scholar]
- Su, M.; Zhang, B.; Ye, Z.; Chen, K.; Guo, J.; Gu, X.; Shen, J. Pulp volatiles measured by an electronic nose are related to harvest seasons, TSS concentration and TSS/TA ratio among 39 peaches and nectarines. Sci. Hortic. 2013, 150, 146–153. [Google Scholar]
- Zhang, H.; Chang, M.; Wang, J.; Ye, S. Evaluation of peach quality indices using an electronic nose by MLR, QPST and BP network. Sens. Actuators B Chem. 2008, 134, 332–338. [Google Scholar]
- Zhang, H.; Wang, J.; Ye, S.; Chang, M. Application of electronic nose and statistical analysis to predict quality indices of peach. Food Bioprocess Technol. 2012, 5, 65–72. [Google Scholar]
- Liu, H.; Ceng, F.K.; Wang, Q.H.; Wu, H.S.; Tan, L.H. Studies on the chemical and flavor qualities of white pepper (Piper nigrum L.) derived from fine new genotypes. Eur. J. Food Res. Technol. 2013, 273, 245–251. [Google Scholar]
- Garcia Breijo, E.; Guarrasi, V.; Masot Peris, R.; Alcaniz Fillol, M.; Olguin Pinatti, C. Odour sampling system with modifiable parameters applied to fruit classification. J. Food Eng. 2013, 116, 277–285. [Google Scholar]
- Li, J.; Peng, Z.; Xue, Y.; Chen, S.; Zhang, P. Discrimination of maturity and storage life for ‘Mopan’ persimmon by electronic nose technique. Acta Hortic. 2013, 996, 385–390. [Google Scholar]
- Torri, L.; Sinelli, N.; Limbo, S. Shelf life evaluation of fresh-cut pineapple by using an electronic nose. Postharvest Biol. Technol. 2010, 56, 239–245. [Google Scholar]
- Nugroho, J.; Rahayoe, S.; Oka, A.A. Applications of electronic nose to determine the maturity of sapodilla using pattern recognition system. Acta Hortic. 2013, 1011, 87–93. [Google Scholar]
- Supriyadi; Shimizu, K.; Suzuki, M.; Yoshida, K.; Muto, T.; Fujita, A.; Tomita, N.; Watanabe, N. Maturity discrimination of snake fruit (Salacca edulis Reinw.) cv. Pondoh based on volatiles analysis using an electronic nose device equipped with a sensor array and fingerprint mass spectrometry. Flavour Fragr. J. 2004, 19, 44–50. [Google Scholar]
- Akakabe, Y.; Sakamoto, M.; Ikeda, Y.; Tanaka, M. Identification and characterization of volatile components of the Japanese sour citrus fruit Citrus nagato-yuzukichi Tanaka. Biosci. Biotechnol. Biochem. 2008, 72, 1965–1968. [Google Scholar]
- Marquez Cardozo, C.J.; Cartagena Valezuela, J.R.; Correa Londono, G.A. Determination of Soursop (Annona muricata L. cv. Elita) fruit volatiles during ripening by electronic nose and gas chromatography coupled to mass spectroscopy. Rev. Fac. Nac. Agron. Med. 2013, 66, 7117–7128. [Google Scholar]
- Agulheiro-Santos, A.C. Quality of strawberry ‘Camarosa’ with different levels of nitrogen fertilization. Acta Hortic. 2009, 842, 907–910. [Google Scholar]
- Du, X.; Bai, J.; Plotto, A.; Baldwin, E.; Whitaker, V.; Rouseff, R. Electronic nose for detecting strawberry fruit maturity. Proc. Fla. State Hort. Soc. 2010, 123, 259–263. [Google Scholar]
- Qiu, S.; Wang, J.; Gao, L. Discrimination and characterization of strawberry juice based on electronic nose and tongue: Comparison of different juice processing approaches by LDA, PLSR, RF, and SVM. J. Agric. Food Chem. 2014, 62, 6426–6434. [Google Scholar]
- Berna, A.Z.; Buysens, S.; Di Natale, C.; Grun, I.U.; Lammertyn, J.; Nicolai, B.M. Relating sensory analysis with electronic nose and headspace fingerprint MS for tomato aroma profiling. Postharvest Biol. Technol. 2005, 36, 143–155. [Google Scholar]
- Berna, A.Z.; Lammertyn, J.; Saevels, S.; di Natale, C.; Nicolai, B.M. Electronic nose systems to study shelf life and cultivar effect on tomato aroma profile. Sens. Actuators B Chem. 2004, 97, 324–333. [Google Scholar]
- Cheng, S.; Wang, J.; Wang, Y.; Wei, Z. Discrimination of tomato plant with different levels of mechanical damage by electronic nose. Trans. Chin. Soc. Agric. Eng. 2012, 28, 102–106. [Google Scholar]
- Hernandez Gomez, A.; Hu, G.; Wang, J.; Garcia Pereira, A. Evaluation of tomato maturity by electronic nose. Comput. Electron. Agric. 2006, 54, 44–52. [Google Scholar]
- Hernandez Gomez, A.; Wang, J.; Hu, G.; Garcia Pereira, A. Monitoring storage shelf life of tomato using electronic nose technique. J. Food Eng. 2008, 85, 625–631. [Google Scholar]
- Hong, X.; Wang, J. Application of e-nose and e-tongue to measure the freshness of cherry tomatoes squeezed for juice consumption. Anal. Methods 2014, 6, 3133–3138. [Google Scholar]
- Maul, F.; Sargent, S.A.; Huber, D.J. Non-destructive quality screening of tomato fruit using “electronic nose” technology. Proc. Fla State Hortic. Soc. 1997, 110, 188–194. [Google Scholar]
- Maul, F.; Sargent, S.A.; Balaban, M.O. Aroma volatile profiles from ripe tomatoes are influenced by physiological maturity at harvest: An application for electronic nose technology. J. Am. Soc. Hort. Sci. 1998, 126, 1094–1101. [Google Scholar]
- Sinesio, F.; di Natale, C.; Quaglia, G.B.; Bucarelli, F.M.; Moneta, E.; Macagnano, A.; Paolesse, R.; D'Amico, A. Use of electronic nose and trained sensory panel in the evaluation of tomato quality. J. Sci. Food Agric. 2000, 80, 63–71. [Google Scholar]
- Wang, J.; Zhou, Y. Electronic-nose technique: Potential for monitoring maturity and shelf life of tomatoes. N. Z. J. Agric. Res. 2007, 50, 1219–1228. [Google Scholar]
- Li, G.; Jia, H.; Wang, Q.; Zhang, M.; Teng, Y.W. Changes of aromatic composition in Youhongli (Pyrus ussuriensis) fruit during fruit ripening. J. Fruit Sci. 2012, 29, 11–16. [Google Scholar]
- Guohua, H.; Yuling, W.; Dandan, Y.; Wenwen, D.; Linshan, Z.; Lvye, W. Study of peach freshness predictive method based on electronic nose. Food Control 2012, 28, 25–32. [Google Scholar]
- Zhang, H.; Wang, J. Evaluation of peach quality attribute using an electronic nose. Sens. Mater. 2009, 21, 419–431. [Google Scholar]
- Chatonnet, P.; Dubourdieu, D. Using electronic odor sensor to discriminate among oak barrel toasting levels. J. Agric. Food Chem. 1999, 47, 4319–4322. [Google Scholar]
- Di Natale, C.; Paolesse, R.; Burgio, M.; Martinelli, E.; Pennazza, G.; D'Amico, A. Application of metalloporphyrins-based gas and liquid sensor arrays to the analysis of red wine. Anal. Chim. Acta 2004, 513, 49–56. [Google Scholar]
- Buratti, S.; Benedetti, S.; Scampicchio, M.; Pangerod, E.C. Characterization and classification of Italian Barbera wines by using an electronic nose and an amperometric electronic tongue. Anal. Chim. Acta 2004, 525, 133–139. [Google Scholar]
- Penza, M.; Cassano, G. Recognition of adulteration of Italian wines by thin-film multisensory array and artificial neural networks. Anal. Chim. Acta 2004, 509, 159–177. [Google Scholar]
- McKellar, R.C.; Rupasinghe, H.P.V.; Lu, X.; Knight, K.P. The electronic nose as a tool for the classification of fruit and grape wines from different Ontario wineries. J. Food Sci. Agric. 2005, 85, 2391–2396. [Google Scholar]
- Lozano, J.; Santos, J.P.; Horrillo, M.C. Classification of white wine aromas with an electronic nose. Talanta 2005, 67, 610–616. [Google Scholar]
- Martin, A.; Mallikarjunan, K.; Zoecklein, B.W. Discrimination of wines produced from Cabernet Sauvignon grapes treated with aqueous ethanol post-bloom using an electronic nose. Int. J. Food Eng. 2008, 4. [Google Scholar] [CrossRef]
- Guarrasi, V.; Farina, V.; San Biagio, P.L.; Germana, M.A. Naso elettronico con sensori MOS per discriminare 3 cultivar di Eriobotrya japonica Lindl. Italus Hortus 2010, 17, 103–108. [Google Scholar]
- Guohua, H.; Jiaojiao, J.; Shanggui, D.; Xiao, Y.; Mengtian, Z.; Minmin, W.; Dandan, Y. Winter jujube (Zizyphus jujube Mill.) quality forecasting method based on electronic nose. Food Chem. 2015, 170, 484–491. [Google Scholar]
- Lewis, R.A. CRC Dictionary of Agricultural Sciences; CRC Press: Boca Raton, FL, USA, 2002; p. 375. [Google Scholar]
- Mamatha, B.Z.; Prakash, M. Studies on Pepper (Piper nigrum L.) cultivars by sensory and instrumental techniques. Z. Arznei- Gewurzpflanzen 2011, 16, 176–180. [Google Scholar]
- Wilson, A.D. Development of an electronic-nose technology for the rapid detection of agricultural pesticide residues. Proceedings of the annual meeting of American Phytopathological Society, Providence, RI, USA, 4–8 August 2012.
- Wilson, A.D. Identification of insecticide residues with a conducting-polymer electronic nose. Chem. Sens. 2014, 4, 1–10. [Google Scholar]
- Wilson, A.D. Fungicide residue identification and discrimination using a conducting polymer electronic-nose. Proceedings of the Fourth International Conference on Sensor Device Technologies and Applications, Barcelona, Spain, 25–31 August 2014; Xpert Publishing Services: Wilmington, DE, USA, 2013; pp. 116–121. [Google Scholar]
| Fruit Type | Chemical Class | Example Compounds | Chemical Structure | Reference |
|---|---|---|---|---|
| Apple | Aliphatic esters | ethyl butanoate | 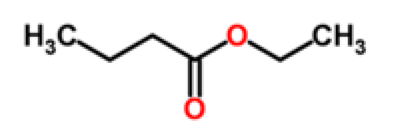 | [18–20] |
| Apricots | Aliphatic alcohols | 1-hexanol |  | [21,22] |
| Banana | Aliphatic esters | isoamyl acetate |  | [23] |
| Caraway | Terpenoids | carvone |  | [24,25] |
| Cantaloupe | Thiobutyrates | S-methyl thiobutanoate |  | [26] |
| Kiwifruit | Aliphatic aldehydes | hexanal |  | [27] |
| Peach | Lactones | γ-decalactone |  | [28] |
| Peach | Aliphatic esters | hexyl acetate |  | [29] |
| Pineapple | Organic acids | hexanoic acid |  | [30] |
| Raspberry | Aliphatic ketones | raspberry ketone |  | [31,32] |
| Strawberry | Furanone lactones | furaneol |  | [17] |
| Tomato | Apocarotenoids | β-ionone |  | [33] |
| Fruit Type | Cultivars/Varieties | Principal Volatile Compounds in Aroma † | References |
|---|---|---|---|
| Apple | Cox orange | Acetaldehyde, ethyl butanoate, ethyl methyl propanoate, 2-methyl butanol | [18] |
| Elstar | Ethyl butanoate, ethyl 2-methyl butanoate | [18] | |
| Fuji | Ethyl 2-methyl butanoate, 2-methyl butyl acetate, hexyl acetate | [18] | |
| Pink Lady | Butyl acetate, hexyl acetate, 2-methylbutyl acetate, hexyl butanoate, hexyl 2-methyl butanoate, hexyl hexanoate | [20] | |
| Banana | Cavendish | (E)-2-hexenal, acetoin | [38] |
| Frayssinette | 2, 3-Butanediol, solerol | [39] | |
| Plantain | (E)-2-hexenal, hexanal | [23,38,40] | |
| Blackberry | Black Diamond | Furaneol, 2-heptanol, β-ionone, linalool | [41,42] |
| Marion | Furaneol, hexanal, β-ionone, linalool | [41,42] | |
| Blueberries | Primadonna, Jewel | Many aliphatic esters, C6-aldehydes | [43] |
| Snowchaser, FL02-40 | Primarily terpenoids, less aliphatic esters | [43] | |
| Grape | Cabernet Sauvignon | Benzene derivatives, monoterpenes, and sesquiterpenes, (also primarily alcohols) | [44–46] |
| Muscat | Citral, citronellol, diendiol I, diendiol II, geraniol, linalool, rose oxide, nerol | [44–46] | |
| Riesling | Geraniol, α-muurolene, (also primarily esters and aldehydes) | [44–46] | |
| Mango (Columbian) | Haden Irwin, Manila | δ-3-Carene | [47] |
| Hilacha, Vallenato | α-Pinene | [47] | |
| Van Dyke | α-Phellandrene | [47] | |
| Yulima | Terpinolene | [47] | |
| Pineapple | Cayenne | Ethyl 2-methylbutanoate, ethyl hexanoate, 2, 5-dimethyl-4-hydroxy-3(2H)-furanone (DMHF), decanal, ethyl 3-(methylthio) propionate, ethyl butanoate, (E)-3-ethyl hexenoate | [48] |
| Tainong No. 4 | Furaneol, 3-(methylthio) propanoic acid methyl ester, 3-(methylthio) propanoic acid ethyl ester, δ-octalactone | [49] | |
| Tainong No. 6 | Ethyl-2-methylbutyrate, methyl-2-methylbutyrate, 3-(methylthio) propanoic acid ethyl ester, ethyl hexanoate, decanal | [50] |
†Principal chemicals (VOCs) found in complex fruit volatile mixtures are listed in alphabetical order, not in order of relative abundance by quantity within mixtures analyzed from individual fruit cultivars or varieties.
| Common Name | Scientific Name | Family | E-Nose Type † | Discrimination Type | Reference | |
|---|---|---|---|---|---|---|
| Apple | Malus domestica Borkr | Rosaceae | FOX 4000 1 | Post-harvest treatments | [83] | |
| Smart Nose 2 | Shelf life | [84] | ||||
| Prototype MOS 3 | Maturity stage at harvest | [85] | ||||
| Prototype MOS | Shelf life | [86] | ||||
| Prototype MOS | Shelf life | [87] | ||||
| Prototype MOS | Shelf life | [88] | ||||
| Prototype QMBs 4 | Shelf life | [89] | ||||
| Prototype MOS | Prediction of storage time | [90] | ||||
| Cyranose 320 5 | Aroma profile during deteriorative shelf life | [91] | ||||
| Unspecified | Cultivar effect | [92] | ||||
| Cyranose 320 | Maturity stage at harvest | [93] | ||||
| FOX 4000 | Cultivar effect | [94] | ||||
| Libra Nose 6 | Maturity stage at harvest | [95] | ||||
| Libra Nose | Shelf life | [96] | ||||
| Unspecified | Quality assessment | [97] | ||||
| FOX 4000 | Maturity stage at harvest | [98] | ||||
| Apricot | Prunus armeniaca L. | Rosaceae | EOS835 7 | Ripening stage after harvest | [99] | |
| PEN2 8 | Cultivar effect | [100] | ||||
| FOX 4000 | Cultivar effect | [101] | ||||
| Banana | Musa x paradisiaca L. | Musaceae | Prototype MOS | Ripening stage after harvest | [102] | |
| Bell pepper | Capsicum annuum L. | Solanaceae | Unspecified | Quality assessment | [103] | |
| Bergamot | Citrus bergamia Risso and Poiteau | Rutaceae | ISE Nose 2000 9 | Cultivar effect; geographic effect; adulteration | [104] | |
| Blackberry | Rubus glaucus Benth | Rosaceae | PEN3 6 | Maturity stage at harvest | [105] | |
| Unspecified | Maturity stage at harvest | [106] | ||||
| Bilberry | Vaccinium meridionale Swartz | Ericaceae | PEN3 | Maturity stage at harvest | [105] | |
| Blueberry | Vaccinium spp. | Ericaceae | Prototype MOS | Ripening stage after harvest; quality control | [82] | |
| Cucumber | Cucumis sativus L. | Cucurbitaceae | FOX 4000 | Genotypic effect | [107] | |
| Date | Phoenix dactylifera L. | Arecaceae | FOX 4000 | Cultivar effect | [108] | |
| Durian | Durio spp. | Malvaceae | Unspecified | Maturity stage at harvest | [109] | |
| Grape | Vitis vinifera L. | Vitaceae | Cyranose 320 | Maturity stage at harvest | [110] | |
| enQbe 10 | Dehydration time | [111] | ||||
| enQbe | Dehydration time | [112] | ||||
| enQbe | Dehydration time | [113] | ||||
| enQbe | Post-harvest treatments | [114] | ||||
| zNose 11 | Canopy side effect | [115] | ||||
| Cyranose 320 | ||||||
| Hazelnut | Corylus avellana L. | Betulaceae | E-Nose 4000 12 | Cultivar effect | [116] | |
| Moses II 13 | Cultivar effect | [117] | ||||
| Loquat | Eriobotrya japonica (Thunb.) Lindl. | Rosaceae | Unspecified | Cultivar effect | [118] | |
| Mandarin | Citrus reticulate Blanco | Rutaceae | PEN2 | Maturity stage at harvest | [119] | |
| PEN2 | Post-harvest treatments | [120] | ||||
| PEN2 | Maturity stage at harvest | [121] | ||||
| Mango | Mangifera indica L. | Anacardiaceae | Unspecified | Maturity stage at harvest | [122] | |
| FOX 4000 | Cultivar effect, post-harvest treatments | [123] | ||||
| FOX 4000 | Cultivar effect, maturity stage at harvest, shelf life | [124] | ||||
| Cyranose 320 | Maturity stage at harvest | [125] | ||||
| Muskmelon | Cucumis melo L. | Cucurbitaceae | Not specified | Maturity stage at harvest | [81] | |
| Onion, spring onion garlic, shallot, leek | Allium spp. | Liliaceae | Aromascan CP 14 | Species effect | [126] | |
| Onion | Allium cepa L. | Liliaceae | Aromascan | Fertilization, soil type effect | [127] | |
| ISENose 2000 15 | Ecotype effect | [128] | ||||
| Orange | Citrus x sinensis (L.) Osbeck | Rutaceae | Libra Nose | Shelf life | [129] | |
| Pear | Pyrus communis L. | Rosaceae | Prototype MOS | Maturity stage at harvest | [85] | |
| Prototype MOS | Shelf life | [87] | ||||
| Aromascan | Maturity stage at harvest | [130] | ||||
| Prototype MOS | Maturity stage at harvest; quality assessment | [131] | ||||
| Prototype MOS | Maturity stage at harvest; quality assessment | [132] | ||||
| Peach | Prunus persica (L.) Batsch | Rosaceae | PEN2 | Shelf life and cultivar effect | [133] | |
| Prototype MOS | Maturity stage at harvest | [85] | ||||
| Libra Nose | Sensorial assessment | [134] | ||||
| Libra Nose | Cultivar effect, quality assessment | [135] | ||||
| Prototype MOS | Shelf life, quality assessment | [87] | ||||
| EOS 835 | Cultivar effect, shelf life | [136] | ||||
| EOS 835 | Cultivar effect | [137] | ||||
| PEN3 | Maturity stage at harvest | [138] | ||||
| FOX 4000 | Cultivar effect | [139] | ||||
| FOX 4000 | Prediction of harvest time, quality assessment | [140] | ||||
| Prototype MOS | Prediction of harvest time, quality assessment | [141] | ||||
| Prototype MOS | Prediction of harvest time, quality assessment | [142] | ||||
| FOX 4000 | Shelf life | [61] | ||||
| Pepper | Piper nigrum L. | Piperaceae | Alpha Gemini | Genotype effect | [143] | |
| Persimmon | Diospirus Kaki L.f. | Ebenaceae | Prototype MOS | Cultivar effect | [144] | |
| Ebenace | PEN3 | Maturity stage at harvest and shelf life | [145] | |||
| Pineapple | Ananas comosus (L.) Merr. | Bromeliaceae | PEN2 | Shelf life | [146] | |
| Sapodilla | Manilkara zapota (L.)P. Royen | Sapotaceae | Prototype MOS | Maturity stage at harvest | [147] | |
| Snake fruit | Salacca edulis Reinw. | Arecaceae | FOX 4000 | Maturity stage at harvest | [148] | |
| Sour citrus | Citrus nagato-yuzukichi Tanaka | Rutaceae | Unspecified | Species effect | [149] | |
| Soursoup | Annona muricata L. | Annonaceae | PEN3 | Maturity stage at harvest | [150] | |
| Strawberry | Fragaria x ananassa Duch. | Rosaceae | Unspecified | Fertilizations effect | [151] | |
| PEN2 | Processing approaches effect | [152] | ||||
| PEN2 | Processing approaches effect | [153] | ||||
| Tomato | Solanum lycopersicon L. | Solanaceae | Unspecified | Cultivar effect, shelf life | [154] | |
| Libra Nose | Cultivar effect, shelf life | [155] | ||||
| Unspecified | Mechanical deterioration effect | [156] | ||||
| PEN2 | Maturity stage at harvest | [157] | ||||
| PEN2 | Post-harvest treatments effect | [158] | ||||
| Cherry tomato | Lycopersicon esculentum var. cerasiforme | Solanaceae | PEN2 | Shelf life of processed fruits | [159] | |
| Tomato | Solanum lycopersicon L. | Solanaceae | e-Nose 4000 12 | Harvesting and postharvest handling treatments effect | [160] | |
| e-Nose 4000 | Post-harvest treatments | [161] | ||||
| enQbe | Cultivation techniques effect | [162] | ||||
| Unspecified | Maturity stage at harvest and shelf life | [163] | ||||
| Ussurian pear | Pyrus ussuriensis Maxim. | Rosaceae | Unspecified | Maturity stage at harvest | [164] | |
†Electronic nose (e-nose) names, types and manufacturers: 1 = Alpha MOS, Toulouse, France; 2 = SmartNose BV, Amersfoort, The Netherlands; 3 = Self-made prototype equipped with an array of commercial MOS gas sensors; 4 = Quartz microbalances (QMB) gas sensors; 5 = Cyrano Sciences Inc., Pasadena, CA, USA; 6 = Technobiochip, Marciana, Italy; 7 = Sacmi Imola s.c.a.r.l., Imola, Italy; 8 = Airsense Analytics GmbH, Schwerin, Germany; 9 = ISE, Pisa, Italia; 10 = University of Rome ‘Tor Vergata’, Rome, Italy; 11 = Electronic Sensor Technology, Newbury Park, CA, USA; 12 = EEV Inc., Amsford, NJ, USA; 13 = Lennartz Electronic GmbH, Tübingen, Germany; 14 = Osmetech Inc., Wobum, MA, USA; 15 = Labservice Analytica, Bologna, Italy. Unspecified e-nose types were not determinable from descriptions given in the methods section.
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baietto, M.; Wilson, A.D. Electronic-Nose Applications for Fruit Identification, Ripeness and Quality Grading. Sensors 2015, 15, 899-931. https://doi.org/10.3390/s150100899
Baietto M, Wilson AD. Electronic-Nose Applications for Fruit Identification, Ripeness and Quality Grading. Sensors. 2015; 15(1):899-931. https://doi.org/10.3390/s150100899
Chicago/Turabian StyleBaietto, Manuela, and Alphus D. Wilson. 2015. "Electronic-Nose Applications for Fruit Identification, Ripeness and Quality Grading" Sensors 15, no. 1: 899-931. https://doi.org/10.3390/s150100899
APA StyleBaietto, M., & Wilson, A. D. (2015). Electronic-Nose Applications for Fruit Identification, Ripeness and Quality Grading. Sensors, 15(1), 899-931. https://doi.org/10.3390/s150100899






