Disposable Screen Printed Electrochemical Sensors: Tools for Environmental Monitoring
Abstract
: Screen printing technology is a widely used technique for the fabrication of electrochemical sensors. This methodology is likely to underpin the progressive drive towards miniaturized, sensitive and portable devices, and has already established its route from “lab-to-market” for a plethora of sensors. The application of these sensors for analysis of environmental samples has been the major focus of research in this field. As a consequence, this work will focus on recent important advances in the design and fabrication of disposable screen printed sensors for the electrochemical detection of environmental contaminants. Special emphasis is given on sensor fabrication methodology, operating details and performance characteristics for environmental applications.1. Introduction
A major part of analytical research activity is devoted to the development of new and robust methodologies. For example, new analytical tools are required for economical and real time monitoring of environmental pollutants, and for prevention of toxic materials in the environment. Progress in the field of analytical chemistry is aimed at bringing the analytical data close to the production operations [1–3]. Such advances offer improved analytical methods with reduced environmental impact. A real time field detection system is highly desirable for continuous environmental monitoring to overcome the limitations such as sample collection and transport to a central laboratory, problems associated with commonly used methods for environmental pollutants. In this context, real time methods offer a rapid return of the chemical profile (alarm tools for sudden discharge) with minimized errors and costs as compared to the offsite laboratory-based analyses [1]. The development of portable approaches and devices with reduced sample volume is of considerable interest for both centralized and decentralized (field) analyses. This review paper highlights recent advances, primarily from the authors' laboratories, aimed at designing electrochemical systems for meeting the needs of analytical chemistry. Electrochemical devices offer unique properties to address the challenges of analytical chemistry. The advantages of electrochemical devices include possibility of miniaturization and portability, sensitivity, selectivity, a wide linear range, minimal space and power requirement and cost effective instrumentation. Devices based on the electrochemical detection are well established for many years. The past decades have seen enormous progress in electro-analytical chemistry with the development of ultra-microelectrodes, tailored interfaces, molecular devices and smart sensors. These developments have resulted in substantial popularity of electro-analyses and to their expansion into new phases and environments [4,5].
As we enter the 21 century, we do not want to rely on cumbersome electrochemical cells and bulky electrodes but rather would like to have fast, small, easy to use, portable, economical and disposable electrode systems. A vast array of electrodes for on site and in situ environmental monitoring has been developed during recent years [6–10]. Several representative examples, illustrating the scope, power, versatility and application of such miniaturized electrodes for environmental monitoring are described in the coming sections. This review paper as whole will focus on the trends in screen printed electrode design, screen printed electrode fabrication processes, types of screen printed electrodes and finally their environmental applications.
2. Towards Easy to Use, Disposable and Portable Screen Printed Electrodes
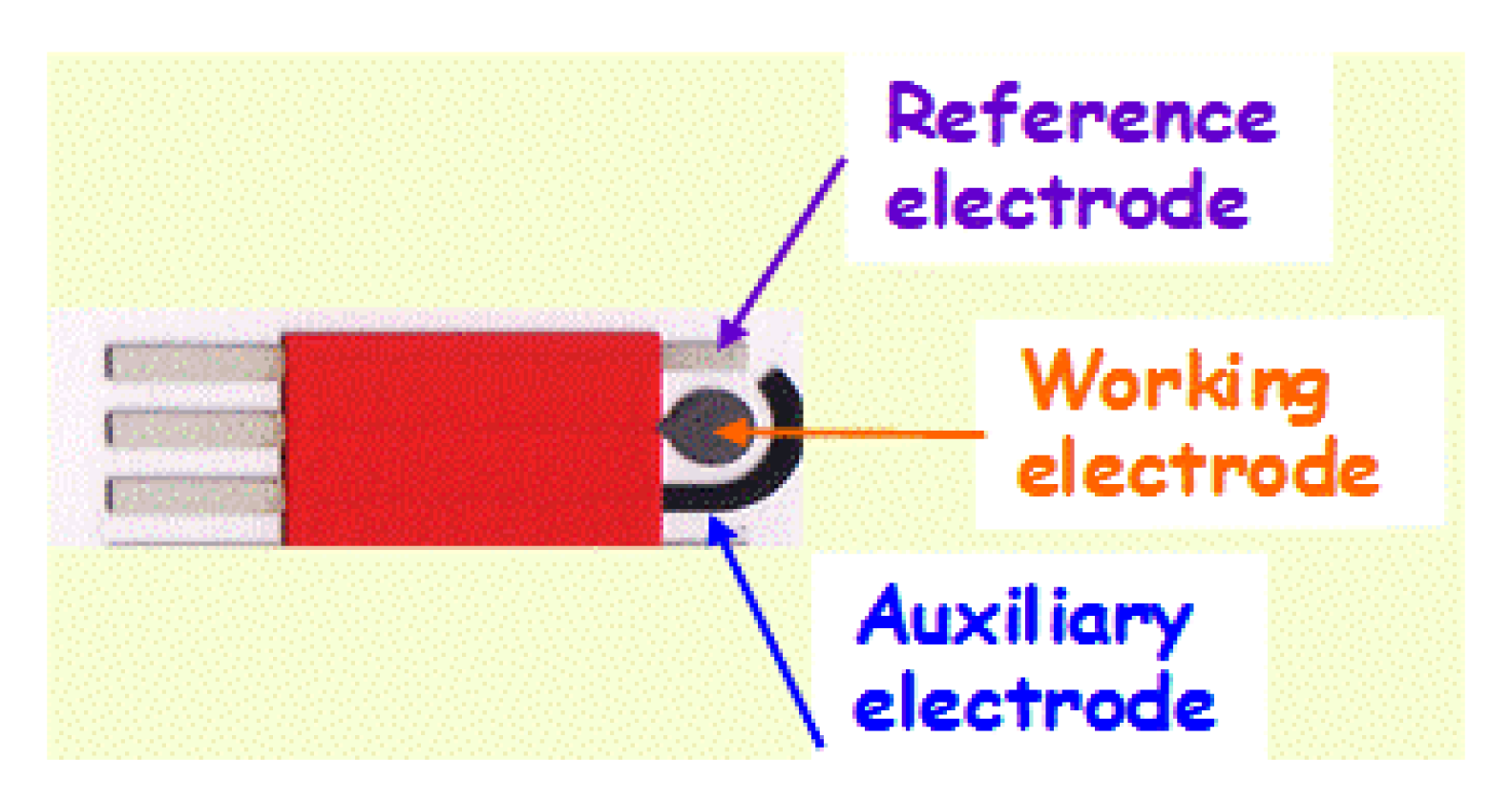
The elimination of bulky materials and instruments from the analytical protocol is a major thrust of analytical chemistry. The performance of analytical methods is directly related to the material of the working electrode. For many years, mercury was used as the most suitable electrode material due to its very attractive behavior and highly reproducible, renewable and smooth surface. These distinct properties of the mercury drop electrode led to the Nobel Prize in Chemistry awarded to Heyerovský in 1959. Both doping and hanging electrodes have been widely used in various polarographic and electrochemical techniques [1]. With the advancements in electro-analytical science, various non-mercury electrodes have also been examined. For example, bismuth and carbon electrodes started to be used in electro-analysis more than three decades ago due to their low background current, wide potential range, chemical inertness and suitability for various sensing and detection applications [11–13]. Recently, miniaturization of the solid electrodes was used to get several fundamental and practical advantages including such as a dramatic reduction in sample volume, portability and cost effectiveness. To address the needs of on-site analysis, it was necessary to move away from the commonly used cumbersome electrodes and cells. The exploitation of new fabrication techniques allows the replacement of traditional beaker type electrochemical cells and bulky electrodes with easy to use sensors. Fabrication of printed devices on bendable substrates has enabled the development of a wide range of new electrode systems. Screen printing technology is a well established technique for the fabrication of economical, portable and disposable electrode systems [14,15]. The whole electrode system, including reference, counter and working electrodes can be printed on the same substrate surface [8] (Figure 1). One prominent commercialization of screen printed electrode is the glucose biosensor used for diabetes which represents a billion dollar per year global market [16–18]. Society is in constant state of growth and development, and it is evident that demands for sensing devices related to the environment will increase with the passage of time. In order to achieve this, accurate, portable and rapid devices are highly needed. Decentralized analyses are necessary and thus traditional analytical methods cannot cope with these requirements.
Screen printed electrodes not only address the issue of cost effectiveness but also satisfy the requirement of portability, a progress towards decentralized analysis. The adaptability of screen printed electrodes is of vital importance in the area of research, the ability to modify electrodes with ease through different inks commercially available for the reference, counter and working electrode, allows for highly specific and finally calibrated electrodes to be produced for specific target analytes [8,19,20]. Many kinds of screen printed electrode modifiers exist for environmental monitoring such as noble metals, inorganic nanomaterials, proteins, enzymes and DNA sequences [21]. Screen printed electrodes combine the properties of ease of use and portability with simple, inexpensive analytical methods [22,23]. Consequently, screen printed electrodes can be easily adapted to in situ environmental monitoring to achieve improved performance, as has been demonstrated over the past several years.
3. Working Principle of a Screen Printed Electrochemical Sensor
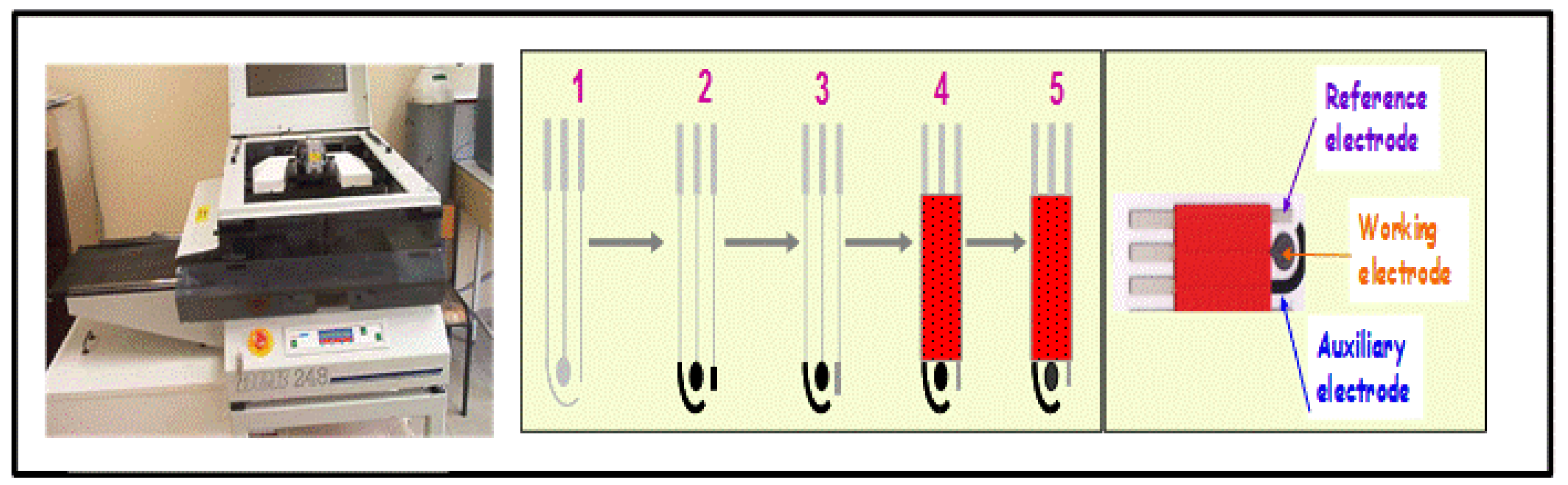
Screen printed methodology offers an attractive way to design new generation electrochemical sensors. Scientists from across different fields have shown their interest in designing low cost and reliable screen printed electrochemical sensors. A detailed description of screen printed electrode fabrication methodology was already reported in a review paper by Li et al. [9]. Briefly, a screen printed electrode comprises a chemically inert substrate on which three electrodes, including working electrode, reference electrode and counter electrode, are printed through screen printing methodology. The working electrode is the principal electrode on which electrochemical reactions are performed, while the reference electrode and counter electrode are used to complete the electronic circuit. Figure 2 represents the stepwise fabrication of a screen printed electrode. The chemical or biological event on the screen printed electrode is converted into a detectable signal with the integration of a transducer element. Among the different transduction techniques, the electrochemical method of detection has attracted more interest for the design of low cost devices. Electrochemical methods of detection include amperometric (based on the current measurement), potentiometric (based on the voltage or potential differences) and conductometric (based on the conductivity or resistance). Among the electrochemical sensing techniques, amperometric detection is widely adopted due to its high sensitivity and applicability.
The fabrication of an electrochemical screen printed sensor usually involves three steps: fabrication of the screen printed electrode, surface design of the screen printed electrode and subsequently utilization for a sensing application. The next section will focus on the fabrication strategies employed to design efficient electrochemical sensors in term of analytical characteristics.
4. Dynamics of Screen Printed Electrochemical Sensors
The inks used in screen printed electrode fabrication consist of particles, polymeric binder and other additives for improved dispersion, printing and adhesion process. The exact ink formulation and composition are patented by the respective companies, and are not disclosed to the users. The variation in the ink composition such as types, size or loading of particles strongly influence the electron transfer process and change the overall performance of the designed screen printed sensor [24–27]. However, screen printed electrodes surface can be very easily modified with a variety of materials and structurally related materials to compensate these limitations. Although these screen printed sensors have found widespread applications, the fundamental understanding of the electrochemical reactivity at the screen printed electrode is still rarely studied and addressed. In this regards, Sljukie et al. have shown that the performance of a macro-screen printed sensor can be improved by the use of ultrasound through increase in mass transport of the analyte and removal of surface active species. [28] Choudhry et al., have for the first time explored the fact that the electrode morphology can be changed dramatically by varying the concentration of the polymeric binder [29]. The same group has demonstrated that a bespoke screen printed electrode can be modified with electro-active palladium for the electrochemical oxidation of hydrazine where the unmodified electrode exhibited slow electron transfer [30]. It has also been shown that non-linear diffusion over an electrode surface may affect the individual contributions of edge and basal plane materials, affecting the relative area of the electrode surfaces [31]. Recently, Choudhry and Banks have designed a screen printed electrode substrate modified with nickel nanoparticles supported on boron diamond. These nickel-modified electrodes were shown as a potential analytical tool for the detection of alcohols [32].
The other fundamental expect of understanding screen printed electrodes reactivity is to explore the creation of surface oxygen functionalities, and the use of mediators on the electrode surface [4]. The next section will highlight some of the important advances made on screen printed electrodes creating oxygen functionalities and edge plane like sites, along with some chemical modifications.
4.1. Preanodized Screen Printed Carbon Electrode
The research on several carbon materials, especially carbon nanotubes and graphene, has revolutionized the scope of screen printed electrodes in electro-analysis. Compton's group observed enhanced electrocatalytic properties of CNT towards several target analytes attributed to the defect/edge plane-like sites [33,34]. Zen et al. demonstrated the creation of defect/edge-like sites and oxygen functionalities on the screen printed electrode surface through a simple pre-anodization process. Prasad et al. performed a comparative study on the role of oxygen functionalities and edge plane sites created achieved through a pre-anodization process and oxygen plasma treated screen printed electrodes [4]. Consequently, the pre-anodized electrode showed better characteristics in terms of reduction in overpotential and separation of oxidation peaks for the detection of uric acid and dopamine compared to the oxygen plasma-treated screen printed electrodes [35,36]. Similarly, the versatility of the pre-anodized screen printed electrode has been demonstrated in different media for enzymeless detection, detection of poorly electro-active analytes and direct electron transfer-related researches [37,38].
4.2. Mediator Integrated Screen Printed Electrode
There are many target analytes which have no significant electroactivity or near impossible to get electrochemical signals. In such cases, the use of an electrocatalyst mediator and methodologies to improve the sensor performance are very common. The appropriate selection of the mediator can improve the selectivity and lower the working potential for electrocatalysis. Redox mediators such as metal/metal complexes and pure organic polymers can be used as electrode modifiers. The viable approaches to immobilize the mediator on the screen-printed electrode include drop casting, physical attachment, and covalent binding or mixing into carbon paste. The mediator mixing approach has found more applications, and has been the mainly used technique for many decades, since the pioneering work of Adams et al., Various types of mediators such as Meldola's Blue, Prussian Blue, crown ethers, cobalt phthalocyanine and nickel hexacyanoferrate have been successfully integrated into the screen printed ink to design sensors for many target analytes. Ionic liquids have been used in analytical chemistry and carbon composites due to their physiochemical properties and biocompatible nature [39,40], but they were never been used in screen printed electrodes fabrication until the notable work by Ping et al., who incorporated a variety of ionic liquids into screen printed electrodes [41]. This work was further extended by Ren et al. in the fabrication of DNA sensors to achieve nano-level sensitivity [42]. Recently, carbon nanotubes-mediated screen printed electrodes have been used to increase the electrochemically active area of screen printed electrodes, subsequently employed in the detection of p-aminophenol [43]. This work has provided a base to use other carbon nanotubes such as single walled carbon nanotubes and multi-walled carbon nanotube derivatives in designing screen printed carbon electrodes, with the possibility of accessing mass produced and reproducible nanotube-modified screen printed electrodes.
4.3. Metal Oxide Based Screen Printed Electrodes
Despite their various advantages, mediator approaches have the drawback of instability, and the fact they are not easily mass produced reproducibly, posing the problem of optimization of the sensors. As an alternative, metal oxides, including ruthenium oxide, copper oxide, nickel oxide, manganese oxide and bismuth oxide, have been used in the modification of electrodes [8,44], and the subsequently modified electrodes were used for many sensing applications. Among all the oxides, bismuth oxide is well documented to enhance the electro-analytical performance of the sensors, with reduced toxic effects. Since the pioneering work of Wang et al., bismuth-modified electrodes have been extensively explored for diverse applications. Bismuth nanopowder immobilized with Nafion, and electrochemically oxidized bismuth oxides are normally used to obtain a uniform layer of bismuth on the electrode surface. The electrochemical deposition of the target species on the bismuth layer rather than the underlying graphite electrode results in the improved analytical performance of the sensors. Bismuth domains, as preferential nucleation sites, are distributed across the electrode surface producing their own diffusion zones and minimizing the effect of surface coverage which may result in an increased electron transfer resistance thus reducing the sensor sensitivity [45,46]. An advantageous approach for screen printed electrode modification is the one in which any metal oxide can be readily incorporated onto an electrode surface, allowing a true platform technology.
5. Screen Printed Sensors for Environmental Monitoring
Screen printed electrodes have been employed as a tool to design disposable and portable electrochemical sensors for environmental monitoring, such as water quality tests, organic compound analyses, heavy metals detection and gas pollutants (Figure 3).
5.1. Water Quality Tests
It is of vital importance to monitor source water and the aquatic systems that can be contaminated by industrial waste, sewage treatment plants and runoff from urban and agricultural lands. Water quality monitoring is mainly based on the measurement of the physical, chemical and bacteriological characteristics of water. The physical elements of analysis include monitoring of temperature, pH and conductivity, while chemical analyses measure oxygen, alkalinity, nitrogen and phosphorus compounds. Thousands of emerging contaminants may be present in water resources, including those used for drinking water production, therefore, the development of cost-effective devices for on-line and continuous monitoring of water quality is highly desirable. In this context, the use of screen printed electrodes to monitor pH changes has attracted great attention to replace the commonly used methods for testing pH changes. Koncki et al., designed a plastic, fully screen-printed, disposable pH sensor based on ruthenium dioxide by the application of a thick-film technology. The electrodes enable fast measurements, with good sensitivity in acidic and neutral solutions [47]. pH sensors with both printed reference and working electrodes on one substrate are reported in the literature [46], however this concept led to printing of the three electrode system for pH sensing with enhanced sensitivity as compared to the two electrode system [48]. Kampouris et al. designed a screen printed pH sensor by incorporating the pH sensitive phenanthraquinone moiety which undergoes a Nernstian potential shift with pH, and the pH insensitive dimethylferrocene one which acts as an internal reference. This generic approach offered a calibration-less and reproducible approach for portable pH measurements with the possibility of miniaturization, allowing incorporation into existing sensing devices [49]. Betelu et al., investigated the applicability of CeO2-based screen printed electrodes for monitoring the pH of the COx pore water. However, this study was limited to the pH range between 5.5 and 13.2 [50]. Xiong et al. designed a calibration-less sensor based on nitrosophenyl-modified edge plane pyrolytic electrodes and screen printed electrodes to monitor pH changes [51]. Dissolved oxygen concentration is also another parameter that can be employed to test water quality. Zen et al., developed an efficient photocatalytic amperometric sensor for the determination of dissolved oxygen in phosphate buffer solution using a disposable copper-plated screen-printed carbon electrode. Real sample assays for groundwater and tap water were also consistent with those measured by a commercial dissolved oxygen meter [52]. Various modified screen printed sensors have been demonstrated as potential candidates to measure the chemical oxygen demand and biochemical chemical oxygen demand for various environmental studies [53].
Nitrate is also an important analyte for environmental and human health monitoring thus its detection and quantification is very important. In this regards, some screen printed electrodes have been designed and used to detect low levels of toxic ions. Moreover, microelectrodes in combination with screen printing technology have been employed to measure the nitrate level in water samples [54,55]. For example, Lin et al., fabricated poly (3,4-ethylenedioxythiophene) and PEDOT/multi-wall carbon nanotubes (PEDOT/MWCNTs)-modified screen-printed carbon electrodes (SPCEs) and studied their catalytic properties for nitrite measurement. The developed sensor was also applied to the determination of nitrite concentration in tap water samples [56]. Monchindu et al., electropolymerised aniline doped with polyvinyl sulphonate on screen printed carbon electrodes. The designed electrochemical sensors exhibited good analytical characteristics for nitrate detection [57]. Lin et al., investigated the oxidative electrochemistry of nitrite on a poly(3,4-ethylenedioxythiophene)/iron phthalocyanine/multi-wall carbon nanotubes-modified screen-printed carbon electrode. The developed sensor was also applied for the determination of nitrite in tap water samples [58]. Metters et al. reported the fabrication of screen printed graphite micro-band electrodes which were electrochemically characterized and critically explored in electro-analytical applications for the sensing of nitrite [55] .Saljukis et al., fabricated manganese dioxide screen printed graphite electrodes for electro-analytical sensing purposes. The prepared sensors exhibited attractive performances as electrocatalysts for the sensing of nitrite ions with detection limits comparable or lower than those obtainable with other electrochemical sensors [59].
Similarly, the hydrophilic nature of phosphate ions makes them difficult to detect in water analysis. Ion selective electrodes, due to their ability to measure various species in turbid and colored medium, have been appeared as the prominent tools to measure phosphate ions for routine water sample analysis. Screen printed carbon paste and conventional PVC membrane electrodes have been integrated in ion selective sensors for phosphate ion analysis [60]. Some of the recently developed screen printed sensors for water quality are listed in Table 1.
5.2. Organic Compounds
Phenols are organic compounds broadly employed in the chemical, petrochemical, pharmaceutical, pesticide, pulp and paper, textile, metallurgic, resin and plastic, and pulp and paper industries. Phenol poisoning by skin absorption, inhalation of vapors or ingestion causes accumulation and damage to the brain, kidneys, liver, muscle, and eyes, as well as necrosis [21]. Therefore, detection of phenolic compounds and their derivatives is highly desirable to meet the corresponding environmental challenges. Even though the standardized methods are able to obtain accurate results for a wide range of phenolic compounds, conventional approaches are time-consuming and cost-intensive. Furthermore, they require large volumes of toxic organic solvents such as methylene chloride, acetone, and methanol. Consequently, there is a demand for the development of reliable, portable, sensitive, simple and cost-effective methods for the fast detection of phenolic compounds. Electrochemical sensors based on screen printed electrodes have been used as low cost, simple, sensitive and disposable tools for in situ monitoring of phenolic compounds. The possibility of direct electrochemical oxidation of these phenolic compounds at the screen printed electrode facilitates their detection [68], Many modification strategies and immobilization methods have been reported in the literature to design innovative electrochemical sensors for monitoring phenolic compounds [69–72].
Pesticides are released intentionally into the environment, and through various processes can contaminate the environment. Although pesticides are associated with many health hazards, there is a lack of monitoring of these contaminants. Traditional chromatographic methods—high-performance liquid chromatography, capillary electrophoresis, and mass spectrometry—are effective for the analysis of pesticides in the environment, but have certain limitations such as complexity, time-consuming sample preparation, and the requirement of expensive apparatus and trained persons to operate them. Over the past decades, acetylcholinesterase (AChE) inhibition-based biosensors have emerged as simple, rapid, and ultra-sensitive tools for pesticide analysis in environmental monitoring, food safety, and quality control. These biosensors have the potential to complement or replace the classical analytical methods by simplifying or eliminating sample preparation and making field-testing easier and faster with a significant decrease in cost per analysis [73,74]. Based on the inhibition mechanism of the pesticide, various electrochemical biosensors based on screen printed electrodes have been constructed to analyse water and soil samples for the presence of pesticides. Prussian Blue, carbon nanotubes, cobalt phthalocyanine and conductive polymers have been successfully integrated as mediators in screen printed electrochemical biosensors for pesticide detection [75–78].
Despite the use of modern, less persistent agrochemicals, herbicide residues and herbicide metabolites in water are a serious environmental problem. Even when used appropriately, water soluble herbicides can be found in surface waters, ground waters, and tap water. For this reason, the monitoring of herbicides and herbicide metabolites is important to ensure the quality of water. Electrochemical immunosensors based on screen printed carbon electrodes are used for single shot determination of herbicides, eliminating the cleaning and reuse of components [79,80]. However, the immunoassays undergo some drawbacks such as the time consuming antibody production process and the possibility of cross-reactivity. Alternatively, photosynthetic electrochemical biosensors based on screen printed electrodes have been proposed, and successfully implemented for herbicide detection [81–83]. Polycyclic aromatic hydrocarbons (PAHs) are a large group of organic compounds with two or more fused aromatic rings. They have a relatively low solubility in water, but are highly lipophilic. Aromatic compounds can interact with graphite walls and thus stack onto carbon materials through non-covalent binding. After concentrating polyaromatic hydrocarbons on screen printed electrodes, an operating potential can be applied for the individual electrochemical detection of a specific aromatic compound [84–86]. Similarly, immunosensor approaches based on screen printed electrodes have also been reported in the literature for a mixture of individual polyaromatic hydrocarbon compounds. Nevertheless, the very similar structures of polyaromatic hydrocarbon compounds make difficult the production of a specific antibody for only one polyaromatic hydrocarbons. Future work may focus on the integration of various antibodies within a single screen printed sensor to get different signals and detailed information regarding polyaromatic hydrocarbon mixtures. Sensitive and decentralized analysis of antibiotic residues in environmental samples is also high desirable. Tetracyclines are important classes of antibiotic that are detected by employing screen printed electrodes. Immunoassays in combination with nanoparticles on screen printed electrodes have shown great potential in the development of high sample throughput screening systems for antibiotics in environmental samples [87,88]. Table 2 provides examples of the some of the recently developed screen printed sensors for organic compounds (Table 2).
5.3. Heavy Metals
Due to the major negative impact of heavy metal ions toward human health and the environment, even at low concentrations, the development of simple, fast and not expensive detection methods for heavy metals is a major challenge for scientists. Among the different analytical methods for the analysis of heavy metal ions, the methods based on electrochemical sensors are widely applied for the detection of metals. Among toxic heavy metals, lead continues to be one of the most problematic. Despite considerable efforts to identify and eliminate Pb exposure sources, this metal still remains a significant health concern. Pb(II) is one of the heavy metals that has been detected with improved sensitivity by using modified carbon, bismuth, gold or other materials. These modifiers were integrated onto the surface of screen printed electrodes to make portable and disposable devices, improving their suitability for on-site analysis [100–103]. However, such approaches exhibit shortcomings such as necessity of acid or alkaline working media. Alternatively, mercury has the potential to perform analysis over a wide range of pH, and can be used as a possible screen printed electrode modifier for trace metal detections. As a proof of concept, mercury-modified screen printed electrodes have been employed to detect low levels of Cd(II) in environmental samples [104,105].
Mercury ions, the most stable form of inorganic mercury, are highly toxic environmental pollutants and have serious medical effects. Therefore, it is highly desirable to develop sensitive methods for the detection of Hg2+. Indeed, there have been numerous reports on optical Hg2+ detection by using Hg2+ sensitive fluorophores or chromophores, however, most of these fluorophores or chromophore-based Hg2+ sensors only work in organic media, which cannot be directly used to detect mercury contaminants in aqueous media. Bare gold or modified gold electrodes are normally used for the detection of Hg2+ due to their strong affinity for Hg2+ [106,107]. Commercial screen printed electrodes are reported for simple detection of Hg2+ in water samples. Nanostructured carbon black and screen printed electrodes modified with conducting polymer layers have also been designed for the trace level measurement of Hg2+ in water samples [108,109]. Arsenic is also a common compound found in drinking water, especially in some Asian countries. The toxicity of arsenic is greatly dependent on its oxidation state since As(III) is 50 times more toxic than arsenate due to its reactions with enzymes involved in human metabolism. Many detection methods have been developed for determination of such levels of arsenic. Among these methods, electrochemical methods provide accurate measurements of low concentrations of metal ions at ppb levels with rapid analysis times and low cost instrumentation. Screen printed electrodes modified with nanoparticles have been utilized to detect arsenic in water environments [110]. However, to avoid interferences from other metals, enzymatic biosensors based on screen printed electrodes for the measurement of arsenic in water samples have also been reported in the literature [111]. Designing screen printed sensors for simultaneous detection of various metals is also interesting for time and cost reasons. Screen printed electrodes modified with gold nanoparticles/gold films have also been reported for stripping voltammetric determination of mercury (II) and lead (II) [112]. Selected and recently developed screen printed sensors for heavy metals detection are listed in Table 3.
5.4. Gas Pollutants
The air pollution caused by exhaust gases from automobiles has become a critical issue. In some regions, fossil fuel combustion is a problem as well. The principal gases that cause air pollution from automobiles are nitrogen oxide and carbon monoxide. Conventional and traditional methods to detect the levels of toxic gases include color reactions, chemiluminescence and IR absorption approaches. In comparison to these described methods, electrochemical gas sensor based on screen printed electrodes can provide low cost, easy to use and portable devices for environmental analysis.
Carbon monoxide is a colorless, odorless, tasteless and poisonous gas mainly produced by the combustion of fossil fuels. Bare gold, nanoparticles particles-modified carbon and SnO2-modified carbon electrodes based on screen printing technology have been employed to detect the levels of this gas in environmental samples [130,131]. These devices have the potential to be used for in situ measurement and for continuous monitoring. Nitrogen oxide is a prominent air pollutant produced during high temperature combustion processes. The symptoms of nitrogen oxide poising appear several hours after its inhalation and require sensitive methods to detect it at low levels. Tin-doped and indium oxide thin films on screen printed electrodes have been used for the detection of nitrogen oxide in the air samples [132,133]. Volatile organic compounds including formaldehyde, acetone and methanol pose harmful effects to human health and contaminate the environment. Screen printed nanocomposite films integrated with multi-walled carbon nanotubes and silicon binders have been used to measure organic gases [134,135].
5.5. Other Environmental Pollutants
The presence of bacteria may also pose some enteric disease problems and indirectly results in economic losses. Enzyme-labeled and impedimetric immunoassays based on screen printed electrodes have been developed for bacterial detection in river and tap water samples without pre-concentration steps [136]. Screen printed micro-system have been designed for pathogen detection that serve as both functional and structural components, to improve the simplicity of the fabrication steps [137]. Radio-elements are also considered as radiological and chemical toxic compounds, with their presence in the aquatic system needs to be monitored. Screen printing technology has efficiently contributed in the detection of radioelements, and screen printed sensors to monitor uranium have been reported in the literature [138,139].
6. Conclusions and Future Prospects
As discussed in this review paper, there have been many exciting developments in the use of screen printing to design new types of electrochemical sensors. The combination of modern electrochemical systems with screen printing technology along with breakthroughs in micro-electronoics and miniaturization permits the introduction of powerful and potential analytical tools for effective monitoring of environmental pollutants. Such real time on-site monitoring methodologies have successfully addressed the time constraints associated with classical laboratory analysis. With the passage of time, electrochemical devices are becoming more and more sophisticated and versatile while dramatically shrinking in size and weight. Screen printed methodologies offer the advantage of production of simple, economical, disposable, portable and mass produced devices suitable for on-site analysis of environmental pollutants. Disposable screen printed electrodes have extensively improved the sensitivity and selectivity of the analytical approaches, especially in the detection of certain environmental analytes that were difficult and challenging to measure with conventional and traditional techniques. The field of screen printed electrodes, however, continues to grow and find new application domains. It is expected that future work shall focus on the integration of nanomaterials in the screen printed electrodes to improve the electron transfer rates, thus enhancing the analytical performance of the sensors. Furthermore, microchip formats may find application to improve the miniaturization process to decrease the analysis time, sample volumes and reagent consumption and enhance portability and for on-site analysis.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wang, J. Real-Time Electrochemical Monitoring: Toward Green Analytical Chemistry. Acc. Chem. Res. 2002, 35, 811–816. [Google Scholar]
- Anastas, P.T. Green Chemistry the Role of Analytical Methodology Development. Crit. Rev. Anal. Chem. 1999, 29, 167–175. [Google Scholar]
- Workman, J.; Creasy, K.E.; Doherty, S.; Bond, L.; Koch, M.; Ullman, A.; Veltkamp, D.J. Process Analytical Chemistry. Anal. Chem. 2001, 73, 2705–2718. [Google Scholar]
- Thiyagarajan, N.; Chang, J.-L.; Senthilkumar, K.; Zen, J.-M. Disposable electrochemical sensors: A mini review. Electrochem. Commun. 2014, 38, 86–90. [Google Scholar]
- Honeychurch, K.C.; Hart, J.P. Screen-printed electrochemical sensors for monitoring metal pollutants. TrAC, Trends Anal. Chem. 2003, 22, 456–469. [Google Scholar]
- Bakker, E.; Bühlmann, P.; Pretsch, E. Polymer Membrane Ion-Selective Electrodes—What are the Limits? Electroanalysis 1999, 11, 915–933. [Google Scholar]
- Wang, J. Remote electrochemical sensors for monitoring inorganic and organic pollutants. TrAC, Trends Anal. Chem. 1997, 16, 84–88. [Google Scholar]
- Metters, J.P.; Kadara, R.O.; Banks, C.E. New directions in screen printed electroanalytical sensors: An overview of recent developments. Analyst 2011, 136, 1067–1076. [Google Scholar]
- Li, M.; Li, Y.-T.; Li, D.-W.; Long, Y.-T. Recent developments and applications of screen-printed electrodes in environmental assays—A review. Anal. Chim. Acta 2012, 734, 31–44. [Google Scholar]
- Renedo, O.D.; Alonso-Lomillo, M.A.; Martínez, M.J.A. Recent developments in the field of screen-printed electrodes and their related applications. Talanta 2007, 73, 202–219. [Google Scholar]
- Wang, J.; Lu, J.; Hocevar, S.B.; Farias, P.A.M.; Ogorevc, B. Bismuth-Coated Carbon Electrodes for Anodic Stripping Voltammetry. Anal. Chem. 2000, 72, 3218–3222. [Google Scholar]
- Murray, R.W.; Ewing, A.G.; Durst, R.A. Chemically modified electrodes. Molecular design for electroanalysis. Anal. Chem. 1987, 59, 379A–390A. [Google Scholar]
- Hayat, A.; Yang, C.; Rhouati, A.; Marty, J. Recent Advances and Achievements in Nanomaterial-Based, and Structure Switchable Aptasensing Platforms for Ochratoxin A Detection. Sensors 2013, 13, 15187–15208. [Google Scholar]
- Hayat, A.; Andreescu, S.; Marty, J.-L. Design of PEG-aptamer two piece macromolecules as convenient and integrated sensing platform: Application to the label free detection of small size molecules. Biosens. Bioelectron. 2013, 45, 168–173. [Google Scholar]
- Hayat, A.; Haider, W.; Rolland, M.; Marty, J.-L. Electrochemical grafting of long spacer arms of hexamethyldiamine on a screen printed carbon electrode surface: Application in target induced ochratoxin A electrochemical aptasensor. Analyst 2013, 138, 2951–2957. [Google Scholar]
- Heller, A.; Feldman, B. Electrochemical Glucose Sensors and Their Applications in Diabetes Management. Chem. Rev. 2008, 108, 2482–2505. [Google Scholar]
- Newman, J.D.; Turner, A.P.F. Home blood glucose biosensors: A commercial perspective. Biosens. Bioelectron. 2005, 20, 2435–2453. [Google Scholar]
- Wilson, R.; Turner, A.P.F. Glucose oxidase: An ideal enzyme. Biosens. Bioelectron. 1992, 7, 165–185. [Google Scholar]
- Hart, J.P.; Wring, S.A. Recent developments in the design and application of screen-printed electrochemical sensors for biomedical, environmental and industrial analyses. TrAC, Trends Anal. Chem. 1997, 16, 89–103. [Google Scholar]
- Hayat, A.; Marty, J.-L.; Radi, A.-E. Novel Amperometric Hydrogen Peroxide Biosensor Based on Horseradish Peroxidase Azide Covalently Immobilized on Ethynyl-Modified Screen-Printed Carbon Electrode via Click Chemistry. Electroanalysis 2012, 24, 1446–1452. [Google Scholar]
- Su, W.-Y.; Wang, S.-M.; Cheng, S.-H. Electrochemically pretreated screen-printed carbon electrodes for the simultaneous determination of aminophenol isomers. J. Electroanal. Chem. 2011, 651, 166–172. [Google Scholar]
- Hayat, A.; Barthelmebs, L.; Marty, J.L. Electrochemical impedimetric immunosensor for the detection of okadaic acid in mussel sample. Sens. Actuators B Chem. 2012, 171, 810–815. [Google Scholar]
- Hayat, A.; Sassolas, A.; Marty, J.L.; Radi, A. Highly sensitive ochratoxin A impedimetric aptasensor based on the immobilization of azido-aptamer onto electrografted binary film via click chemistry. Talanta 2013, 103, 14–19. [Google Scholar]
- Fan, Z.; Ho, J.C.; Takahashi, T.; Yerushalmi, R.; Takei, K.; Ford, A.C.; Chueh, Y.-L.; Javey, A. Toward the Development of Printable Nanowire Electronics and Sensors. Adv. Mater. 2009, 21, 3730–3743. [Google Scholar]
- Khairy, M.; Kadara, R.O.; Banks, C.E. Electroanalytical sensing of nitrite at shallow recessed screen printed microelectrode arrays. Anal. Methods 2010, 2, 851–854. [Google Scholar]
- Kadara, R.O.; Jenkinson, N.; Banks, C.E. Screen printed recessed microelectrode arrays. Sens. Actuators B 2009, 142, 342–346. [Google Scholar]
- Fanjul-Bolado, P.; Hernández-Santos, D.; Lamas-Ardisana, P.J.; Martín-Pernía, A.; Costa-García, A. Electrochemical characterization of screen-printed and conventional carbon paste electrodes. Electrochim. Acta 2008, 53, 3635–3642. [Google Scholar]
- Šljukić, B.; Malakhova, N.A.; Brainina, K.Z.; Banks, C.E.; Compton, R.G. Screen Printed Electrodes and Screen Printed Modified Electrodes Benefit from Insonation. Electroanalysis 2006, 18, 928–930. [Google Scholar]
- Choudhry, N.A.; Kampouris, D.K.; Kadara, R.O.; Jenkinson, N.; Banks, C.E. Next generation screen printed electrochemical platforms: Non-enzymatic sensing of carbohydrates using copper (ii) oxide screen printed electrodes. Anal. Methods 2009, 1, 183–187. [Google Scholar]
- Choudhry, N.A.; Kadara, R.O.; Jenkinson, N.; Banks, C.E. Screen printed electrodes provide micro-domain sites for fabricating disposable electro-catalytic ensembles. Electrochem. Commun. 2010, 12, 406–409. [Google Scholar]
- Davies, T.J.; Hyde, M.E.; Compton, R.G. Nanotrench Arrays Reveal Insight into Graphite Electrochemistry. Angew. Chem. 2005, 117, 5251–5256. [Google Scholar]
- Banks, C.E.; Compton, R.G. New electrodes for old: From carbon nanotubes to edge plane pyrolytic graphite. Analyst 2006, 131, 15–21. [Google Scholar]
- Banks, C.E.; Crossley, A.; Salter, C.; Wilkins, S.J.; Compton, R.G. Carbon Nanotubes Contain Metal Impurities Which Are Responsible for the “Electrocatalysis” Seen at Some Nanotube-Modified Electrodes. Angew. Chem. Int. Ed. 2006, 45, 2533–2537. [Google Scholar]
- Shi, K.; Shiu, K.-K. Determination of Uric Acid at Electrochemically Activated Glassy Carbon Electrode. Electroanalysis 2001, 13, 1319–1325. [Google Scholar]
- Chen, J.C.; Chung, H.H.; Hsu, C.T.; Tsai, D.M.; Kumar, A.S.; Zen, J.M. A disposable single-use electrochemical sensor for the detection of uric acid in human whole blood. Sens. Actuators B 2005, 110, 364–369. [Google Scholar]
- Tsai, T.-H.; Thiagarajan, S.; Chen, S.-M. Detection of Melamine in Milk Powder and Human Urine. J. Agric. Food Chem. 2010, 58, 4537–4544. [Google Scholar]
- Yang, T.-H.; Hung, C.-L.; Ke, J.-H.; Zen, J.-M. An electrochemically preanodized screen-printed carbon electrode for achieving direct electron transfer to glucose oxidase. Electrochem. Commun. 2008, 10, 1094–1097. [Google Scholar]
- Zen, J.-M.; SenthillKumar, A.; Tsai, D.-M. Recent Updates of Chemically Modified Electrodes in Analytical Chemistry. Electroanalysis 2003, 15, 1073–1087. [Google Scholar]
- Kozub, B.R.; Compton, R.G. Voltammetric studies of the redox mediator, cobalt phthalocyanine, with regard to its claimed electrocatalytic properties. Sens. Actuators B 2010, 147, 350–358. [Google Scholar]
- Ping, J.; Wu, J.; Ying, Y. Development of an ionic liquid modified screen-printed graphite electrode and its sensing in determination of dopamine. Electrochem. Commun. 2010, 12, 1738–1741. [Google Scholar]
- Ren, R.; Leng, C.; Zhang, S. A chronocoulometric DNA sensor based on screen-printed electrode doped with ionic liquid and polyaniline nanotubes. Biosens. Bioelectron. 2010, 25, 2089–2094. [Google Scholar]
- Lamas-Ardisana, P.J.; Queipo, P.; Fanjul-Bolado, P.; Costa-García, A. Multiwalled carbon nanotube modified screen-printed electrodes for the detection of p-aminophenol: Optimisation and application in alkaline phosphatase-based assays. Anal. Chim. Acta 2008, 615, 30–38. [Google Scholar]
- Wang, J.; Naser, N.; Angnes, L.; Wu, H.; Chen, L. Metal-dispersed carbon paste electrodes. Anal. Chem. 1992, 64, 1285–1288. [Google Scholar]
- Wang, J. Stripping Analysis at Bismuth Electrodes: A Review. Electroanalysis 2005, 17, 1341–1346. [Google Scholar]
- Economou, A. Bismuth-film electrodes: Recent developments and potentialities for electroanalysis. TrAC, Trends Anal. Chem. 2005, 24, 334–340. [Google Scholar]
- Müller, A.; Brinz, T.; Simon, U. Preparation and Measurement of Combinatorial Screen Printed Libraries for the Electrochemical Analysis of Liquids. J. Comb. Chem. 2008, 11, 138–142. [Google Scholar]
- Koncki, R.; Mascini, M. Screen-printed ruthenium dioxide electrodes for pH measurements. Anal. Chim. Acta 1997, 351, 143–149. [Google Scholar]
- Hallam, P.M.; Kampouris, D.K.; Kadara, R.O.; Jenkinson, N.; Banks, C.E. Nickel oxide screen printed electrodes for the sensing of hydroxide ions in aqueous solutions. Anal. Methods 2010, 2, 1152–1155. [Google Scholar]
- Kampouris, D.K.; Kadara, R.O.; Jenkinson, N.; Banks, C.E. Screen printed electrochemical platforms for pH sensing. Anal. Methods 2009, 1, 25–28. [Google Scholar]
- Betelu, S.; Polychronopoulou, K.; Rebholz, C.; Ignatiadis, I. Novel CeO2-based screen-printed potentiometric electrodes for pH monitoring. Talanta 2011, 87, 126–135. [Google Scholar]
- Xiong, L.; Batchelor-McAuley, C.; Compton, R.G. Calibrationless pH sensors based on nitrosophenyl and ferrocenyl co-modified screen printed electrodes. Sens. Actuators B 2011, 159, 251–255. [Google Scholar]
- Zen, J.-M.; Song, Y.-S.; Chung, H.-H.; Hsu, C.-T.; Senthil Kumar, A. Photoelectrochemical Oxygen Sensor Using Copper-Plated Screen-Printed Carbon Electrodes. Anal. Chem. 2002, 74, 6126–6130. [Google Scholar]
- Zheng, R.-J.; Fang, Y.-M.; Qin, S.-F.; Song, J.; Wu, A.-H.; Sun, J.-J. A dissolved oxygen sensor based on hot electron induced cathodic electrochemiluminescence at a disposable CdS modified screen-printed carbon electrode. Sens. Actuators B 2011, 157, 488–493. [Google Scholar]
- Chang, J.-L.; Zen, J.-M. A poly(dimethylsiloxane)-based electrochemical cell coupled with disposable screen printed edge band ultramicroelectrodes for use in flow injection analysis. Electrochem. Commun. 2007, 9, 2744–2750. [Google Scholar]
- Metters, J.P.; Kadara, R.O.; Banks, C.E. Electroanalytical properties of screen printed graphite microband electrodes. Sens. Actuators B 2012, 169, 136–143. [Google Scholar]
- Lin, C.-Y.; Vasantha, V.S.; Ho, K.-C. Detection of nitrite using poly(3,4-ethylenedioxythiophene) modified SPCEs. Sens. Actuators B 2009, 140, 51–57. [Google Scholar]
- Muchindu, M.; Waryo, T.; Arotiba, O.; Kazimierska, E.; Morrin, A.; Killard, A.J.; Smyth, M.R.; Jahed, N.; Kgarebe, B.; Baker, P.G.L.; et al. Electrochemical nitrite nanosensor developed with amine- and sulphate-functionalised polystyrene latex beads self-assembled on polyaniline. Electrochimica Acta 2010, 55, 4274–4280. [Google Scholar]
- Lin, C.-Y.; Balamurugan, A.; Lai, Y.-H.; Ho, K.-C. A novel poly(3,4-ethylenedioxythiophene)/iron phthalocyanine/multi-wall carbon nanotubes nanocomposite with high electrocatalytic activity for nitrite oxidation. Talanta 2010, 82, 1905–1911. [Google Scholar]
- Sljukic, B.R.; Kadara, R.O.; Banks, C.E. Disposable manganese oxide screen printed electrodes for electroanalytical sensing. Anal. Methods 2011, 3, 105–109. [Google Scholar]
- Khaled, E.; Hassan, H.N.A.; Girgis, A.; Metelka, R. Construction of novel simple phosphate screen-printed and carbon paste ion-selective electrodes. Talanta 2008, 77, 737–743. [Google Scholar]
- Muller, A.; Brinz, T.; Simon, U. Preparation and measurement of combinatorial screen printed libraries for the electrochemical analysis of liquids. J. Comb. Chem. 2009, 11, 138–42. [Google Scholar]
- Malha, S.I.R.; Mandli, J.; Ourari, A.; Amine, A. Carbon Black-Modified Electrodes as Sensitive Tools for the Electrochemical Detection of Nitrite and Nitrate. Electroanalysis 2013, 25, 2289–2297. [Google Scholar]
- Gilbert, L.; Jenkins, A.T.; Browning, S.; Hart, J.P. Development of an amperometric assay for phosphate ions in urine based on a chemically modified screen-printed carbon electrode. Anal. Biochem. 2009, 393, 242–247. [Google Scholar]
- Gilbert, L.; Jenkins, A.T.A.; Browning, S.; Hart, J.P. Development of an amperometric, screen-printed, single-enzyme phosphate ion biosensor and its application to the analysis of biomedical and environmental samples. Sens. Actuators B 2011, 160, 1322–1327. [Google Scholar]
- Karousos, N.; Chong, L.C.; Ewen, C.; Livingstone, C.; Davis, J. Evaluation of a multifunctional indicator for the electroanalytical determination of nitrite. Electrochim. Acta 2005, 50, 1879–1884. [Google Scholar]
- Quan, D.; Shim, J.H.; Kim, J.D.; Park, H.S.; Cha, G.S.; Nam, H. Electrochemical Determination of Nitrate with Nitrate Reductase-Immobilized Electrodes under Ambient Air. Anal. Chem. 2005, 77, 4467–4473. [Google Scholar]
- Plumeré, N.; Henig, J.; Campbell, W.H. Enzyme-Catalyzed O2 Removal System for Electrochemical Analysis under Ambient Air: Application in an Amperometric Nitrate Biosensor. Anal. Chem. 2012, 84, 2141–2146. [Google Scholar]
- Brugnera, M.F.; Trindade, M.A.G.; Zanoni, M.V.B. Detection of Bisphenol A on a Screen-Printed Carbon Electrode in CTAB Micellar Medium. Anal. Lett. 2010, 43, 2823–2836. [Google Scholar]
- Li, D.; Li, D.-W.; Fossey, J.S.; Long, Y.-T. Portable Surface-Enhanced Raman Scattering Sensor for Rapid Detection of Aniline and Phenol Derivatives by On-Site Electrostatic Preconcentration. Anal. Chem. 2010, 82, 9299–9305. [Google Scholar]
- Song, W.; Li, D.-W.; Li, Y.-T.; Li, Y.; Long, Y.-T. Disposable biosensor based on graphene oxide conjugated with tyrosinase assembled gold nanoparticles. Biosens. Bioelectron. 2011, 26, 3181–3186. [Google Scholar]
- Alkasir, R.S.J.; Ganesana, M.; Won, Y.-H.; Stanciu, L.; Andreescu, S. Enzyme functionalized nanoparticles for electrochemical biosensors: A comparative study with applications for the detection of bisphenol A. Biosens. Bioelectron. 2010, 26, 43–49. [Google Scholar]
- Ibarra-Escutia, P.; Gómez, J.J.; Calas-Blanchard, C.; Marty, J.L.; Ramírez-Silva, M.T. Amperometric biosensor based on a high resolution photopolymer deposited onto a screen-printed electrode for phenolic compounds monitoring in tea infusions. Talanta 2010, 81, 1636–1642. [Google Scholar]
- Ben Oujji, N.; Bakas, I.; Istamboulié, G.; Ait-Ichou, I.; Ait-Addi, E.; Rouillon, R.; Noguer, T. Acetylcholinesterase Immobilized on Magnetic Beads for Pesticides Detection: Application to Olive Oil Analysis. Sensors 2012, 12, 7893–7904. [Google Scholar]
- Gan, N.; Yang, X.; Xie, D.; Wu, Y.; Wen, W. A Disposable Organophosphorus Pesticides Enzyme Biosensor Based on Magnetic Composite Nano-Particles Modified Screen Printed Carbon Electrode. Sensors 2010, 10, 625–638. [Google Scholar]
- Istamboulie, G.; Sikora, T.; Jubete, E.; Ochoteco, E.; Marty, J.-L.; Noguer, T. Screen-printed poly(3,4-ethylenedioxythiophene) (PEDOT): A new electrochemical mediator for acetylcholinesterase-based biosensors. Talanta 2010, 82, 957–961. [Google Scholar]
- Won, Y.-H.; Jang, H.S.; Kim, S.M.; Stach, E.; Ganesana, M.; Andreescu, S.; Stanciu, L.A. Biomagnetic Glasses: Preparation, Characterization, and Biosensor Applications. Langmuir 2009, 26, 4320–4326. [Google Scholar]
- Arduini, F.; Ricci, F.; Tuta, C.S.; Moscone, D.; Amine, A.; Palleschi, G. Detection of carbamic and organophosphorous pesticides in water samples using a cholinesterase biosensor based on Prussian Blue-modified screen-printed electrode. Anal. Chim. Acta 2006, 580, 155–162. [Google Scholar]
- Kumar, J.; D'Souza, S.F. Microbial biosensor for detection of methyl parathion using screen printed carbon electrode and cyclic voltammetry. Biosens. Bioelectron. 2011, 26, 4289–4293. [Google Scholar]
- Baskeyfield, D.E.H.; Davis, F.; Magan, N.; Tothill, I.E. A membrane-based immunosensor for the analysis of the herbicide isoproturon. Anal. Chim. Acta 2011, 699, 223–231. [Google Scholar]
- Nangia, Y.; Bhalla, V.; Kumar, B.; Suri, C.R. Electrochemical stripping voltammetry of gold ions for development of ultra-sensitive immunoassay for chlorsulfuron. Electrochem. Commun. 2012, 14, 51–54. [Google Scholar]
- Bhalla, V.; Zazubovich, V. Self-assembly and sensor response of photosynthetic reaction centers on screen-printed electrodes. Anal. Chim. Acta 2011, 707, 184–190. [Google Scholar]
- Shitanda, I.; Takamatsu, S.; Watanabe, K.; Itagaki, M. Amperometric screen-printed algal biosensor with flow injection analysis system for detection of environmental toxic compounds. Electrochim. Acta 2009, 54, 4933–4936. [Google Scholar]
- Zamaleeva, A.I.; Sharipova, I.R.; Shamagsumova, R.V.; Ivanov, A.N.; Evtugyn, G.A.; Ishmuchametova, D.G.; Fakhrullin, R.F. A whole-cell amperometric herbicide biosensor based on magnetically functionalised microalgae and screen-printed electrodes. Anal. Methods 2011, 3, 509–513. [Google Scholar]
- Leyton, P.; Gómez-Jeria, J.S.; Sanchez-Cortes, S.; Domingo, C.; Campos-Vallette, M. Carbon Nanotube Bundles as Molecular Assemblies for the Detection of Polycyclic Aromatic Hydrocarbons: Surface-Enhanced Resonance Raman Spectroscopy and Theoretical Studies. J. Phys. Chem. B 2006, 110, 6470–6474. [Google Scholar]
- Honeychurch, K.C.; Hart, J.P.; Kirsch, N. Voltammetric, chromatographic and mass spectral elucidation of the redox reactions of 1-hydroxypyrene occurring at a screen-printed carbon electrode. Electrochim. Acta 2004, 49, 1141–1149. [Google Scholar]
- Fähnrich, K.A.; Pravda, M.; Guilbault, G.G. Disposable amperometric immunosensor for the detection of polycyclic aromatic hydrocarbons (PAHs) using screen-printed electrodes. Biosens. Bioelectron. 2003, 18, 73–82. [Google Scholar]
- Masawat, P.; Slater, J.M. The determination of tetracycline residues in food using a disposable screen-printed gold electrode (SPGE). Sens. Actuators B 2007, 124, 127–132. [Google Scholar]
- Centi, S.; Stoica, A.I.; Laschi, S.; Mascini, M. Development of an Electrochemical Immunoassay Based on the Use of an Eight-Electrodes Screen-Printed Array Coupled with Magnetic Beads for the Detection of Antimicrobial Sulfonamides in Honey. Electroanalysis 2010, 22, 1881–1888. [Google Scholar]
- Crew, A.; Lonsdale, D.; Byrd, N.; Pittson, R.; Hart, J.P. A screen-printed, amperometric biosensor array incorporated into a novel automated system for the simultaneous determination of organophosphate pesticides. Biosens. Bioelectron. 2011, 26, 2847–2851. [Google Scholar]
- Arduini, F.; Guidone, S.; Amine, A.; Palleschi, G.; Moscone, D. Acetylcholinesterase biosensor based on self-assembled monolayer-modified gold-screen printed electrodes for organophosphorus insecticide detection. Sens. Actuators B 2013, 179, 201–208. [Google Scholar]
- Alonso, G.A.; Muñoz, R.; Marty, J.-L. Automatic Electronic Tongue for On-Line Detection and Quantification of Organophosphorus and Carbamate Pesticides Using Enzymatic Screen Printed Biosensors. Anal. Lett. 2012, 46, 1743–1757. [Google Scholar]
- Ivanov, A.N.; Younusov, R.R.; Evtugyn, G.A.; Arduini, F.; Moscone, D.; Palleschi, G. Acetylcholinesterase biosensor based on single-walled carbon nanotubes—Co phtalocyanine for organophosphorus pesticides detection. Talanta 2011, 85, 216–221. [Google Scholar]
- Li, H.; Li, J.; Yang, Z.; Xu, Q.; Hu, X. A novel photoelectrochemical sensor for the organophosphorus pesticide dichlofenthion based on nanometer-sized titania coupled with a screen-printed electrode. Anal. Chem. 2011, 83, 5290–5295. [Google Scholar]
- Mayorga-Martinez, C.C.; Cadevall, M.; Guix, M.; Ros, J.; Merkoci, A. Bismuth nanoparticles for phenolic compounds biosensing application. Biosens. Bioelectron. 2013, 40, 57–62. [Google Scholar]
- Mayorga, C.; Pino, F.; Kurbanoglua, S.; Rivas, L.; Ozkan, S.A.; Merkoci, A. Iridium oxide nanoparticles induced dual catalytic/inhibition based detection of phenol and pesticide compounds. J. Mater. Chem. B 2014, 2, 2233–2239. [Google Scholar]
- Nadifiyine, S.; Haddam, M.; Mandli, J.; Chadel, S.; Blanchard, C.C.; Marty, J.L.; Amine, A. Amperometric Biosensor Based on Tyrosinase Immobilized on to a Carbon Black Paste Electrode for Phenol Determination in Olive Oil. Anal. Lett. 2013, 46, 2705–2726. [Google Scholar]
- Lu, L.; Zhang, L.; Zhang, X.; Huan, S.; Shen, G.; Yu, R. A novel tyrosinase biosensor based on hydroxyapatite–chitosan nanocomposite for the detection of phenolic compounds. Anal. Chim. Acta 2010, 665, 146–151. [Google Scholar]
- Moczko, E.; Istamboulie, G.; Calas-Blanchard, C.; Rouillon, R.; Noguer, T. Biosensor employing screen-printed PEDOT:PSS for sensitive detection of phenolic compounds in water. J. Polym. Sci. Part A 2012, 50, 2286–2292. [Google Scholar]
- Chai, Y.; Niu, X.; Chen, C.; Zhao, H.; Lan, M. Carbamate Insecticide Sensing Based on Acetylcholinesterase/Prussian Blue-Multi-Walled Carbon Nanotubes/Screen-Printed Electrodes. Anal. Lett. 2013, 46, 803–817. [Google Scholar]
- Arduini, F.; Calvo, J.Q.; Palleschi, G.; Moscone, D.; Amine, A. Bismuth-modified electrodes for lead detection. TrAC, Trends Anal. Chem. 2010, 29, 1295–1304. [Google Scholar]
- Krystofova, O.; Trnkova, L.; Adam, V.; Zehnalek, J.; Hubalek, J.; Babula, P.; Kizek, R. Electrochemical Microsensors for the Detection of Cadmium(II) and Lead(II) Ions in Plants. Sensors 2010, 10, 5308–5328. [Google Scholar]
- Honeychurch, K.C.; Al-Berezanchi, S.; Hart, J.P. The voltammetric behaviour of lead at a microband screen-printed carbon electrode and its determination in acetate leachates from glazed ceramic plates. Talanta 2011, 84, 717–723. [Google Scholar]
- Mazumdar, D.; Liu, J.; Lu, G.; Zhou, J.; Lu, Y. Easy-to-use dipstick tests for detection of lead in paints using non-cross-linked gold nanoparticle-DNAzyme conjugates. Chem. Commun. 2010, 46, 1416–1418. [Google Scholar]
- Zaouak, O.; Authier, L.; Cugnet, C.; Castetbon, A.; Potin-Gautier, M. Electroanalytical Device for Cadmium Speciation in Waters. Part 1: Development and Characterization of a Reliable Screen-Printed Sensor. Electroanalysis 2010, 22, 1151–1158. [Google Scholar]
- Betelu, S.; Parat, C.; Petrucciani, N.; Castetbon, A.; Authier, L.; Potin-Gautier, M. Semicontinuous Monitoring of Cadmium and Lead with a Screen-Printed Sensor Modified by a Membrane. Electroanalysis 2007, 19, 399–402. [Google Scholar]
- Giacomino, A.; Abollino, O.; Malandrino, M.; Mentasti, E. Parameters affecting the determination of mercury by anodic stripping voltammetry using a gold electrode. Talanta 2008, 75, 266–273. [Google Scholar]
- Gong, J.; Zhou, T.; Song, D.; Zhang, L.; Hu, X. Stripping Voltammetric Detection of Mercury(II) Based on a Bimetallic Au–Pt Inorganic–Organic Hybrid Nanocomposite Modified Glassy Carbon Electrode. Anal. Chem. 2009, 82, 567–573. [Google Scholar]
- Bernalte, E.; Sánchez, C.M.; Gil, E.P. Determination of mercury in ambient water samples by anodic stripping voltammetry on screen-printed gold electrodes. Anal. Chim. Acta 2011, 689, 60–64. [Google Scholar]
- Somerset, V.S.; Hernandez, L.H.; Iwuoha, E.I. Stripping voltammetric measurement of trace metal ions using screen-printed carbon and modified carbon paste electrodes on river water from the Eerste-Kuils River System. J. Environ. Sci. Health Part A 2010, 46, 17–32. [Google Scholar]
- Sanllorente-Méndez, S.; Domínguez-Renedo, O.; Arcos-Martínez, M.J. Determination of Arsenic(III) Using Platinum Nanoparticle-Modified Screen-Printed Carbon-Based Electrodes. Electroanalysis 2009, 21, 635–639. [Google Scholar]
- Sanllorente-Méndez, S.; Domínguez-Renedo, O.; Arcos-Martínez, M.J. Immobilization of Acetylcholinesterase on Screen-Printed Electrodes. Application to the Determination of Arsenic(III). Sensors 2010, 10, 2119–2128. [Google Scholar]
- Mandil, A.; Idrissi, L.; Amine, A. Stripping voltammetric determination of mercury(II) and lead(II) using screen-printed electrodes modified with gold films, and metal ion preconcentration with thiol-modified magnetic particles. Microchim. Acta 2010, 170, 299–305. [Google Scholar]
- Maczuga, M.; Economou, A.; Bobrowski, A.; Prodromidis, M.I. Novel screen-printed antimony and tin voltammetric sensors for anodic stripping detection of Pb(II) and Cd(II). Electrochim. Acta 2013, 114, 758–765. [Google Scholar]
- Andreuccetti, C.; Bettazzi, F.; Giorgi, C.; Laschi, S.; Marrazza, G.; Mascini, M.; Palchetti, I. Macrocyclic Polyamine Modified Screen-Printed Electrodes for Copper(II) Detection. In Sensors; Baldini, F., D'Amico, A., Di Natale, C., Siciliano, P., Seeber, R., De Stefano, L., Bizzarri, R., Andò, B., Eds.; Springer: New York, NY, USA, 2014; Volume 162, pp. 471–474. [Google Scholar]
- Bouden, S.; Bellakhal, N.; Chaussé, A.; Vautrin-Ul, C. Performances of carbon-based screen-printed electrodes modified by diazonium salts with various carboxylic functions for trace metal sensors. Electrochem. Commun. 2014, 41, 68–71. [Google Scholar]
- Chen, C.; Niu, X.; Chai, Y.; Zhao, H.; Lan, M. Bismuth-based porous screen-printed carbon electrode with enhanced sensitivity for trace heavy metal detection by stripping voltammetry. Sens. Actuators B 2013, 178, 339–342. [Google Scholar]
- Jian, J.-M.; Liu, Y.-Y.; Zhang, Y.-L.; Guo, X.-S.; Cai, Q. Fast and Sensitive Detection of Pb2+ in Foods Using Disposable Screen-Printed Electrode Modified by Reduced Graphene Oxide. Sensors 2013, 13, 13063–13075. [Google Scholar]
- Fu, L.; Li, X.; Yu, J.; Ye, J. Facile and Simultaneous Stripping Determination of Zinc, Cadmium and Lead on Disposable Multiwalled Carbon Nanotubes Modified Screen-Printed Electrode. Electroanalysis 2013, 25, 567–572. [Google Scholar]
- Wei, Y.; Yang, R.; Liu, J.-H.; Huang, X.-J. Selective detection toward Hg(II) and Pb(II) using polypyrrole/carbonaceous nanospheres modified screen-printed electrode. Electrochim. Acta 2013, 105, 218–223. [Google Scholar]
- Gich, M.; Fernandez-Sanchez, C.; Cotet, L.C.; Niu, P.; Roig, A. Facile synthesis of porous bismuth-carbon nanocomposites for the sensitive detection of heavy metals. J. Mater. Chem. A 2013, 1, 11410–11418. [Google Scholar]
- Chen, C.; Niu, X.; Chai, Y.; Zhao, H.; Lan, M.; Zhu, Y.; Wei, G. Determination of Lead(II) Using Screen-Printed Bismuth-Antimony Film Electrode. Electroanalysis 2013, 25, 1446–1452. [Google Scholar]
- Bouden, S.; Chaussé, A.; Dorbes, S.; El Tall, O.; Bellakhal, N.; Dachraoui, M.; Vautrin-Ul, C. Trace lead analysis based on carbon-screen-printed-electrodes modified via 4-carboxy-phenyl diazonium salt electroreduction. Talanta 2013, 106, 414–421. [Google Scholar]
- Punrat, E.; Chuanuwatanakul, S.; Kaneta, T.; Motomizu, S.; Chailapakul, O. Method development for the determination of arsenic by sequential injection/anodic stripping voltammetry using long-lasting gold-modified screen-printed carbon electrode. Talanta 2013, 116, 1018–1025. [Google Scholar]
- Khairy, M.; Kampouris, D.K.; Kadara, R.O.; Banks, C.E. Gold Nanoparticle Modified Screen Printed Electrodes for the Trace Sensing of Arsenic(III) in the Presence of Copper(II). Electroanalysis 2010, 22, 2496–2501. [Google Scholar]
- Aragay, G.; Pons, J.; Merkoci, A. Enhanced electrochemical detection of heavy metals at heated graphite nanoparticle-based screen-printed electrodes. J. Mater. Chem. 2011, 21, 4326–4331. [Google Scholar]
- Bernalte, E.; Marín Sánchez, C.; Pinilla Gil, E. Gold nanoparticles-modified screen-printed carbon electrodes for anodic stripping voltammetric determination of mercury in ambient water samples. Sens. Actuators B 2012, 161, 669–674. [Google Scholar]
- Song, W.; Zhang, L.; Shi, L.; Li, D.-W.; Li, Y.; Long, Y.-T. Simultaneous determination of cadmium(II), lead(II) and copper(II) by using a screen-printed electrode modified with mercury nano-droplets. Microchim. Acta 2010, 169, 321–326. [Google Scholar]
- Fang, H.-L.; Zheng, H.-X.; Ou, M.-Y.; Meng, Q.; Fan, D.-H.; Wang, W. One-step sensing lead in surface waters with screen printed electrode. Sens. Actuators B 2011, 153, 369–372. [Google Scholar]
- Henríquez, C.; Laglera, L.M.; Alpizar, M.J.; Calvo, J.; Arduini, F.; Cerdà, V. Cadmium determination in natural water samples with an automatic multisyringe flow injection system coupled to a flow-through screen printed electrode. Talanta 2012, 96, 140–146. [Google Scholar]
- Morata, A.; Viricelle, J.P.; Tarancón, A.; Dezanneau, G.; Pijolat, C.; Peiro, F.; Morante, J.R. Development and characterisation of a screen-printed mixed potential gas sensor. Sens. Actuators B 2008, 130, 561–566. [Google Scholar]
- Chou, C.-H.; Chang, J.-L.; Zen, J.-M. Effective analysis of gaseous formaldehyde based on a platinum-deposited screen-printed edge band ultramicroelectrode coated with Nafion as solid polymer electrolyte. Sens. Actuators B 2010, 147, 669–675. [Google Scholar]
- Fergus, J.W. Materials for high temperature electrochemical NOx gas sensors. Sens. Actuators B 2007, 121, 652–663. [Google Scholar]
- Mbarek, H.; Saadoun, M.; Bessaïs, B. Porous screen printed indium tin oxide (ITO) for NOx gas sensing. Phys. Status Solidi C 2007, 4, 1903–1907. [Google Scholar]
- Khadayate, R.S.; Sali, J.V.; Patil, P.P. Acetone vapor sensing properties of screen printed WO3 thick films. Talanta 2007, 72, 1077–1081. [Google Scholar]
- Cantalini, C.; Valentini, L.; Lozzi, L.; Armentano, I.; Kenny, J.M.; Santucci, S. NO2 gas sensitivity of carbon nanotubes obtained by plasma enhanced chemical vapor deposition. Sens. Actuators B 2003, 93, 333–337. [Google Scholar]
- Lin, Y.-H.; Chen, S.-H.; Chuang, Y.-C.; Lu, Y.-C.; Shen, T.Y.; Chang, C.A.; Lin, C.-S. Disposable amperometric immunosensing strips fabricated by Au nanoparticles-modified screen-printed carbon electrodes for the detection of foodborne pathogen Escherichia coli O157:H7. Biosens. Bioelectron. 2008, 23, 1832–1837. [Google Scholar]
- Mata, D.; Bejarano, D.; Botero, M.L.; Lozano, P.; Constantí, M.; Katakis, I. Screen-printed integrated microsystem for the electrochemical detection of pathogens. Electrochim. Acta 2010, 55, 4261–4266. [Google Scholar]
- Kostaki, V.T.; Florou, A.B.; Prodromidis, M.I. Electrochemically induced chemical sensor properties in graphite screen-printed electrodes: The case of a chemical sensor for uranium. Electrochim. Acta 2011, 56, 8857–8860. [Google Scholar]
- Betelu, S.; Vautrin-Ul, C.; Ly, J.; Chaussé, A. Screen-printed electrografted electrode for trace uranium analysis. Talanta 2009, 80, 372–376. [Google Scholar]

| Analyte | Modifier | Detection Method | Ref. |
|---|---|---|---|
| Liquids | Iridium and ruthenium oxide | pH sensor | [61] |
| Liquids | Phenanthraquinone moiety | pH sensor | [49] |
| Hydroxide ions | Nickel oxide bulk | pH sensor | [48] |
| Dissolved oxygen | CdS modified | Cathodic electrochemiluminescenc | [53] |
| Nitrite | Poly(dimethylsiloxane) | Amperometric detection | [54] |
| Nitrite | Shallow recessed unmodified | Amperometric detection | [25] |
| Phosphate | Bisthiourea ionophores | Amperometric detection | [60] |
| Nitrite | Carbon Black | Multi-electrochemical methods | [62] |
| Phosphate | Electrocatalyst cobalt phthalocyanine | Amperometric | [63] |
| Phosphate | Cobalt phthalocyanine | Amperometric | [64] |
| Nitrate | Modified screen printed electrodes | Electrochemical detection | [65] |
| Nitrate | polymer (poly(vinyl alcohol)) modified | Amperometric | [66] |
| Nitrate | commercial screen-printed electrochemical cell | Amperometric | [67] |
| Analyte | Modifier | Detection Method | Ref. |
|---|---|---|---|
| Organophosphate | Poly(3,4-ethylenedioxythiophene) (PEDOT) | Amprometric | [76] |
| Organophosphate pesticides | Cobalt phthalocyanine | Chronoamperometry | [89] |
| Organophosphorus | Cysteamine self-assembled monolayer | Amprometric | [90] |
| Organophosphorus and Carbamate Pesticides | Unmodified | Amperometry, flow system | [91] |
| Aminophenol isomers | Untreated SPCE | Voltammetric | [21] |
| Organophosphorus Pesticide | Single-walled carbon nanotubes— Co phtalocyanine | Amperometry | [79] |
| Organophosphorus Pesticide Dichlofenthion | Nanometer-Sized Titania | Photoelectrochemical | [81] |
| Herbicide isoproturon | Unmodified | Amperometric | [92] |
| Herbicide | Magnetic nanoparticles | Amperometric | [83] |
| Picric acid and atrazine | Self-assembled monolayer | Photo-electrochemical | [93] |
| Chlorsulfuron | Gold (Au) metal ions | Stripping voltammetry | [80] |
| Phenol and catechol | Bismuth nanoparticles | Amperometric measurements | [94] |
| Phenol and pesticide | Iridium oxide nanoparticles | Electrochemical measurement | [95] |
| Phenol | Carbon Black Paste | Amperometric | [96] |
| Phenolic compounds | Nano-HA-chitosan nanocomposite-modified gold electrode | Amperometric | [97] |
| Phenolic compounds | Screen-printed PEDOT:PSS electrodes | Amperometric | [98] |
| Carbamate Insecticide | Prussian Blue-Multi-Walled Carbon Nanotubes | Amperometric | [99] |
| Analyte | Modifier | Detection Method | Ref. |
|---|---|---|---|
| Pb2+ and Cd2+ | screen-printed antimony and tin | anodic stripping detection | [113] |
| Cu2+ | Macrocyclic Polyamine Modified Screen-Printed Electrodes | Square wave anodic stripping voltammetry | [114] |
| Cd2+, Cu2+ | Diazonium modified electtrodes | Amperometric detection | [115] |
| Pb2+ and Cd2+ | Bismuth-coated | Stripping voltammetry | [116] |
| Pb2+ | Reduced graphene oxide | Square wave anodic stripping voltammetry | [117] |
| Zn2+, Cd2+ and Pb2+ | Multiwalled carbon nanotubes | Differential pulse stripping voltammetry | [118] |
| Hg2+ and Pb2+ | Polypyrrole/carbonaceous nanospheres | Square wave anodic stripping voltammetry | [119] |
| Pb2+ and Cd2+ | Bismuth–carbon nanocomposites | Differential electrochemical methods | [120] |
| Pb2+ | Bismuth-antimony film | Stripping voltammetric | [121] |
| Pb2+ | 4-carboxyphenyl-grafted | Anodic Square Wave Voltammetry | [122] |
| As(III) | Gold electrode | Sequential injection/anodic stripping voltammetry | [123] |
| As(III) | Nanoparticles | Linear sweep voltammetric | [124] |
| As(III) | Modified screen printed electrodes | Amperometric | [111] |
| Cd2+, Pb2+, Cu2+ and Hg2+ ions | Heated graphitenanoparticle | Electrochemical stripping | [125] |
| Hg2+ | Gold nanoparticles-modified | Square wave anodic stripping voltammetry | [126] |
| Pb2+, Cu2+ and Cd2+ | Mercury nano-droplets | Square wave anodic stripping voltammetry | [127] |
| Pb2+ | Paper disk impregnated | One-step electrochemical detection | [128] |
| Cd2+ | Nafion. Cd | Square Wave Anodic Stripping Voltammetry | [129] |
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hayat, A.; Marty, J.L. Disposable Screen Printed Electrochemical Sensors: Tools for Environmental Monitoring. Sensors 2014, 14, 10432-10453. https://doi.org/10.3390/s140610432
Hayat A, Marty JL. Disposable Screen Printed Electrochemical Sensors: Tools for Environmental Monitoring. Sensors. 2014; 14(6):10432-10453. https://doi.org/10.3390/s140610432
Chicago/Turabian StyleHayat, Akhtar, and Jean Louis Marty. 2014. "Disposable Screen Printed Electrochemical Sensors: Tools for Environmental Monitoring" Sensors 14, no. 6: 10432-10453. https://doi.org/10.3390/s140610432
APA StyleHayat, A., & Marty, J. L. (2014). Disposable Screen Printed Electrochemical Sensors: Tools for Environmental Monitoring. Sensors, 14(6), 10432-10453. https://doi.org/10.3390/s140610432





