Metal Oxide Semi-Conductor Gas Sensors in Environmental Monitoring
Abstract
:1. Introduction
1.1. Basic Theory
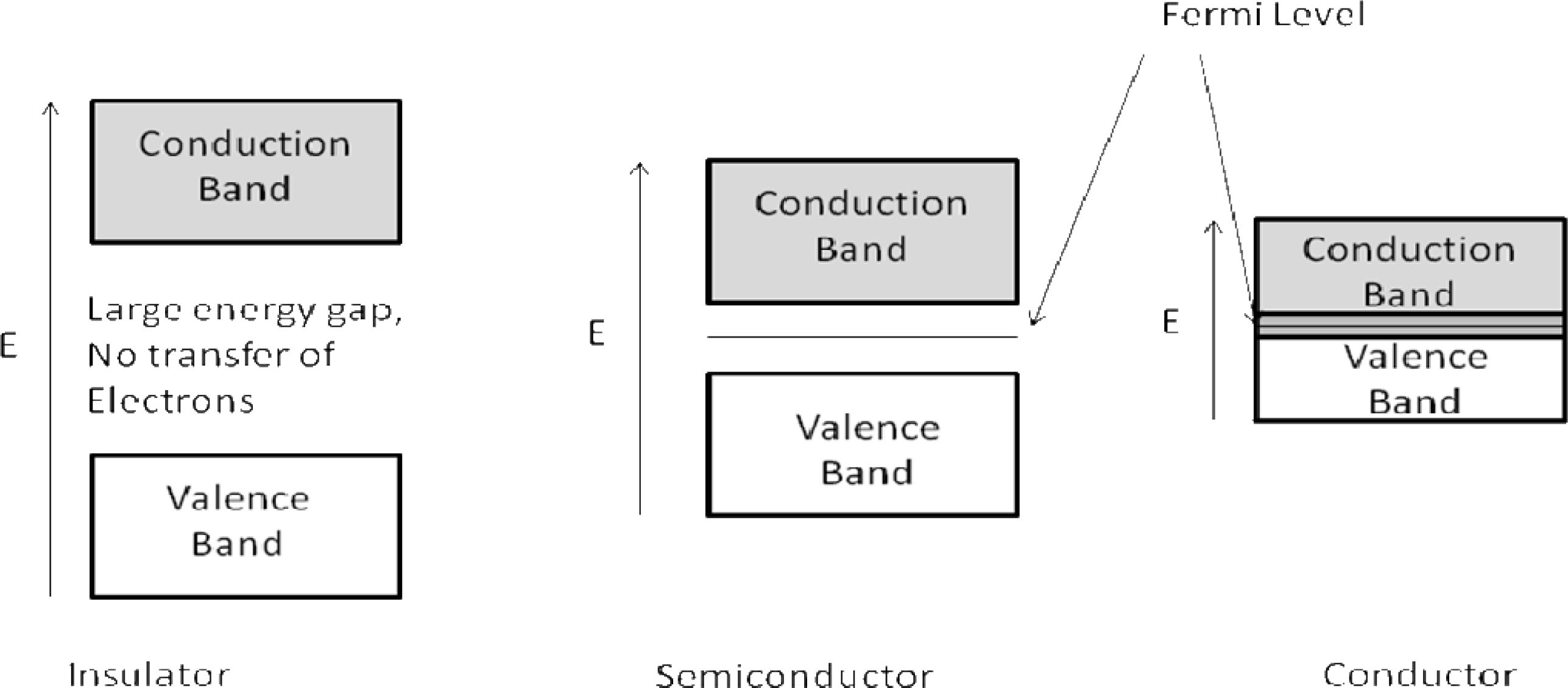
1.1.1. Band Theory
1.1.2. Band Theory Applied to Sensors
1.2. A Model for Gas Interaction
1.2.1. A p-Type Sensor Response
1.2.2. The Equivalent Circuit Model
1.3. Sensor Response Disruption
1.3.1. Ozone
1.3.2. Water
1.3.3. Volatile Organic Compounds
1.4. Factors Influencing Sensor Design
1.5. Techniques for Sensor Fabrication
1.5.1. Screen Printing
1.5.2. Chemical Vapour Deposition (CVD)
1.5.3. Spray Pyrolysis
1.5.4. Sol-Gel
1.5.5. Physical Vapour Deposition (PVD)
1.5.6. Drop Coating
1.5.7. Comparison of Synthetic Techniques
2. Carbon Monoxide Sensors
2.1. Reasons for Detecting Carbon Monoxide
2.2. Advances in Carbon Monoxide Sensors
2.3. Outlook for MOS Carbon Monoxide Sensors
3. Carbon Dioxide Sensors
3.1. Reasons for Detecting Carbon Dioxide
3.2. Advances in Carbon Dioxide Sensors
3.3. Outlook for MOS Carbon Dioxide Sensors
4. Nitrogen Oxide (NOx) Sensors
4.1. Reasons for Detecting Nitrous Oxides
4.2. Advances in Nitrous Oxide sensors
4.3. Outlook for MOS Nitrous Oxide Sensors
5. Ammonia Sensors
5.1. Reasons for Detecting Ammonia
5.2. Advances in Ammonia Sensors
5.3. Outlook for MOS Ammonia Sensors
6. Recent Work Conducted at University College London
6.1. Zeolite Modified Sensors
6.2. Carbon Dioxide Sensors
6.3. Electric Fields and Chemical Vapour Deposition (ElFi CVD)
7. Conclusions
References
- Seiyama, T.; Kato, A.; Fujiishi, K.; Nagatani, M. A new detector for gaseous components using semiconductive thin films. Anal. Chem 1962, 34, 1502–1503. [Google Scholar]
- Elmi, I.; Zampolli, S.; Cozzani, E.; Mancarella, F.; Cardinali, G.C. Development of ultra-low-power consumption MOX sensors with ppb-level VOC detection capabilities for emerging applications. Sens. Actuat. B-Chem 2008, 135, 342–351. [Google Scholar]
- Guo, Y.; Zhang, X.W.; Han, G.R. Investigation of structure and properties of N-doped TiO2 thin films grown by APCVD. Mater. Sci. Eng.: B 2006, 135, 83–87. [Google Scholar]
- Gurlo, A.; Bârsan, N.; Ivanovskaya, M.; Weimar, U.; Göpel, W. In2O3 and MoO3–In2O3 thin film semiconductor sensors: interaction with NO2 and O3. Sens. Actuat. B-Chem 1998, 47, 92–99. [Google Scholar]
- Kida, T.; Nishiyama, A.; Yuasa, M.; Shimanoe, K.; Yamazoe, N. Highly sensitive NO2 sensors using lamellar-structured WO3 particles prepared by an acidification method. Sens. Actuat. B-Chem 2009, 135, 568–574. [Google Scholar]
- Williams, D.E. Semiconducting oxides as gas-sensitive resistors. Sens. Actuat. B-Chem 1999, 57, 1–19. [Google Scholar]
- Martin, R. Electronic Structure: Basic Theory and Practical Methods; Cambridge University Press: Cambridge, UK, 2004. [Google Scholar]
- Shriver, A. Inorganic Chemistry; Oxford University Press: Oxford, UK, 2006. [Google Scholar]
- Yamazoe, N.; Fuchigami, J.; Kishikawa, M.; Seiyama, T. Interactions of tin oxide surface with O2, H2O and H2. Surf. Sci 1979, 86, 335–344. [Google Scholar]
- Barsan, N.; Schweizer-Berberich, M.; Göpel, W. Fundamental and practical aspects in the design of nanoscaled SnO2 gas sensors: a status report. Fresenius J. Anal. Chem 1999, 365, 287–304. [Google Scholar]
- Barsan, N.; Weimar, U. Understanding the fundamental principles of metal oxide based gas sensors; the example of CO sensing with SnO2 sensors in the presence of humidity. J. Phys. Cond. Matt 2003, 15, R813. [Google Scholar]
- Binions, R.; Davies, H.; Afonja, A.; Dungey, S.; Lewis, D.; Williams, D.E.; Parkin, I.P. Zeolite modified discriminating gas sensors. J. Electrochem. Soc 2009, 156, J46–J51. [Google Scholar]
- Naisbitt, S.C.; Pratt, K.F.E.; Williams, D.E.; Parkin, I.P. A microstructural model of semiconducting gas sensor response: The effects of sintering temperature on the response of chromium titanate (CTO) to carbon monoxide. Sens. Actuat. B-Chem 2006, 114, 969–977. [Google Scholar]
- Vincenzi, D.; Butturi, M.A.; Guidi, V.; Carotta, M.C.; Martinelli, G.; Guarnieri, V.; Brida, S.; Margesin, B.; Giacomozzi, F.; Zen, M.; Giusti, D.; Soncini, G.; Vasiliev, A.A.; Pisliakov, A.V. Gas-sensing device implemented on a micromachined membrane: A combination of thick-film and very large scale integrated technologies. J. Vac. Sci. Technol. B 2000, 18, 2441–2445. [Google Scholar]
- Patel, N.G.; Patel, P.D.; Vaishnav, V.S. Indium tin oxide (ITO) thin film gas sensor for detection of methanol at room temperature. Sens. Actuat. B-Chem 2003, 96, 180–189. [Google Scholar]
- Korotcenkov, G.; Blinov, I.; Brinzari, V.; Stetter, J.R. Effect of air humidity on gas response of SnO2 thin film ozone sensors. Sens. Actuat. B-Chem 2007, 122, 519–526. [Google Scholar]
- Wang, Z.; Hu, X. Fabrication and electrochromic properties of spin-coated TiO2 thin films from peroxo-polytitanic acid. Thin Solid Films 1999, 352, 62–65. [Google Scholar]
- Vaishanv, V.S.; Patel, P.D.; Patel, N.G. Indium tin oxide thin-film sensor for detection of Volatile Organic Compounds (VOCs). Mater. Manufact. Proces 2006, 21, 257–261. [Google Scholar]
- Binions, R.; Kanu, S.S. Thin films for solar control applications. Proc. Royal Soc. Lon. A 2010, 466, 19–44. [Google Scholar]
- Binions, R.; Carmalt, C.J.; Parkin, I.P. Aerosol-assisted chemical vapour deposition of sodium fluoride thin films. Thin Solid Films 2004, 469–470, 416–419. [Google Scholar]
- Cross, W.B.; Parkin, I.P. Aerosol assisted chemical vapour deposition of tungsten oxide films from polyoxotungstate precursors: active photocatalyst. Chem. Comm 2003, 9, 1696–1697. [Google Scholar]
- Parkin, I.P.; Binions, R.; Piccirillo, C.; Blackman, C.S.; Manning, T.D. thermochromic coatings for intelligent architectural glazing. J. Nano Res 2008, 2, 1–20. [Google Scholar]
- Ashraf, S.; Binions, R.; Blackman, C.S.; Parkin, I.P. The APCVD of tungsten oxide thin films from reaction of WCl6 with ethanol and results on their gas-sensing properties. Polyhedron 2007, 26, 1493–1498. [Google Scholar]
- Shaw, G.; Parkin, I.P.; Pratt, K.F.E.; Williams, D.E. Control of semiconducting oxide gas-sensor microstructure by application of an electric field during aerosol-assisted chemical vapour deposition. J. Mater. Chem 2005, 15, 149–154. [Google Scholar]
- Shaw, G.A.; Parkin, I.P.; Williams, D.E. Control of semiconducting oxide gas-sensor microstructure by application of an electric field during aerosol-assisted chemical vapour deposition. J. Mater. Chem 2005, 15, 149–154. [Google Scholar]
- Blackman, C.S.; Parkin, I.P. Atmospheric pressure chemical vapor deposition of crystalline monoclinic WO3 and WO3-x thin films from reaction of WCl6 with O-containing solvents and their photochromic and electrochromic properties. Chem. Mater 2005, 17, 1583–1590. [Google Scholar]
- Ashraf, S.; Blackman, C.S.; Palgrave, R.G.; Naisbitt, S.C.; Parkin, I.P. Aerosol assisted chemical vapour deposition of WO3 thin films from tungsten hexacarbonyl and their gas sensing properties. J. Mater Chem 2007, 17, 3708–3713. [Google Scholar]
- Ashraf, S.; Blackman, C.S.; Naisbitt, S.C.; Parkin, I.P. The gas-sensing properties of WO3-x thin films deposited via the atmospheric pressure chemical vapour deposition (APCVD) of WCl6 with ethanol. Mes. Sci. Technol 2008, 19, 025203. [Google Scholar]
- Choy, K.L. Chemical vapour deposition of coatings. Prog. Mater. Sci 2003, 48, 57–170. [Google Scholar]
- Tischner, A.; Maier, T.; Stepper, C.; Köck, A. Ultrathin SnO2 gas sensors fabricated by spray pyrolysis for the detection of humidity and carbon monoxide. Sens. Actuat. B-Chem 2008, 134, 796–802. [Google Scholar]
- Goldberg, H.D.; Brown, R.B.; Liu, D.P.; Meyerhoff, M.E. Screen printing: a technology for the batch fabrication of integrated chemical-sensor arrays. Sens. Actuat. B-Chem 1994, 21, 171–183. [Google Scholar]
- Yadava, L.; Verma, R.; Dwivedi, R. Sensing properties of CdS-doped tin oxide thick film gas sensor. Sens. Actuat. B-Chem 2009, 144, 37–42. [Google Scholar]
- Alhomoudi, I.A.; Newaz, G. Residual stresses and Raman shift relation in anatase TiO2 thin film. Thin Solid Films 2009, 517, 4372–4378. [Google Scholar]
- Boulmani, R.; Bendahan, M.; Lambert-Mauriat, C.; Gillet, M.; Aguir, K. Correlation between rf-sputtering parameters and WO3 sensor response towards ozone. Sens. Actuat. B-Chem 2007, 125, 622–627. [Google Scholar]
- Michel, H.J.; Leiste, H.; Halbritter, J. Structural and electrical characterization of PVD-deposited SnO2 films for gas-sensor application. Sens. Actuat. B-Chem 1995, 25, 568–572. [Google Scholar]
- Gupta, S.; Roy, R.K.; Chowdhury, M.P.; Pal, A.K. Synthesis of SnO2/Pd composite films by PVD route for a liquid petroleum gas sensor. Vacuum 2004, 75, 111–119. [Google Scholar]
- Cerdà, J.; Cirere, A.; Vilà, A.; Cornet, A.; Morante, J.R. Deposition on micromachined silicon substrates of gas sensitive layers obtained by a wet chemical route: a CO/CH4 high performance sensor. Thin Solid Films 2001, 391, 265–269. [Google Scholar]
- Brezmes, J.; Llobet, E.; Vilanova, X.; Saiz, G.; Correig, X. Fruit ripeness monitoring using an Electronic Nose. Sens. Actuat. B-Chem 2000, 69, 223–229. [Google Scholar]
- Binions, R.; Parkin, I.P. A comparison of the gas sensing properties of solid state metal oxide semiconductor gas sensors produced by atmospheric pressure chemical vapour deposition and screen printing. Mes. Sci. Technol 2007, 18, 190–200. [Google Scholar]
- Cobb, C.; Cetzel, C.R. Unintentional carbon monoxide—Related deaths in the United States, 1979 through 1988. J. Am. Med. Assoc 1991, 266, 659–663. [Google Scholar]
- Meredith, C.T. Correction: Comroe and Dripps revisited. Brit. Med. J 1988, 296, 110. [Google Scholar]
- Runyan, C.C.; Johnson, C.; Yang, J.; Waller, A.; Perkis, D.; Marshall, S. Risk and protective factors for fires, burns, and carbon monoxide poisoning in U.S. households. Am. J. Prev. Med 2005, 28, 102–108. [Google Scholar]
- Varon, J.; Marik, P.E.; Fromm, R.E., Jr.; Gueler, A. Carbon monoxide poisoning: A review for clinicians. J. Emer. Med 1999, 17, 87–93. [Google Scholar]
- Recommendations for Occupational Safety and Health: Compendium of Policy Documents and Statements; U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, National Institute for Occupational Safety and Health, DHHS (NIOSH): Cincinnati, OH, USA, 1992; Publication No. 92–100.
- Wiegleb, G.; Heitbaum, J. Semiconductor gas sensor for detecting NO and CO traces in ambient air of road traffic. Sens. Actuat. B-Chem 1994, 17, 93–99. [Google Scholar]
- Barbi, G.B.; Santos, J.P.; Serrini, P.; Gibson, P.N.; Horrillo, M.C.; Manes, L. Ultrafine grain-size tin-oxide films for carbon monoxide monitoring in urban environments. Sens. Actuat. B-Chem 1995, 25, 559–563. [Google Scholar]
- Riviere, B.; Viricelle, J.P.; Pijolat, C. Development of tin oxide material by screen-printing technology for micro-machined gas sensors. Sens. Actuat. B-Chem 2003, 93, 531–537. [Google Scholar]
- Li, X.; Ramasamy, R.; Dutta, P.K. Study of the resistance behavior of anatase and rutile thick films towards carbon monoxide and oxygen at high temperatures and possibilities for sensing applications. Sens. Actuat. B-Chem 2009, 143, 308–315. [Google Scholar]
- Izu, N.; Nishizaki, S.; Itoh, T.; Nishibori, M.; Shin, W.; Matsubara, I. Gas response, response time and selectivity of a resistive CO sensor based on two connected CeO2 thick films with various particle sizes. Sens. Actuat. B-Chem 2009, 136, 364–370. [Google Scholar]
- Durrani, S.M.A.; Al-Kuhaili, M.F.; Bakhtiari, I.A. Carbon monoxide gas-sensing properties of electron-beam deposited cerium oxide thin films. Sens. Actuat. B-Chem 2008, 134, 934–939. [Google Scholar]
- Wiegleb, G.; Heitbaum, J. Semiconductor gas sensor for detecting NO and CO traces in ambient air of road traffic. Sens. Actuat. B-Chem 1994, 17, 93–99. [Google Scholar]
- NOAA ESRL Data. Available online: ftp://ftp.cmdl.noaa.gov/ccg/co2/trends/co2_mm_mlo.txt (accessed on 18 March 2010).
- Code of Federal Regualtions; The Superintendent of Documents, U.S. Government Printing Office: Washington, DC, USA, 1994.
- Trends in Atmospheric Carbon Dioxide. Available online: http://www.esrl.noaa.gov/gmd/ccgg/trends/ (accessed on 16 March 2010).
- Patel, N.G.; Makhija, K.K.; Panchal, C.J. Fabrication of carbon dioxide gas sensor and its alarm system using Indium Tin Oxide (ITO) thin films. Sens. Actuat. B-Chem 1994, 21, 193–197. [Google Scholar]
- Hoefer, U.; Kühner, G.; Schweizer, W.; Sulz, G.; Steiner, K. CO and CO2 thin-film SnO2 gas sensors on Si substrates. Sens. Actuat. B-Chem 1994, 22, 115–119. [Google Scholar]
- Kim, D.H.; Yoon, J.Y.; Park, H.C.; Kim, K.H. CO2-sensing characteristics of SnO2 thick film by coating lanthanum oxide. Sens. Actuat. B-Chem 2000, 62, 61–66. [Google Scholar]
- Lee, M.S.; Meyer, J.U. A new process for fabricating CO2-sensing layers based on BaTiO3 and additives. Sens. Actuat. B-Chem 2000, 68, 293–299. [Google Scholar]
- Marsal, A.; Dezanneau, G.; Cornet, A.; Morante, J.R. A new CO2 gas sensing material. Sens. Actuat. B-Chem 2003, 95, 266–270. [Google Scholar]
- Marsal, A.; Cornet, A.; Morante, J.R. Study of the CO and humidity interference in La doped tin oxide CO2 gas sensor. Sens. Actuat. B-Chem 2003, 94, 324–329. [Google Scholar]
- Barth, P.J.; Muller, B.; Wagner, U.; Bittinger, A. Quantitative analysis of parenchymal and vascular alterations in NO2-induced lung injury in rats. Eur. Resp. J 1995, 8, 1115–1121. [Google Scholar]
- Wegmann, M.; Fehrenbach, A.; Heimann, S.; Fehrenbach, H.; Renz, H.; Garn, H.; Herz, U. NO2-induced airway inflammation is associated with progressive airflow limitation and development of emphysema-like lesions in C57BL/6 mice. Expt. Tox. Path 2005, 56, 341–350. [Google Scholar]
- Ponka, A.; Virtanen, M. Chronic bronchitis, emphysema, and low-level air pollution in Helsinki, 1987–1989. Env. Res 1994, 65, 207–217. [Google Scholar]
- Akiyama, M.; Tamaki, J.; Miura, N.; Yamazoe, N. Tungsten oxide-based semiconductor sensor highly sensitive to NO and NO2. Chem. Lett 1991, 20, 1611–1614. [Google Scholar]
- Tamaki, J.; Zhang, Z.; Fujimori, K.; Akiyama, M.; Harada, T.; Miura, N.; Yamazoe, N. Grain-size effects in tungsten oxide-based sensor for nitrogen oxides. J. Electrochem. Soc 1994, 141, 2207–2210. [Google Scholar]
- Cantalini, C.; Pelino, M.; Sun, H.T.; Faccio, M.; Santucci, S.; Lozzi, L.; Passacantando, M. Cross sensitivity and stability of NO2 sensors from WO3 thin film. Sens. Actuat. B-Chem 1996, 35, 112–118. [Google Scholar]
- Chung, Y.K.; Kim, M.H.; Um, W.S.; Lee, H.S.; Song, J.K.; Choi, S.C.; Yi, K.M.; Lee, M.J.; Chung, K.W. Gas sensing properties of WO3 thick film for NO2 gas dependent on process condition. Sens. Actuat. B-Chem 1999, 60, 49–56. [Google Scholar]
- Lee, D.S.; Han, S.D.; Huh, J.S.; Lee, D.D. Nitrogen oxides-sensing characteristics of WO3-based nanocrystalline thick film gas sensor. Sens. Actuat. B-Chem 1999, 60, 57–63. [Google Scholar]
- Comini, E.; Sberveglieri, G.; Ferroni, M.; Guidi, V.; Martinelli, G. NO2 monitoring with a novel p-type material: TiO. Sens. Actuat. B-Chem 2000, 68, 175–183. [Google Scholar]
- Kanazawa, E.; Sakai, G.; Shimanoe, K.; Kanmura, Y.; Teraoka, Y.; Miura, N.; Yamazoe, N. Metal oxide semiconductor N2O sensor for medical use. Sens. Actuat. B-Chem 2001, 77, 72–77. [Google Scholar]
- Shieh, J.; Feng, H.M.; Hon, M.H.; Juang, H.Y. WO3 and W---Ti---O thin-film gas sensors prepared by sol–gel dip-coating. Sens. Actuat. B-Chem 2002, 86, 75–80. [Google Scholar]
- Tomchenko, A.A.; Harmer, G.P.; Marquis, B.T.; Allen, J.W. Semiconducting metal oxide sensor array for the selective detection of combustion gases. Sens. Actuat. B-Chem 2003, 93, 126–134. [Google Scholar]
- Williams, D.E.; Salmond, J.; Yung, Y.F.; Akali, J.; Wright, B.; Wilson, J.; Henshaw, G.S.; Wells, D.B.; Ding, G.; Wagner, J.; Laing, G. Development of low-cost ozone and nitrogen dioxide measurement instruments suitable for use in an air quality monitoring network. Proceedings of the 215th ECS Meeting, San Francisco, CA, USA, May 24–29, 2009; pp. 251–254.
- Ammonia, in Ullmann’s Encyclopedia of Industrial Chemistry; Appl, M. (Ed.) Wiley-VCH: Weinheim, Germany, 2006.
- Nanto, H.; Minami, T.; Takata, S. Zinc□oxide thin□film ammonia gas sensors with high sensitivity and excellent selectivity. J. Appl. Phys 1986, 60, 482–485. [Google Scholar]
- Wang, X.; Miura, N.; Yamazoe, N. Study of WO3-based sensing materials for NH3 and NO detection. Sens. Actuat. B-Chem 2000, 66, 74–76. [Google Scholar]
- Karunagaran, B.; Uthirakumar, P.; Chung, S.J.; Velumani, S.; Suh, E.K. TiO2 thin film gas sensor for monitoring ammonia. Mater. Charact 2007, 58, 680–684. [Google Scholar]
- Wagh, M.S.; Jain, G.H.; Patil, D.R.; Patil, S.A.; Patil, L.A. Modified zinc oxide thick film resistors as NH3 gas sensor. Sens. Actuat. B-Chem 2006, 115, 128–133. [Google Scholar]
- Hagen, G.; Dubbe, A.; Rettig, F.; Jerger, A.; Birkhofer, T.; Müller, R.; Plog, C.; Moos, R. Selective impedance based gas sensors for hydrocarbons using ZSM-5 zeolite films with chromium(III)oxide interface. Sens. Actuat. B-Chem 2006, 119, 441–448. [Google Scholar]
- Vilaseca, M.; Coronas, J.; Cirera, A.; Cornet, A.; Morante, J.; Santamarià, J. Use of zeolite films to improve the selectivity of reactive gas sensors. Catal. Today 2003, 82, 179–185. [Google Scholar]
- Hugon, O.; Sauvan, M.; Benech, P.; Pijolat, C.; Lefebvre, F. Gas separation with a zeolite filter, application to the selectivity enhancement of chemical sensors. Sens. Actuat. B-Chem 2000, 67, 235–243. [Google Scholar]
- Sahner, K.; Schönauer, D.; Kuchinke, P.; Moos, R. Zeolite cover layer for selectivity enhancement of p-type semiconducting hydrocarbon sensors. Sens. Actuat. B-Chem 2008, 133, 502–508. [Google Scholar]
- Afonja, A.; Dungey, S.; Binions, R.; Parkin, I.; Lewis, D.; Williams, D. Gas sensing properties of composite tungsten trioxide—Zeolite thick films. Electrochem. Soc. Trans 2009, 16, 77–84. [Google Scholar]
- Binions, R.; Davis, H.; Afonja, A.; Dungey, S.; Lewis, D.; Williams, D.; Parkin, I. Zeolite modified gas sensors. Electrochem. Soc. Trans 2008, 16, 275–286. [Google Scholar]
- Binions, R.; Afonja, A.; Dungey, S.; Lewis, D.; Parkin, I.; Williams, D.E. Zeolite moidification: towards discriminating gas sensors. Electrochem. Soc. Trans 2009, 19, 241–250. [Google Scholar]























| Classification | Oxidising Gases | Reducing Gases |
|---|---|---|
| n-type | Resistance increase | Resistance decrease |
| p-type | Resistance decrease | Resistance increase |
| Carbon Monoxide Concentration (ppm) | COHb Level (%) | Signs and Symptoms |
|---|---|---|
| 35 | <10 | Headache and dizziness within 6 to 8 hours of exposure |
| 100 | >10 | Slight headache in 2 to 3 hours |
| 200 | 20 | Slight headache within 2 to 3 hours; loss of judgement |
| 400 | 25 | Frontal headache within 1 to 2 hours |
| 800 | 30 | Dizziness, nausea and convulsions within 45 minutes, insensible within 2 hours |
| 1,600 | 40 | Headache, tachycardia, dizziness and nausea within 20 minutes; death within 30 minutes |
| 3,200 | 50 | Headache dizziness and nausea in 5 to 10 minutes; death within 30 minutes |
| 6,400 | 60 | Headache and dizziness in 1 to 2 minutes; convulsions, respiratory arrest, and death in less than 20 minutes |
| 12,800 | >70 | Death in less than 3 minutes |
| CO2 Concentration and Exposure time | Effect on Health (symptoms) |
|---|---|
| 0.035% | Approximate atmospheric concentration, no noticeable effect. |
| 3.3–5.4% for 15 mins | Increased depth of breathing |
| 7.5% for 15 mins | Feeling of an inability to breathe, increased pulse rate, headache, dizziness, sweating, restlessness, disorientation, and visual distortion. |
| 3%, for over 15 hours | Decreased night vision, colour sensitivity |
| 10%, 1.5 mins | Eye flickering, increased muscle activity, twitching |
| 10+% | Difficulty in breathing, impaired hearing, nausea, vomiting, a strangling sensation, sweating, after 15 minutes a loss of consciousness. |
| 30% | Unconsciousness, convulsions. Several deaths attributed to CO2 at concentrations of more than 20% |
| Gas Species | ΔR for ZnO | ΔR Al-doped ZnO (2% wt) | ΔR In-doped ZnO (2% wt) | ΔR Ga-doped ZnO (2% wt) |
|---|---|---|---|---|
| Ammonia | −72 | −94 | −88 | −82 |
| Hydrogen | 35 | 13 | 15 | 49 |
| Butane | 21 | 0 | 0 | 2 |
| Methane | 0 | 0 | 0 | 0 |
| Ethyl Alcohol | 69 | 54 | 39 | 40 |
| Acetone | 89 | 84 | 62 | 68 |
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fine, G.F.; Cavanagh, L.M.; Afonja, A.; Binions, R. Metal Oxide Semi-Conductor Gas Sensors in Environmental Monitoring. Sensors 2010, 10, 5469-5502. https://doi.org/10.3390/s100605469
Fine GF, Cavanagh LM, Afonja A, Binions R. Metal Oxide Semi-Conductor Gas Sensors in Environmental Monitoring. Sensors. 2010; 10(6):5469-5502. https://doi.org/10.3390/s100605469
Chicago/Turabian StyleFine, George F., Leon M. Cavanagh, Ayo Afonja, and Russell Binions. 2010. "Metal Oxide Semi-Conductor Gas Sensors in Environmental Monitoring" Sensors 10, no. 6: 5469-5502. https://doi.org/10.3390/s100605469
APA StyleFine, G. F., Cavanagh, L. M., Afonja, A., & Binions, R. (2010). Metal Oxide Semi-Conductor Gas Sensors in Environmental Monitoring. Sensors, 10(6), 5469-5502. https://doi.org/10.3390/s100605469




