Metal Oxide Gas Sensors: Sensitivity and Influencing Factors
Abstract
:1. Introduction
2. Metal Oxides for Gas Sensors
- Transition-metal oxides (Fe2O3, NiO, Cr2O3, etc.)
- Non-transition-metal oxides, which include (a) pre-transition-metal oxides (Al2O3, etc.) and (b) post-transition-metal oxides (ZnO, SnO2, etc.).
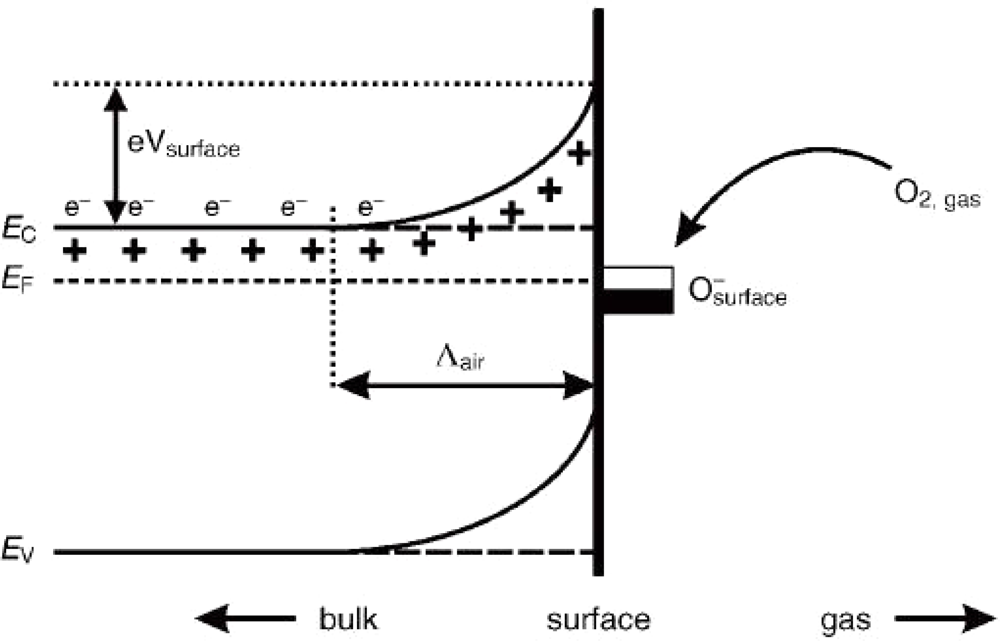
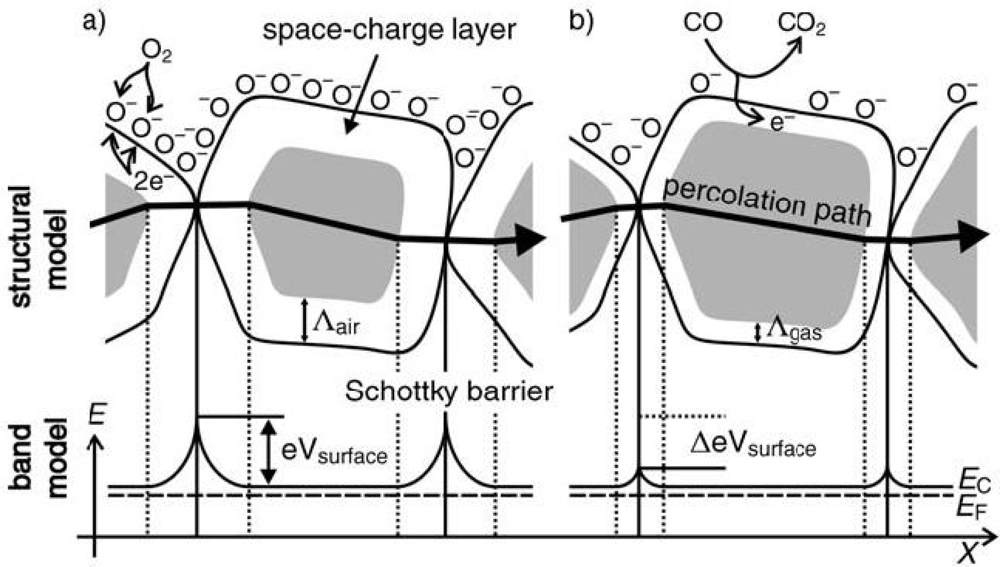
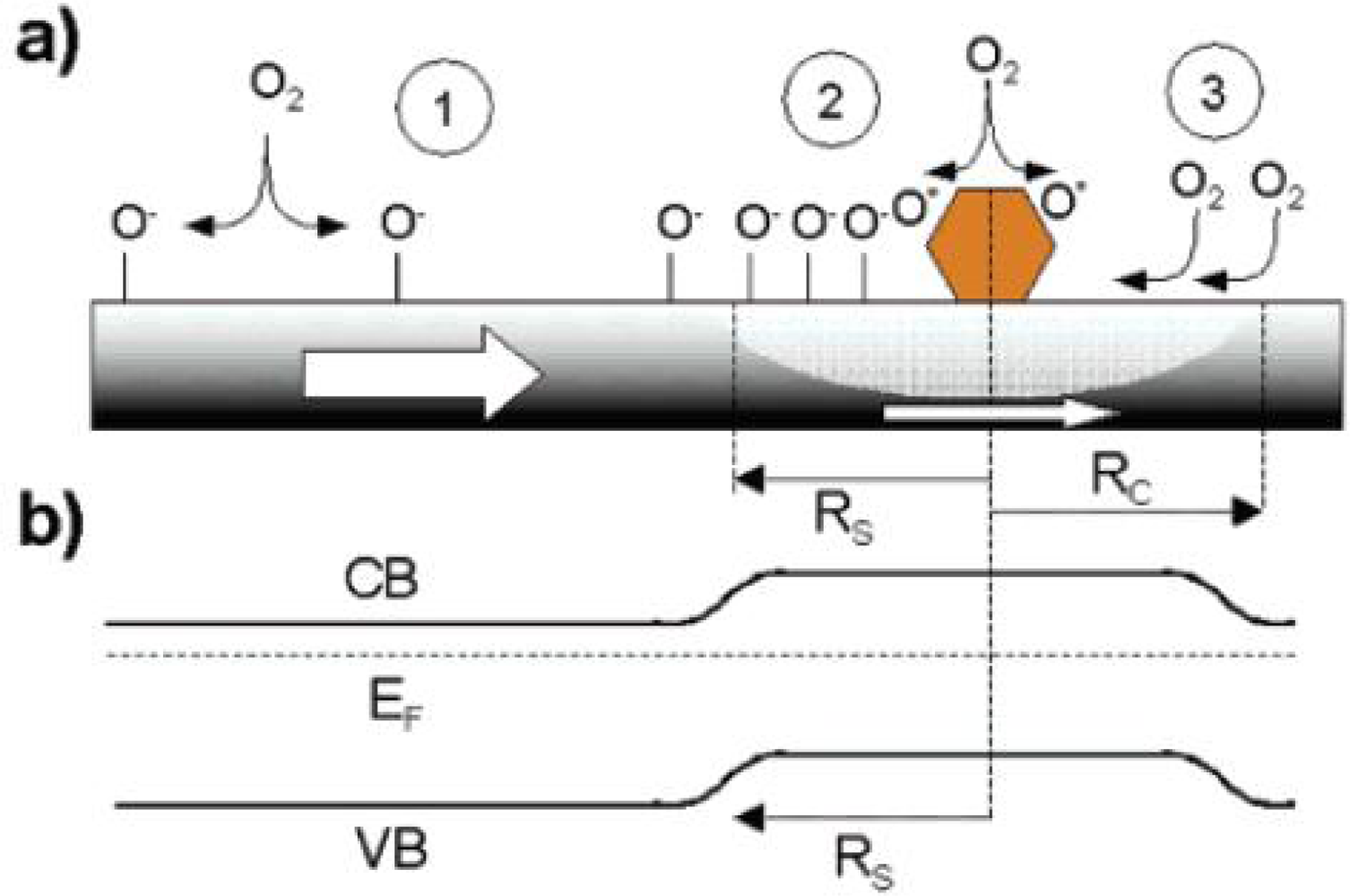
3. Sensing Mechanism
4. Factors Influencing the Sensitivity
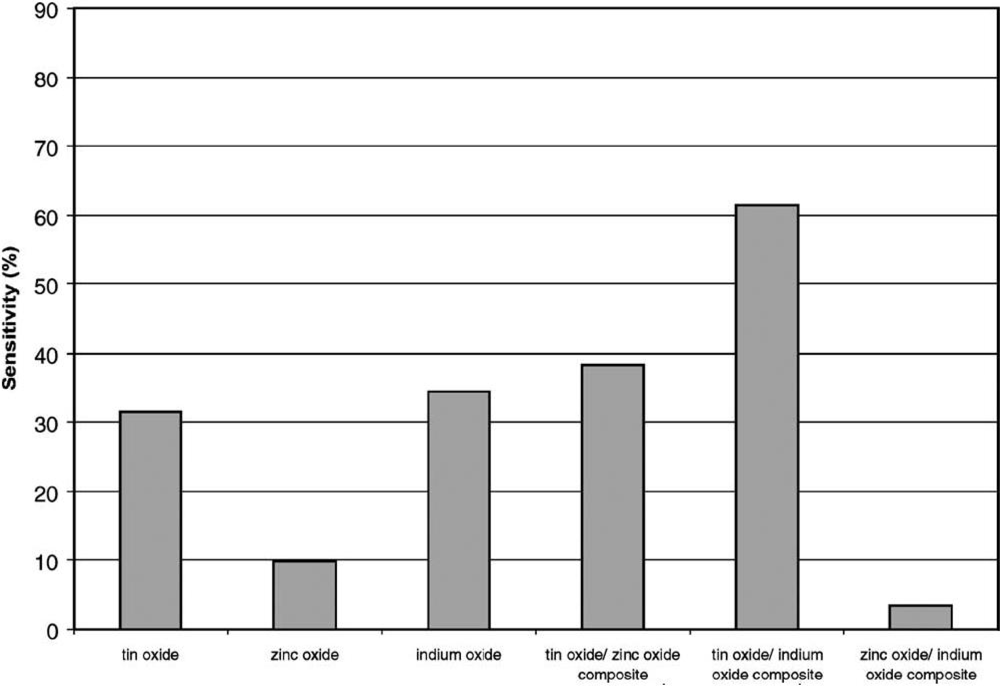
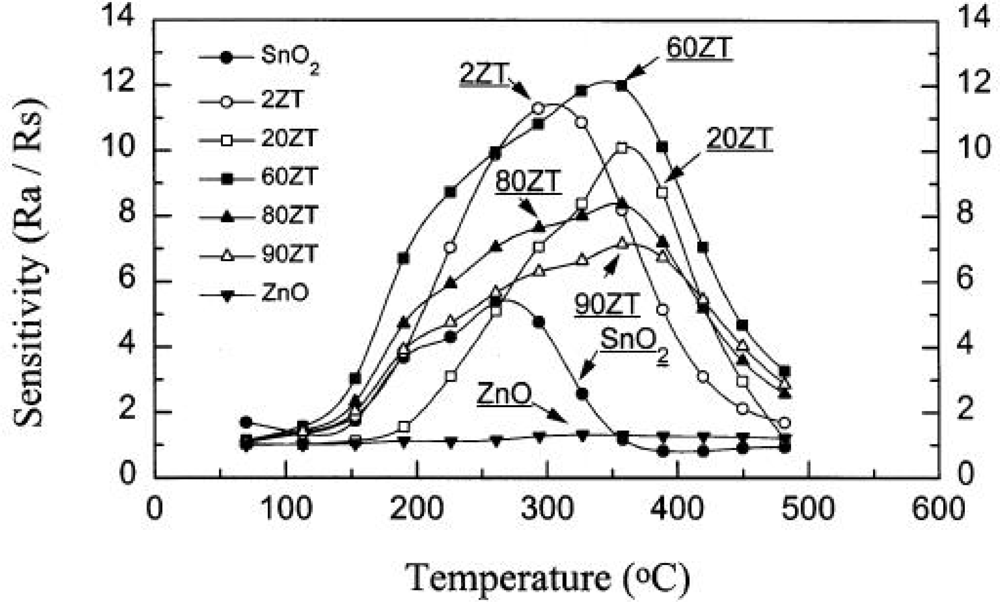
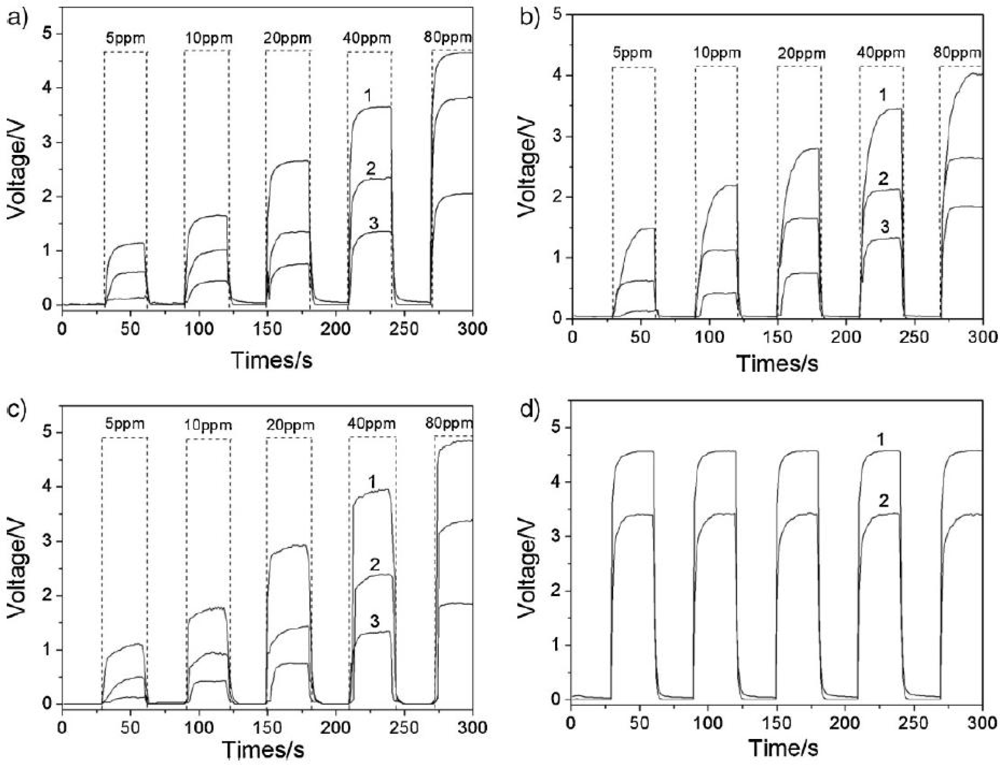
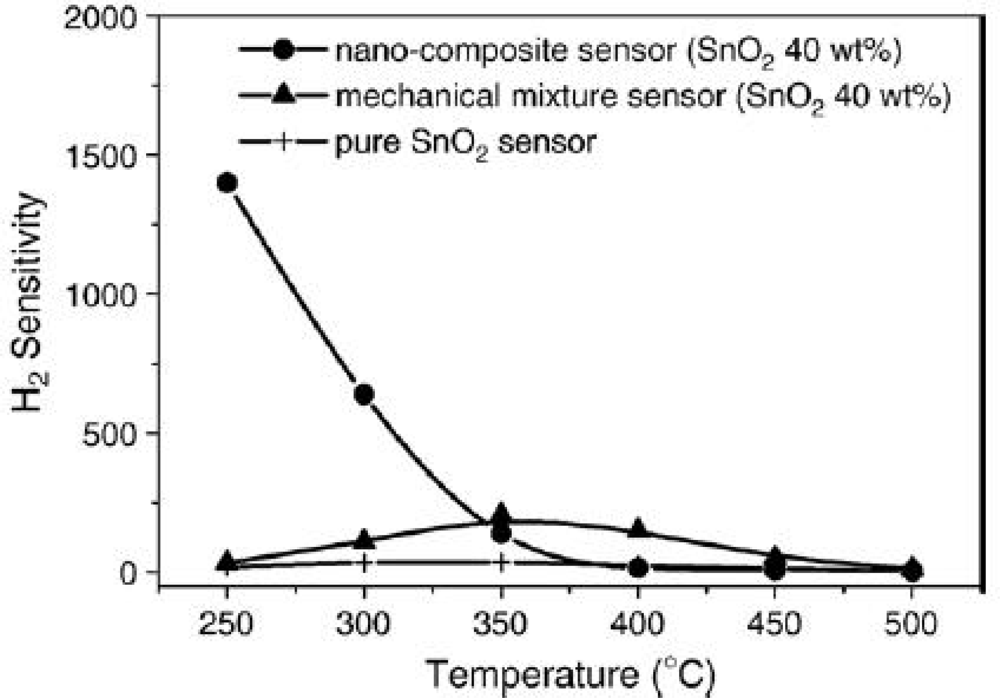
4.1. Chemical Composition
4.2. Surface Modification by Noble Metal Particles
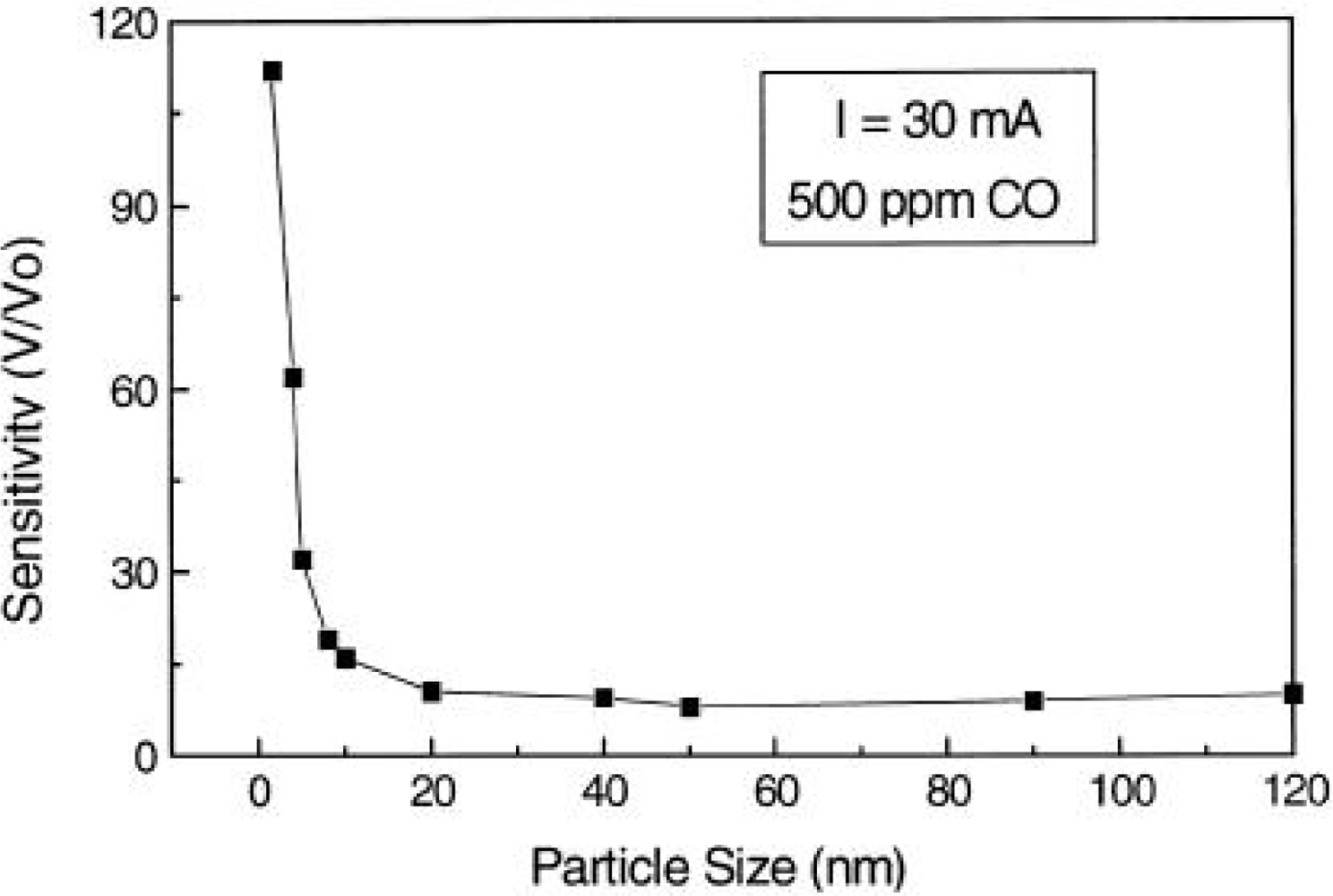
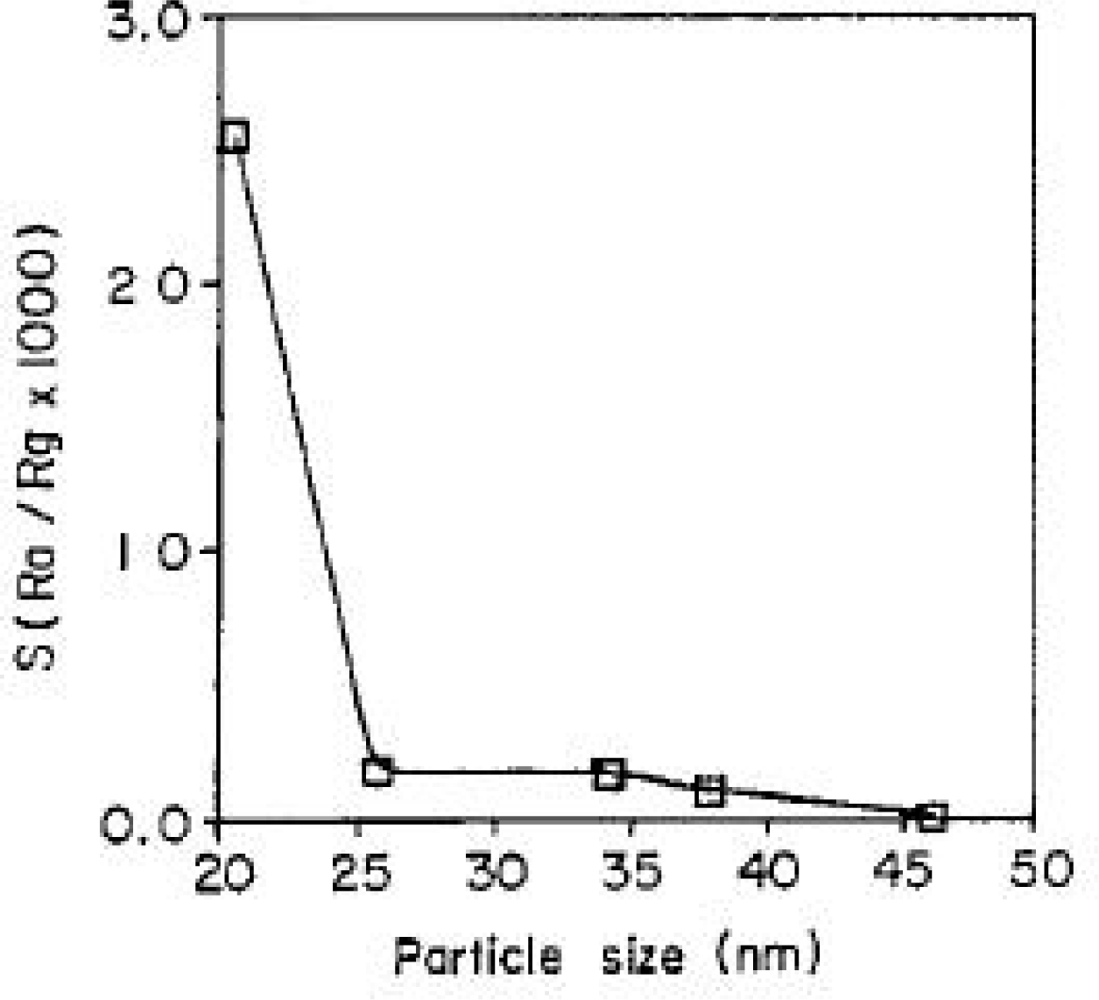
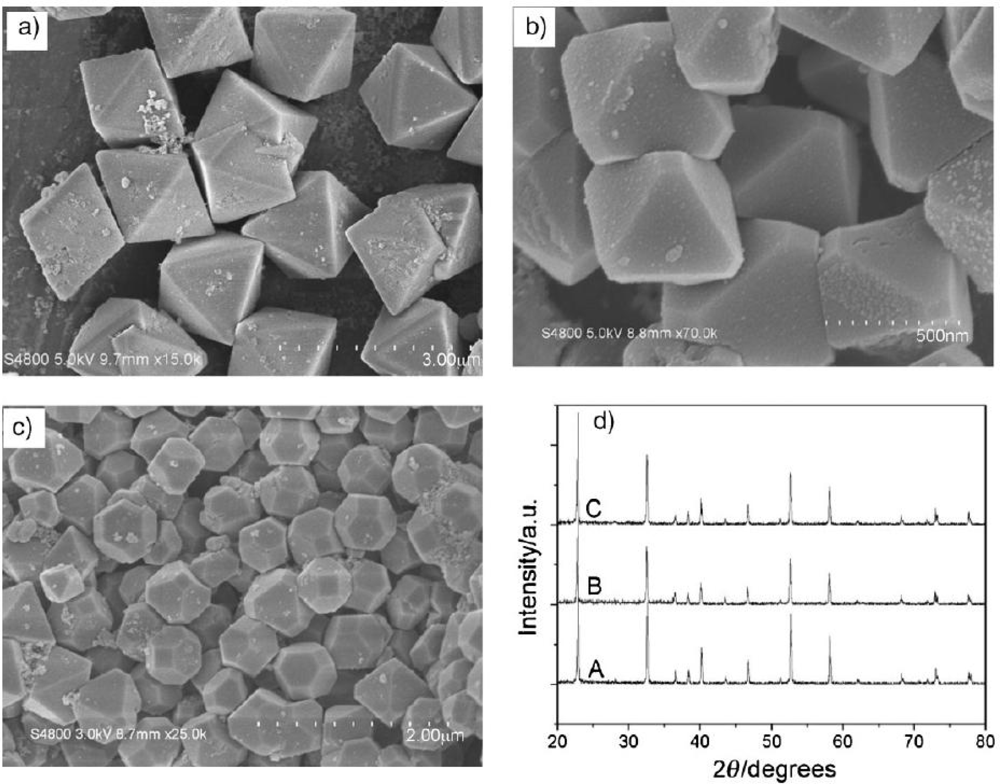
4.3. Microstructure
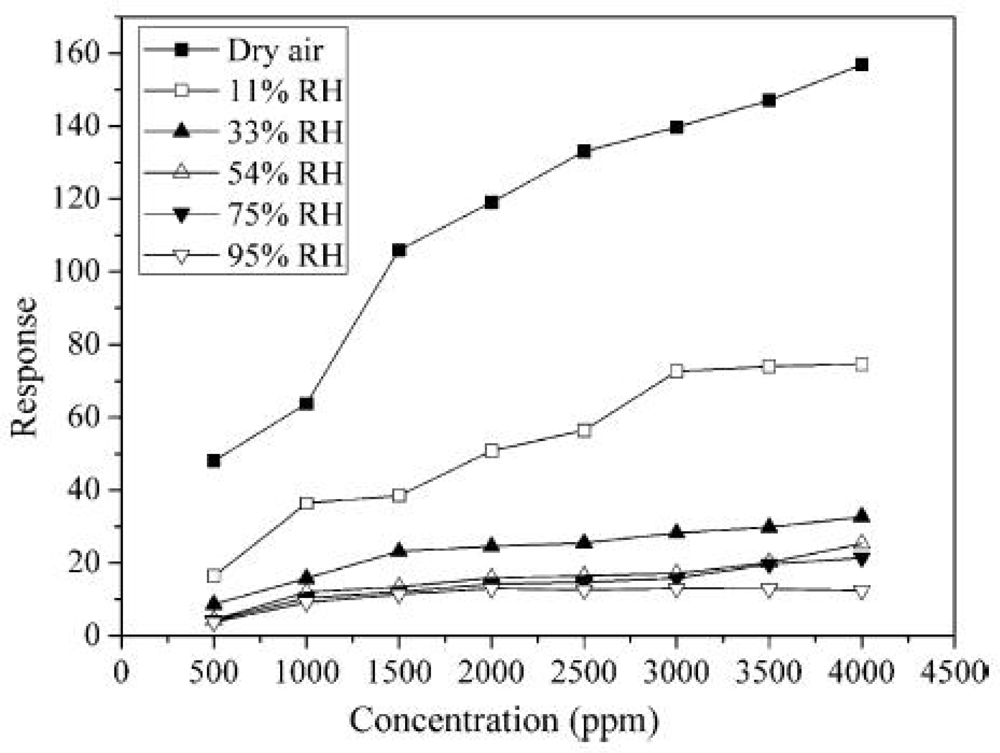
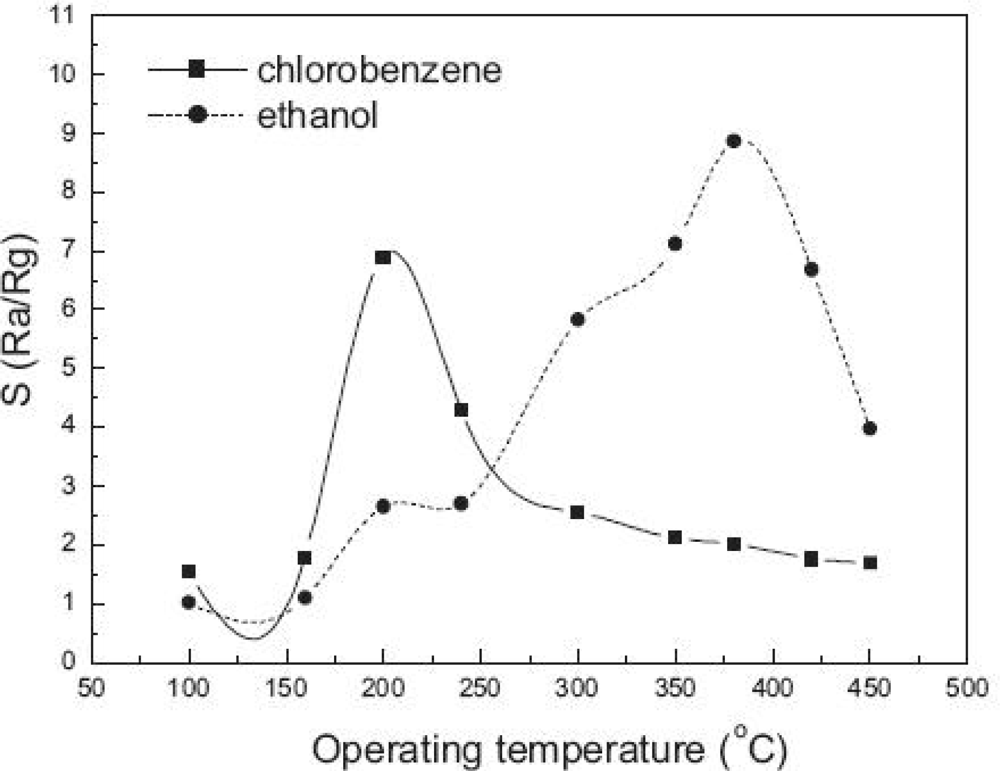
4.4. Humidity and Temperature
5. Conclusions
Acknowledgments
References and Notes
- Korotcenkov, G. Metal Oxides for Solid-State Gas Sensors: What Determines Our Choice? Mater. Sci. Eng. B 2007, 139, 1–23. [Google Scholar]
- Barsan, N.; Koziej, D.; Weimar, U. Metal Oxide-Based Gas Sensor Research: How to? Sens. Actuat. B 2007, 121, 18–35. [Google Scholar]
- Korotcenkov, G. The Role of Morphology and Crystallographic Structure of Metal Oxides in Response of Conductometric-Type Gas Sensors. Mater. Sci. Eng R 2008, 61, 1–39. [Google Scholar]
- Batzill, M. Surface Science Studies of Gas Sensing Materials: SnO2. Sensors 2006, 6, 1345–1366. [Google Scholar]
- Barsan, N.; Schweizer-Berberich, M.; Göpel, W. Fundamental and Practical Aspects in the Design of Nanoscaled SnO2 Gas Sensors: a Status Report. Fresenius J. Anal. Chem 1999, 365, 287–304. [Google Scholar]
- Park, C.O.; Akbar, S.A. Ceramics for Chemical Sensing. J. Mater. Sci 2003, 38, 4611–4637. [Google Scholar]
- Batzill, M.; Diebold, U. The Surface and Materials Science of Tin Oxide. Prog. Surf. Sci 2005, 79, 47–154. [Google Scholar]
- Moos, R.; Sahner, K.; Fleischer, M.; Guth, U.; Barsan, N.; Weimar, U. Solid State Gas Sensor Research in Germany—a Status Report. Sensors 2009, 9, 4323–4365. [Google Scholar]
- Rumyantseva, M.N.; Gas’kov, A.M. Chemical Modification of Nanocrystalline Metal Oxides: Effect of the Real Structure and Surface Chemistry on the Sensor Properties. Russ. Chem. Bull 2008, 57, 1106–1125. [Google Scholar]
- Lee, J.H. Gas Sensors Using Hierarchical and Hollow Oxide Nanostructures: Overview. Sens. Actuat. B 2009, 140, 319–336. [Google Scholar]
- Fergus, J.W. Perovskite Oxides for Semiconductor-Based Gas Sensors. Sens. Actuat. B 2007, 123, 1169–1179. [Google Scholar]
- Lu, J.G.; Chang, P.C.; Fan, Z.Y. Quasi-One-Dimensional Metal Oxide Materials-Synthesis, Properties and Applications. Mater. Sci. Eng. R 2006, 52, 49–91. [Google Scholar]
- Orton, J.W.; Powell, M.J. The Hall Effect in Polycrystalline and Powdered Semiconductors. Rep. Prog. Phys 1980, 43, 1263–1307. [Google Scholar]
- Rothschild, A.; Komem, Y. On the Relationship between the Grain Size and Gas-Sensitivity of Chemo-Resistive Metal-Oxide Gas Sensors with Nanosized Grains. J. Electroceram 2004, 13, 697–701. [Google Scholar]
- Rothschild, A.; Komem, Y. The Effect of Grain Size on the Sensitivity of Nanocrystalline Metal-Oxide Gas Sensors. J. Appl. Phys 2004, 95, 6374–6380. [Google Scholar]
- Comini, E.; Baratto, C.; Faglia, G.; Ferroni, M.; Vomiero, A.; Sberveglieri, G. Quasi-One Dimensional Metal Oxide Semiconductors: Preparation, Characterization and Application as Chemical Sensors. Prog.Mater. Sci 2009, 54, 1–67. [Google Scholar]
- Korotcenkov, G. Gas Response Control through Structural and Chemical Modification of Metal Oxide Films: State of the Art and Approaches. Sens. Actuat. B 2005, 107, 209–232. [Google Scholar]
- Yamazoe, N.; Shimanoe, K. New Perspectives of Gas Sensor Technology. Sens. Actuat. B 2009, 138, 100–107. [Google Scholar]
- Tiemann, M. Porous Metal Oxides as Gas Sensors. Chem. Eur. J 2007, 13, 8376–8388. [Google Scholar]
- Eranna, G.; Joshi, B.C.; Runthala, D.P.; Gupta, R.P. Oxide Materials for Development of Integrated Gas Sensors-A Comprehensive Review. Crit. Rev. Solid State Mater. Sci 2004, 29, 111–188. [Google Scholar]
- Kanazawa, E.; Sakai, G.; Shimanoe, K.; Kanmura, Y.; Teraoka, Y.; Miura, N.; Yamazoe, N. Metal Oxide Semiconductor N2O Sensor for Medical Use. Sens. Actuators B 2001, 77, 72–77. [Google Scholar]
- Henrich, V.E.; Cox, P.A. The Surface Science of Metal Oxides; Cambridge University Press: Cambridge, UK, 1994. [Google Scholar]
- Franke, M.E.; Koplin, T.J.; Simon, U. Metal and Metal Oxide Nanoparticles in Chemiresistors: Does the Nanoscale Matter? Small 2006, 2, 36–50. [Google Scholar]
- Schierbaum, K.D.; Weimar, U.; Göpel, W.; Kowalkowski, R. Conductance, Work Function and Catalytic Activity of SnO2-Based Gas Sensors. Sens. Actuat. B 1991, 3, 205–214. [Google Scholar]
- Göpel, W.; Schierbaum, K.D. SnO2 Sensors-Current Status and Future Prospects. Sens. Actuat. B 1995, 26, 1–12. [Google Scholar]
- Yu, J.H.; Choi, G.M. Electrical and CO Gas Sensing Properties of ZnO-SnO2 Composites. Sens. Actuat. B 1998, 52, 251–256. [Google Scholar]
- De Lacy Costello, B.P.J.; Ewen, R.J.; Jones, P.R.H.; Ratcliffe, N.M.; Wat, R.K.M. A Study of the Catalytic and Vapour-Sensing Properties of Zinc Oxide and Tin Dioxide in Relation to 1-Butanol and Dimethyldisulphide. Sens. Actuat. B 1999, 61, 199–207. [Google Scholar]
- Zhu, C.L.; Chen, Y.J.; Wang, R.X.; Wang, L.J.; Cao, M.S.; Shi, X.L. Synthesis and Enhanced Ethanol Sensing Properties of α-Fe2O3/ZnO Heteronanostructures. Sens. Actuat. B 2009, 140, 185–189. [Google Scholar]
- Yoon, D.H.; Yu, J.H.; Choi, G.M. CO Gas Sensing Properties of ZnO-CuO Composite. Sens. Actuat. B 1998, 46, 15–23. [Google Scholar]
- Teterycz, H.; Klimkiewicz, R.; Licznerski, B.W. A New Metal Oxide Catalyst in Alcohol Condensation. Appl. Catal. A: Gen 2001, 214, 243–249. [Google Scholar]
- Meixner, H.; Lampe, U. Metal Oxide Sensors. Sens. Actuat. B 1996, 33, 198–202. [Google Scholar]
- De Lacy Costello, B.P.J.; Ewen, R.J.; Ratcliffe, N.M.; Sivanand, P.S. Thick Film Organic Vapour Sensors Based On Binary Mixtures of Metal Oxides. Sens. Actuat. B 2003, 92, 159–166. [Google Scholar]
- Ivanovskaya, M.; Kotsikau, D.; Faglia, G.; Nelli, P. Influence of Chemical Composition and Structural Factors of Fe2O3/In2O3 Sensors on Their Selectivity and Sensitivity to Ethanol. Sens. Actuat. B 2003, 96, 498–503. [Google Scholar]
- Hu, Y.; Zhou, X.; Han, Q.; Cao, Q.; Huang, Y. Sensing Properties of CuO-ZnO Heterojunction Gas Sensors. Mater.Sci. Eng. B 2003, 99, 41–43. [Google Scholar]
- Ling, Z.; Leach, C. The Effect of Relative Humidity on the NO2 Sensitivity of a SnO2/WO3 Heterojunction Gas Sensor. Sens. Actuat. B 2004, 102, 102–106. [Google Scholar]
- Aygün, S.; Cann, D. Hydrogen Sensitivity of Doped CuO/ZnO Heterocontact Sensors. Sens. Actuat. B 2005, 106, 837–842. [Google Scholar]
- Patil, L.A.; Patil, D.R. Heterocontact Type CuO-Modified SnO2 Sensor for the Detection of a ppm Level H2S Gas at Room Temperature. Sens. Actuat. B 2006, 120, 316–323. [Google Scholar]
- Herrán, J.; Mandayo, G.G.; Castaño, E. Physical Behaviour of BaTiO3-CuO Thin-Film under Carbon Dioxide Atmospheres. Sens. Actuat. B 2007, 127, 370–375. [Google Scholar]
- Haridas, D.; Gupta, V.; Sreenivas, K. Enhanced Catalytic Activity of Nanoscale Platinum Islands Loaded onto SnO2 Thin Film for Sensitive LPG Gas Sensors. Bull. Mater. Sci 2008, 31, 397–400. [Google Scholar]
- Hyodo, T.; Baba, Y.; Wada, K.; Shimizu, Y.; Egashira, M. Hydrogen Sensing Properties of SnO2 Varistors Loaded with SiO2 by Surface Chemical Modification with Diethoxydimethylsilane. Sens. Actuat. B 2000, 64, 175–181. [Google Scholar]
- Lu, Y.; Li, J.; Han, J.; Ng, H.-T.; Binder, C.; Partridge, C.; Meyyappan, M. Room Temperature Methane Detection Using Palladium Loaded Single-Walled Carbon Nanotube Sensors. Chem. Phys. Lett 2004, 391, 344–348. [Google Scholar]
- Wang, D.; Ma, Z.; Dai, S.; Liu, J.; Nie, Z.; Engelhard, M.H.; Huo, Q.; Wang, C.; Kou, R. Low-Temperature Synthesis of Tunable Mesoporous Crystalline Transition Metal Oxides and Applications as Au Catalyst Supports. J. Phys. Chem. C 2008, 112, 13499–13509. [Google Scholar]
- Haridas, D.; Sreenivas, K.; Gupta, V. Improved Response Characteristics of SnO2 Thin Film Loaded with Nanoscale Catalysts for LPG Detection. Sens. Actuat. B 2008, 133, 270–275. [Google Scholar]
- Shimizu, Y.; Matsunaga, N.; Hyodo, T.; Egashira, M. Improvement of SO2 Sensing Properties of WO3 by Noble metal Loading. Sens. Actuat. B 2001, 77, 35–40. [Google Scholar]
- Ruiz, A.M.; Cornet, A.; Shimanoe, K.; Morante, J.R.; Yamazoe, N. Effects of Various Metal Additives on the Gas Sensing Performances of TiO2 Nanocrystals Obtained from Hydrothermal Treatments. Sens. Actuat. B 2005, 108, 34–40. [Google Scholar]
- Kolmakov, A.; Klenov, D.O.; Lilach, Y.; Stemmer, S.; Moskovits, M. Enhanced Gas Sensing by Individual SnO2 Nanowires and Nanobelts Functionalized with Pd Catalyst Particles. Nano Lett 2005, 5, 667–673. [Google Scholar]
- Henry, C.R.; Chapon, C.; Duriez, C. Precursor State in the Chemisorption of CO on Supported Palladium Clusters. J. Chem. Phys 1991, 95, 700–705. [Google Scholar]
- Tsu, K.; Boudart, M. Recombination of Atoms at the Surface of Thermocouple Probes. Can. J. Chem 1961, 39, 1239–1246. [Google Scholar]
- Boudart, M. On the Nature of Spilt-over hydrogen. J. Mol. Catal. A: Chem 1999, 138, 319–321. [Google Scholar]
- Lu, F.; Liu, Y.; Dong, M.; Wang, X.P. Nanosized Tin Oxide as the Novel Material with Simultaneous Detection towards CO, H2 and CH4. Sens. Actuat. B 2000, 66, 225–227. [Google Scholar]
- Ansari, S.G.; Boroojerdian, P.; Sainkar, S.R.; Karekar, R.N.; Aiyer, R.C.; Kulkarni, S.K. Grain Size Effects on H2 Gas Sensitivity of Thick Film Resistor Using SnO2 Nanoparticles. Thin Solid Films 1997, 295, 271–276. [Google Scholar]
- Barsan, N.; Weimar, U. Conduction Model of Metal Oxide Gas Sensors. J. Electroceram 2001, 7, 143–167. [Google Scholar]
- Xu, C.; Tamaki, J.; Miura, N.; Yamazoe, N. Grain Size Effects on Gas Sensitivity of Porous SnO2-Based Elements. Sens. Actuat. B 1991, 3, 147–155. [Google Scholar]
- Timmer, B.; Olthuis, W.; Van den Berg, A. Ammonia Sensors and Their Applications-a Review. Sens. Actuat. B 2005, 107, 666–677. [Google Scholar]
- Wang, X.; Yee, S.S.; Carey, W.P. Transition between Neck-Controlled and Grain-Boundary-Controlled Sensitivity of Metal-Oxide Gas Sensors. Sens. Actuat. B 1995, 25, 454–457. [Google Scholar]
- Barsan, N. Conduction Models in Gas-Sensing SnO2 Layers: Grain-Size Effects and Ambient Atmosphere Influence. Sens. Actuators B 1994, 17, 241–246. [Google Scholar]
- Liu, H.; Gong, S.P.; Hu, Y.X.; Liu, J.Q.; Zhou, D.X. Properties and Mechanism Study of SnO2 Nanocrystals for H2S Thick-Film Sensors. Sens. Actuat. B 2009, 140, 190–195. [Google Scholar]
- Korotcenkov, G.; Brinzari, V.; Ivanov, M.; Cerneavschi, A.; Rodriguez, J.; Cirera, A.; Cornet, A.; Morante, J. Structural Stability of Indium Oxide Films Deposited by Spray Pyrolysis During Thermal Annealing. Thin Solid Films 2005, 479, 38–51. [Google Scholar]
- Rao, C.N.R.; Kulkarni, G.U.; Thomas, P.J.; Edwards, P.P. Size-Dependent Chemistry: Properties of Nanocrystals. Chem. Eur. J 2002, 8, 28–35. [Google Scholar]
- Geng, B.; Fang, C.; Zhan, F.; Yu, N. Synthesis of Polyhedral ZnSnO3 Microcrystals with Controlled Exposed Facets and Their Selective Gas-Sensing Properties. Small 2008, 4, 1337–1343. [Google Scholar]
- Devi, G.S.; Hyodo, T.; Shimizu, Y.; Egashira, M. Synthesis of Mesoporous TiO2-Based Powders and Their Gas-Sensing Properties. Sens. Actuat. B 2002, 87, 122–129. [Google Scholar]
- Cabot, A.; Arbiol, J.; Cornet, A.; Morante, J.R.; Chen, F.; Liu, M. Mesoporous Catalytic Filters for Semiconductor Gas Sensors. Thin Solid Films 2003, 436, 64–69. [Google Scholar]
- Ciesla, U.; Demuth, D.; Leon, R.; Petroff, P.; Stucky, G.; Unger, K.; Schuth, F. Surfactant Controlled Preparation of Mesostructured Transition-Metal Oxide Compounds. Chem. Commun 1994, 1387–1388. [Google Scholar] [CrossRef]
- Hudson, M.J.; Knowles, J.A. Preparation and Characterisation of Mesoporous, High-Surface-Area Zirconium (IV) Oxide. J. Mater. Chem 1996, 6, 89–95. [Google Scholar]
- Yang, J.; Hidajat, K.; Kawi, S. Synthesis of Nano-SnO2/SBA-15 Composite as a Highly Sensitive Semiconductor Oxide Gas Sensor. Mater. Lett 2008, 62, 1441–1443. [Google Scholar]
- Liu, X.W.; Li, J.W.; Zhou, L.; Huang, D.S.; Zhou, Y.P. Adsorption of CO2, CH4 and N2 on Ordered Mesoporous Silica Molecular Sieve. Chem. Phys. Lett 2005, 415, 198–201. [Google Scholar]
- Kim, J.C.; Jun, H.K.; Huh, J.-S.; Lee, D.D. Tin Oxide-Based Methane Gas Sensor Promoted by Alumina-Supported Pd Catalyst. Sens. Actuators B 1997, 45, 271–277. [Google Scholar]
- Ogawa, H.; Nishikawa, M.; Abe, A. Hall Measurement Studies and an Electrical Conduction Model of Tin Oxide Ultrafine Particle Films. J. Appl. Phys 1982, 53, 4448–4455. [Google Scholar]
- Sysoev, V.V.; Schneider, T.; Goschnick, J.; Kiselev, I.; Habicht, W.; Hahn, H.; Strelcov, E.; Kolmakov, A. Percolating SnO2 Nanowire Network as a Stable Gas Sensor: Direct Comparison of Long-Term Performance versus SnO2 Nanoparticle Films. Sens. Actuat. B 2009, 139, 699–703. [Google Scholar]
- Traversa, E. Ceramic Sensors for Humidity Detection: The State-of-the-art and Future Developments. Sens. Actuat. B 1995, 23, 135–156. [Google Scholar]
- McCafferty, E.; Zettlemoyer, A.C. Adsorption of Water Vapour on α-Fe2O3. Discuss. Faraday Soc 1971, 52, 239–263. [Google Scholar]
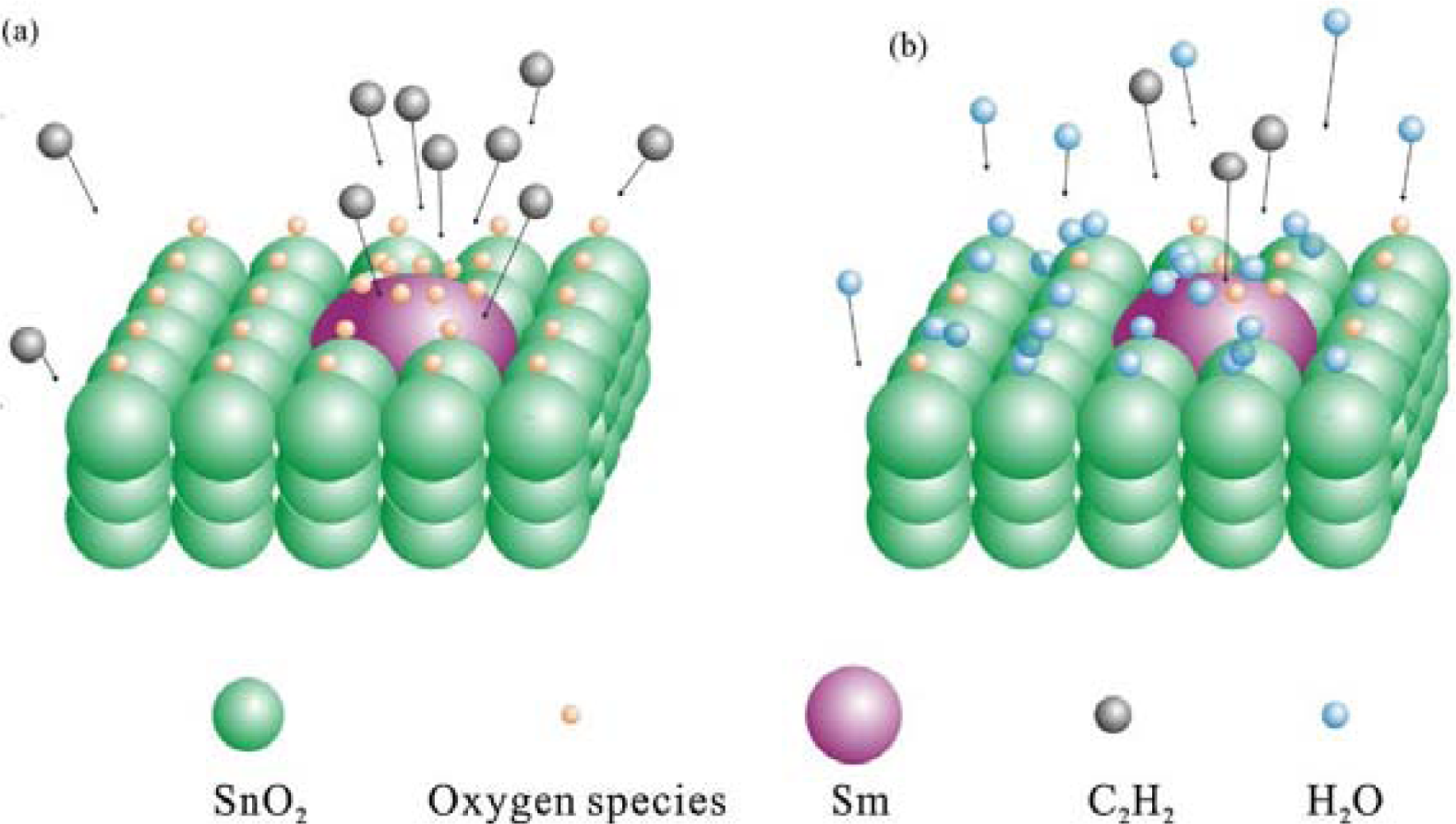
- Qi, Q.; Zhang, T.; Zheng, X.; Fan, H.; Liu, L.; Wang, R.; Zeng, Y. Electrical Response of Sm2O3-Doped SnO2 to C2H2 and Effect of Humidity Interference. Sens. Actuat. B 2008, 134, 36–42. [Google Scholar]
- Gong, J.; Chen, Q.; Lian, M.; Liu, N.; Stevenson, R.G.; Adamic, F. Micromachined Nanocrystalline Silver Doped SnO2 H2S Sensor. Sens. Actuat. B 2006, 114, 32–39. [Google Scholar]
- Egashira, M.; Kawasumi, S.; Kagawa, S.; Seiyama, T. Temperature Programmed Desorption Study of Water Absorbed on Metal Oxides. I. Anatase and Rutile. Bull. Chem. Soc. Jpn 1978, 51, 3144–3149. [Google Scholar]
- Tischner, A.; Maier, T.; Stepper, C.; Köck, A. Ultrathin SnO2 Gas Sensors Fabricated by Spray Pyrolysis for the Detection of Humidity and Carbon Monoxide. Sens. Actuat. B 2008, 134, 796–802. [Google Scholar]
- Jing, Z.; Zhan, J. Fabrication and Gas-Sensing Properties of Porous ZnO Nanoplates. Adv. Mater 2008, 20, 4547–4551. [Google Scholar]
- Malyshev, V.V.; Pislyakov, A.V. Investigation of Gas-Sensitivity of Sensor Structures to Hydrogen in a Wide Range of Temperature, Concentration and Humidity of Gas Medium. Sens. Actuat. B 2008, 134, 913–921. [Google Scholar]
- Kim, I.-D.; Rothschild, A.; Lee, B.H.; Kim, D.Y.; Jo, S.M.; Tuller, H.L. Ultrasensitive Chemiresistors Based on Electrospun TiO2 Nanofibers. Nano Lett 2006, 6, 2009–2013. [Google Scholar]
- Cao, M.; Wang, Y.; Chen, T.; Antonietti, M.; Niederberger, M.A. Highly Sensitive and Fast-Responding Ethanol Sensor Based on CdIn2O4 Nanocrystals Synthesized by a Nonaqueous Sol-Gel Route. Chem. Mater 2008, 20, 5781–5786. [Google Scholar]
- Duy, N.V.; Hieu, N.V.; Huy, P.H.; Chien, N.D.; Thamilselvan, M.; Yi, J. Mixed SnO2/TiO2 Included with Carbon Nanotubes for Gas-Sensing Application. Physica E 2008, 41, 258–263. [Google Scholar]

© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, C.; Yin, L.; Zhang, L.; Xiang, D.; Gao, R. Metal Oxide Gas Sensors: Sensitivity and Influencing Factors. Sensors 2010, 10, 2088-2106. https://doi.org/10.3390/s100302088
Wang C, Yin L, Zhang L, Xiang D, Gao R. Metal Oxide Gas Sensors: Sensitivity and Influencing Factors. Sensors. 2010; 10(3):2088-2106. https://doi.org/10.3390/s100302088
Chicago/Turabian StyleWang, Chengxiang, Longwei Yin, Luyuan Zhang, Dong Xiang, and Rui Gao. 2010. "Metal Oxide Gas Sensors: Sensitivity and Influencing Factors" Sensors 10, no. 3: 2088-2106. https://doi.org/10.3390/s100302088
APA StyleWang, C., Yin, L., Zhang, L., Xiang, D., & Gao, R. (2010). Metal Oxide Gas Sensors: Sensitivity and Influencing Factors. Sensors, 10(3), 2088-2106. https://doi.org/10.3390/s100302088



