Signal Amplification by Enzymatic Reaction in an Immunosensor Based on Localized Surface Plasmon Resonance (LSPR)
Abstract
:1. Introduction
2. Results and Discussion
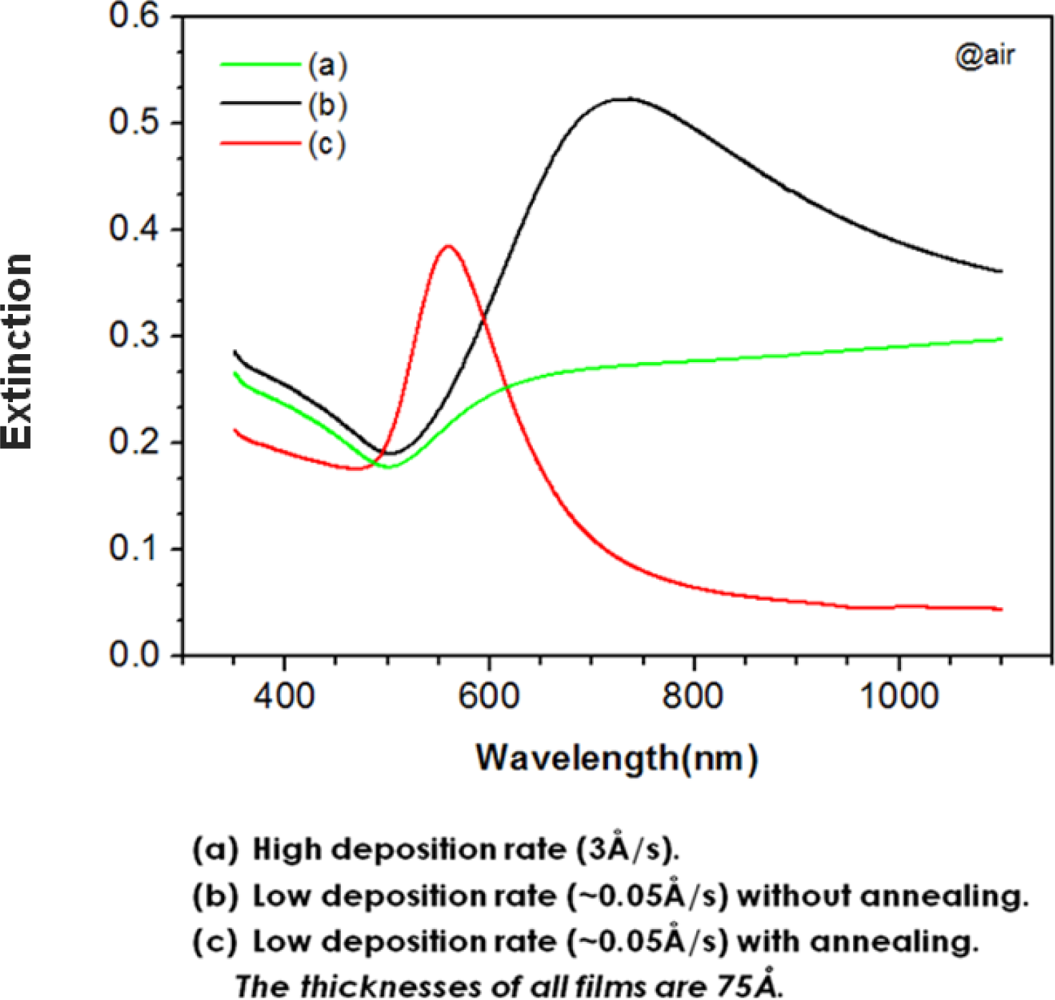
2.1. Annealing Effect on the Morphology and LSPR Spectrum of a Thin Gold Film
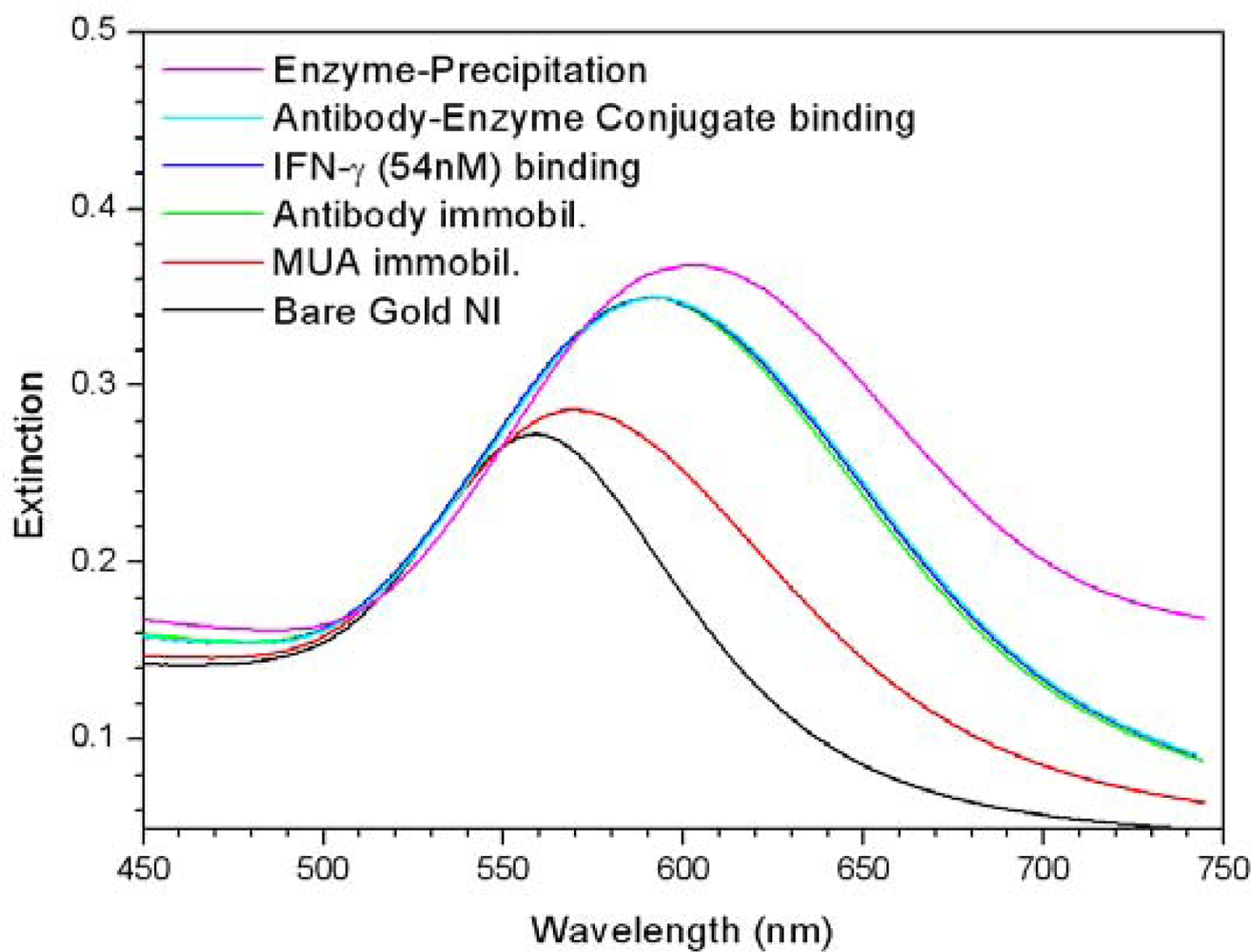
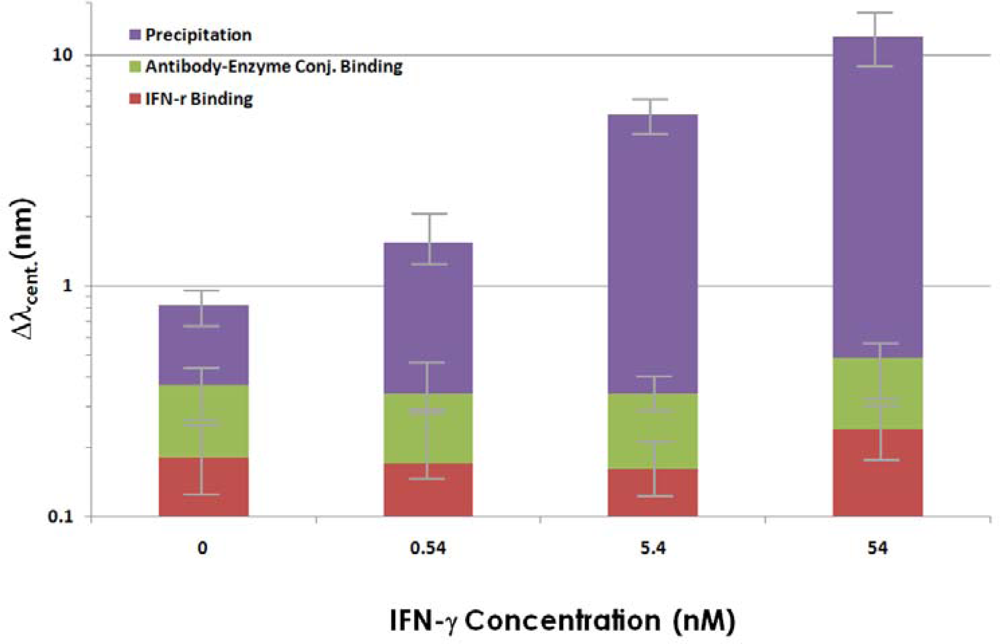
2.2. Signal Amplification using Enzymatic Precipitation in Immunoassay with Gold NI Chip
3. Experimental Section
3.1. Formation of Fold Nano-Island(NI) Films on Glass Substrates
3.2. Modifications of Gold NI Surfaces and Immobilization of IFN-γ antibody on Gold NI Surfaces
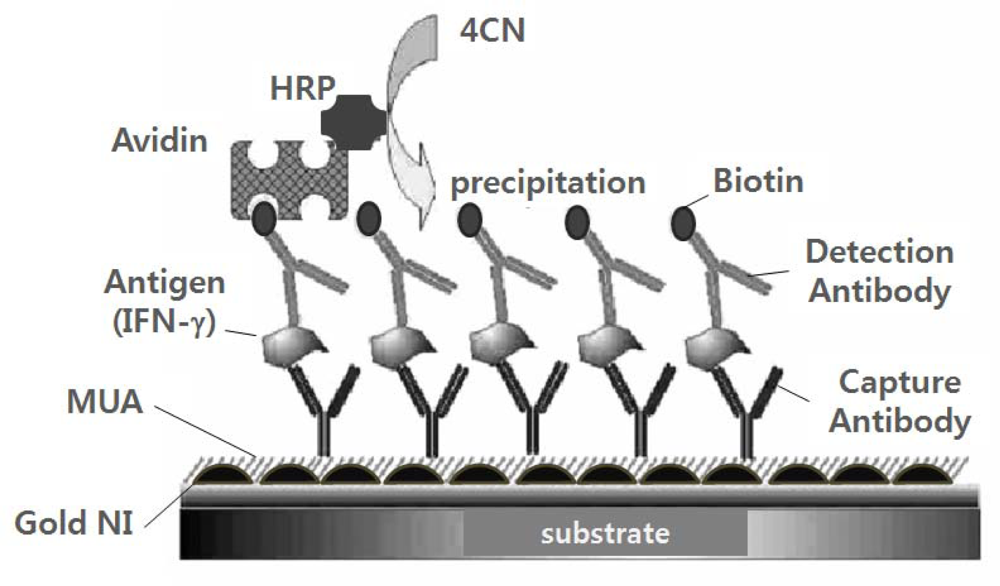
3.3. Signal Amplification Using an Enzymatic Precipitating Reaction on the Gold NI Surface
3.4. Measurements of LSP Bands and Surface Morphologies
4. Conclusions
Acknowledgments
References and Notes
- Nath, N.; Chilkoti, A. A colorimetric gold nanoparticle sensor to interrogate biomolecular interactions in real time on surface. Anal. Chem 2002, 74, 504–509. [Google Scholar]
- Frederix, F.; Friedt, J.; Choi, K.; Laureyn, W.; Campitelli, A.; Mondelaers, D.; Maes, G.; Borghs, G. Biosensing based on light absorption of nanoscaled gold and silver particles. Anal. Chem 2003, 75, 6894–6900. [Google Scholar]
- Lahav, M.; Vaskevich, A.; Rubinstein, I. Biological sensing using transmission surface plasmon resonance spectroscopy. Langmuir 2004, 20, 7365–7367. [Google Scholar]
- Shin, Y.; Lee, J.; Park, M.; Kim, M.; Chung, B.H.; Pyo, H.; Maeng, M. Analysis of recombinant protein expression using localized surface plasmon resonance (LSPR). Biosens. Bioelectron 2007, 22, 2301–2307. [Google Scholar]
- Hiep, H.M.; Endo, T.; Saito, M.; Chikae, M.; Kim, D.K.; Yamamura, S.; Takamura, Y.; Tamiya, E. Label-free detection of melittin binding to a membrane using electrochemical localized surface plasmon resonance. Anal. Chem 2008, 80, 1859–1864. [Google Scholar]
- Haes, A.J.; Hall, W.P.; Chang, L.; Klein, W.L.; Van Duyne, R.P. A localized surface plasmon resonance biosensor: first steps toward an assay for alzheimer’s disease. Nano lett 2004, 4, 1029–1034. [Google Scholar]
- Endo, T.; Kerman, K.; Nagatani, N.; Takamura, Y.; Tamiya, E. Label-free detection of peptide nucleic acid−dna hybridization using localized surface plasmon resonance based optical biosensor. Anal. Chem 2005, 77, 6976–6984. [Google Scholar]
- Yonzon, C.R.; Stuart, D.A.; Zhang, X.; McFarland, A.D.; Haynes, C.L.; Van Duyne, R.P. Toward advanced chemical and biological nanosensors-an overview. Talanta 2005, 67, 438–448. [Google Scholar]
- Kalyuzhny, G.; Vaskevich, A.; Schneeweiss, M.A.; Rubinstein, I. Transmission surface plasmon resonance (T-SPR) measurements for monitoring adsorption on ultrathin gold island films. Chem. Eur. J 2002, 8, 3849–3857. [Google Scholar]
- Doron-Mor, I.; Barkay, Z.; Filip-Granit, N.; Vaskevich, A.; Rubinstein, I. Ultrathin gold island films on silanized glass. Morphology and optical properties. Chem. Mater 2004, 16, 3476–3483. [Google Scholar]
- Dahlin, A.B.; Tegenfeldt, J.O.; Höök, F. Improving the instrumental resolution of sensors based on localized surface plasmon resonance. Anal. Chem 2006, 78, 4416–4423. [Google Scholar]
- Raether, H. Surface Plasmons on Smooth and Rough Surfaces and on Gratings; Springer-Verlag: Berlin Heidelberg, Germany, 1988; pp. 4–7. [Google Scholar]
- Homola, J.; Yee, S.S.; Gauglitz, G. Surface plasmon resonance sensors: review. Sens. Actuat. B 1999, 54, 3–15. [Google Scholar]
- Kim, M.; Shin, Y.; Jung, J.; Ro, H.; Chung, B.H. Enhanced sensitivity of surface plasmon resonance (SPR) immunoassays using a peroxidase-catalyzed precipitation reaction and application to a protein microarray. J. Immun. Method 2005, 297, 125–132. [Google Scholar]
- Patolsky, F.; Zayats, M.; Katz, E.; Willner, I. Precipitation of an insoluble product on enzyme monolayer electrodes for biosensor applications: characterization by faradaic impedance spectroscopy, cyclic voltammetry, and microgravimetric quartz crystal microbalance analyses. Anal. Chem 1999, 71, 3171–3180. [Google Scholar]

© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, T.-H.; Lee, S.-W.; Jung, J.-A.; Ahn, J.; Kim, M.-G.; Shin, Y.-B. Signal Amplification by Enzymatic Reaction in an Immunosensor Based on Localized Surface Plasmon Resonance (LSPR). Sensors 2010, 10, 2045-2053. https://doi.org/10.3390/s100302045
Lee T-H, Lee S-W, Jung J-A, Ahn J, Kim M-G, Shin Y-B. Signal Amplification by Enzymatic Reaction in an Immunosensor Based on Localized Surface Plasmon Resonance (LSPR). Sensors. 2010; 10(3):2045-2053. https://doi.org/10.3390/s100302045
Chicago/Turabian StyleLee, Tae-Han, Seung-Woo Lee, Ji-Ae Jung, Junhyoung Ahn, Min-Gon Kim, and Yong-Beom Shin. 2010. "Signal Amplification by Enzymatic Reaction in an Immunosensor Based on Localized Surface Plasmon Resonance (LSPR)" Sensors 10, no. 3: 2045-2053. https://doi.org/10.3390/s100302045
APA StyleLee, T.-H., Lee, S.-W., Jung, J.-A., Ahn, J., Kim, M.-G., & Shin, Y.-B. (2010). Signal Amplification by Enzymatic Reaction in an Immunosensor Based on Localized Surface Plasmon Resonance (LSPR). Sensors, 10(3), 2045-2053. https://doi.org/10.3390/s100302045



