Abstract
(1) Background: Airborne microbes are an integral part of a cave ecosystem. Cave allochtonous airborne microbiota, which occurs mainly during aerosolization from an underground river, from animals, and from visitors, is particularly pronounced in show caves. The impacts of tourists and natural river aerosolization on the cave air were estimated in large cave spaces within the Škocjan Caves; (2) Methods: Simultaneously with the measurements of atmospheric parameters, cultivable airborne bacteria were impacted, counted and identified using MALDI-TOF MS (Matrix-Assisted Laser Desorption/Ionization Time-Of-Flight Mass Spectrometry); (3) Results: A mix of bacteria typically associated with humans and with natural habitats, including a large percentage of non-identified isolates, was found in the cave air. Few of the isolates were attributed to Risk Group 2. A strong positive correlation between tourist numbers and the rise in the concentration of airborne bacteria was indicated. Concentration of airborne bacteria rises to particularly high levels close to the underground river during periods of high discharge. A 10-times lower discharge reflected an approximately 20-times lower concentration of airborne bacteria; (4) Conclusions: Caves that are open and visited contain a diverse airborne microbiota originating from different sources. Enormous cave chambers that display relatively dynamic cave climate conditions do not normally support the enhancement of airborne bacterial concentrations.
1. Introduction
Some microorganisms have their own ability to move which, on the micro scale, serves to support active movement, searching for nutrients and new ecological niches [1]. For long-distance transport, microbes and other minute particles use one of three modes of dispersion: air (anemochoric), water (hydrochloric), or biological vectors (biochoric), the latter via either animal or human vectors [2]. When airborne, microbes can travel reasonably great distances and can have a global impact on ecosystems [3]. Results of laboratory experiments provide fundamental data on the transport and fate of fine particles, but in some cases it is appropriate to adopt a natural model system.
An example of natural, more or less confined, systems is provided by caves, which are advantageous for some physiological and ecological studies because they lack various environmental stressors, such as UV irradiation and desiccation. On the other hand, elevated concentrations of radon and its decay products might have a significant impact on airborne microbiota [4]. Some microorganisms are already airborne when they enter caves from the external surface, whereas others become airborne due to the splashing of water or the presence and activity of animals and humans. This latter effect is best observed in show caves, where visitors introduce and spread many inanimate and living particles [5,6,7,8]. An example from an indoor environment, a children’s classroom, showed that the emission rates of microbes from human skin can range from 0.8 to 35 × 106 bacterial cells per person-hour and from 3 to 57 × 106 fungal cells per person-hour [9]. Dissemination of detached skin scales is not their only influence because humans impact the air quality directly, by breathing, coughing, and sneezing. Related droplets and aerosols contain numerous microorganisms originating from the upper respiratory tract and from the oral mucosa, and these are part of the normal, persistent, or transient microbiota. Clothes and lint can also serve as a source for airborne bacteria and fungi. Once they are incorporated within an aerosol, microorganisms persist in the air for relatively long periods, and some of them have the potential to cause infections in susceptible hosts. Frequently-encountered bacterial genera are Staphylococcus, Bacillus, Kocuria, Micrococcus, Corynebacterium, and Arthrobacter, alongside fungi from genera, such as Candida, Aureobasidium, Penicillium, and Phoma [10], and many other microorganisms were revealed using culture-independent techniques [11]. These microbes can remain viable on various surfaces for periods ranging from days to months, depending upon the species and the environmental conditions, particularly the temperature and relative humidity [12].
The airborne microbiota, together with microenvironmental and climatic disturbances caused by human activities [4,8,13,14], can contribute to biodeterioration of many susceptible items preserved underground, especially artefacts of cultural heritage, such as the prehistoric paintings and rock art in Altamira Cave and Nerja Cave in Spain [15], and in Lascaux Cave in France [16]. Taking a broader view, the importance of aerobiology for cave ecology lies in the characterization of introduced biomass, its impacts upon cave biota, survival rate, and dispersal limitation, and any interaction with the cave-autochtonous microbes. The study provided estimates of the qualitative and quantitative impact of tourists on cave air, and the impact of natural aerosolization driven by the underground river upon the total airborne biomass in a large cave system.
2. Materials and Methods
2.1. Cave Description and Sites of Bioaerosol Impacting
Within the Classical Karst of Slovenia, the Škocjan Caves (Škocjanske jame, 45°39′53.33 N 13°59′40.44 E) are part of a complex geomorphological feature composed of four caves and deep, vertical-sided, collapse dolines formed by the Reka River [17,18,19]. The Reka River drains water from a surface catchment of more than 350 km2. Its average discharge upstream of its sink into the Škocjan Cave is 8.95 m3/s, but during floods it can reach 387 m3/s. From the Škocjan Caves, the Reka River flows underground through the karst massif towards the Timavo spring, some 35 km away in Italy. The Škocjan Caves are 6.2 km long and 250 m deep, with large galleries and chambers, and the underground canyon carrying the Reka River [20]. They are formed in Cretaceous and Paleocene limestone, close to the contact with impermeable Eocene flysch rocks that consist of quartz-sandstone, conglomerate and marl [21]. Canyon passages have developed in massive and thick-bedded limestones, mostly along just a few tectonized bedding planes and along distinct fractured zones [22].
The site is on the UNESCO World Heritage List and is recognized as an underground karst wetland under the Ramsar Wetland Classification System. More than 5000 individuals of twelve different bat species have been identified in the Škocjan Caves [23]. A 2.6 km section of the Škocjan Caves is open to tourists throughout the year and attracts approximately 140,000 visitors per year. The number of organized tourist visits during the year depends on the season. Eight regular visits are organized during the peak season (June to September), four in the shoulder season (April, May, October) and two during the off-season (January to March, November and December). In contrast with the dry passages of the tourist route (Tiha jama), the river canyon (Šumeča jama) is subject to flooding and aerosolization by the Reka River (Figure 1).
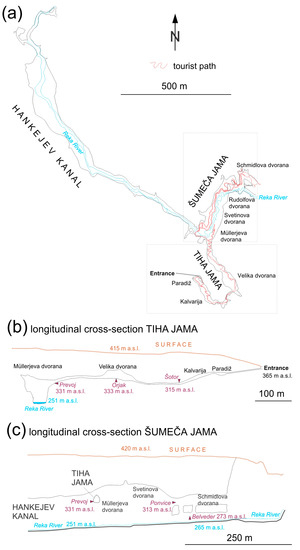
Figure 1.
Map of the Škocjan Caves (a) with the designated locations of bioaerosol sampling sites in the Tiha jama section (Šotor, Orjak) along the tourist footpath (tourist impact, (b)); and in the Šumeča jama section (Prevoj, Ponvice, Belveder, with the Reka River impact, (c)). The ground plan is from the Cave cadastre of the Karst Research Institute ZRC SAZU (drawn by Franjo Drole).
Due to the specific cave morphology in the Tiha jama section, bioaerosols deriving from tourists provide most of the allochtonous input to the cave air whereas, in the Šumeča jama section, the impact on the cave air is attributed mainly to the Reka River (Figure 1). Approximately five metres from the tourist footpath in Tiha jama, air was sampled at a site named Šotor at the end of a large chamber (Kalvarija) with an estimated volume of 23,144 m3 (22 January 2012, 1 April 2012, 3 June 2012, 22 July 2012, 22 August 2012, 26 September 2013, 3 December 2013, 1 July 2014, and 11 November 2014). Additionally, in the Tiha jama section, an additional site close to a stalagmite known as “Orjak” was sampled (13 November 2014) in the Velika dvorana chamber (estimated volume: 64,960 m3).
To obtain details of the background concentration of airborne bacteria, each day’s air sampling started one hour before the cave was open for tourists, and continued up to 1.5 h after the final tourist group had passed the sampling site. During the high tourist season, several tourist groups usually followed each other through the cave. The number of passing tourists were recorded.
In the Šumeča jama section of the cave, three sampling sites were selected to obtain a vertical transect above the Reka River: Belveder (Be)—the closest to the Reka River (~8 m); Ponvice (Po)—40 m above Belveder; and Prevoj (Pr) at the entry point to the Šumeča jama section, 58 m above Be (Figure 1). On 12 November 2014, the discharge of the Reka River upstream of its ponor was increasing (165–208 m3/s), on 27 August 2015 it was in a recession period (0.201–0.385 m3/s), as was also the case on 23 November 2016 (22.3–22.9 m3/s). Data on discharge were provided by the Slovenian Environment Agency, from the national hydrological network on the Reka River upstream of its ponor for the Reka-Cerkvenikov mlin station and the Reka-Škocjan station [24]).
2.2. Atmospheric Conditions and Cultivation of Airborne Bacteria
Temperature was measured continuously (with a data acquisition period of 30 s) before and during tourist visits using a portable Kestrel 4500 PocketWeather Tracker (Boothwyn, PA, USA) (accuracy: ±1.0 °C; range: 45 to 125 °C), and atmospheric carbon dioxide was measured continuously (with a data acquisition period of 30 s) by a MI70 Vaisala handheld carbon dioxide metre (Vaisala Oyj, Helsinki, Finland) (probe: GMP222; accuracy: ±1.5% of range; range: 0–3000 ppm). Simultaneously with these measurements at the site, a Mas-100 Air Sample Device (Merck, Darmstadt, Germany) (with a constant flow of 100 L/min) was used to inoculate microbiological media with 500 or 1000 L of air. Bioaerosols were sampled 1.5 m above ground level using a portable platform. The perforated plate of the impactor was sterilized with 96% ethanol before inoculating microbiological media. Impacting of the air onto Petri plates with the microbiological media was carried out continuously by changing plates, and lasted up to six hours. Sampling started a few hours before the tourist visits, continued during the passing of the tourist groups, and even after tourists vacated the underground chamber. To minimise the impact of the research team, the operator was not present at the sampling location.
A 1.5% nutrient agar (NA, Sigma-Aldrich, St. Louis, MO, USA) was used to estimate and characterize the culturable airborne bacteria, because it is commonly used in microbiology, and it ensured compatibility with previous studies [4,5]. No antimicrobial substances were added to the media. After impacting, the NA plates were cultivated at 37 ± 1 °C for 24 h. Counted colony-forming units (CFU) values were corrected statistically according to Feller [25] and expressed as CFU per cubic metre. For MALDI-TOF MS (matrix-assisted laser desorption/ionization time-of-flight mass spectrometry) identification, distinct morphotypes from the NA plates (sampled at Šotor on 1 July 2014, 11 November 2014, and 12 November 2014; at Orjak on 13 November 2014; and in the Šumeča jama section on 12 November 2014) were subsequently inoculated on 5.0% defibrinated sheep-blood agar (BA) and incubated at 37 ± 1 °C for 24 to 48 h. BA was prepared immediate before use at the Institute of Microbiology and Immunology, Faculty of Medicine, University of Ljubljana: per 1000 mL: 15 g agar (Sigma-Aldrich), brain-heart infusion broth (Becton Dickinson, Sparks, MD, USA), and 50 mL defibrinated reoxygenated sheep blood (BioGnost, Zagreb, Croatia). Pure cultures from BA never older than 48 h were subjected to MALDI-TOF MS identification. BA is an enriched medium that allows growth of the majority of medically-important bacteria.
2.3. Characterisation of Isolates
MALDI-TOF MS is an emerging method for microbial identification, characterisation, and typing, particularly in clinical microbiology [26,27]. Outside routine diagnostics it is used for epidemiological studies, typing, and detection of antimicrobial resistance and biological warfare agents. The sample—a bacterial colony or lysed bacterial cells—is overlaid with a photo-absorptive matrix that co-crystalizes with the sample and enables the initial ionisation step. The most frequently used matrices in microbiology are α-cyano-4-hydroxy-cinnamic acid (HCCA), 2,5-dihydroxy benzoic acid (DHB), and 3,5-dimethoxy-4-hydroxycinnamic acid (sinapinic acid). The separation of ionised molecules is based on the mass-to-charge ratio (m/z), which is represented in a graph plotting m/z and the relative intensity. The obtained mass spectrum of an unknown microorganism is compared to the mass spectra in the database. For species level identification, a mass range m/z of 2–20 kDa is used, which represents mainly ribosomal proteins, along with a few housekeeping proteins [26,27,28]. Matches of peak positions and their intensities in the mentioned mass range between sample and database spectra are used to generate a match score [27]. Pre-treatment protocols (formic acid extraction, formic acid—ethanol extraction etc.) are needed in cases when extraction of intracellular proteins by matrix alone is not sufficient. The greatest limitation of MALDI-TOF MS is the size of the spectral database, because identification of the isolate is only possible if the database contains the specific type of spectrum [26].
Bacterial isolates on BA were subjected to identification using MALDI-TOF MS with an on-target formic acid extraction technique. A colony was smeared onto the MALDI steel plate and overlaid with 1 μL of 70% formic acid. After drying, the sample was overlaid with 1 μL of saturated α-cyano-4-hydroxy-cinnamic acid (HCCA) matrix solution in 50% acetonitrile-2.5% trifluoroacetic acid (Bruker Daltonik, Bremen, Germany) and left to dry before subsequent analysis with Bruker MALDI Biotyper RTC software version 3.1 (Bruker Daltonik, Bremen, Germany). The Bruker bacterial test standard (Bruker Daltonik) was used for calibration according to the manufacturer’s instructions. The quality of identification was assessed using the manufacturer’s score value. A score of ≥2.000 indicated reliable species level identification, a score of 1.700 to 1.999 indicated reliable identification to the genus level, and a score of <1.700 was interpreted as no identification. Microbial risk groups were assessed according to the Risk Group Database of the American Biological Safety Association [29].
2.4. Statistical Evaluation
Statistical analyses were performed using PAST [30] for species accumulation curve and Daniel’s XL Toolbox, an open-source add-in for Microsoft Excel (Version 6.60, licensed under the Apache License, Version 2.0, Daniel Kraus, Würzburg, Germany) for linear regression.
3. Results and Discussion
3.1. Tourist-Derived Cave Air Biomass
A previous study on aerosol nanoparticles revealed that the Škocjan Caves are well ventilated with notable impacts both from polluted atmospheric air and from tourist presence, particularly in summer [31]. In the current study, the presence of tourists did not inevitably reflect as a clear peak of carbon dioxide (e.g., 22 July 2012, 22 August 2012, 26 September 2013, 11 November 2014, Table 1) that could be attributed to the specificity of the selected microlocation and the impact of the external climate [5]. Additionally, the highest measured concentration of carbon dioxide (2480 ppm) did not correspond to the highest number of visitors (1 July 2014). In caves, the concentration of carbon dioxide is generally lower during the winter, reaching a peak during the summer [32]. The concentration of airborne bacteria during tourist visits always exceeded the bacterial level before the tourists’ presence (Table 1). The lowest concentration of airborne bacteria was recorded when the Tiha jama section was closed to tourists for 44 days (3 December 2013). This value can be regarded as an approximation of the cave air biomass without any tourist influence.

Table 1.
Air temperature, concentrations of carbon dioxide, and airborne bacteria at the Šotor site (Tiha jama) before and after the arrival of tourists, with the number of tourists passing the sampling station; data from 2012 compiled from the previous study [5].
The biomass indicator of cultivable airborne microorganisms that was employed clearly pointed out their human source during tourist visits. After tourists reached the sampling station there was a statistically significant strong positive correlation between the number of tourists and the increase of the concentration of airborne bacteria above the background level (Figure 2). Taking into consideration the recommended limiting value of 300 CFU/m3 for total microbial counts for indoor commercial and residential environments [33], the linear relationship between the increase in the concentration of airborne bacteria and the tourist counts, the impact of just 370 tourists could raise microbe levels to the limiting value.
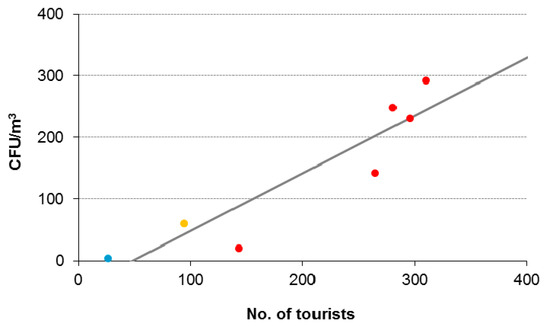
Figure 2.
Relationship between tourist counts (n = 7) and the increase in concentration of airborne bacteria from their background level at the Šotor sampling site (increase in biomass expressed in CFU/m3 = 0.94 × tourist count—45.28; p = 0.001; blue—off-season, orange—shoulder season, red—peak season; see Material and Methods and Table 1 for details).
After tourists vacated the underground chamber, the concentration of airborne bacteria slowly dropped due to air circulation. In such large and relatively open cave systems, natural air movements and the circulation induced by heat emission from lamps enhance the translocation of minute particles, but the question of dispersal limitation in such conditions remains open. It seems that, generally, dispersal limitation can be a key process in structuring the air microbiome [34]. Both biomass and biomass diversity, particularly of fungi, are higher in small and/or closed underground chambers [6]. In the comparatively far smaller Altamira Cave with its Palaeolithic paintings, a notable level of bacteria related to humans was reported, and the use of protective gloves, masks, and clothing was recommended in considering the case for re-opening the cave to the public [13]. For such relatively small and highly vulnerable sites, with rock art susceptible to biodeterioration, there was even a proposal for a tentative index of fungal hazard [14].
3.2. Diversity of Human-Related Airborne Bacteria
Altogether, 868 isolates were subjected to identification by MALDI-TOF MS and 88 (~10%) were discarded from further analysis by the Biotyper RTC software. Two-hundred and eleven processed isolates were from periods preceding tourist presence and 569 were from periods of tourist presence. For both groups, a similar proportion of colonies were identified at species level (no tourists—23.7%; with tourists—21.1%) and at the genus level (no tourists—26.5%; with tourists—28.8%), with a large proportion of unidentified colonies (no tourists—49.8%; with tourists—50.1%).
Several airborne bacteria were identified solely in the air sampled before tourist visits, and some of them might represent the natural background without direct tourist impact: Arthrobacter arilaitensis, Kocuria polaris, Paenibacillus amyolyticus, P. polymixa, Pseudomonas antarctica, P. cedrina ssp. cedrina, P. jessenii, P. marginalis, Staphylococcus equorum, S. haemolyticus, S. pasteuri, S. warneri, and Streptomyces badius (Table 2). More diversified microbiota were sampled during tourist visits (Table 2 and Table 3). The presence of some airborne microbes might also be associated with the presence of bats, because the Škocjan Caves host several colonies, exceeding 5000 individuals, year-round [35]. In a study of Ardales Show Cave (Spain), with its Palaeolithic paintings, the majority of the culturable airborne bacteria were Gram-positive before (Streptomyces), and even after, a visit (Bacillus, Streptomyces). Bacillus spp. were frequently present, but with a lower abundance during the cave closure periods. Furthermore, some bacteria, e.g., Arthrobacter, Micrococcus, Pseudomonas, showed no relevant patterns in their distribution [8].

Table 2.
Identification of airborne bacterial isolates (MALDI-TOF MS log score value >2.000, species level identification) at Šotor and Orjak (*) sampling station before and during tourist visits, with the number of passing tourists (in brackets) in the Šumeča jama section during high Reka River discharge (∆).

Table 3.
Identification of airborne bacterial isolates (MALDI-TOF MS log score values of 1.700–1.999, genus level identification) at Šotor and Orjak (*) sampling stations before and during tourist visits with the number of passing tourists (in brackets) in the Šumeča jama section during the high Reka River discharge (∆).
Some of the identified airborne bacterial species are not directly related with the human microbiome (e.g., Acidovorax facilis, Kocuria polaris, Microbacterium hydrocarbonoxydans, Paenibacillus polymyxa, Pseudomonas antarctica, Psychrobacillus psychrotolerans, Rhodococcus erythropolis, Streptomyces badius) and are typical inhabitants of soil and aquatic environments. On the other hand, some belong to a typically human and animal biota, e.g., Acinetobacter haemolyticus, A. johnsonii, A. junii, A. parvus, Dietzia maris, Pseudochrobactrum asaccharolyticum, Staphylococcus capitis, S. epidermidis, and S. haemolyticus, S. warneri, Table 4).

Table 4.
Characteristics of identified bacterial species.
Based on various national criteria (e.g., Australia, Belgium, Switzerland, Germany, New Zealand, Singapore, and the United Kingdom, Table 4) for assessment of microbial risk groups available at the American Biological Safety Association, few isolates were attributed to Risk Group 2 (organisms that can cause disease in humans, but the disease is treatable, Table 4). Their entry point is usually by inhalation of aerosolized cells and spores or direct contact with contaminated surfaces. These identified microorganisms are associated with the normal human microbiome and can cause disease only in patients that have a pronounced susceptibility to opportunistic infections.
The diversity of airborne bacteria revealed by MALDI-TOF MS was not high; this is related directly to the limited availability in the database of mass spectra for environmental isolates, including those from caves [43] and the selected medium and cultivation conditions. A sample rarefaction analysis confirmed that only a small proportion of the airborne bacteria was revealed in the Tiha jama section during the 2014 sampling campaign (Figure 3).
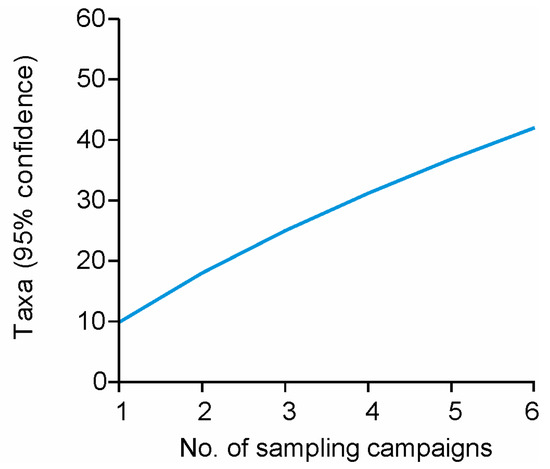
Figure 3.
Estimated bacterial species richness after six sampling campaigns at the Šotor and Orjak sampling stations before, and during, tourist visits in 2014.
3.3. Attribution of River Aerosolization to Airborne Biomass
The Škocjan Caves are also an excellent example for underground, naturally-induced, aerosol formation due to splashing and mist deriving from the underground river. Large open spaces along the Reka River canyon (Figure 1A,C) are reflected in large variations of temperature and carbon dioxide among the sampling sites within a vertical transect (Figure 4). In the Šumeča jama section (Figure 1), the important factors affecting the air temperature are the temperature of the Reka River, its discharge, and the distance from the cave entrance and vertical position within a cave transect [44]. In comparison to past organic pollution of the Reka River [45], the situation is, nowadays, improved although there are some recorded events with elevated indicators of microbial fecal pollution (unpublished data).
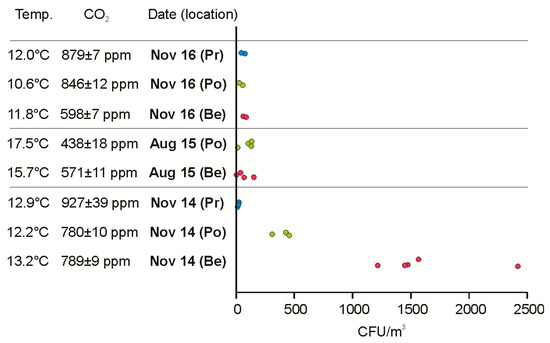
Figure 4.
Atmospheric conditions and concentrations of airborne bacteria at three sampling locations Belveder (Be), Ponvice (Po), and Prevoj (Pr) along the underground channel of the Reka River, with different discharges in November 2014 (~200 m3/s), August 2015 (~0.2 m3/s), and November 2016 (~20 m3/s).
The high discharge of the Reka River (~200 m3/s) was responsible for an elevated aerosolization rate of bacteria from the river and consequently high concentration of airborne bacteria close to the river (Belveder site with the highest concentration of 2418 CFU/m3, Figure 4). The impact of the river in a vertical transect above the Belveder site gradually diminished (the highest concentration at the Ponvice station was 455 CFU/m3 and that at the Prevoj station 23 CFU/m3, 12 November 2014, Figure 1 and Figure 4). In the air, six bacterial species (Table 2) and an additional six genera (Acinetobacter, Bacillus, Brevundimonas, Carnobacterium, Pseudomonas, Stenotrophomonas, Table 3) were identified. B. cereus was dominant at the Belveder and Ponvice sampling stations where P. extremorientalis was also particularly abundant.
During an approximately 10-times lower discharge episode in the autumn of 2016, the concentration of airborne bacteria at the Belveder site was lower (~20-times), but within a similar range to that on 27 August 2015, when the Reka River had low discharge and air temperatures were far higher (Figure 4). It is likely that not only the discharge influences the concentration of airborne bacteria, but also other atmospheric conditions, such as temperature. For some microbes replication in the air was already proven [46], and caves lacking UV and desiccation stress, and with high levels of subcellular minute particles and organic molecules, can be supportive of microbial replication.
Airborne microbes and other minute organic particles also represent a source of nutrients for cave biota. In some caves, such as the Škocjan Caves, there are three major sources of airborne bacteria, autochtonous to the underground, human- and river-derived. At a certain point these subpopulations interact with each other (e.g., Prevoj sampling station), which introduces an additional ecological aspect to the cave ecosystem, e.g., gene transfer.
4. Conclusions
Cave air acts as a sink for externally-introduced airborne biomass that is available to the cave ecosystem. In show caves tourists are a significant vector and source of airborne bacteria. Tourist numbers correlate directly with the increase of airborne biomass, but vast cave spaces, such as those in the Škocjan Caves, prevent long-lasting on-site concentration of bacterial biomass. These aerosolized bacteria float or persist on surfaces for relatively long periods and form a reservoir for the colonization/infection of susceptible hosts. The discharge of the underground river impacts directly upon the formation of aerosols, the distribution of which in a cave is governed by cave morphology and thermal convection.
Acknowledgments
The authors acknowledge the monitoring plan for Škocjan Caves (2012–2015), and projects “Natural resources of karst show caves: a balance among protection, exploitation, and promotion (no. J7-7100)” and “Karst research for sustainable use of Škocjan Caves as World Heritage (no. L7-8268)” supported by the Slovenian Research Agency. The authors also acknowledge Andrej Mihevc for his comments on an earlier version of the manuscript, Andrej Kozinc for fieldwork support, Franjo Drole for assistance with diagrams and cave cartography, and David Lowe for language editing assistance.
Author Contributions
J.M., A.O.-M., and S.Š. conceived and designed the experiments; J.M. and R.T. performed the experiments; J.M., A.O.-M., R.T., and T.M. analysed the data; J.M. and T.M. contributed reagents and materials; and J.M., A.O.-M., S.Š., R.T., and T.M. wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- Hibbing, M.; Fuqua, C.; Parsek, M.; Peterson, S. Bacterial competition: Surviving and thriving in the microbial jungle. Nat. Rev. Microbiol. 2010, 8, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.; Bhangar, S.; Pasut, W.; Arens, E.; Taylor, J.; Lindow, S.; Nazaroff, W.; Bruns, T. Chamber bioaerosol study: Outdoor air and human occupants as sources of indoor airborne microbes. PLoS ONE 2015, 10, e0128022. [Google Scholar] [CrossRef] [PubMed]
- Kellogg, C.; Griffin, D. Aerobiology and the global transport of desert dust. Trends Ecol. Evol. 2006, 21, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Mulec, J.; Vaupotič, J.; Walochnik, J. Prokaryotic and eukaryotic airborne microorganisms as tracers of microclimatic changes in the underground (Postojna Cave, Slovenia). Microb. Ecol. 2012, 64, 654–667. [Google Scholar] [CrossRef] [PubMed]
- Mulec, J. Human impact on underground cultural and natural heritage sites, biological parameters of monitoring and remediation actions for insensitive surfaces: Case of Slovenian show caves. J. Nat. Conserv. 2014, 22, 132–141. [Google Scholar] [CrossRef]
- Wang, W; Ma, X.; Ma, Y.; Mao, L.; Wu, F.; Ma, X.; An, L.; Feng, H. Seasonal dynamics of airborne fungi in different caves of the Mogao Grottoes, Dunhuang, China. Int. Biodeter. Biodegr. 2010, 64, 461–466. [Google Scholar] [CrossRef]
- Wang, W.; Ma, Y.; Ma, X.; Wu, F.; Ma, X.; An, L.; Feng, H. Seasonal variations of airborne bacteria in the Mogao Grottoes, Dunhuang, China. Int. Biodeter. Biodegr. 2010, 64, 309–315. [Google Scholar] [CrossRef]
- Fernandez-Cortes, A.; Cuezva, S.; Sanchez-Moral, S.; Canaveras, J.; Porca, E.; Jurado, V.; Martin-Sanchez, P.M.; Saiz-Jimenez, C. Detection of human-induced environmental disturbances in a show cave. Environ. Sci. Pollut. R. 2011, 18, 1037–1045. [Google Scholar] [CrossRef] [PubMed]
- Hospodsky, D.; Yamamoto, N.; Nazaroff, W.; Miller, D.; Gorthala, S.; Peccia, J. Characterizing airborne fungal and bacterial concentrations and emission rates in six occupied children’s classrooms. Indoor Air 2015, 25, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Setlhare, G.; Malebo, N.; Shale, K.; Lues, R. Identification of airborne microbiota in selected areas in a health-care setting in South Africa. BMC Microbiol. 2014, 14, 100. [Google Scholar] [CrossRef] [PubMed]
- Bowers, R.M.; Clements, N.; Emerson, J.B.; Wiedinmyer, C.; Hannigan, M.P.; Fierer, N. Seasonal variability in bacterial and fungal diversity of the near-surface atmosphere. Environ. Sci. Technol. 2013, 47, 12097–12106. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.; Schwebke, I.; Kampf, G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect. Dis. 2006, 6, 130. [Google Scholar] [CrossRef] [PubMed]
- Jurado, V.; Laiz, L.; Sanchez-Moral, S.; Saiz-Jimenez, C. Pathogenic microorganisms related to human visits in Altamira Cave, Spain. In The Conservation of Subterranean Cultural Heritage; Saiz-Jimenez, C., Ed.; CRC Press/Balkema: Boca Raton, FL, USA; London, UK; New York, NY, USA; Leiden, The Netherlands, 2014; pp. 229–239. [Google Scholar]
- Porca, E.; Jurado, V.; Martin-Sanchez, P.; Hermosin, B.; Bastian, F.; Alabouvette, C.; Saiz-Jimenez, C. Aerobiology: An ecological indicator for early detection and control of fungal outbreaks in caves. Ecol. Indic. 2011, 11, 1594–1598. [Google Scholar] [CrossRef]
- Sanchez-Moral, S.; Cuezva, S.; Garcia-Anton, E.; Fernandez-Cortes, A.; Elez, J.; Benavente, D.; Canaveras, J.C.; Jurado, V.; Rogerio Candelera, M.A.; Saiz-Jimenez, C. Microclimatic monitoring in Altamira Cave: Two decades of scientific projects for its conservation. In The Conservation of Subterranean Cultural Heritage; Saiz-Jimenez, C., Ed.; CRC Press/Balkema: Boca Raton, FL, USA; London, UK; New York, NY, USA; Leiden, The Netherlands, 2014; pp. 139–144. [Google Scholar]
- Geneste, J.M.; Mauriac, M. The Conservation of Lascaux Cave, France. In The Conservation of Subterranean Cultural Heritage; Saiz-Jimenez, C., Ed.; CRC Press/Balkema: Boca Raton, FL, USA; London, UK; New York, NY, USA; Leiden, The Netherlands, 2014; pp. 165–172. [Google Scholar]
- Gams, I.; Nagel, J. Kras: Zgodovinski, Naravoslovni in Geografski Oris (Kras/Karst: Historical, Natural and Geographical Description); Slovenska matica: Ljubljana, Yugoslavia, 1974; p. 358. [Google Scholar]
- Habič, P.; Knez, M.; Kogovšek, J.; Kranjc, A.; Mihevc, A.; Slabe, T.; Šebela, S.; Zupan, N. Škocjanske Jame speleological revue. Int. J. Speleol. 1989, 18, 1–42. [Google Scholar] [CrossRef]
- Mihevc, A. Škocjanske jame—Prispevek k speleogenezi (Škocjan Caves—A contribution to speleogenesis). Naše Jame 1999, 40, 11–27. [Google Scholar]
- Mihevc, A. Speleogeneza Divaškega Krasa; Založba ZRC: Ljubljana, Slovenia, 2001; p. 180. [Google Scholar]
- Gospodarič, R. O Geologiji in Speleogenezi Škocjanskih Jam. Geološki Zbornik 1983, 4, 163–172. [Google Scholar]
- Knez, M. The influence of bedding-planes on the development of Karst caves (a study of Velika Dolina at Škocjanske Jame caves, Slovenia). Carbonate Evaporite 1998, 13, 121–131. [Google Scholar] [CrossRef]
- Presetnik, P.; Koselj, K.; Zagmajster, M.; Zupančič, N.; Jazbec, K.; Žibrat, U.; Petrinjak, A.; Hudoklin, A. Atlas Netopirjev (Chiroptera) Slovenije (Atlas of Bats (Chiroptera) of Slovenia). Atlas Faunae et Florae Sloveniae 2; Center za kartografijo favne in flore: Miklavž na Dravskem polju, Slovenia, 2009; p. 152. [Google Scholar]
- Vode, Podatki Samodejnih Hidroloških Postaj. Available online: http://www.arso.gov.si/vode/podatki/amp/ (accessed on 24 November 2016).
- Feller, W. An Introduction to the Probability Theory and Its Application; John Wiley and Sons: New York, NY, USA, 1950; 175p. [Google Scholar]
- Singhal, N.; Kumar, M.; Kanaujia, P.K.; Viridi, J.S. MALDI-TOF mass spectrometry: An emerging technology for microbial identification and diagnosis. Front. Microbial. 2015, 6, 791. [Google Scholar] [CrossRef] [PubMed]
- Santos, I.C.; Hildenbrand, Z.L.; Schung, K.A. Applications of MALDI-TOF MS in environmental microbiology. Analyst 2016, 141, 2827–2837. [Google Scholar] [CrossRef] [PubMed]
- Cassagne, C.; Normand, A.C.; L’Ollivier, C.; Ranque, S.; Piarroux, R. Performance of MALDI-TOF MS platforms for fungal identification. Mycoses 2016, 59, 678–690. [Google Scholar] [CrossRef] [PubMed]
- Risk Group Database. Available online: https://my.absa.org/Riskgroups (accessed on 11 May 2017).
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. Past: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Grgić, I.; Iskra, I.; Podkrajšek, B.; Debevec Gerjevič, V. Measurements of aerosol particles in the Škocjan Caves, Slovenia. Environ. Sci. Pollut. R. 2014, 21, 1915–1923. [Google Scholar] [CrossRef] [PubMed]
- Šebela, S.; Prelovšek, M.; Turk, J. Impact of peak period visits on the Postojna Cave (Slovenia) microclimate. Theor. Appl. Climatol. 2013, 111, 51–64. [Google Scholar] [CrossRef]
- Robertson, L.D. Monitoring viable fungal and bacterial bioaerosol concentrations to identify acceptable levels for common indoor environments. Indoor Built Environ. 1997, 6, 295–300. [Google Scholar] [CrossRef]
- Adams, R.; Miletto, M.; Taylor, J.; Bruns, T. Dispersal in microbes: Fungi in indoor air are dominated by outdoor air and show dispersal limitation at short distances. ISME J. 2013, 7, 1262–1273. [Google Scholar] [CrossRef] [PubMed]
- Mulec, J.; Dietersdorfer, E.; Ustunturk-Onan, M.; Walochnik, J. Acanthamoeba and other free-living amoebae in bat guano, an extreme habitat. Parasitol. Res. 2016, 115, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Garrity, G. (Ed.) Bergey’s Manual® of Systematic Bacteriology: Volume 2: The Proteobacteria, Part A Introductory Essays; Springer Science & Business Media: New Delhi, India, 2005; p. 304. [Google Scholar]
- Goodfellow, M.; Kämpfer, P.; Busse, H.J.; Trujillo, M.E.; Suzuki, K.I.; Ludwig, W.; Whitman, W.B. (Eds.) Bergey’s Manual® of Systematic Bacteriology: Volume 3: The Actinobacteria, Part A and B; Springer: New York, NY, USA, 2012; p. 2083. [Google Scholar]
- Reddy, G.S.; Prakash, J.S.; Prabahar, V.; Matsumoto, G.I.; Stackebrandt, E.; Shivaji, S. Kocuria polaris sp. nov., an orange-pigmented psychrophilic bacterium isolated from an Antarctic cyanobacterial mat sample. Int. J. Syst. Evol. Microbiol. 2003, 53, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Sedláček, I. Taxonomie Prokaryot; Masarykova Univerzita: Brno, Czech Republic, 2007; p. 270. [Google Scholar]
- Stackebrandt, E.; Koch, C.; Gvozdiak, O.; Schumann, P. Taxonomic dissection of the genus micrococcus: Kocuria gen. nov., Nesterenkonia gen. nov., Kytococcus gen. nov., Dermacoccus gen. nov., and Micrococcus Cohn 1872 gen. emend. Int. J. Syst. Evol. Microbiol. 1995, 45, 682–692. [Google Scholar] [CrossRef] [PubMed]
- Tvrzová, L.; Schumann, P.; Sedláček, I.; Páčová, Z.; Spröer, C.; Verbarg, S.; Kroppenstedt, R.M. Reclassification of strain CCM 132, previously classified as Kocuria varians, as Kocuria carniphila sp. nov. Int. J. Syst. Evol. Microbiol. 2005, 55, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Vos, P.; Garrity, G.; Jones, D.; Krieg, N.R.; Ludwig, W.; Rainey, F.A.; Schleifer, K.H.; Whitman, W. (Eds.) Bergey’s Manual of Systematic Bacteriology: Volume 3: The Frmicutes; Springer Science & Business Media: New York, NY, USA, 2009; p. 1450. [Google Scholar]
- Mulec, J.; Oarga-Mulec, A.; Tomazin, R.; Matos, T. Characterization and fluorescence of yellow biofilms in karst caves, southwest Slovenia. Int. J. Speleol. 2015, 44, 107–114. [Google Scholar] [CrossRef]
- Kranjc, A.; Opara, B. Temperature monitoring in Škocjanske jame caves. Acta Carsol. 2002, 31, 85–96. [Google Scholar]
- Kogovšek, J.; Kranjc, A. Pollution by surface water: The Reka River. In Karst Hydrogeology and Human Activities: Impacts, Consequences and Implications, International Contributions to Hydrogeology 20; Drew, D.P., Hötzl, H., Eds.; Balkema: Rotterdam, The Netherlands; Brookfield, VT, USA, 1999; pp. 148–153. [Google Scholar]
- Di Giorgio, C.; Krempff, A.; Guiraud, H.; Binder, P.; Tiret, C.; Dumenil, G. Atmospheric pollution by airborne microorganisms in the city of Marseilles. Atmos. Environ. 1996, 30, 155–160. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).