Variation in Incidence and Severity of Injuries among Crown-of-Thorns Starfish (Acanthaster cf. solaris) on Australia’s Great Barrier Reef
Abstract
:1. Introduction
2. Materials and Methods
Data Analyses
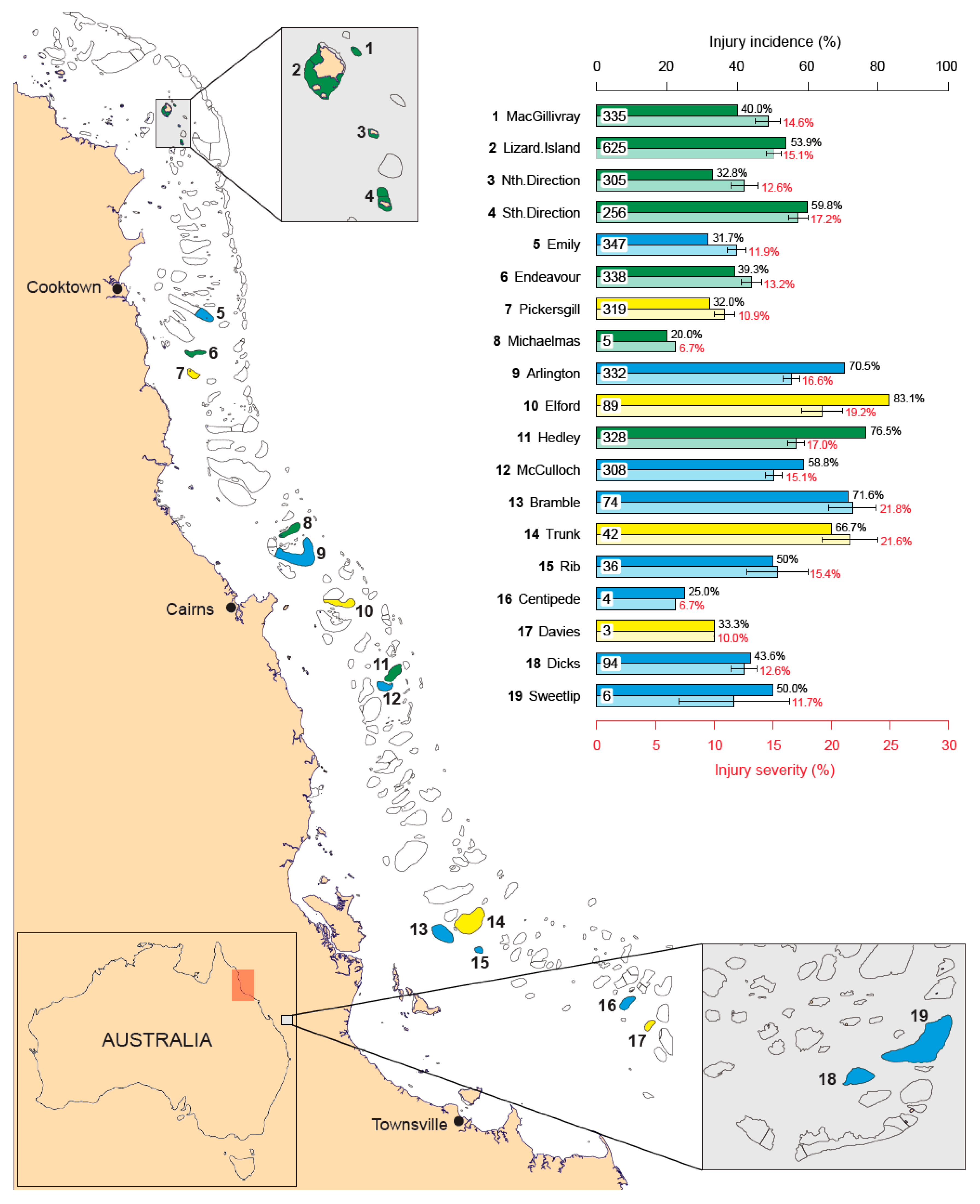
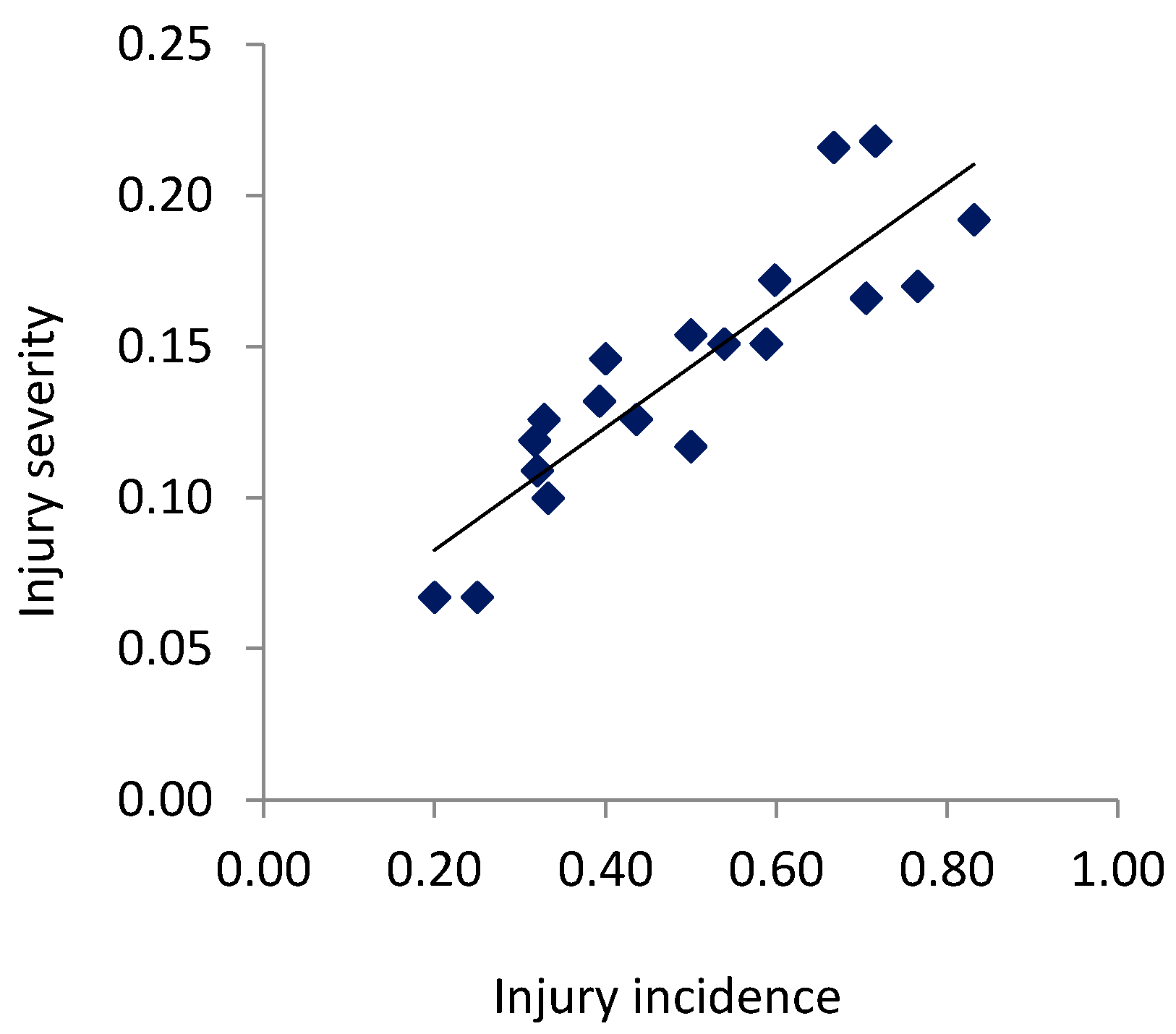
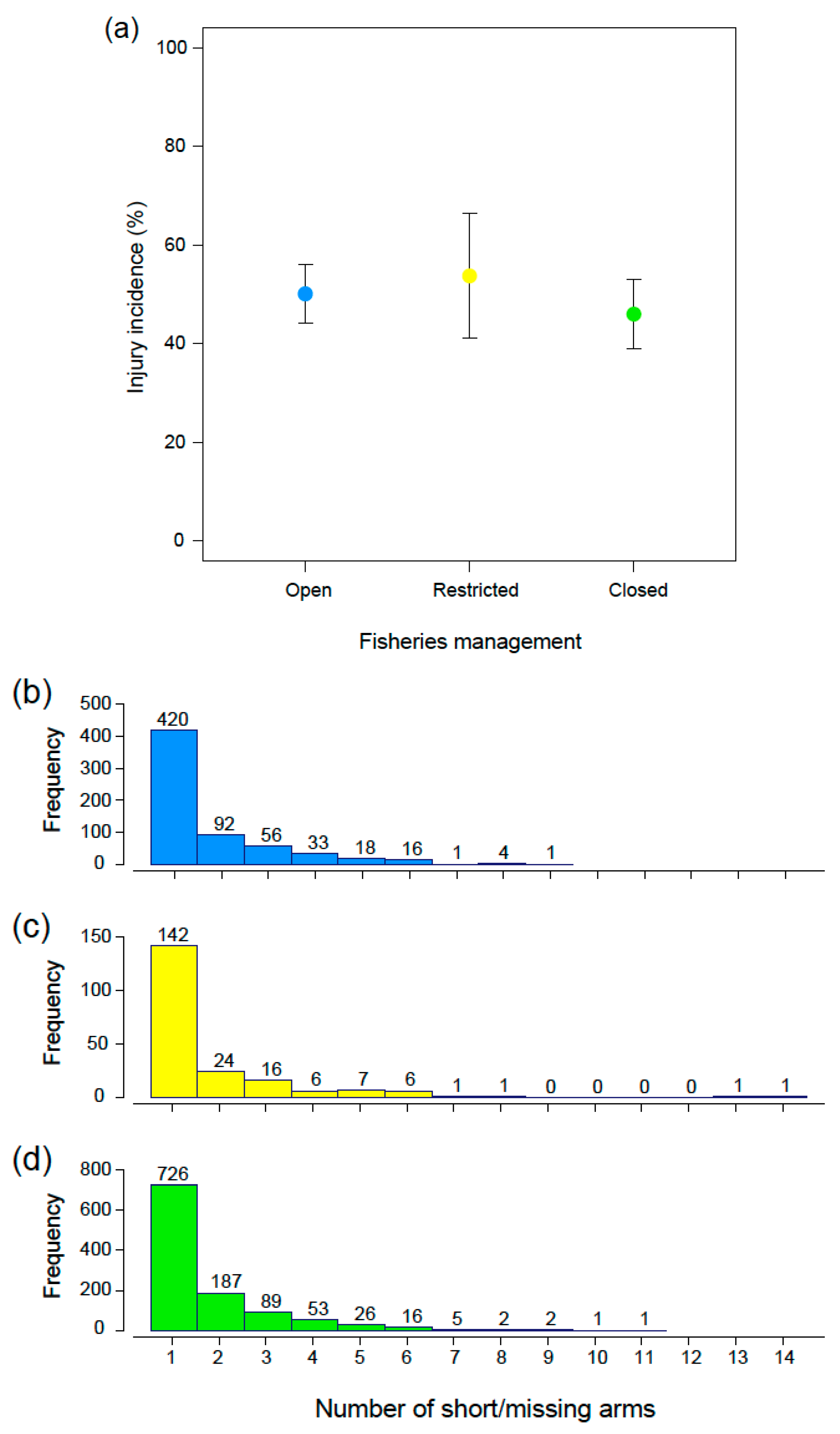
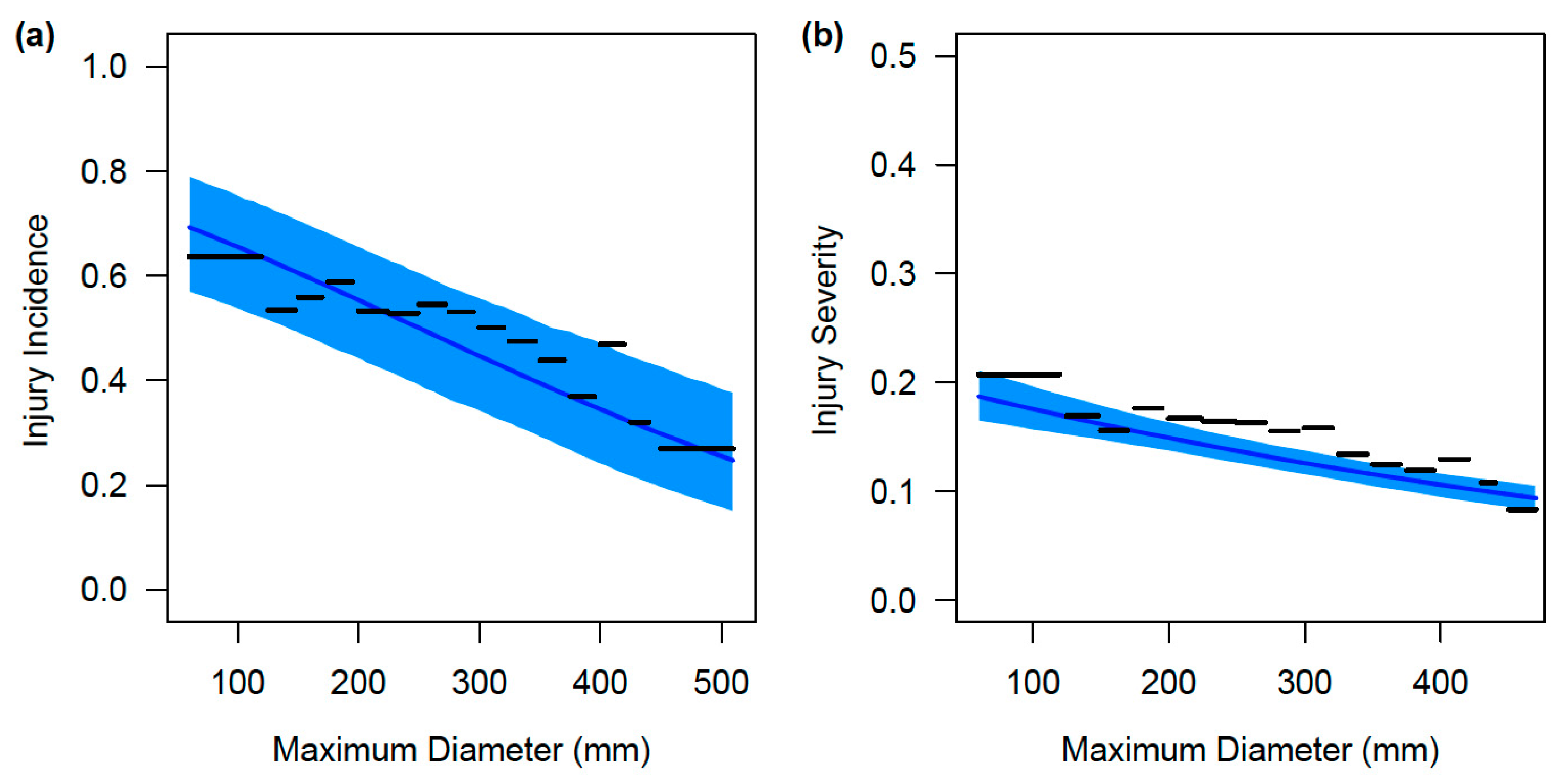
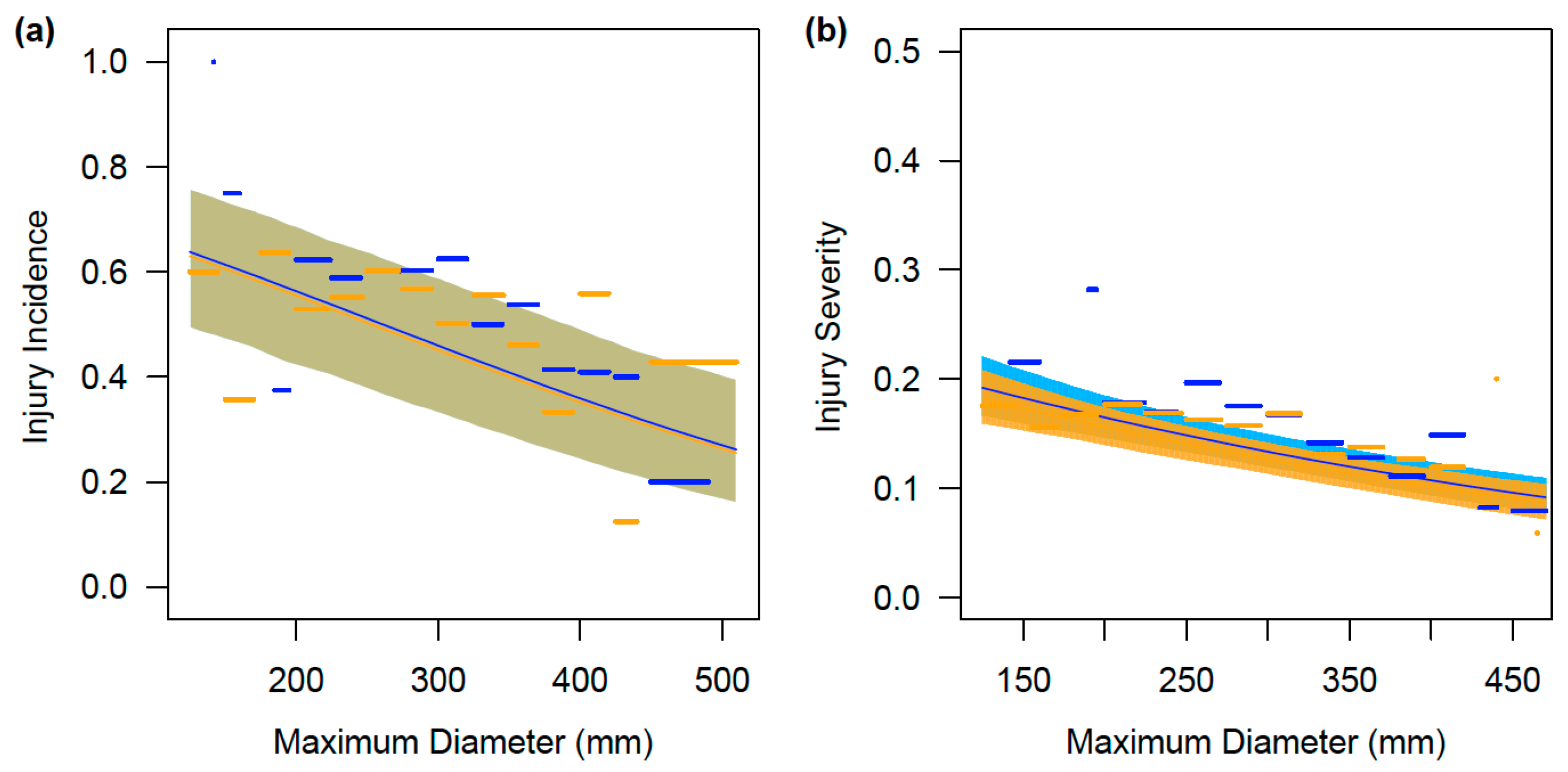
3. Results
3.1. Incidence of Injuries
3.2. Size-Based Variation
3.3. Gender-Based Differences in Incidence and Severity of Injuries
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Murdoch, W.W.; Oaten, A. Predation and population stability. Adv. Ecol. Res. 1975, 9, 1–131. [Google Scholar]
- Carpenter, J.R. Insect outbreaks in Europe. J. Anim. Ecol. 1940, 9, 108–147. [Google Scholar] [CrossRef]
- Holling, C.S. The functional response of predators to prey density and its role in mimicry and population regulation. Mem. Entomol. Soc. Can. 1965, 45, 5–60. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. Paradox of enrichment: Destabilization of exploitation ecosystems in ecological time. Science 1971, 171, 385–387. [Google Scholar] [CrossRef] [PubMed]
- Stiling, P. Density-dependent processes and key factors in insect populations. J. Anim. Ecol. 1988, 57, 581–594. [Google Scholar] [CrossRef]
- Clark, L.R. Predation by birds in relation to the population density of Cardiaspina albitextura (Psyllidae). Aust. J. Zool. 1964, 12, 349–361. [Google Scholar] [CrossRef]
- Uthicke, S.; Schaffelke, B.; Byrne, M. A boom-bust phylum? Ecological and evolutionary consequences of density variations in echinoderms. Ecol. Monogr. 2009, 79, 3–24. [Google Scholar]
- Singleton, G.R. Population dynamics of an outbreak of house mice (Mus domesticus) in the Mallee Wheatlands of Australia—Hypothesis of plague formation. J. Zool. 1989, 219, 495–515. [Google Scholar] [CrossRef]
- Pratchett, M.S.; Caballes, C.F.; Rivera-Posada, J.A.; Sweatman, H.P.A. Limits to understanding and managing outbreaks of crown-of-thorns stafish (Acanthaster spp.). Oceanogr. Mar. Biol. Annu. Rev. 2014, 52, 133–199. [Google Scholar]
- Kayal, M.; Vercelloni, J.; de Loma, T.L.; Bosserelle, P.; Chancerelle, Y.; Geoffroy, S.; Stievenart, C.; Michonneau, F.; Penin, L.; Planes, S.; et al. Predator Crown-of-Thorns Starfish (Acanthaster planci) Outbreak, Mass Mortality of Corals, and Cascading Effects on Reef Fish and Benthic Communities. PLoS ONE 2012, 7, e47363. [Google Scholar] [CrossRef] [PubMed]
- Endean, R. Report on Investigations Made into Aspects of the Current Acanthaster planci (Crown-of-Thorns) Infestations of Certain Reefs of the Great Barrier Reef; Queensland Department of Primary Industries: Brisbane, Australia, 1969. [Google Scholar]
- Cowan, Z.L.; Pratchett, M.S.; Messmer, V.; Ling, S. Known predators of crown-of-thorns starfish (Acanthaster spp.) and their role in mitigating, if not preventing, population outbreaks. Diversity 2017, 9, 7. [Google Scholar]
- Ormond, R.F.G.; Bradbury, R.H.; Brainbridge, S.; Fabricius, K.; Kessing, J.K.; de Vantier, L.M.; Medlay, P.; Steven, A.D.L. Test of a model for regulation of crown-of-thorns starfish by fish predators. In Acanthaster and the Coral Reef: A Theoretical Perspective; Bradbury, R.H., Ed.; Springer: Heidelberg, Germany, 1990; pp. 189–207. [Google Scholar]
- Sweatman, H. No-take reserves protect coral reefs from predatory starfish. Curr. Biol. 2008, 18, R598–R599. [Google Scholar] [CrossRef] [PubMed]
- Dulvy, N.K.; Freckleton, R.P.; Polunin, N.V.C. Coral reef cascades and the indirect effects of predator removal by exploitation. Ecol. Lett. 2004, 7, 410–416. [Google Scholar] [CrossRef]
- Rivera-Posada, J.; Caballes, C.F.; Pratchett, M.S. Size-related variation in arm damage frequency in the crown-of-thorns sea star, Acanthaster planci. J. Coast. Life Med. 2014, 2, 187–195. [Google Scholar]
- McCallum, H.I.; Endean, R.; Cameron, A.M. Sublethal damage to Acanthaster planci as an index of predation pressure. Mar. Ecol. Prog. Ser. 1989, 56, 29–36. [Google Scholar] [CrossRef]
- Bos, A.R.; Gumanao, G.S.; Salac, F.N. A newly discovered predator of the crown-of-thorns starfish. Coral Reefs 2008, 27, 581. [Google Scholar] [CrossRef]
- Messmer, V.; Pratchett, M.S.; Clark, T.D. Capacity for regeneration in crown of thorns starfish, Acanthaster planci. Coral Reefs 2013, 32, 461. [Google Scholar] [CrossRef]
- Simpson, C.J.; Grey, K.A. Crown-of-Thorns Starfish (Acanthaster planci) in the Dampier Archipelago, Western Australia; Technical Series; Perth Environmental Protection Authority: Perth, Australia, 1988. [Google Scholar]
- Lawrence, J.M.; Vasquez, J. The effect of sublethal predation on the biology of echinoderms. Oceanol. Acta 1996, 19, 431. [Google Scholar]
- Lawrence, J.M. Arm loss and regeneration in Asteroidea (Echinodermata). In Echinoderm Research; Scalera-Liaci, L., Canicatti, C., Eds.; Balkema: Rotterdam, The Netherlands, 1991; pp. 39–52. [Google Scholar]
- Harris, R.N. Nonlethal injury to organisms as a mechanism of population regulation. Am. Nat. 1989, 134, 835–847. [Google Scholar] [CrossRef]
- Bolker, B.M.; Brooks, M.E.; Clark, C.J.; Geange, S.W.; Poulson, J.R.; Stevens, M.H.H.; White, J.-S.S. Generalized linear mixed models: A practical guide for ecology and evolution. Trends Ecol. Evol. 2009, 24, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Zuur, A.F.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer: New York, NY, USA, 2009. [Google Scholar]
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using LME4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Pisapia, C.; Burn, D.; Yoosuf, R.; Najeeb, A.; Anderson, K.D.; Pratchett, M.S. Coral recovery in the central Maldives archipelago since the last major mass-bleaching, in 1998. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- McCook, L.J.; Ayling, T.; Cappo, M.; Choat, J.H.; Evans, R.D.; De Freitas, D.M.; Heupel, M.; Hughes, T.P.; Jones, G.P.; Mapstone, B.; et al. Adaptive management of the Great Barrier Reef: A globally significant demonstration of the benefits of networks of marine reserves. Proc. Natl. Acad. Sci. USA 2010, 107, 18278–18285. [Google Scholar] [CrossRef] [PubMed]
- Messmer, V.; Pratchett, M.S.; Hoey, A.S.; Tobin, A.J.; Coker, D.J.; Cooke, S.J.; Clark, T.D. Global warming may disproportionately affect larger adults in a predatory coral reef fish. Glob. Chang. Biol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Zajac, R.N. Sublethal predation on Polydora cornuta (Polychaeta, Spionidae)—Patterns of tissue loss in a field population, predator functional response and potential demographic impacts. Mar. Biol. 1995, 123, 531–541. [Google Scholar] [CrossRef]
- Barrios, J.V.; Gaymer, C.F.; Vasquez, J.A.; Brokordt, K.B. Effect of the degree of autotomy on feeding, growth, and reproductive capacity in the multi-armed sea star Heliaster helianthus. J. Exp. Mar. Biol. Ecol. 2008, 361, 21–27. [Google Scholar] [CrossRef]
- Maginnis, T.L. The costs of autotomy and regeneration in animals: A review and framework for future research. Behav. Ecol. 2006, 17, 857–872. [Google Scholar] [CrossRef]
- Bernardo, J.; Agosta, S.J. Evolutionary implications of hierarchical impacts of nonlethal injury on reproduction, including maternal effects. Biol. J. Linn. Soc. 2005, 86, 309–331. [Google Scholar] [CrossRef]
- Caballes, C.F.; Pratchett, M.S.; Kerr, A.M.; Rivera-Posada, J.A. The role of maternal nutrition on oocyte size and quality, with respect to early larval development in the coral-eating starfish, Acanthaster planci. PLoS ONE 2016, 11, e0158007. [Google Scholar] [CrossRef]
- Pearson, R.G.; Endean, R. A preliminary study of the coral predator Acanthaster planci (L.) (Asteroidea) on the Great Barrier Reef. Fish Notes 1969, 3, 27–68. [Google Scholar]
- Stump, R.J.W. An Investigation to Describe the Population Dynamics of Acanthaster planci (L.) around Lizard Island, Cairns Section, Great Barrier Reef Marine Park; CRC Reef Research Centre: Townsville, Australia, 1996. [Google Scholar]
- Scharf, F.S.; Juanes, F.; Rountree, R.A. Predator size-prey size relationships of marine fish predators: Interspecific variation and effects of ontogeny and body size on trophic-niche breadth. Mar. Ecol. Prog. Ser. 2000, 208, 229–248. [Google Scholar] [CrossRef]
- Berglund, A.; Rosenqvist, G. Reproductive costs in the prawn Palaemon adspersus: Effects on growth and predator vulnerability. Oikos 1986, 46, 349–354. [Google Scholar] [CrossRef]
- Nordström, M.C.; Aarnio, K.; Törnroos, A.; Bonsdorff, E. Nestedness of trophic links and biological traits in a marine food web. Ecosphere 2015, 6, 161. [Google Scholar] [CrossRef]
- Fuiman, L.A. The interplay of ontogeny and scaling in the interactions of fish larvae adn their predators. J. Fish Biol. 1994, 45 (Suppl. SA), 55–79. [Google Scholar] [CrossRef]
- Lundvall, D.; Svanbäck, R.; Persson, L.; Byström, P. Size-dependent predation in piscivores: Interactions between predator foraging adn prey avoidance abilities. Can. J. Fish. Aquat. Sci. 1999, 56, 1285–1292. [Google Scholar] [CrossRef]
- McClanahan, T.R.; Muthinga, N.A. Patterns of predation on a sea urchin, Echinometra mathei (de Blainville), on Kenyan coral reefs. J. Exp. Mar. Biol. Ecol. 1989, 126, 77–94. [Google Scholar] [CrossRef]
- Ling, S.D.; Johnson, C.R.; Frusher, S.D.; Ridgway, K.R. Overfishing reduces resilience of kelp beds to climate-driven catastrophic phase shift. Proc. Natl. Acad. Sci. USA 2009, 106, 22341–22345. [Google Scholar] [CrossRef] [PubMed]
- Wilmes, J.; Matthews, S.; Schultz, D.; Messmer, V.; Hoey, A.S.; Pratchett, M.S. Modelling growth of juvenile crown-of-thorns starfish (0+ year cohort) on the northern Great Barrier Reef. Diversity 2017, 9, 1. [Google Scholar] [CrossRef]
- Svensson, I. Reproductive costs in two sex-role reversed pipefish species (Sygnathidae). J. Anim. Ecol. 1988, 57, 929–942. [Google Scholar] [CrossRef]

| Data | Candidate Model Effects | AIC | Likelihood Test * |
|---|---|---|---|
| Incidence (n = 3846) | Zone * Size + Observer + (1|Reef) | 4937.8 | Chi = 1.0, df = 2, p = 0.617 |
| Zone * Size + Observer + (Size|Reef) | 4935.4 | ||
| Zone + Size + Observer + (Size|Reef) | 4933.2 | ||
| Size + Observer + (Size|Reef) | 4930.1 | ||
| Severity ** (n = 1797) | Zone * Size + Observer | 5847.6 | Chi = 3.1, df = 4, p = 0.541 |
| Zone + Size + Observer | 5846.0 | ||
| Size | 5841.1 | ||
| Gender (Incidence) ** (n = 2553) | Zone + Size * Sex + Observer | 3287.6 | Chi = 3.2, df = 2, p = 0.204 |
| Zone + Size + Sex + Observer | 3285.8 | ||
| Size + Sex | 3285.0 | ||
| Gender (Severity) ** (n = 1263) | Zone + Size * Sex + Observer | 4210.1 | Chi = 1.2, df = 4, p = 0.814 |
| Zone + Size + Sex + Observer | 4204.1 | ||
| Size + Sex | 4177.2 |
| Data | Variable | Estimate ± SE | z-Value | p-Value |
|---|---|---|---|---|
| Incidence | (Intercept) | 1.0708 ± 0.3239 | 3.3055 | 0.0009 |
| Size | −0.0043 ± 0.0009 | −4.8358 | 0.0000 | |
| Observer2 | 1.1351 ± 0.3793 | 2.9929 | 0.0028 | |
| Observer3 | −0.0872 ± 0.3248 | −0.2685 | 0.7883 | |
| Severity | (Intercept) | −1.3512 ± 0.0925 | −14.6038 | 0.0000 |
| Size | −0.0020 ± 0.0003 | −7.0685 | 0.0000 | |
| Gender (incidence) | (Intercept) | 1.0882 ± 0.3639 | 2.9907 | 0.0028 |
| Size | −0.0042 ± 0.0008 | −5.4448 | 0.0000 | |
| Male | −0.0343 ± 0.0874 | −0.3922 | 0.6949 | |
| Observer2 | 0.9585 ± 0.3945 | 2.4300 | 0.0151 | |
| Observer3 | 0.0255 ± 0.3693 | 0.0690 | 0.9450 | |
| Gender (severity) | (Intercept) | −1.1265 ± 0.1258 | −8.9549 | 0.0000 |
| Size | −0.0025 ± 0.0004 | −6.5331 | 0.0000 | |
| Male | −0.0623 ± 0.0394 | −1.5818 | 0.1137 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Messmer, V.; Pratchett, M.; Chong-Seng, K. Variation in Incidence and Severity of Injuries among Crown-of-Thorns Starfish (Acanthaster cf. solaris) on Australia’s Great Barrier Reef. Diversity 2017, 9, 12. https://doi.org/10.3390/d9010012
Messmer V, Pratchett M, Chong-Seng K. Variation in Incidence and Severity of Injuries among Crown-of-Thorns Starfish (Acanthaster cf. solaris) on Australia’s Great Barrier Reef. Diversity. 2017; 9(1):12. https://doi.org/10.3390/d9010012
Chicago/Turabian StyleMessmer, Vanessa, Morgan Pratchett, and Karen Chong-Seng. 2017. "Variation in Incidence and Severity of Injuries among Crown-of-Thorns Starfish (Acanthaster cf. solaris) on Australia’s Great Barrier Reef" Diversity 9, no. 1: 12. https://doi.org/10.3390/d9010012
APA StyleMessmer, V., Pratchett, M., & Chong-Seng, K. (2017). Variation in Incidence and Severity of Injuries among Crown-of-Thorns Starfish (Acanthaster cf. solaris) on Australia’s Great Barrier Reef. Diversity, 9(1), 12. https://doi.org/10.3390/d9010012






