No Evidence for Temporal Variation in a Cryptic Species Community of Freshwater Amphipods of the Hyalella azteca Species Complex
Abstract
: The co-occurrence of cryptic species of Hyalella amphipods is a challenge to our traditional views of how species assemble. Since these species have similar morphologies, it is not evident that they have developed phenotypic differences that would allow them to occupy different ecological niches. We examined the structure of a community of Hyalella amphipods in the littoral zone of a boreal lake to verify if temporal variation was present in relative abundances. Morphological and molecular analyses using the mitochondrial cytochrome c oxidase I (COI) gene enabled us to detect three cryptic species at the study site. No temporal variation was observed in the community, as one cryptic species was always more abundant than the two others. The relative abundances of each species in the community appeared constant at least for the open-water season, both for adult and juvenile amphipods. Niche differences are still to be found among these species, but it is suggested that migration from nearby sites may be an important factor explaining the species co-occurrence.1. Introduction
Cryptic species are two or more species that share similar morphologies but have high genetic differences between them suggesting their distinct species status [1-3]. The fact that many cryptic species live in sympatry challenges our traditional view of how species assemble [4-6]. To coexist at the local scale, species must be ecologically distinct in a manner that makes intraspecific competition more important for the population of a species than interspecific competition [7]. If this is the case, a species should be able to increase when rare in the community (referred to as the invasibility criterion, which is a fundamental prerequisite for stable species coexistence [7,8]). To be ecologically distinct, species develop trade-offs that enable them to occupy different ecological niches [9]; these trade-offs are often linked with morphological differences between competing species [10,11]. It is not evident that cryptic species living in sympatry exhibit such differences. Fine morphological differences, however, may allow species to occupy different niches. Moreover, differences in other phenotypes (i.e. physiology and behavior) or life history traits may allow species to exploit different niches and foster stable coexistence. [6,12]. Alternatively, cryptic species living in sympatry may only be co-occurring rather than coexisting, e.g., their persistence together is not necessarily indefinite [5,13]. In that case, co-occurring cryptic species could possibly be ecologically similar; however, without migration from other sites, all species but one are expected to be eventually driven to extinction via neutral dynamics [7,13]. Co-occurring cryptic species could also be maintained in a local area by migration of individuals from other areas via source-sink dynamics [5,7,13]. In that case, species could persist at a local site despite poor performance because of the migration of new recruits from the source.
Amphipods of the Hyalella azteca (Saussure) species complex are freshwater benthic crustaceans considered to be omnivorous detritivores living in macrophytes communities, on sediments and on organic debris [14-16]. Traditionally, H. azteca amphipods were considered on a morphological basis as one species found all over North America, Central America and northern South America [17,18]. However, recent genetic studies using different molecular markers [12,19-22] and interbreeding trials [23] revealed that they were in fact a cryptic species complex composed of at least 33 provisional species [21]; this number is probably an underestimate as it was found by a study limited to the southern Great Basin region of California and Nevada [21]. Cryptic species of Hyalella amphipods often live in sympatry at the same site in a water body [6,21]. A recent study by Wellborn and Cothran [6] suggests that Hyalella cryptic species can, in some cases, exhibit niche partitioning despite being similar in morphology and life history [6,12]. For example, different Hyalella cryptic species can have different predation risks and different spatial distributions both across a distance-from-shore gradient and a vertical depth gradient [6]. It was suggested that Hyalella cryptic species could coexist in the littoral zone of a lake via a trade-off between predator avoidance and resource exploitation [6]. However, predation risk (by fish and by macroinvertebrates) [24,25] and the use and the availability of resources (food and substrates like macrophytes and leaves) [26-29] also vary with season in the littoral zone of lakes [30]. Seasonal changes in the environmental conditions can also affect the spatial distribution of amphipods in lakes. For instance, it has been suggested that Hyalella amphipods can migrate to deeper, colder water in summer to optimize their metabolism when temperature rises in the littoral zone [31,32]. The impact of such temporal variation has not been investigated in Hyalella cryptic species communities. If the dominant species in a habitat changes with season, it could suggest that Hyalella amphipods coexist at a site via a temporal trade-off. For non-cryptic species, seasonal succession in growth patterns and life cycles keyed to the predictable input of leaves in autumn are observed for insect shredders in streams [33] and have been recently suggested for their lakes counterparts [29]. For amphipods, temporal succession has at least been reported for non-cryptic species of the genus Gammarus that live in Danish brackish waters [34]. Other cryptic species complex such as Capitella polychaetes [35] and monogont rotifers [36,37] can exhibit seasonal successions which are explained to a large extent by differential adaptation to different environmental conditions. However, in all these cases, there are periods when the distributions of at least two species overlap spatially, especially for the most morphologically similar species.
This study aims at assessing whether relative abundances of Hyalella cryptic species in a community are constant or variable through time in a boreal lake. Specific objectives were to determine the number of Hyalella cryptic species present in Lac des Baies, a large Canadian boreal lake, to verify the occurrence throughout the open-water (ice-free) season of morphological differences between the species that could ease their coexistence via niche diversification and to evaluate the temporal variation throughout the open-water season of the relative abundance of each cryptic species in the community.
2. Material and Methods
2.1. Determination of the Number of Species in Lac des Baies
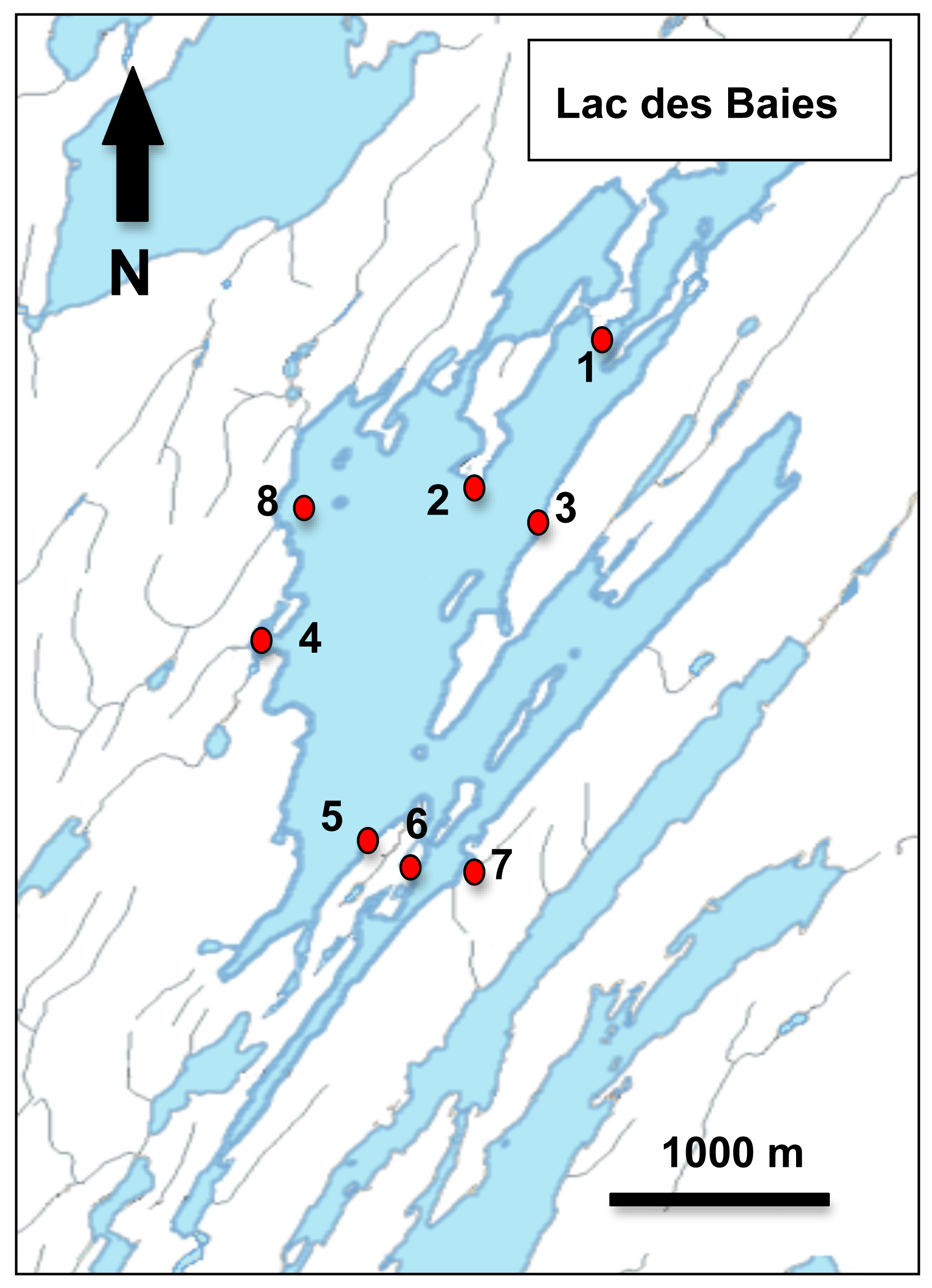
Lac des Baies is an oligotrophic lake of 569.8 hectares located in Eastern Québec, Canada (Figure 1). It is a large lake surrounded by boreal forest with both deciduous and evergreen trees. The lake has many bays more or less isolated from each other.
To estimate the number of Hyalella cryptic species and their distribution within Lac des Baies, amphipods were sampled from nine sites in the lake in August 2007 (site 1 to 5) and July 2008 (site 6, 7a, 7b and 8) using a dip net (500 μm mesh size) for littoral sites or an Ekman dredge for profundal sites (Table 1). For sampling with the dip net, sediments were kicked to suspend the benthos into the water and the dip net was swept into the suspended sediments to collect the amphipods. Each sample was preserved in 95% ethanol. These amphipods were subjected to morphological and molecular analyses.
2.2. Morphological Variation Among Species
Each amphipod was first identified under a dissecting microscope as H. azteca using the morphological traits described in Bousfield [17,38,39]. Gammarus lacustris is also present in Lac des Baies [40]; H. azteca amphipods are easily distinguished from other amphipods by their small adult body size, their second pair of antennae 1.2 × longer than the first pair, the presence of spines on pleon segments 1 and 2 (except for the inermis form) and the distinct shapes of the second gnathopod between males and females. After identification, body length was measured following the curve of the body from the first segment of the pereon to the tip of the urosome using a Leica MZ16 A image analyzer (Bannockburn, Illinois, USA) with the Northern Eclipse 6.0 software from Empix Imaging Inc (Mississauga, Ontario, Canada). Body length is an important trait that varies among species of the H. azteca complex [12,23]. The total number of segments on the two pairs of antennae was counted as an index of age (adults > 21 segments) [14,41]. The size and the shape of the second gnathopod were examined to determine the sex of the individuals; females have small and parachelate second gnathopods while adult males usually have large subchelate second gnathopods [17]. Some males had small second gnathopods only slightly larger than female gnathopods; this difference was noted to verify if it was linked to different species. This gnathopod is subject to sexual selection in Hyalella amphipods [42-44].
2.3. Molecular Analyses
Total DNA was extracted using a QuickExtract DNA Extraction Kit from Epicentre Biotechnologies (Madison, Wisconsin, USA). Depending on the size of the individual, a quarter to a half of an amphipod was used for the extraction. A 710-bp fragment of the mitochondrial cytochrome c oxidase I (COI) gene was amplified using the LCO1490 and HCO2198 universal primers from Folmer et al. [45]. The HCO2198 primer was labeled with HEX fluorescent (AlphaDNA, Montréal, Québec, Canada). The polymerase chain reaction (PCR) contained 1 μL of DNA template, 2.5 μL of 10× PCR buffer, 0.25 μM of each primer, 25 mM MgCl2, 0.25 μM of each dNTP and 1.5 units of Taq DNA polymerase in a final volume of 25 μL. PCR conditions were 3 min at 94 °C followed by 5 cycles of 60 s at 94 °C, 90 s at 45 °C and 60 s at 72 °C; followed by 35 cycles of 60 s at 94 °C, 90 s at 51 °C and 60 s at 72 °C; followed by 5 min at 72 °C (Biometra Uno-II, Goettingen, Germany).
To discriminate haplotypes (variants of COI), PCR products were run on denaturing gradient gel electrophoresis (DGGE) using a CBS DGGE-4001-Rev.B apparatus (Del Mar, California, USA). Briefly, 30% polyacrylamide (acrylamide: bisacrylamide 37.5 : 1), 1 mm thick gels with a linear denaturant gradient of 10 to 100% (where 100% denaturant = 40 % formamide and 7 M urea), were run for 15 h at 120 V in a 60 °C bath of 1× TAE buffer (40 mM Tris, 40 mM acetic acid, 1 mM EDTA, pH 7.4). Fluorescent dye-labeled alleles were detected on a FMBIO III fluorescent scanner (Hitachi Software Engineering America Ltd., San Bruno, California, USA) and analyzed using the ImageAnalysis v.3.0.0.21 software (Miraibio, San Francisco, California, USA). Each haplotype found with DGGE was also sequenced using the LCO1490 primer (McGill University and Genome Quebec Innovation Centre, Montréal, Québec, Canada).
COI sequences were aligned using the CLUSTALW Multiple alignment module in BioEdit version 7.0.9.0 [46]. Pairwise sequence divergences among haplotypes were calculated using Kimura's 2-parameter model in MEGA version 4 [47]. To discriminate cryptic species, we used a species screening threshold (SST) of 3.75% as calculated by Witt et al. [21] for H. azteca populations of the southern Great Basin region of California and Nevada. Note that SSTs and DNA barcoding using COI do not constitute a species concept and must be considered as tools to discriminate biological units corresponding to provisional species that need further investigation [21]. COI sequences were also compared with other sequences of the Hyalella cryptic species complex found on the GenBank database ( http://www.ncbi.nlm.nih.gov/).
2.4. Relative Abundances of Species Over Time
To compare the relative abundances of each species over time, amphipods were sampled each month from May to November 2007 at site 1 (Table 1). This site was chosen because we had previously determined that it had the greatest number of amphipods in a study on different populations of Hyalella amphipods in Lac des Baies [40]. This site seems to be representative of the most common habitat in the littoral zone of the lake; it is devoid of macrophytes year round and is composed of sandy sediments. Different food resources were available across seasons; during the summer months (June-September), periphyton growing on sediments was probably the main food available for amphipods while during autumn months (September-November), leaves from the riparian zone of the lake became available [40]. Predation risks and competition from other taxa were not estimated during our study, but it is clear that the insect larvae community changed during the season at the study site. For example, dragonfly and damselfly larvae were more abundant in spring (May and June) than at other periods of the year; during autumn (September to November), Hyalella amphipods were the main component of the macroinvertebrate community at the study site (K. Dionne, personal observation). Physical and chemical parameters were measured each month using a YSI 556 MPS multiparametric sonde from YSI (Yellow Springs, Ohio, USA). Water temperature peaked at 20.3 °C in July and was at its lowest point in November at 5.7 °C; pH was stable for all months with a mean of 7.4 while dissolved oxygen decreased gradually from 11.5 mg L−1 in May to 6.1 mg L−1 in September and finished to a point of 10.6 mg L−1 in November.
For the determination of the relative abundances of each cryptic species in the community, three replicates were collected at a depth of about 0.3 m in areas of about six square-meters with a distance of three meters between them. For each replicate, sediments were kicked during three minutes to suspend the benthos into the water, and a dip net (500 μm mesh size) was swept into the suspended sediments during that time. Sampling was always performed by the same person to keep the sampling effort constant. Each replicate was preserved in 95% ethanol before sorting in the laboratory for the amphipods. For each replicate, all amphipods were counted and submitted to morphological analyses. Then, for each replicate, ten amphipods were randomly chosen for molecular analyses.
To evaluate temporal variation in the community, the relative abundance of each species was compared between months using ANOVA. The month was the factor and the relative abundance of one species was the dependent variable. Similar analyses were made separately on adults and on juveniles. Cryptic species of Hyalella amphipods can exhibit different life history traits [12,23]; this could foster different proportions for each species in the community of juveniles. Also, different relative abundances in the community of adults and juveniles could be possible if one or more species were not residents of the study site since juveniles and adults do not have the same mortality risks [48] and might not have the same capacity to establish in a sink site. All statistical analyses were performed on the SAS 9.2 software (SAS Institute Inc, Cary, North Carolina, USA).
Probably due to problems of preservation, we were unable to extract total DNA from the amphipods sampled in September and October 2007. Instead, we extracted DNA from 30 adult amphipods sampled for another study [40] using the same methods on the same dates and at the same site. Unfortunately, the animals used for that study were pooled across replicates, therefore, the September and October samples were excluded from the ANOVA on relative abundances of species over time. To explore the complete data set, we pooled samples within months and used chi-squared tests to evaluate temporal variation in amphipod relative abundances.
3. Results
3.1. Determination of the Number of Species in Lac des Baies
All amphipods used in the molecular analyses belonged morphologically to H. azteca. Five COI haplotypes were detected in Lac des Baies using molecular analyses on a total of 282 amphipods (GenBank accession numbers JN161817-JN161821). Haplotypes 2, 3 and 4 had paired divergences of 1% or below, suggesting that they belonged to the same species (Table 2). Other COI haplotypes had divergences over 20% between them, suggesting the occurrence of three cryptic species: species A (haplotype 1), species B (haplotypes 2, 3 and 4) and species C (haplotype 5) (Table 2). Comparisons with other sequences found on GenBank revealed that species A was genetically similar to haplotype 1–5 from clade 1 of Witt et al. [49] (COI divergence = 0.6%), species B was genetically similar to the haplotype from the B Clade of Wellborn and Broughton [22] occurring in Duck Lake, Michigan (COI divergences varying between 1.0% and 1.8%) and species C was genetically similar to haplotype 6–3 from clade 6 of Witt et al. [49] (COI divergence = 0.8%). For all sites, at least two cryptic species were found living in sympatry (Table 1). Very few amphipods, essentially G. lacustris, were found living in the profundal zone of the lake. G. lacustris was also present in the littoral zone at site 6.
3.2. Morphological Variation Among Species
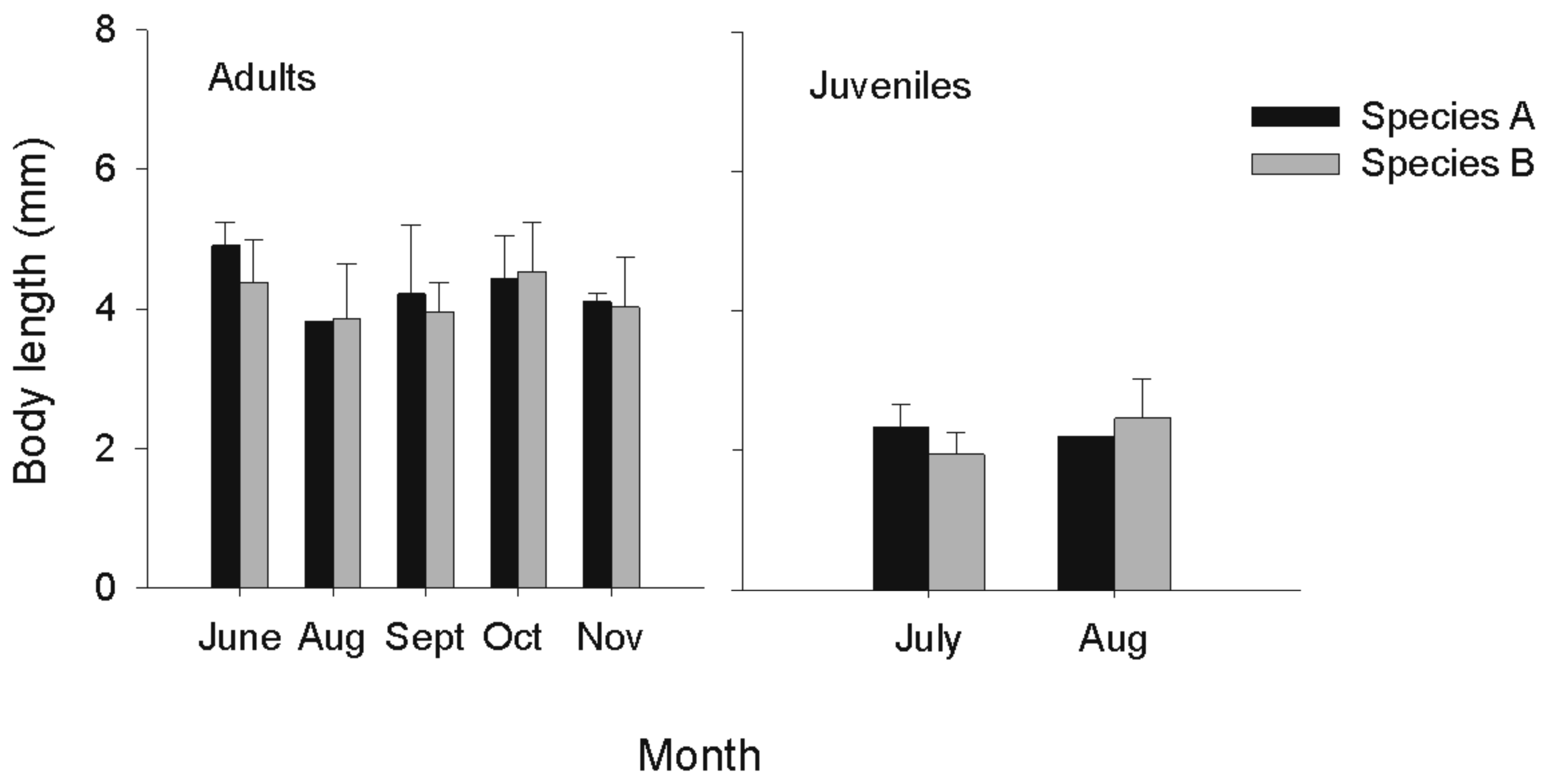
No morphological differences were found among Hyalella species for any month. All Hyalella amphipods had two spines on pleon segments 1 and 2, excluding the presence of the form inermis. Throughout the open-water season, all species had some adult males with small second gnathopods (species A = 3, species B = 5, species C = 2, on a total of 81 males), suggesting inter-individual variation rather than an inter-specific difference. Since species A and C were rare (total number of adult individuals = 20 and 4, respectively), we could not statistically compare their body lengths with species B. However, by visually comparing the mean body lengths of species A and B for each month (Figure 2), one can see a large overlap in body lengths for adults and juveniles of these species for all months.
3.3. Relative Abundances of Species Over Time
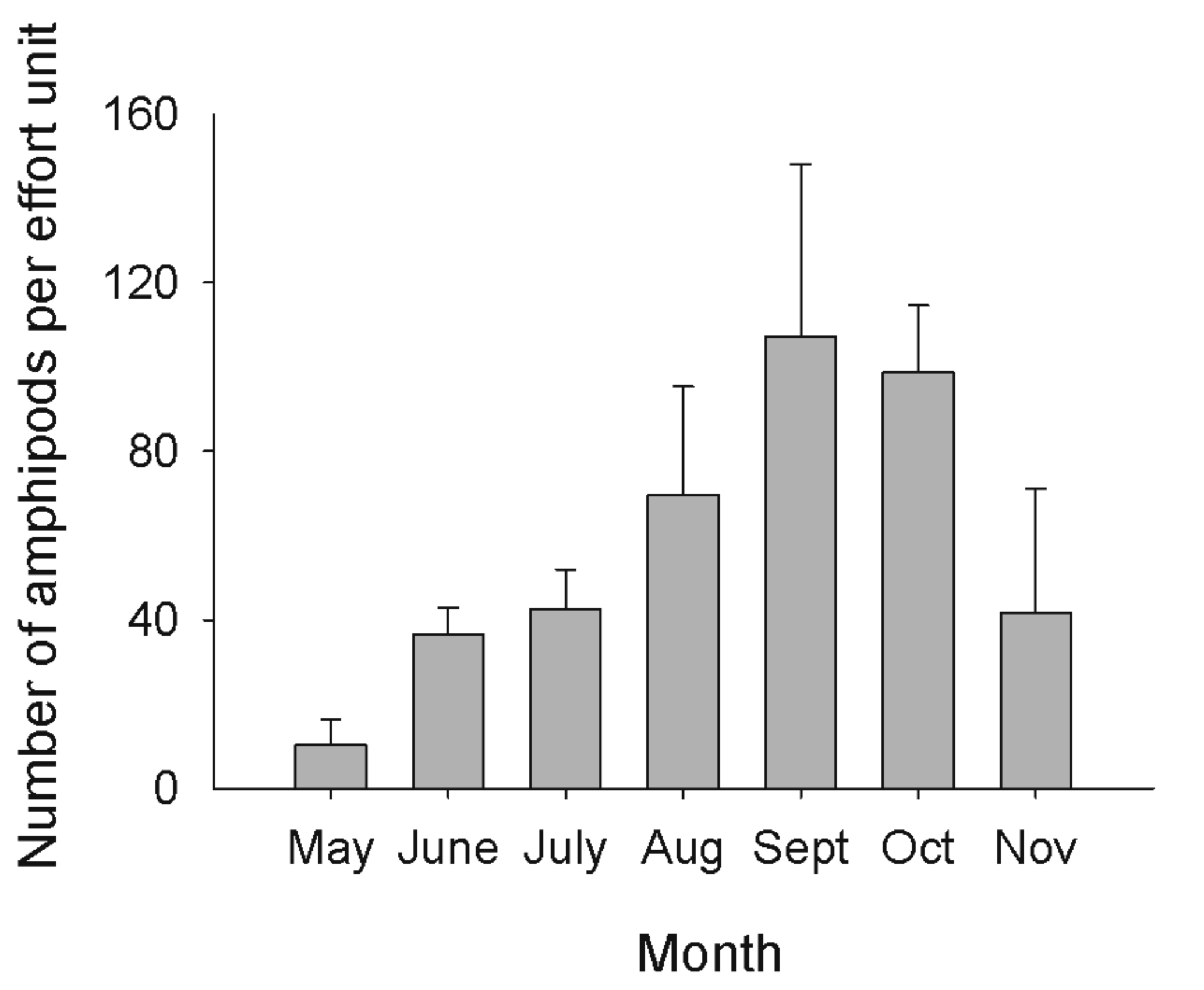
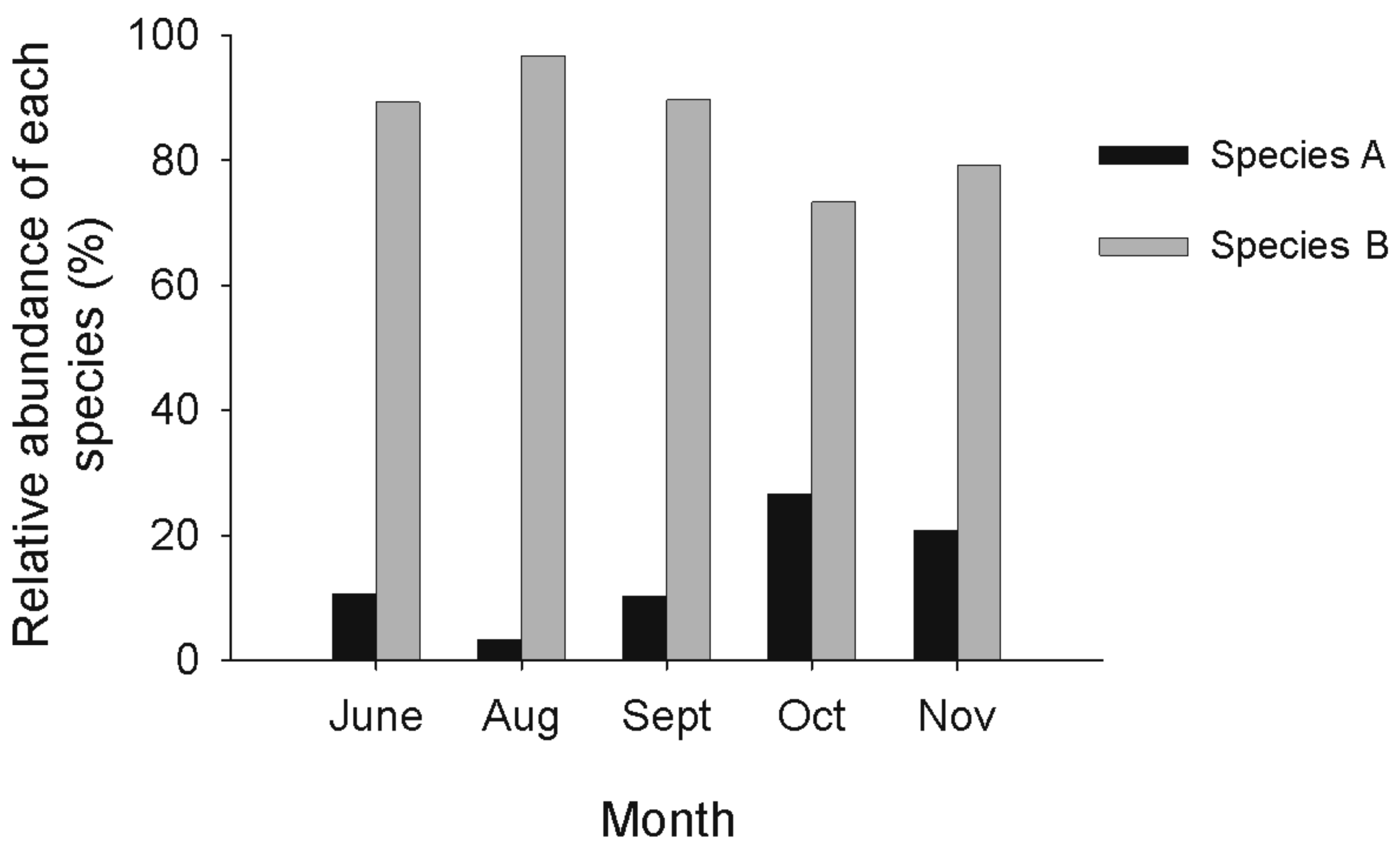
From May to November 2007, 1240 amphipods were sampled at site 1. Abundances were at their lowest in May, increased to reach a peak in September and then decreased in October and November (Figure 3). The samples of amphipods used for molecular analyses represented from 9% (in September) to 28% (in August) of the total number of amphipods. May was not included in the analyses since not enough amphipods were present at the site. In July, adults were seldom found and juveniles were dominant in the community. Juveniles were mainly present at the site in July and August. Species C was excluded from the analyses since it was only represented by one or two individuals per month. One G. lacustris was found in October.
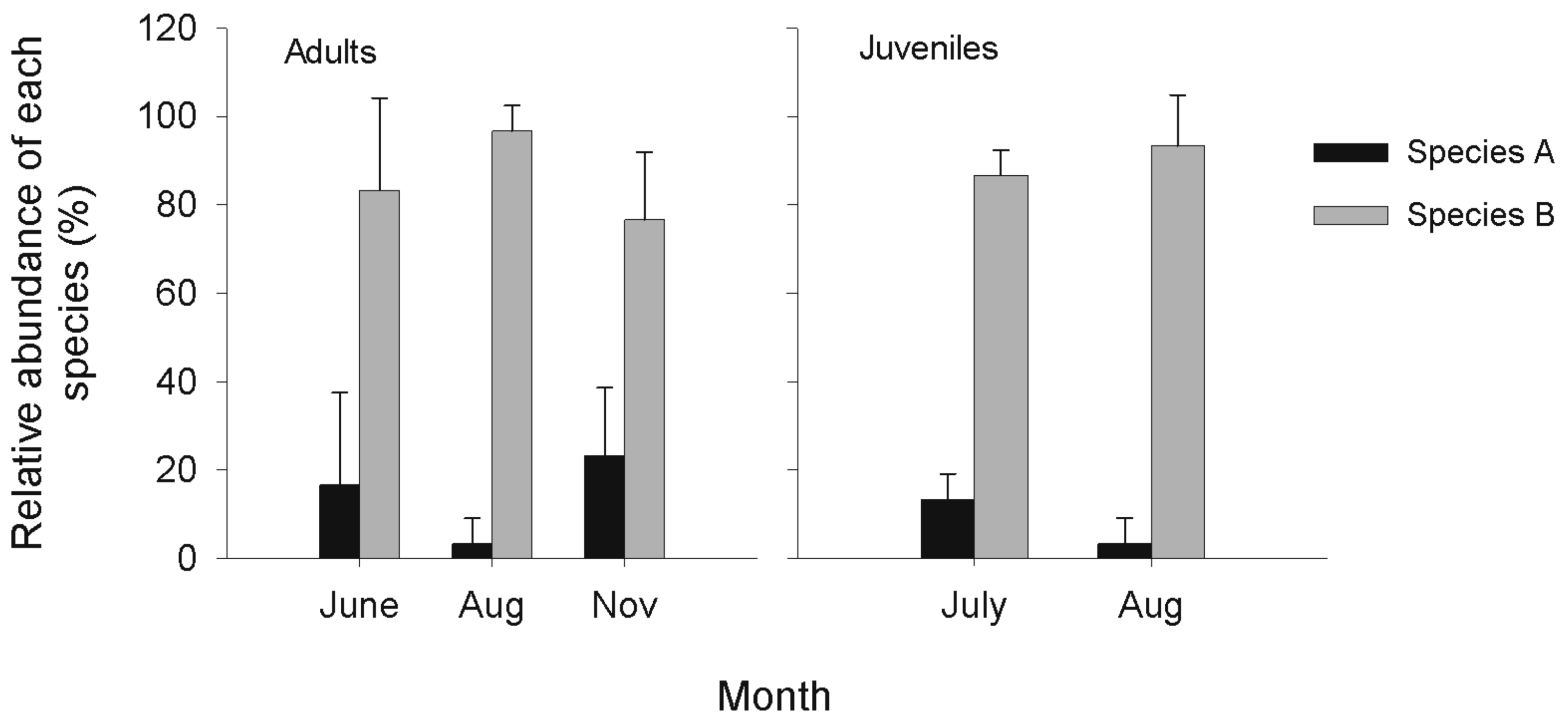
No significant variation was observed among months in the relative abundance of each species in the community, both for adults (reduced data set) (ANOVA F = 1.33, df = 2, P = 0.33) and juveniles (ANOVA F = 0.80, df = 1, P = 0.42). Both adults and juveniles of species B were more abundant than species A for all months (Figure 4). By including adult amphipods sampled with the protocol of Dionne [40] in September and October (complete data set), still no significant variation was observed among months in the relative abundance of each species (χ2 = 8.28, df= 4, P = 0.08) (Figure 5).
4. Discussion
4.1. Species Identification
Three cryptic species of the H. azteca species complex were found in Lac des Baies. Morphological measurements are consistent with the morphological descriptions given by Witt et al. [49] and Wellborn and Broughton [22] for related haplotypes. Body length measurements suggest that all species belong to the small ecomorph of the Hyalella complex [22,23]. Genetic divergences for COI were high between species (over 20%); such high genetic divergences for sympatric H. azteca cryptic species were also reported by Wellborn and Cothran [6] and Witt et al. [21]. Further studies are needed to assess if differences occur in life history traits of these species or if differences exist in their morphology, physiology or behavior, as it is the case for other communities of H. azteca amphipods [6,12].
4.2. Constant Composition of the Community
Despite environmental variations, the composition of the Hyalella species complex remained constant during the open-water season in the littoral zone of Lac des Baies. Species B was always more abundant than species A and C. It is not clear if Hyalella cryptic species of Lac des Baies coexist via undetected trade-offs that enable them to occupy different ecological niches or if they only co-occur at the site. However, the fact that these species were also found together at other sites of the lake that have similar and different environmental conditions than the study site suggests that their ecological niches probably overlap importantly. In comparison, G. lacustris had a more restricted niche in the lake; it was mainly found in shallow water (a few centimeters deep) in well-developed leaf litters or in the profundal zone of the lake (K. Dionne, personal observation). Further studies comparing the relative abundances of each Hyalella cryptic species among different habitats of the lake could be informative on the specific niche of each species.
Migration from nearby sites may explain the presence of species at low abundances at a given site. The fairly low contributions of species C and to a lesser extent species A to the community would suggest that these species are not permanent residents at the study site. As mentioned by Wellborn and Cothran [6], the community structure of a local site could be maintained year round via source-sink dynamics [50-52]. In such a system, a species that is a bad competitor in one particular habitat can be rescued from local exclusion by individuals that emigrate from another habitat where this species is a good competitor [51-52]. In Lac des Baies, the fact that species B dominates at the study site suggests that it could be a specialist of this habitat. Wellborn and Cothran [6] reported that species B dominated in the narrow-edge habitat of Sullivan Lake, Michigan, where it could be a good competitor for resources while being particularly vulnerable to predation risk from fish in deeper water [6]. In the present case, it is likely that the rarer species A and C are outcompeted in the habitat of the study site and that the occurrence of these species in the community is only maintained via migration from other habitats where species A and C could be, for instance, predator-avoidance specialists. Since the study site is located on bare sediments, it would be worth comparing its community with communities inhabiting nearby macrophytes located in deeper water (2 m) to verify if species A and C are more abundant in this habitat. Our results also show that adults and juveniles of species A were always more abundant than species C. This could mean that either species A migrates from a nearer site than species C, or that it is more abundant at the source.
Migration is also important in the formation of the amphipod community in the spring. In May, amphipods were seldom found at the study site, whereas they were more abundant in June. Since no juvenile was detected before July, amphipods at the study site were probably individuals that had migrated from nearby sites. Indeed, some studies have reported the occurrence of Hyalella amphipods at different depths in temperate lakes depending on the season and suggest their ability to perform extensive intra-lake migrations [31,32,53]. In winter, Hyalella amphipods are absent in the frozen narrow edges of lakes; these animals do not possess any resting stage capable of surviving in such harsh conditions. This means that when ice melts in spring, the whole littoral zone can be colonized by amphipods returning from their winter refuge, but also by amphipods from other locations of the lake. In that latter case, the species composition of the community could change from one year to another, especially if little differences exist in dispersal capacity and in ecological niche between species. Over long distances, amphipods probably act as passive dispersers [54], so their migration rates could be greatly influenced by seasonal events such as spring and autumn mixings and occasional great winds [31]. These events could explain why, despite being statistically non-significant, species A became slightly more abundant in the community in October and November, and why we found a lone G. lacustris in October.
Our study shows that cryptic species of Hyalella amphipods can co-occur in the littoral zone of a boreal lake without temporal variation in their community. However, since our study is only focused on one site during one open-water season, its scale do not suffice to completely explain the composition of the community of Hyalella amphipods during the open-water season. Processes related to metacommunities that may be important in explaining the co-occurrence of species act both at the local (site) and regional (lake) scales [13,52,55,56]. A lake is a heterogeneous ecosystem that contains many habitats that vary both in space and time; this variability can help species to coexist by offering them a diversity of niches to exploit. This could also favor cryptic species co-occurrence by offering them various possibilities to actively or passively migrate in the lake. Further studies should compare relative abundances of cryptic species among multiple habitats at different periods of a year and among many years. Such studies would help to comprehend the effects of the spatiotemporal variability of habitats on cryptic species assemblage and its interaction with neutral and metacommunity-related processes.
| Site | Latitude/Longitude | Depth (m) | Current velocity | Substrate | Species (N) |
|---|---|---|---|---|---|
| 1 | 48°11′22″N 68°39′24″W | 0.3 | Low | Sand with organic detritus | sp. A (1), sp. B (28), sp. C (1) |
| 2 | 48°10′76″N 68°39′72″W | 0.3 | Low | Sand with organic detritus | sp. A (2), sp. B (4) |
| 3 | 48°10′68″N 68°39′43″W | 0.3 | High | Rocks | sp. A (1), sp. B (1) |
| 4 | 48°09′96″N 68°40′65″W | 0.3 | Low | Sand with organic detritus | sp. A (1), sp. B (16), sp. C (2) |
| 5 | 48°09′58″N 68°40′05″W | 0.3 | Medium | Rocks | sp. A (4), sp. B (1) |
| 6 | 48°09′31″N 68°40′15″W | 0.3 | Low | Mud with organic detritus | sp. B (9), sp. C (4) |
| 7a | 48°09′48″N 68°39′81″W | 0.3 | Low | Sand with macrophytes | sp. A (2), sp. B (2), sp. C (2) |
| 7b | 48°09′48″N 68°39′81″W | 0.3 | Low | Sand with organic detritus | sp. B (14), sp. C (1) |
| 8 | 48°10′30″N 68°40′00″W | 13.0 | Medium | Mud | sp. B (2), sp. C (4) |
| Haplotypes | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 2 | 22.3 | |||
| 3 | 22.9 | 0.8 | ||
| 4 | 23.2 | 1.0 | 1.0 | |
| 5 | 25.9 | 20.6 | 20.3 | 20.4 |
Acknowledgments
We thank Richard Cloutier for allowing us to use his equipments for the morphological analyses. We are grateful to Beatrix Beisner, Dominique Gravel and three anonymous reviewers for their comments on an earlier version of this manuscript. Special thanks to Chantal Lacoste, Katherine Gareau, Catherine Ménard and Martin Pelletier for their assistance in the field, to Robert Chabot and Marion Trudel for their help with the morphological identification of G. lacustris and to the Réserve faunique Duchénier personnel for the access in the study site. This work was supported by a grant from the Natural Sciences and Engineering Research Council of Canada to CN and FD and a scholarship from the Fonds Québécois de la Recherche sur la Nature et les Technologies to KD. This study is a contribution to the research programs of the Centre d'études nordiques and Boréas.
References
- Knowlton, K. Sibling species in the sea. Annu. Rev. Ecol. Syst. 1993, 24, 189–216. [Google Scholar]
- Sáez, A.G.; Lozano, E. Body doubles. Nature 2005, 433, 111. [Google Scholar]
- Bickford, D.; Lohman, D.J.; Sodhi, N.S.; Ng, P.K.L.; Meier, R.; Winker, K.; Ingram, K.K.; Das, I. Cryptic species as a window on diversity and conservation. Trends Ecol. Evol. 2006, 22, 148–155. [Google Scholar]
- Zhang, D.-Y.; Lin, K.; Hanski, I. Coexistence of cryptic species. Ecol. Lett. 2004, 7, 165–169. [Google Scholar]
- McPeek, M.A.; Gomulkiewicz, R. Assembling and depleting species richness in metacommunities: Insights from ecology, population genetics and macroevolution. In Metacommunities: Spatial Dynamics and Ecological Communities; Holyoak, M., Leibold, M.A., Holt, R.D., Eds.; University of Chicago Press: Chicago, USA, 2005; pp. 355–373. [Google Scholar]
- Wellborn, G.A.; Cothran, R.D. Niche diversity in crustacean cryptic species: Complementarity in spatial distribution and predation risk. Oecologia 2007, 154, 175–183. [Google Scholar]
- Chesson, P. Mechanisms of maintenance of species diversity. Annu. Rev. Ecol. Syst. 2000, 31, 343–366. [Google Scholar]
- Siepielski, A.M.; McPeek, M.A. On the evidence for species coexistence: A critique of the coexistence program. Ecology 2010, 91, 3153–3164. [Google Scholar]
- Silvertown, J. Plant coexistence and the niche. Trends Ecol. Evol. 2004, 19, 605–611. [Google Scholar]
- Schluter, D.; McPhail, J.D. Ecological character displacement and speciation in sticklebacks. Am. Nat. 1992, 140, 85–108. [Google Scholar]
- Grant, P.R.; Grant, B.R. Evolution of character displacement in Darwin's finches. Science 2006, 313, 224–226. [Google Scholar]
- Wellborn, G.A.; Cothran, R.D. Phenotypic similarity and differentiation among sympatric cryptic species in a freshwater amphipod complex. Freshwater Biol. 2004, 49, 1–13. [Google Scholar]
- Leibold, M.A.; McPeek, M.A. Coexistence of the niche and neutral perspectives in community ecology. Ecology 2006, 87, 1399–1410. [Google Scholar]
- Cooper, W.E. Dynamics and production of a natural population of a fresh-water amphipod. Hyalella azteca. Ecol. Monogr. 1965, 35, 377–394. [Google Scholar]
- Hargrave, B.T. The utilization of benthic microflora by Hyalella azteca (Amphipoda). J. Anim. Ecol. 1970, 39, 427–437. [Google Scholar]
- Strong, D.R. Life history variation among populations of an amphipod (Hyalella azteca). Ecology 1972, 53, 1103–1111. [Google Scholar]
- Bousfield, E.L. A contribution to the reclassification of neotropical freshwater hyalellid amphipods (Crustacea: Gammaridea, Talitroidea). Boll. Mus. Civ. St. Nat. Verona 1996, 20, 175–224. [Google Scholar]
- Gonzalez, E.R.; Watling, L. Redescription of Hyalella azteca from its type locality, Vera Cruz, Mexico (Amphipoda: Hyalellidae). J. Crust. Biol. 2002, 22, 173–183. [Google Scholar]
- Hogg, I.D.; Larose, C.; de Lafontaine, Y.; Doe, K.G. Genetic evidence for a Hyalella species complex within the Great Lakes—St. Lawrence River drainage basin: Implications for ecotoxicology and conservation biology. Can. J. Zool. 1998, 76, 1134–1140. [Google Scholar]
- Witt, J.D.S.; Hebert, P.D.N. Cryptic species diversity and evolution in the amphipod genus Hyalella within central glaciated North America: A molecular phylogenetic approach. Can. J. Fish. Aquat. Sci. 2000, 57, 687–698. [Google Scholar]
- Witt, J.D.S.; Threloff, D.L.; Hebert, P.D.N. DNA barcoding reveals extraordinary cryptic diversity in an amphipod genus: Implications for desert spring conservation. Mol. Ecol. 2006, 15, 3073–3082. [Google Scholar]
- Wellborn, G.A.; Broughton, R.E. Diversification on an ecologically constrained adaptative landscape. Mol. Ecol. 2008, 17, 2927–2936. [Google Scholar]
- Wellborn, G.A.; Cothran, R.; Bartholf, S. Life history and allozyme diversification in regional ecomorphs of the Hyalella azteca (Crustacea: Amphipoda) species complex. Biol. J. Linn. Soc. 2005, 84, 161–175. [Google Scholar]
- Gilinsky, E. The role of fish predation and spatial heterogeneity in determining benthic community structure. Ecology 1984, 65, 455–468. [Google Scholar]
- Cummins, K.W.; Merritt, R.W. Ecology and distribution of aquatic insects. In An Introduction to the Aquatic Insects of North America, 3rd ed.; Merritt, R.W., Cummins, K.W., Eds.; Kendall Hunt Publishing: Dubuque, IA, USA, 1996; pp. 74–86. [Google Scholar]
- Rosine, W.N. The distribution of invertebrates on submerged aquatic plant surfaces in Muskee Lake, Colorado. Ecology 1955, 36, 308–314. [Google Scholar]
- Cattaneo, A. Grazing on epiphytes. Limnol. Oceanogr. 1983, 28, 124–132. [Google Scholar]
- Pope, R.J.; Gordon, A.M.; Kaushik, N.K. Leaf litter colonization by invertebrates in the littoral zone of a small oligotrophic lake. Hydrobiologia 1999, 392, 99–112. [Google Scholar]
- Bjelke, U.; Bohman, I.M; Herrmann, J. Temporal niches of shredders in lake littorals with possible implications on ecosystem functioning. Aquat. Ecol. 2005, 39, 41–53. [Google Scholar]
- Wetzel, R.G. Limnology: Lake and River Ecosystems; Academic Press: Waltham, MA, USA, 2001. [Google Scholar]
- Mathias, J.A. Energy flow and secondary production of the amphipods Hyalella azteca and Crangonyx richmondensis occidentalis in Marion Lake, British Columbia. J. Fish. Res. Board Can. 1971, 28, 711–726. [Google Scholar]
- Panov, V.E.; McQueen, D.J. Effects of temperature on individual growth rate and body size of a freshwater amphipod. Can. J. Zool. 1998, 76, 1107–1116. [Google Scholar]
- Cummins, K.W.; Wilzbach, M.A.; Gates, D.M.; Perry, J.B.; Bruce Taliaferro, W. Shredders and riparian vegetation. BioScience 1989, 39, 24–30. [Google Scholar]
- Kolding, S.; Fenchel, T.M. Coexistence and life cycle characteristics of five species of the amphipod genus Gammarus. Oikos 1979, 33, 323–327. [Google Scholar]
- Grassle, J.F.; Grassle, J.P. Temporal adaptations in sibling species of Capitella. In Ecology of Marine Benthos; Coull, B.C., Ed.; University of South Carolina Press: Columbia, SC, USA, 1977; pp. 177–189. [Google Scholar]
- King, C.E.; Serra, M. Seasonal variation as a determinant of population structure in rotifers reproducing by cyclical parthenogenesis. Hydrobiologia 1998, 387/388, 361–372. [Google Scholar]
- Ortells, R.; Gómez, A.; Serra, M. Coexistence of cryptic rotifer species: Ecological and genetic characterization of Brachionus plicatilis. Freshwater Biol. 2003, 48, 2194–2202. [Google Scholar]
- Bousfield, E.L. Fresh-water amphipod crustaceans of glaciated North America. Can. Field Nat. 1958, 72, 55–113. [Google Scholar]
- Bousfield, E.L. Shallow-Water Gammaridean Amphipoda of New England; Cornell University Press: Ithaca, NY, USA, 1973. [Google Scholar]
- Dionne, K. Structure d'une communauté d'amphipodes Hyalella azteca et sa consommation des détritus organiques selon la saison dans la zone littorale d'un lac boréal. M.Sc. thesis, Université du Québec à Rimouski, Rimouski, Canada, 2009. [Google Scholar]
- Geisler, F.S. Studies on the postembryonic development of Hyalella azteca (Saussure). Biol. Bull. 1944, 86, 6–22. [Google Scholar]
- Wellborn, G.A. Selection on a sexually dimorphic trait in ecotypes within the Hyalella azteca species complex (Amphipoda: Hyalellidae). Am. Midl. Nat. 2000, 143, 212–225. [Google Scholar]
- Wellborn, G.A.; Bartholf, S.E. Ecological context and the importance of body and gnathopod size for pairing success in two amphipod ecomorphs. Oecologia 2005, 143, 308–316. [Google Scholar]
- Cothran, R.D.; Kuzmic, A.; Wellborn, G.A.; Relyea, R.A. Phenotypic manipulation provides insights into the function of a sexually selected trait in a freshwater crustacean species complex. Anim. Behav. 2010, 80, 543–549. [Google Scholar]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar]
- Hall, T.A. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol. Biol. Evol. 2007, 24, 1596–1599. [Google Scholar]
- Wellborn, G.A. Size-biased predation and prey life histories: A comparative study of freshwater amphipod populations. Ecology 1994, 75, 2104–2117. [Google Scholar]
- Witt, J.D.S.; Blinn, D.W.; Hebert, P.D.N. The recent evolutionary origin of the phenotypically novel amphipod Hyalella montezuma offers an ecological explanation for morphological stasis in a closely allied species complex. Mol. Ecol. 2003, 12, 405–413. [Google Scholar]
- Shmida, A.; Wilson, M.V. Biological determinants of species diversity. J. Biogeogr. 1985, 12, 1–20. [Google Scholar]
- Mouquet, N.; Loreau, M. Community patterns in source-sink metacommunities. Am. Nat. 2003, 162, 544–557. [Google Scholar]
- Holyoak, M.; Leibold, M.A.; Mouquet, N.; Holt, R.D.; Hoopes, M.F. Metacommunities—A framework for large-scale community ecology. In Metacommunities: Spatial Dynamics and Ecological Communities; Holyoak, M., Leibold, M.A., Holt, R.D., Eds.; University of Chicago Press: Chicago, IL, USA, 2005. [Google Scholar]
- Kruschwitz, L.G. Environmental factors controlling reproduction of the amphipod Hyalella azteca. Proc. Okla. Acad. Sci. 1978, 58, 16–21. [Google Scholar]
- Bilton, D.T.; Freeland, J.R.; Okamura, B. Dispersal in freshwater invertebrates. Annu. Rev. Ecol. Syst. 2001, 32, 159–181. [Google Scholar]
- Leibold, M.A.; Holyoak, M.; Mouquet, N.; Amarasekare, P.; Chase, J.M.; Hoopes, M.F.; Holt, R.D.; Shurin, J.B.; Law, R.; Tilman, D.; Loreau, M.; Gonzalez, A. The metacommunity concept: A framework for multi-scale community ecology. Ecol. Lett. 2004, 7, 601–613. [Google Scholar]
- Leibold, M.A.; Geddes, P. El concepto de nicho en las metacommunidades. Ecol. Austral 2005, 15, 117–129. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dionne, K.; Vergilino, R.; Dufresne, F.; Charles, F.; Nozais, C. No Evidence for Temporal Variation in a Cryptic Species Community of Freshwater Amphipods of the Hyalella azteca Species Complex. Diversity 2011, 3, 390-404. https://doi.org/10.3390/d3030390
Dionne K, Vergilino R, Dufresne F, Charles F, Nozais C. No Evidence for Temporal Variation in a Cryptic Species Community of Freshwater Amphipods of the Hyalella azteca Species Complex. Diversity. 2011; 3(3):390-404. https://doi.org/10.3390/d3030390
Chicago/Turabian StyleDionne, Kaven, Roland Vergilino, France Dufresne, François Charles, and Christian Nozais. 2011. "No Evidence for Temporal Variation in a Cryptic Species Community of Freshwater Amphipods of the Hyalella azteca Species Complex" Diversity 3, no. 3: 390-404. https://doi.org/10.3390/d3030390
APA StyleDionne, K., Vergilino, R., Dufresne, F., Charles, F., & Nozais, C. (2011). No Evidence for Temporal Variation in a Cryptic Species Community of Freshwater Amphipods of the Hyalella azteca Species Complex. Diversity, 3(3), 390-404. https://doi.org/10.3390/d3030390




