Juvenile Coral Abundance Has Decreased by More Than 50% in Only Three Decades on a Small Caribbean Island
Abstract
: A comparison of the community structure of juvenile hermatypic corals of 2 to 37 m depth at the fringing reefs of Curaçao between 1975 and 2005 shows a decline of 54.7% in juvenile coral abundance and a shift in species composition. Agaricia species and Helioseris cucullata, the most common juveniles in 1975, showed the largest decline in juvenile abundance (a 9 and 120 fold decrease in density respectively) with Helioseris cucullata being nearly extirpated locally. In 2005, Porites astreoides contributed most colonies to the juvenile coral community, increasing from 8.2% (in 1975) to 19.9% of the total juvenile community. Between 1975 and 2005, juveniles of brooding species decreased in relative abundance while the abundance of juveniles of broadcast spawning species increased or remained the same. These data illustrate the magnitude of the changes that have occurred in only three decades in the composition of juvenile coral communities.1. Introduction
Over the past 30 years, coral cover on Caribbean reefs has declined from approximately 50% to 10% [1]. A similar, though less dramatic decline, occurred in Curaçao, in the Southern Caribbean. On the leeward side of Curaçao, coral cover declined from approximately 40% to 20% between 1973 and 2003 [2]. Both in Curaçao [3], and across the Caribbean [4–6], coral cover decline has been attributed to the combination of habitat degradation, sedimentation, and eutrophication that result from local industrial activity and near-shore residential and tourist developments. While the decline in coral cover has been documented for reef systems around the world [7], other changes to coral community structure are less well documented, largely due the absence of long-term studies (i.e., datasets over 20 years old) and relevant historic information.
The maintenance and recovery of coral communities depends on the successful establishment, early survival and subsequent growth of coral planulae [8] and overall recruitment success differs between species under the same environmental conditions [9–11]. Variation in local coral recruitment rates is caused by variation in the composition of the local benthic habitat [8,12,13] as well as the size of the adult source population [8,14]. In addition, the total number of coral recruits, juveniles and adults in a given location will decrease during episodic disturbance events such as storms [15]. Recruitment failure can halt, delay or even prevent coral community maintenance and recovery [12,16,17].
Variability in recruitment across several decades (i.e., between 1963 and 1992) has been quantified in the Pacific (Great Barrier Reef: [15]; Hawaii: [17]) but a decline in recruitment rates following changes in adult abundance was not observed. On the Great Barrier Reef local recruitment rates depended primarily on the availability of open space [15], whereas in Hawaii they depended on adult cover in the preceding year [17]. Such seemingly conflicting observations can be explained by differences in the open/closed nature of the study system under consideration [18]. For the Caribbean, there is little information on decadal variation in recruitment rates. Hughes and Tanner [16] showed that the recruitment rates of three dominant coral species in Jamaica declined between 1977 and 1993. They suggested increased algal cover and a decline in the species' source populations as drivers of the decline in recruitment rates through time. On Curaçao, Bak et al. [2] witnessed a 50% decline in coral cover over a 30-year time period from 1973 to 2003. Similarly, Vermeij [10] observed a 5.16-fold decline in coral recruitment to artificial substrata when comparing data taken during 1979–1981 with data taken over the period 1998 to 2004.
“True” recruitment, i.e., the combined outcome of settlement and early post-settlement mortality, is difficult to monitor due to the temporal variability in larval supply, and the problems in finding and identifying true coral settlers, which are less than 2 mm in diameter; [8]). Growth rates collected for various species at Curaçao [10] indicate that coral colonies up to 4 cm in diameter can be up to 13 years old. Juvenile densities are therefore not indicative of recent and potentially stochastic recruitment dynamics, but rather represent the integrated outcome of settlement and postsettlement survivorship over much longer time periods (i.e., greater than 10 years). The fact that juvenile densities represent the integration of approximately ten years of recruitment means that measurements of juvenile abundance are less subject to dramatic interannual fluctuations.
In this article, we compare the abundance and distribution of juvenile corals on Curaçao's leeward reefs in 2005 with a similar data set from 1975 from the same Caribbean island [19]. Using this uniquely detailed historical ecological data on juvenile corals, we quantify the changes that have occurred in the abundance and composition of Caribbean juvenile coral communities over a thirty year period.
2. Materials and Methods
2.1. Sampling Methodology
The abundance of juvenile corals (maximum diameter 4 cm) was determined at six reefs along the Curaçao coast, including the two sites that were studied using similar methods in 1975 [19]: Buoy I (12°07′34″N–68°58′32″W) and Buoy III (12°07′34″N–68°58′41″W). The other sites are: Santa Martha Bay (12°16′04″N–69°07′47″W), Snake Bay (12°08′21″N–68°59′53″W), Piscadera (12°07′21″N–68°58′16″W) and Water Factory (12°06′32″–68°57′16″W). For details on all sites, see [20]. At each site the abundance of juveniles was determined in four depth zones (following the original sampling design described in [19]): the shallow terrace zone (3–9 m), the drop-off zone (9–17 m), the upper slope zone (17–26 m) and the lower slope zone (26–37 m).
At each depth we counted, identified, and measured all juvenile hermatypic corals within a 0.25 m × 0.25 m quadrant that was moved along a 5 m long transect so that all juveniles in a total area of 1.25 m2 were quantified for each transect. In each depth zone, a minimum of eight transects were haphazardly placed, avoiding sand patches and dense monospecific beds of the yellow pencil coral Madracis mirabilis.
2.2. Species Identification
Because of the difficulty of identifying some of the smallest juveniles to species level, they were binned at the genus level for the following species: Madracis spp. (i.e., M. decactis, M. pharensis and M. senaria), Siderastrea spp. (i.e., S. siderea and S. radians, Montastraea spp. (i.e., M. annularis, M. faveolata and M. franksi) and Agaricia spp. (i.e., A. agaricites, A. humilis and A. fragilis). Finally, because solitary juvenile polyps of Scolymia lacera and Mussa angulosa look very similar, they were grouped as “Mussa sp./Scolymia sp.”. Juveniles of the hydrozoan Millepora spp. (i.e., fire coral) were also included in our surveys.
2.3. Statistical Analyses
For each species, estimates of juvenile abundance in 2005 were correlated against depth to determine if a possible relationship existed between the abundance of their juveniles and depth. Potential differences in the distribution of juveniles (all species combined) across space in terms of depth and site were further investigated using a nested ANOVA.
The original site-by-site abundance data from Bak and Engel [19] were lost, therefore we could not compare our data to the 1975 data on a site-by-site basis. From the 1975 dataset, only island-wide averages for all species × depth combinations were available. To compare our two datasets, juvenile density for both sampling periods was first recalculated as the number of individuals per m2 using depth zones, rather than sites, as replicates. We then compared juvenile abundance in 1975 and 2005 using a one way ANOVA using “year” as a predictor variable. In taking this approach, we make the assumption that the data of Bak and Engel [19] provided an average estimate of juvenile abundance along the central leeward coast of Curaçao. To create an appropriate dataset for comparison, we focused our study in 2005 to sites that were also along the central portion of the leeward coast of the island. For more information on the composition of Curaçao's reef communities around the island [20].
3. Results
3.1. Juvenile Abundance and Distribution in 2005
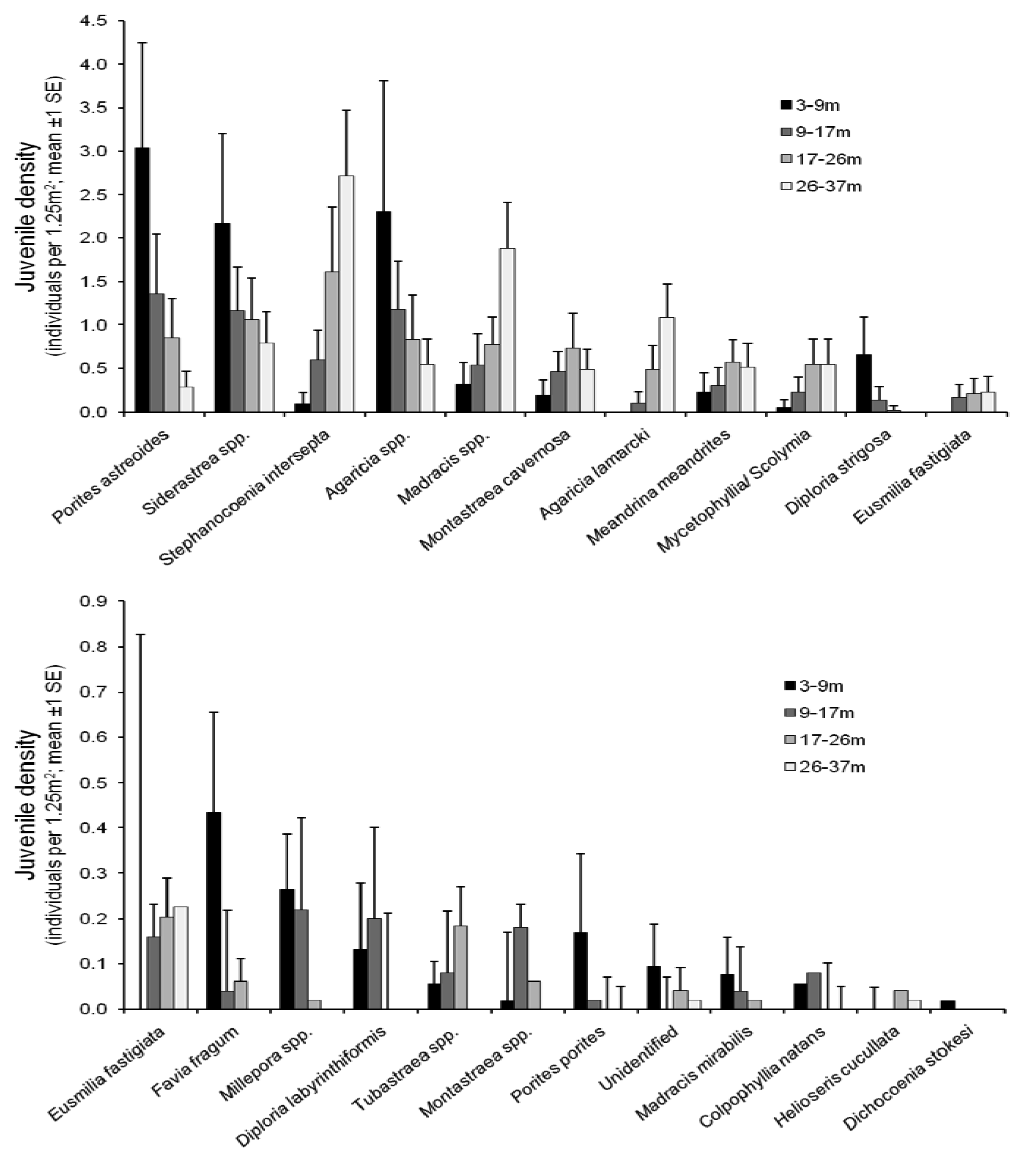
In 2005, the species Agarica spp., Porites astreoides, Stephanocoenia intersepta and Siderastrea spp. were the four most abundant types of coral juveniles on the reefs of Curaçao at depths between 3 and 37 m (Figure 1) and collectively accounted for 58.1% of all juveniles observed (n = 544). Brooding species thus dominated the juvenile community. The most dominant juveniles of broadcast spawning species were Montastraea cavernosa (5.4%) and Colpophyllia natans (0.3%).
The abundances of juvenile corals of some species reduced with increasing depth (Figure 1; Porites porites, P. astreoides, Siderastrea sp., Agaricia sp., Diploria strigosa, Diploria labyrithiformis, Favia fragum, Millepora sp., Madracis mirabilis, C. natans, Dichocoenia stokesi; −0.42 < r < −0.15, p < 0.001, n = 32). Other species increased in abundance with increasing depth (Figure 1); S. intersepta, Madracis sp., Montastraea cavernosa, Agaricia lamarcki, Meandrina meandrites, Mycetophyllia/Scolymia sp., Eusmilia fastigiata and Helioseris cucculata (0.17 < r < 0.54, p < 0.02, n = 32). Montastraea sp. and Tubastrea coccinea showed no clear change in abundance over the depth range considered, i.e., 3–37 m (−0.01 < r < −0.07; p > 0.30, n = 32). Similarly, the relative occurrence of juvenies from broadcast spawning and brooding species did not change with depth. See [19] for species' depth distributions in 1975.
When all species were pooled, no spatial differences in juvenile density across sites could be detected (Nested ANOVA, F18,177 = 1.88, p = 0.10), although the relationship between density and depth varied significantly across sites (Nested ANOVA, F5,177 = 1.88, p < 0.01). All possible relationships between depth and juvenile density were observed for at least one site (Figure 2) suggesting that the distribution of juvenile corals with depth is partially dependent on local environmental factors at the scale of individual sites.
3.2. Historic Comparisons: 1975 and 2005
Across all sites and depths, juvenile coral density averaged 15.3 individuals per m2 in 1975 [19] and 6.9 per m2 in 2005. Using depth zones as replicates, the overall decline in mean coral juvenile abundance was highly significant (ANOVA, F1,6 = 39.85, p < 0.001). Juvenile abundance decreased through time for every depth zone. This trend was driven by the reduction in juvenile abundance of brooding species, whereas the density of juveniles of broadcasting species remained stable or increased (Figure 3). The change in juvenile density thus varied among species (Figure 4).
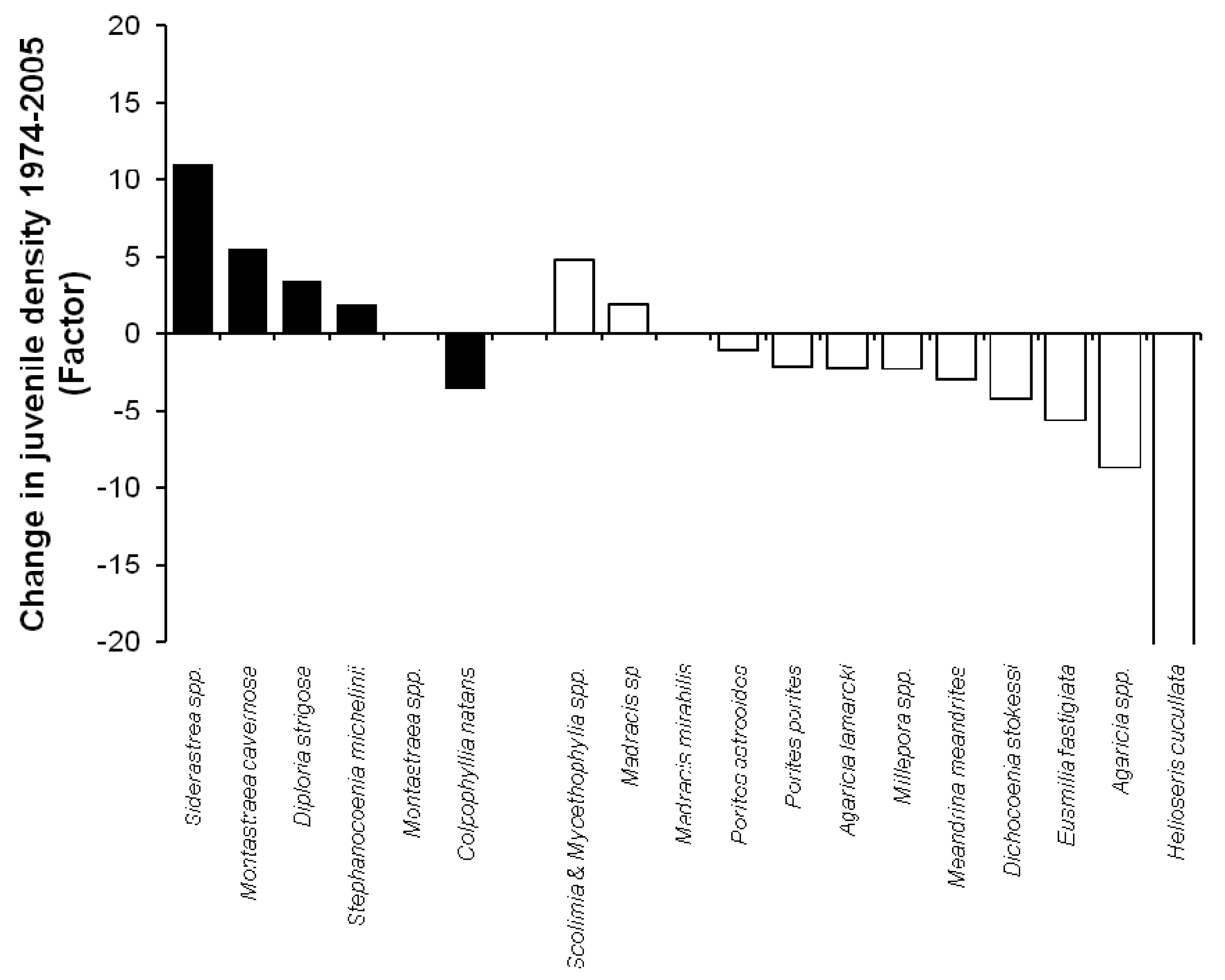
From 1975 to 2005, Agaricia spp. experienced the most dramatic decline, from 56.1% of the total juvenile population in 1975 (n = 518) to 14.3% of the total juvenile population in 2005 (n = 50). Despite this reduction, Agaricia spp. was still one of the most abundant juvenile coral taxa in 2005. The most dramatic decline was observed for Helioseris cucullata, which declined from 9.4% to 0.2% of the total juvenile population. Siderastrea spp. show the largest positive change, increasing from 0.5% to 13.3% of the total juvenile population (Figure 4). Therefore, despite the overall decline in juvenile coral abundance, juveniles of some coral species were found in higher densities in 2005 than in 1975 (Figure 5). Notably, six species have increased in abundance (i.e., number of colonies per m2) since 1975. Four of these species (66.7%) are broadcast spawners. Brooders account for the majority (90%) of the species that were less abundant in 2005 (n = 10).
4. Discussion
This study provided a comparison of the community structure of coral juveniles along the coral reefs of Curaçao's leeward shore, along the depth gradient from 3 m to 37 m. Juvenile coral densities on Curaçao in 2005 (average of 6.9 individuals per m2 across depths) were 1–5 times higher than the densities measured on other Caribbean islands over the last 15 years [21,22], but these values were still lower than those historically measured in the Caribbean region [19,23,24]. The distribution of species across the depth gradient we examined in 2005 (Figure 1) parallels the historical data collected on the same reefs in 1975 [19]. Juvenile coral community composition varied minimally between sites, which is unsurprising due to the uniform nature of the reefs along the central part of the leeward coast of Curaçao [20]. Most of the coral juveniles belonged to four taxa: Porites, Siderastrea, Stephanocoenia and Agaricia, which together accounted for 63.4% of all juveniles observed (n = 544).
The abundance of juveniles serves as a proxy measurement of the integrated outcome of settlement, post-settlement survivorship and disturbance across a multiple year time period. We observed that this metric, total juvenile abundance, declined by 54.7% overall between 1975 and 2005. Importantly, we quantified coral juveniles that were on average approximately 13 yrs old [10] rather than recruits (i.e., coral settlers < 4 mm in diameter). Therefore, the temporal fluctuations that affect recruit supply [8] are less pronounced in the overall abundance of juveniles and the observed decline in juvenile abundance is unlikely to be an artifact from sampling the highly variable recruitment process at the beginning and end of a 30 yr period. Importantly, the decrease of juvenile corals was observed consistently in four different depth zones between 3 and 37 m. (Figure 3). The declining abundance of coral recruits described here on multi-decadal timescales [10], as well as the decreasing densities of coral juveniles on other islands in the Caribbean [16], and decreased growth rates of juvenile corals [25], together suggest that conditions for the successful establishment and growth of coral recruits and juveniles have worsened in recent decades.
Within this dim picture overall, certain species have suffered disproportionately, and are now virtually absent from Caribbean reefs. In our study, juveniles of Helioseris cucullata (formerly known as Leptoseris cucullata) comprised 8.3% of the juvenile population in 1975, making them the second most common juvenile species on Curaçaoan reefs. By 2005, H. cucullata decreased in absolute abundance 120-fold, i.e., from an average of 1.43 juveniles per square meter in 1975 to 0.01 in 2005. In three decades, a once abundant species nearly disappeared from the local species pool. Similar observations were made on Jamaica where recruitment rates of H. cucullata and adult cover of this species both approached zero during the late 1980s [16], while it was known as a dominant species in the juvenile community previously [26,27]. Because this species has a relatively short life span, recruitment is crucial to maintain its population through time [16]. While overall coral recruitment rates appear to have declined over the last three decades, H. cucullata most clearly shows how a once abundant species all but disappeared from Caribbean reefs in a little over three decades.
In the Caribbean, members of the genus Agaricidae typically dominate juvenile coral communities. The abundance of this brooding species, and Porites spp. in 2005 echoes the shift seen in the community structure of other Caribbean reefs (e.g., [28–30]) from long-lived broadcast spawning species to brooding species that are characterized by shorter lifespans and a weedy life history strategy. Importantly, Agaricids have always been the most dominant taxa in juvenile coral communities on Curaçao [19], and on other Caribbean reefs [16,24,31]. Thus, the idea that brooding specie are increasing in dominance is supported by the relative increase in their abundance compared to other coral species. Despite an increase in relative abundance, the absolute numbers of Agaricid juveniles was nevertheless lower in 2005 compared to 30 years earlier (Figure 5).
The corals that presently dominate the adult coral community of Curaçao are members of the Montastraea species complex, Colpophylia natans, and Diploria species [32]. Juvenile abundances of these broadcast spawning species were extremely low in 2005, however similar observations were made over 25 years ago [19,23]. This suggests that low absolute abundance of juveniles per se is not necessarily indicative of recent recruitment failure, but rather a characteristic of long-lived species with infrequent recruitment success. Surprisingly, the absolute density of juveniles of some broadcast spawning species was 2–11 times higher in 2005 than in 1975 (Figure 5) and the same species dominate juvenile communities elsewhere in the Caribbean [21,24,33].
Bak and Engel [19] described three main ecological strategies for corals: massive, long-lived species with infrequent recruitment, short-lived opportunists with high recruitment rates and species whose propagation depends on vegetative rather than sexual reproduction. Interestingly, all but one of the broadcast-spawning species that occurred in higher densities in 2005 in our study were species in which the males release sperm prior to egg release by the females [34]. Therefore, female colonies from these species release fertilized eggs or embryos rather than unfertilized eggs (as is the case for “true” broadcasters). Releasing embryos rather than unfertilized eggs combines benefits of obligate broadcast spawning (i.e., large numbers of outcrossed offspring) and brooding strategies (i.e., increased fertilization success). Based on our observation, this should be considered a fourth life history strategy, distinct from the three described by Bak and Engel. Besides the clear difference in spawning behavior, our data show that corals with this ecological strategy are more robust in degraded habitats than “true” broadcast spawning species.
The observation that some species are now occurring in greater densities whereas others have declined indicates that species-specific factors affected the coral community's response to altered environmental conditions between 1975 and 2005. An increase in juvenile density for some broadcasting species (Siderastrea spp., Montastraea cavernosa, Diploria strigosa and Stephanocoenia michelinii) suggests that space availability could be important in driving their local abundance since the total cover of adult corals of these same species declined by approximately 50% on Curaçao over the same time period [2].
For species with brooding reproductive strategies, habitat degradation (e.g., increased coverage of benthic algae and the increased abundance of potentially pathogenic microbes) [35]) and/or a reduction of the adult pool can explain the decreased densities of juveniles [30]. Brooders generally recruit close to parental colonies [8,21] and they are therefore less likely to escape degraded habitats whose quality declined after their parent colonies established themselves on a given reef. Brooding species, therefore, could therefore be more severely affected by habitat degradation at a reef-by-reef scale. In contrast, broadcast species have the ability for long distance dispersal which allows their planulae to establish themselves outside locally degraded habitats.
In conclusion, whereas many studies have quantified coral degradation by measuring coral cover, few studies have considered how early life history processes contribute to such changes. Based on our data, we suggest that juvenile coral abundance is a useful measure of reef “health” as it reflects the relative success or failure of reef functional processes (recruitment, growth and survival) on a timescale meaningful to both ecology and conservation [36]. While the 54.7% decline in juvenile abundance observed between 1975 and 2005 can both be a cause and a consequence of the decline in adult coral cover on these reefs, it indicates that fundamental processes required for population maintenance and recovery are operating at rates well below their historic baselines. Historical baselines like the one described here clearly illustrate that recent inventories of coral juveniles alone are not sufficient to understand the changes that have occurred on Caribbean reefs over multiple decades. The near-disappearance of once abundant species, such as Helioseris cucullata, in the relatively short time span of 30 years would have been missed if only recent surveys were considered. In addition, the apparent shift from broadcasting species to brooding species could only be falsified through the use of historic data. The overall decline in juvenile coral abundance in Curaçao over a period of 30 years is representative of the profound changes that are taking place in the structure of juvenile coral communities region-wide. Reduced in numbers and subject to increasing stress, these juvenile populations face the heavy burden of serving as the next generation of corals on Caribbean reefs.



Acknowledgments
This study was made possible in part through funding to MJAV from the Government of the (former) Netherlands Antilles and the European Union under the seventh framework program (Grant# 244161). Kristen Marhaver is thanked for correcting linguistical issues in an earlier version of the manuscript.
References and Notes
- Gardner, T.A.; Côté, I.M.; Gill, J.A.; Grant, A.; Watkinson, A.R. Long-term region-wide declines in Caribbean corals. Science 2003, 301, 958–960. [Google Scholar]
- Bak, R.P.M.; Nieuwland, G.; Meesters, E.H. Coral reef crisis in deep and shallow reefs: 30 years of constancy and change in reefs of Curaçao and Bonaire. Coral Reefs 2005, 24, 475–479. [Google Scholar]
- Bak, R.P.M.; Nieuwland, G. Long-term change in coral communities along depth gradients over leeward reefs in the Netherlands Antilles. Bull. Mar. Sci. 1995, 56, 609–619. [Google Scholar]
- Fabricius, K.E. Effects of terrestrial runoff on the ecology of corals and coral reefs: Review and synthesis. Mar. Pollut. Bull. 2005, 50, 125–146. [Google Scholar]
- Nyström, M.; Folke, C.; Moberg, F. Coral reef disturbance and resilience in a human-dominated environment. Trends Ecol. Evol. 2000, 15, 413–417. [Google Scholar]
- Metcalfe, C.D.; Beddows, P.A.; Bouchot, G.G.; Metcalfe, T.L.; Li, H.; van Lavieren, H. Contaminants in the coastal karst aquifer system along the Caribbean coast of the Yucatan Peninsula, Mexico. Environ. Pollut. 2011, 159, 991–997. [Google Scholar]
- Pandolfi, J.M.; Jackson, J.B.C.; Baron, N.; Bradbury, R.H.; Guzman, H.M.; Hughes, T.P.; Kappel, C.V.; Micheli, F.; Ogden, J.C.; Possingham, H.P.; et al. Are U.S. coral reefs on the slippery slope to slime? Science 2005, 307, 1725–1726. [Google Scholar]
- Vermeij, M.J.A.; Sandin, S.A. Density dependent recruitment and mortality in a tropical benthic invertebrate. Ecology 2008, 89, 1994–2004. [Google Scholar]
- Harrison, P.L.; Wallace, C.C. Reproduction, dispersal, recruitment of scleractinian corals. In Coral Reefs: Ecosystems of the World; Dubinsky, Z., Ed.; Elsevier: Amsterdam, The Netherlands, 1995; Volume 25, Chapter 7; pp. 133–208. [Google Scholar]
- Vermeij, M.J.A. Early life-history dynamics of Caribbean coral species on artificial substratum: The importance of competition, growth and variation in life-history strategy. Coral Reefs 2006, 25, 59–71. [Google Scholar]
- Vermeij, M.J.A.; Frade, P.; Jacinto, R.; Debrot, A.O.; Bak, R.P.M. Reproductive mode drives habitat-related differences in population structure of eight Caribbean coral species. Mar. Ecol. Prog. Ser. 2007, 351, 91–102. [Google Scholar]
- Birrell, C.L.; McCook, L.J.; Willis, B.L. Effects of algal turfs and sediment on coral settlement. Mar. Pollut. Bull. 2005, 51, 408–414. [Google Scholar]
- Box, S.J.; Mumby, P.J. Effect of macroalgal competition on growth and survival of juvenile Carbbean corals. Mar. Ecol. Prog. Ser. 2007, 342, 139–149. [Google Scholar]
- Hughes, T.P.; Baird, A.H.; Dindsdale, E.A.; Harriott, V.J.; Moltschaniwskyj, N.A.; Pratchett, M.S.; Tanner, J.E.; Willis, B.L. Detecting regional variation using meta-analysis and large-scale sampling: Latitudinal patterns in recruitment. Ecology 2002, 83, 436–451. [Google Scholar]
- Connell, J.H.; Hughes, T.; Wallace, C.C. A 30-year study of coral abundance, recruitment and disturbance at several scales in space and time. Ecol. Monogr. 1997, 67, 461–488. [Google Scholar]
- Hughes, T.P.; Tanner, J.E. Recruitment failure, life histories, and long-term decline of Caribbean corals. Ecology 2000, 81, 2250–2263. [Google Scholar]
- Coles, S.L.; Brown, E.K. Twenty-five years of change in coral coverage on a hurricane impacted reef in Hawai'i: The importance of recruitment. Coral Reefs 2007, 26, 705–717. [Google Scholar]
- Hixon, M.A.; Pacala, S.W.; Sandin, S.A. Population regulation: Historical context and contemporary challenges of open vs. closed systems. Ecology 2002, 83, 1490–1508. [Google Scholar]
- Bak, R.P.M.; Engel, M.S. Distribution, abundance and survival of juvenile hermatypic corals (Scleractinia) and the importance of life history strategies in the parent coral community. Mar. Biol. 1979, 54, 341–352. [Google Scholar]
- Van Duyl, F.C. Atlas of the Living Reefs of Curaçao and Bonaire (Netherlands Antilles); Naturwetenschappelijke Studiekring voor Suriname en de Nederlandse Antillen: Utrecht, The Netherlands, 1985; Volume 117, pp. 1–38. [Google Scholar]
- Chiappone, M.; Sullivan, K.M. Distribution, abundance and species composition of juvenile scleractinian corals in the Florida reef tract. Bull. Mar. Sci. 1996, 58, 555–569. [Google Scholar]
- Edmunds, P.J.; Carpenter, R.C. Recovery of Diadema leads to reduced macroalgal cover and increased abundance of juvenile corals on a Caribbean reef. Proc. Natl. Acad. Sci. USA 2001, 98, 5067–5071. [Google Scholar]
- Dustan, P. Vitality of reef coral populations off Key Largo, Florida: Recruitment and mortality. Environ. Geol. 1977, 2, 51–58. [Google Scholar]
- Rogers, C.S.; Fitz, H.C.; Gilnack, M.; Beets, J.; Hardin, J. Scleractinian coral recruitment patterns at Salt River Submarine Canyon, St. Croix, US Virgin Islands. Coral Reefs 1984, 3, 69–76. [Google Scholar]
- Edmunds, P.J. Evidence for a decadal-scale decline in the growth rates of juvenile scleractinian corals. Mar. Ecol. Prog. Ser. 2007, 341, 1–13. [Google Scholar]
- Rylaarsdam, K.W. Life histories and abundance patterns of colonial corals on Jamaican reefs. Mar. Ecol. Prog. Ser. 1983, 13, 249–260. [Google Scholar]
- Hughes, T.P. Life histories and population dynamics of early successional corals. Proceedings of The Fifth International Coral Reef Congress, Tahiti, French Polynesia, 27 May–1 June 1985; Gabrie, C., Salvat, B., Eds.; Antenne Museum-Ephe: Moorea, French Polynesia, 1985; 4, pp. 101–106. [Google Scholar]
- Knowlton, N. The future of coral reefs. Proc. Natl. Acad. Sci. USA 2001, 98, 5419–5425. [Google Scholar]
- Precht, W.F.; Miller, S.L. Geological Approaches to Coral Reef Ecology; Aronson, R.B., Ed.; Springer: New York, NY, USA, 2006; Chapter 9; pp. 237–312. [Google Scholar]
- Green, D.H.; Edmunds, P.J.; Carpenter, R.C. Increasing relative abundance of Porites astreoides on Caribbean reefs mediated by an overall decline in coral cover. Mar. Ecol. Prog. Ser. 2008, 359, 1–10. [Google Scholar]
- Edmunds, P.J. Juvenile coral population dynamics track rising seawater temperature on a Caribbean reef. Mar. Ecol. Prog. Ser. 2004, 269, 111–119. [Google Scholar]
- Vermeij, M. Carmabi Foundation; Willemstad, Curaçao, Unpublished work; 2011. [Google Scholar]
- Edmunds, P.J. Patterns in the distribution of juvenile corals and coral reef community structure in St. John, US Virgin Islands. Mar. Ecol. Prog. Ser. 2000, 202, 113–124. [Google Scholar]
- Vermeij, M.J.A.; Barott, K.L.; Johnson, A.E.; Marhaver, K.L. Release of eggs from tentacles in a Caribbean coral. Coral Reefs 2010, 29, 411. [Google Scholar]
- Sandin, S.A.; Smith, J.E.; DeMartini, E.E.; Dinsdale, E.A.; Donner, S.D.; Friedlander, A.M.; Konotchick, T.; Malay, M.; Maragos, J.E.; Obura, D.; et al. Baselines and degradation of coral reefs in the Northern Line Islands. PLoS ONE 2008, 3, e1548. [Google Scholar]
- Rogers, C.S.; Miller, J. Permanent ‘phase shifts’ or reversible declines in coral cover? Lack of recovery of two coral reefs in St. John, US Virgin Islands. Mar. Ecol. Prog. Ser. 2006, 306, 103–114. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vermeij, M.J.A.; Bakker, J.; Hal, N.v.d.; Bak, R.P.M. Juvenile Coral Abundance Has Decreased by More Than 50% in Only Three Decades on a Small Caribbean Island. Diversity 2011, 3, 296-307. https://doi.org/10.3390/d3030296
Vermeij MJA, Bakker J, Hal Nvd, Bak RPM. Juvenile Coral Abundance Has Decreased by More Than 50% in Only Three Decades on a Small Caribbean Island. Diversity. 2011; 3(3):296-307. https://doi.org/10.3390/d3030296
Chicago/Turabian StyleVermeij, Mark J.A., Judith Bakker, Noam van der Hal, and Rolf P.M. Bak. 2011. "Juvenile Coral Abundance Has Decreased by More Than 50% in Only Three Decades on a Small Caribbean Island" Diversity 3, no. 3: 296-307. https://doi.org/10.3390/d3030296
APA StyleVermeij, M. J. A., Bakker, J., Hal, N. v. d., & Bak, R. P. M. (2011). Juvenile Coral Abundance Has Decreased by More Than 50% in Only Three Decades on a Small Caribbean Island. Diversity, 3(3), 296-307. https://doi.org/10.3390/d3030296



