Potential Threat of an Invasive Fish Species for Two Native Newts Inhabiting Wetlands of Europe Vulnerable to Climate Change
Abstract
1. Introduction
2. Materials and Methods
2.1. Occurrence Data Collection
2.2. Environmental Data
2.3. Model Building
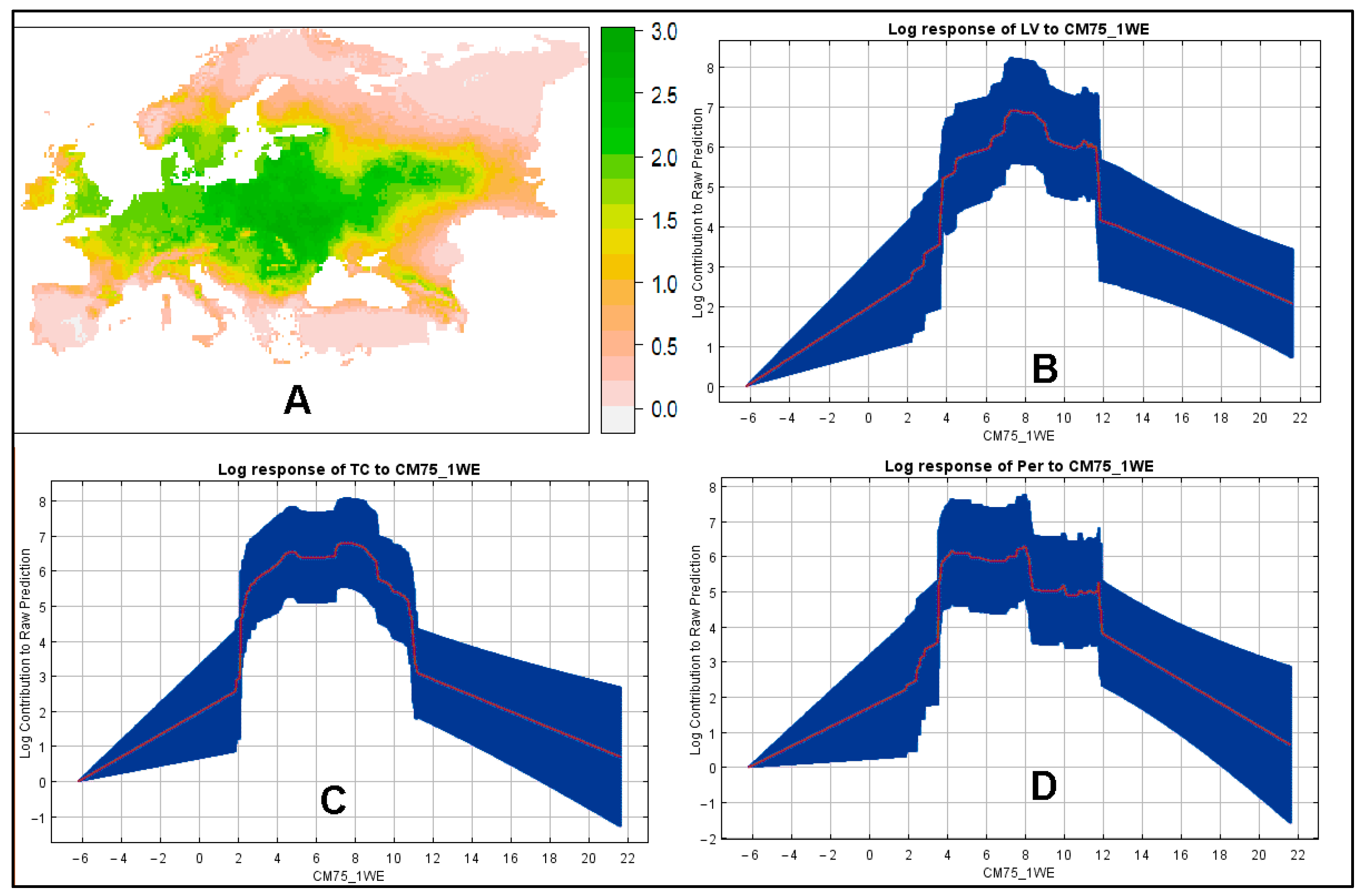
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Catenazzi, A. State of the world’s amphibians. Annu. Rev. Environ. Resour. 2015, 40, 91–119. [Google Scholar] [CrossRef]
- Howard, S.D.; Bickford, D.P. Amphibians over the edge: Silent extinction risk of Data Deficient species. Divers. Distrib. 2014, 20, 837–846. [Google Scholar] [CrossRef]
- Greenberg, D.A.; Palen, W.J. Hydrothermal physiology and climate vulnerability in amphibians. Proc. R. Soc. B Biol. Sci. 2021, 288, 20202273. [Google Scholar] [CrossRef]
- Invasive Alien Species of Union Concern. Available online: https://ec.europa.eu/environment/nature/pdf/IAS_brochure_species.pdf (accessed on 7 January 2022).
- Tytar, V.; Nekrasova, O.; Pupina, A.; Pupins, M.; Oskyrko, O. Long-term bioclimatic modelling the distribution of the fire-bellied toad, Bombina bombina, under the influence of global climate change. Vest. Zool. 2018, 52, 553–556. [Google Scholar] [CrossRef]
- Nekrasova, O.; Marushchak, O.; Pupins, M.; Skute, A.; Tytar, V.; Čeirāns, A. Distribution and potential limiting factors of the European pond turtle (Emys orbicularis) in Eastern Europe. Diversity 2021, 13, 280. [Google Scholar] [CrossRef]
- Nekrasova, O.; Tytar, V.; Pupins, M.; Čeirāns, A. Range expansion of the alien red-eared slider Trachemys scripta (Reptilia, Testudines) in Eastern Europe, with special reference to Latvia and Ukraine. BioInvasions Rec. 2022, 11, 287–295. [Google Scholar] [CrossRef]
- Nekrasova, O.; Tytar, V.; Pupins, M.; Čeirāns, A.; Marushchak, O.; Skute, A. A GIS modeling study of the distribution of viviparous invasive alien fish species in Eastern Europe in terms of global climate change, as exemplified by Poecilia reticulata Peters, 1859 and Gambusia holbrooki Girarg, 1859. Diversity 2021, 13, 385. [Google Scholar] [CrossRef]
- Kuybida, V.V.; Nekrasova, O.D.; Kutsokon, Y.K.; Lopatynska, V.V. Summer fish kills in the Kaniv Reservoir. Hydrobiol. J. 2019, 55, 103–106. [Google Scholar] [CrossRef]
- Tytar, V.; Nekrasova, O.; Pupins, M.; Čeirāns, A.; Skute, A. Modelling the range expansion of pumpkinseed Lepomis gibbosus across Europe, with a special focus on Ukraine and Latvia. North-West. J. Zool. 2022, 18, 143–150. [Google Scholar]
- Pupina, A.; Pupins, M.; Nekrasova, O.; Tytar, V.; Kozynenko, I.; Marushchak, O. Species distribution modelling: Bombina bombina (Linnaeus, 1761) and its important invasive threat Perccottus glenii (Dybowski, 1877) in Latvia under global climate change. J. Env. Res. Eng. Manag. 2018, 74, 79–86. [Google Scholar] [CrossRef]
- Kutsokon, I.; Pukhtayevych, P.; Kolomytsev, G. Spreading of Chinese sleeper (Perccottus glenii Dybowski 1877) in Zhytomyr region (Ukraine). Stud. Biol. 2013, 7, 259–264. [Google Scholar] [CrossRef]
- Hegedis, A.; Lenhardt, M.; Mićković, B.; Cvijanović, G.; Jarić, I.; Gačić, Z. Amur sleeper (Perccottus glenii Dubowski, 1877) spreading in the Danube river basin. J. Appl. Ichthyol. 2007, 23, 705–706. [Google Scholar] [CrossRef]
- Kvach, Y.; Kutsokon, Y.; Demchenko, V.; Yuryshynets, V.; Kudryashov, S.; Abramiuk, I. Post-invasion spread of Chinese sleeper (Perccottus glenii) in the Lower Danube drainage (Budjak region of Ukraine). BioInvasions Rec. 2022, 11, 547–559. [Google Scholar] [CrossRef]
- Reshetnikov, A.N.; Manteifel, Y.B. Newt-fish interactions in Moscow province: A new predatory fish colonizer, Perccottus glenii, transforms metapopulations of newts, Triturus vulgaris and T. cristatus. Adv. Amphib. Res. Former Sov. Union 1997, 2, 1–12. [Google Scholar]
- Košco, J.; Manko, P.; Miklisová, D.; Košuthová, L. Feeding ecology of invasive Perccottus glenii (Perciformes, Odontobutidae) in Slovakia. Czech J. Anim. Sci. 2008, 53, 479–486. [Google Scholar] [CrossRef]
- Reshetnikov, A.N. The introduced fish, rotan (Perccottus glenii), depresses populations of aquatic animals (macroinvertebrates, amphibians, and a fish). Hydrobiologia 2003, 510, 83–90. [Google Scholar] [CrossRef]
- Reshetnikov, A.N. Introduced fish, rotan Percottus glenii–An unavoidable threat for European amphibians. FrogLog 2005, 67, 3–4. [Google Scholar]
- Reshetnikov, A.N. Decreased Triturus cristatus breeding site number as a consequence of Perccottus glenii range expansion. FrogLog 2012, 104, 18. [Google Scholar]
- Horvatić, S.; Zanella, D.; Marčić, Z.; Mustafić, P.; Buj, I.; Onorato, L.; Ivić, L.; Karlović, R.; Ćaleta, M. First report of the Chinese sleeper Perccottus glenii Dybowski, 1877 in the Drava River, Croatia. BioInvasions Rec. 2022, 11, 250–266. [Google Scholar] [CrossRef]
- Fedoniuk, O. Advice for minimizing anthropogenic impact on fauna of amphibians and reptiles in the conditions of western Ukraine. Visnyk Lviv. Univ. Ser. Biol. 2013, 63, 3–12. (In Ukrainian) [Google Scholar]
- Košco, J.; Lusk, S.; Halacka, K.; Lusková, V. The expansion and occurrence of the Amur sleeper (Perccottus glenii) in eastern Slovakia. Folia Zool. 2003, 52, 329–336. [Google Scholar]
- Kutsokon, I.; Tkachenko, M.; Bondarenko, O.; Pupins, M.; Snigirova, A.; Berezovska, V.; Ceirans, A.; Kvach, Y. The role of invasive Chinese sleeper Perccottus glenii Dybowski, 1877 in the Ilgas Nature Reserve ecosystem: An example of a monospecific fish community. BioInvasions Rec. 2021, 10, 396–410. [Google Scholar] [CrossRef]
- Kvach, Y.; Kutsokon, I.; Roman, A.; Ceirans, A.; Pupins, M.; Kirjusina, M. Parasite acquisition by the invasive Chinese sleeper (Perccottus glenii Dybowski, 1877) (Gobiiformes: Odontobutidae) in Latvia and Ukraine. J. Appl. Ichthyol. 2020, 36, 785–794. [Google Scholar] [CrossRef]
- Jorgensen, A.; Sterud, E. The marine pathogenic genotype of Spironucleus barkhanus from farmed salmonids redescribed as Spironucleus salmonicida n. sp. J. Eukaryot. Microbiol. 2006, 53, 531–541. [Google Scholar] [CrossRef]
- Williams, C.F.; Lloyd, D.; Poynton, S.L.; Jorgensen, A.; Millet, C.O.M.; Cable, J. Spironucleus species: Economically-important fish pathogens and enigmatic single-celled eukaryotes. J. Aquac. Res. Dev. 2011, 1, S2. [Google Scholar] [CrossRef]
- Bigun, V.K.; Afanasyev, S.A. Feeding and feeding behavior of invasive fish species in the water bodies of the West Polissya of Ukraine. Hydrobiol. J. 2011, 47, 51–60. [Google Scholar] [CrossRef]
- Marushchak, O.; Nekrasova, O.; Pupins, M.; Tytar, V.; Ceirans, A. The role and importance of the protected areas’ (Emerald Network) development for amphibians and reptiles on the example of Ukraine in the context of various factors’ influence. In Proceedings of the 12th International Scientific and Practical Conference “Environment. Technology. Resources”, Online, 20–22 June 2019; Volume 1, pp. 154–158. [Google Scholar] [CrossRef]
- Kuzmin, S.L. The Amphibians of the Former Soviet Union. Series Faunistica, 12th ed.; Pensoft Publishers: Sofia, Bulgaria, 1999; 538p. [Google Scholar]
- Wielstra, B.E.N.; Babik, W.; Arntzen, J.W. The crested newt Triturus cristatus recolonized temperate Eurasia from an extra-Mediterranean glacial refugium. Biol. J. Linn. Soc. 2015, 114, 574–587. [Google Scholar] [CrossRef]
- Nekrasova, O.; Marushchak, O.; Pupins, M.; Tytar, V.; Georges, J.-Y.; Theissinger, K.; Ceirans, A.; Skute, A. Modeling the influence of invasive fish species Perccottus glenii (Dybowski, 1877) on the distribution of newts in Eastern Europe, exemplified by Lissotriton vulgaris (Linnaeus, 1758) and Triturus cristatus (Laurenti, 1768), using a GIS approach. In Proceedings of the 2nd International Electronic Conference on Diversity (IECD 2022)—New Insights into the Biodiversity of Plants, Animals and Microbes, Basel, Switzerland, 15–31 March 2022; Available online: https://iecd2022.sciforum.net/ (accessed on 6 February 2022).
- Gajjar, J.; Solanki, H. A review: Wetlands as treasure of Earth by providing ecological benefits, threats to wetlands and conservation of wetlands. Int. J. Sci. Res. Sci. Technol. 2021, 8, 136–140. [Google Scholar] [CrossRef]
- Oja, E.; Swartz, L.; Muths, E.; Hossack, B. Amphibian population responses to mitigation: Relative importance of wetland age and design. Ecol. Indic. 2021, 131, 108123. [Google Scholar] [CrossRef]
- García-Muñoz, E.; Gilbert, J.D.; Parra, G.; Guerrero, F. Amphibian diversity as an implement for Mediterranean wetlands conservation. J. Nat. Conserv. 2016, 33, 68–75. [Google Scholar] [CrossRef]
- Kuhns, A.R.; Crawford, J.A.; Phillips, C.A.; Kessler, E.J.; Wright, E.M. Efficacy of created wetlands for amphibian conservation in Illinois. In Proceedings of the 98th ESA Annual Convention 2013, Minneapolis, MN, USA, 4–9 August 2013. [Google Scholar]
- Mulkeen, C.; Gibson-Brabazon, S.; Carlin, C.; Gormally, M. Habitat suitability assessment of constructed wetlands for the smooth newt (Lissotriton vulgaris [Linnaeus, 1758]): A comparison with natural wetlands. Ecol. Eng. 2017, 106, 532–540. [Google Scholar] [CrossRef]
- Vasyliuk, O.; Prylutskyi, O.; Marushchak, O.; Kuzemko, A.; Kutsokon, I.; Nekrasova, O.; Raes, N.; Rusin, M. An extended dataset of occurrences of species listed in Resolution 6 of the Bern Convention from Ukraine. Biodivers. Data J. 2022, 10, e84002. [Google Scholar] [CrossRef]
- Kutsokon, I. The Chinese sleeper (Perccottus glenii Dybowski, 1877) in Ukraine: New data on distribution. J. Appl. Ichthyol. 2017, 33, 1100–1107. [Google Scholar] [CrossRef]
- Kvach, Y.; Kutsokon, Y. The non-indigenous fishes in the fauna of Ukraine: A potentia and actum. BioInvasions Rec. 2017, 6, 269–279. [Google Scholar] [CrossRef]
- Pupina, A.; Pupins, M.; Skute, A.; Pupina, A.; Karklins, A. The distribution of the invasive fish amur sleeper, rotan Perccottus glenii Dybowski, 1877 (Osteichthyes, Odontobutidae), in Latvia. Acta Biol. Univ. Daugavp. 2015, 15, 329–341. [Google Scholar]
- Triturus cristatus (Laurenti, 1768) in GBIF Secretariat (2022). GBIF.org GBIF Occurrence Download. Available online: https://www.gbif.org/zh/species/2431885 (accessed on 6 February 2022).
- Lissotriton vulgaris (Linnaeus, 1758) in GBIF Secretariat (2022). GBIF.org GBIF Occurrence Download. Available online: https://www.gbif.org/species/5218405 (accessed on 6 February 2022).
- Perccottus glenii (Dybowski, 1877) in GBIF Secretariat (2022). GBIF.org GBIF Occurrence Download. Available online: https://www.gbif.org/species/2390064 (accessed on 6 February 2022).
- Osorio-Olvera, L.; Lira-Noriega, A.; Soberón, J.; Peterson, A.T.; Falconi, M.; Contreras-Díaz, R.G. ntbox: An r package with graphical user interface for modelling and evaluating multidimensional ecological niches. Methods Ecol. Evol. 2020, 11, 1199–1206. [Google Scholar] [CrossRef]
- Kriticos, D.J.; Jarošik, V.; Ota, N. Extending the suite of bioclim variables: A proposed registry system and case study using principal components analysis. Methods Ecol. Evol. 2014, 5, 12244. [Google Scholar] [CrossRef]
- Hutchinson, M.; Xu, T.; Houlder, D.; Nix, H.; McMahon, J. ANUCLIM 6.0 User’s Guide; Australian National University, Fenner School of Environment and Society: Canberra, Australia, 2009; 90p. [Google Scholar]
- CliMond. Available online: https://www.climond.org/ (accessed on 27 December 2020).
- Domisch, S.; Amatulli, G.; Jetz, W. Near-global freshwater-specific environmental variables for biodiversity analyses in 1 km resolution. Sci. Data 2015, 2, 150073. [Google Scholar] [CrossRef]
- Nekrasova, O.D.; Tytar, V.M.; Kuybida, V.V. GIS Modeling of Climate Change Vulnerability of Amphibians and Reptiles in Ukraine; Schmalhauzen Institute of Zoology NAS: Kyiv, Ukraine, 2019; 204p. [Google Scholar]
- Phillips, S.J. A brief tutorial on Maxent. ATT Res. 2005, 190, 231–259. [Google Scholar]
- Mas, J.-F.; Soares Filho, B.; Pontius, R.G.; Farfán Gutiérrez, M.; Rodrigues, H. A suite of tools for ROC analysis of spatial models. ISPRS Int. J. Geo-Inf. 2013, 2, 869–887. [Google Scholar] [CrossRef]
- Peterson, A.T.; Papes, M.; Soberón, J. Rethinking receiver operating characteristic analysis applications in ecological niche modeling. Ecol. Model. 2008, 213, 63–72. [Google Scholar] [CrossRef]
- Elith, J.H.; Graham, C.P.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A.; et al. Novel methods improve prediction of species’ distributions from occurrence data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Elith, J.; Phillips, S.J.; Hastie, T.; Dudík, M.; Chee, Y.E.; Yates, C.J. A statistical explanation of MaxEnt for ecologists. Divers. Distrib. 2011, 11, 43–57. [Google Scholar] [CrossRef]
- Zuur, A.F.; Ieno, E.N. A protocol for conducting and presenting results of regression-type analyses. Methods Ecol. Evol. 2016, 7, 636–645. [Google Scholar] [CrossRef]
- Grant, R.A.; Chadwick, E.A.; Halliday, T. The lunar cycle: A cue for amphibian reproductive phenology? Anim. Behav. 2009, 78, 349–357. [Google Scholar] [CrossRef]
- Kusano, T.; Inoue, M. Long-term trends toward earlier breeding of Japanese amphibians. J. Herpetol. 2008, 42, 608–614. [Google Scholar] [CrossRef]
- Klaus, S.P.; Lougheed, S.C. Changes in breeding phenology of eastern Ontario frogs over four decades. Ecol. Evol. 2013, 3, 835–845. [Google Scholar] [CrossRef]
- Litvinchuk, S.N.; Borkin, L.J. Distribution, ecology and conservation status of Danube crested newt, Triturus dobrogicus (Amphibia, Salamandridae) on the territory of Ukraine and Moldova. Vestn. Zool. 2002, 36, 35–44. [Google Scholar]
- Nekrasova, O.; Smirnov, N.; Marushchak, O.; Korshunov, O.; Kotserzhynska, I. Change in the conservation status and location of the newt Triturus cristatus (Amphibia, Salamandridae) in Ukraine. In Materials for the 4th Edition of the Red Book of Ukraine; Fauna/Series: «Conservation Biology in Ukraine»; Ukrainian Nature Conservation Group: Kyiv, Ukraine, 2019; Volume 7, pp. 378–386. [Google Scholar]
- Falaschi, M.; Giachello, S.; Lo Parrino, E.; Muraro, M.; Manenti, R.; Ficetola, G.F. Long-term drivers of persistence and colonization dynamics in spatially structured amphibian populations. Conserv. Biol. 2021, 35, 1530–1539. [Google Scholar] [CrossRef]
- Falaschi, M.; Muraro, M.; Gibertini, C.; Monache, D.D.; Lo Parrino, E.; Faraci, F.; Belluardo, F.; Di Nicola, M.R.; Manenti, R.; Ficetola, G.F. Explaining declines of newt abundance in northern Italy. Freshw. Biol. 2022, 67, 1174–1187. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pupins, M.; Nekrasova, O.; Marushchak, O.; Tytar, V.; Theissinger, K.; Čeirāns, A.; Skute, A.; Georges, J.-Y. Potential Threat of an Invasive Fish Species for Two Native Newts Inhabiting Wetlands of Europe Vulnerable to Climate Change. Diversity 2023, 15, 201. https://doi.org/10.3390/d15020201
Pupins M, Nekrasova O, Marushchak O, Tytar V, Theissinger K, Čeirāns A, Skute A, Georges J-Y. Potential Threat of an Invasive Fish Species for Two Native Newts Inhabiting Wetlands of Europe Vulnerable to Climate Change. Diversity. 2023; 15(2):201. https://doi.org/10.3390/d15020201
Chicago/Turabian StylePupins, Mihails, Oksana Nekrasova, Oleksii Marushchak, Volodymyr Tytar, Kathrin Theissinger, Andris Čeirāns, Arturs Skute, and Jean-Yves Georges. 2023. "Potential Threat of an Invasive Fish Species for Two Native Newts Inhabiting Wetlands of Europe Vulnerable to Climate Change" Diversity 15, no. 2: 201. https://doi.org/10.3390/d15020201
APA StylePupins, M., Nekrasova, O., Marushchak, O., Tytar, V., Theissinger, K., Čeirāns, A., Skute, A., & Georges, J.-Y. (2023). Potential Threat of an Invasive Fish Species for Two Native Newts Inhabiting Wetlands of Europe Vulnerable to Climate Change. Diversity, 15(2), 201. https://doi.org/10.3390/d15020201






