Bioclimatic Preferences of the Great Bustard in a Steppe Region
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Study Species
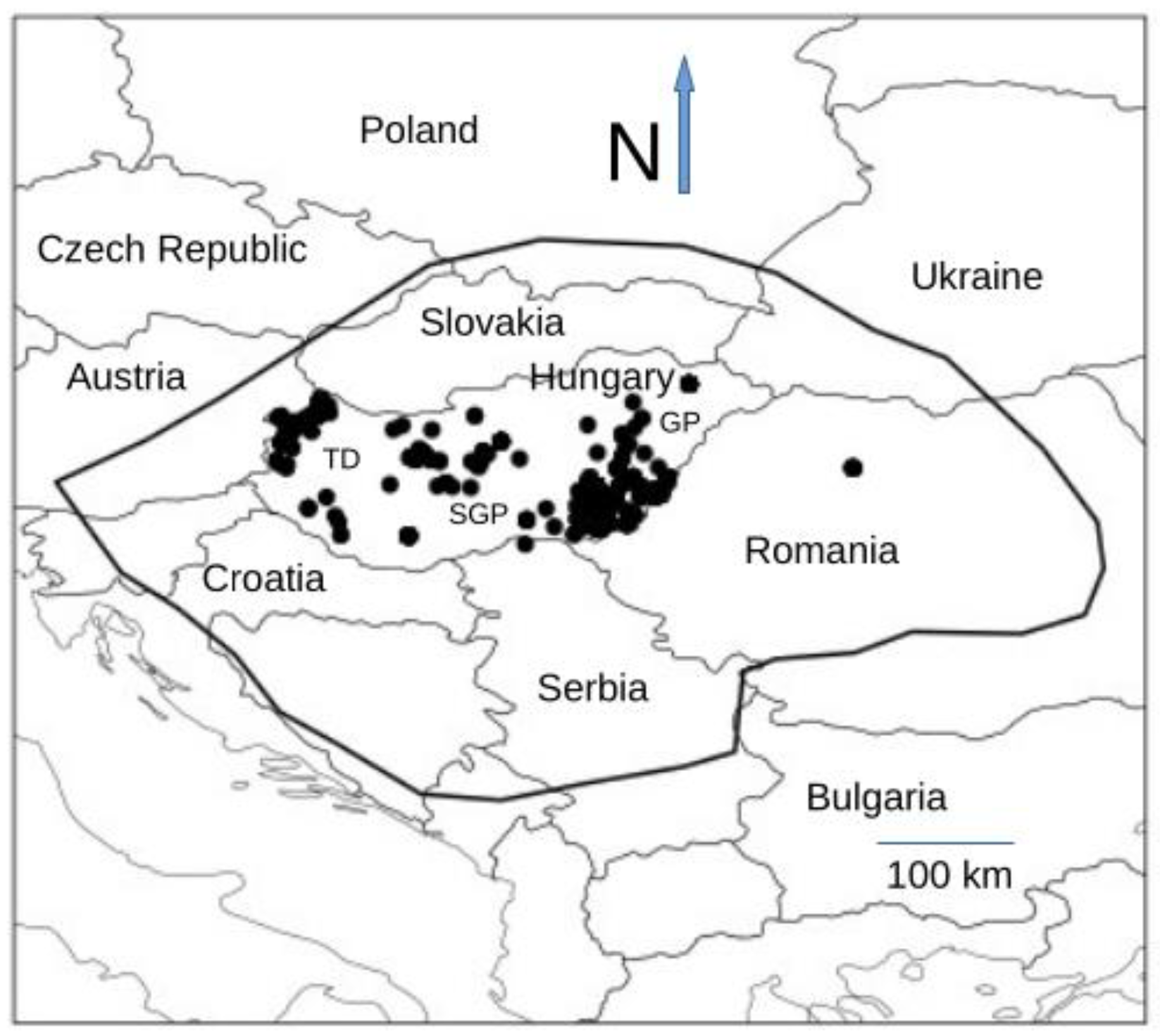
2.3. Occurrence Records
2.4. Species Distribution Modeling
3. Results
3.1. Occurrence Data
3.2. Species Distribution Model
3.3. Historical Distribution Range
3.4. Future Distribution Range
4. Discussion
Bioclimatic Preferences
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. References for the Historical Occurrences of the Great Bustard within the Carpathian Region
Appendix B. Occurrence records of the Great Bustard between 1828–2014 within the Carpathian Basin
Appendix C. Pearson’s Correlation Coefficients among Rasters of Bioclimatic Variables
| bio_1 | bio_10 | bio_11 | bio_12 | bio_13 | bio_14 | bio_15 | bio_16 | bio_17 | bio_18 | bio_19 | bio_2 | bio_3 | bio_4 | bio_5 | bio_6 | bio_7 | bio_8 | bio_9 | |
| bio_1 | 1.000 | 0.975 | 0.929 | −0.332 | −0.506 | −0.189 | −0.424 | −0.527 | −0.172 | −0.798 | 0.027 | 0.463 | 0.296 | 0.209 | 0.969 | 0.857 | 0.333 | 0.401 | 0.580 |
| bio_10 | 0.975 | 1.000 | 0.829 | −0.463 | −0.601 | −0.321 | −0.324 | −0.623 | −0.310 | −0.815 | −0.128 | 0.530 | 0.183 | 0.416 | 0.991 | 0.739 | 0.511 | 0.517 | 0.471 |
| bio_11 | 0.929 | 0.829 | 1.000 | −0.049 | −0.268 | 0.073 | −0.544 | −0.280 | 0.100 | −0.674 | 0.310 | 0.263 | 0.424 | −0.163 | 0.825 | 0.976 | −0.022 | 0.144 | 0.719 |
| bio_12 | −0.332 | −0.463 | −0.049 | 1.000 | 0.897 | 0.906 | −0.304 | 0.906 | 0.924 | 0.629 | 0.881 | −0.479 | 0.137 | −0.740 | −0.453 | 0.049 | −0.712 | −0.761 | 0.241 |
| bio_13 | −0.506 | −0.601 | −0.268 | 0.897 | 1.000 | 0.687 | 0.095 | 0.988 | 0.709 | 0.751 | 0.707 | −0.521 | 0.002 | −0.622 | −0.611 | −0.185 | −0.649 | −0.730 | 0.104 |
| bio_14 | −0.189 | −0.321 | 0.073 | 0.906 | 0.687 | 1.000 | −0.599 | 0.684 | 0.994 | 0.419 | 0.886 | −0.384 | 0.192 | −0.688 | −0.298 | 0.174 | −0.642 | −0.725 | 0.288 |
| bio_15 | −0.424 | −0.324 | −0.544 | −0.304 | 0.095 | −0.599 | 1.000 | 0.109 | −0.592 | 0.279 | −0.528 | −0.090 | −0.355 | 0.321 | −0.371 | −0.584 | 0.189 | 0.122 | −0.405 |
| bio_16 | −0.527 | −0.623 | −0.280 | 0.906 | 0.988 | 0.684 | 0.109 | 1.000 | 0.708 | 0.775 | 0.695 | −0.536 | 0.001 | −0.641 | −0.631 | −0.194 | −0.667 | −0.727 | 0.091 |
| bio_17 | −0.172 | −0.310 | 0.100 | 0.924 | 0.709 | 0.994 | −0.592 | 0.708 | 1.000 | 0.415 | 0.915 | −0.394 | 0.204 | −0.713 | −0.289 | 0.201 | −0.663 | −0.731 | 0.332 |
| bio_18 | −0.798 | −0.815 | −0.674 | 0.629 | 0.751 | 0.419 | 0.279 | 0.775 | 0.415 | 1.000 | 0.209 | −0.376 | −0.090 | −0.350 | −0.801 | −0.612 | −0.395 | −0.362 | −0.458 |
| bio_19 | 0.027 | −0.128 | 0.310 | 0.881 | 0.707 | 0.886 | −0.528 | 0.695 | 0.915 | 0.209 | 1.000 | −0.403 | 0.211 | −0.728 | −0.126 | 0.401 | −0.676 | −0.754 | 0.573 |
| bio_2 | 0.463 | 0.530 | 0.263 | −0.479 | −0.521 | −0.384 | −0.090 | −0.536 | −0.394 | −0.376 | −0.403 | 1.000 | 0.592 | 0.488 | 0.606 | 0.090 | 0.760 | 0.573 | −0.062 |
| bio_3 | 0.296 | 0.183 | 0.424 | 0.137 | 0.002 | 0.192 | −0.355 | 0.001 | 0.204 | −0.090 | 0.211 | 0.592 | 1.000 | −0.383 | 0.269 | 0.361 | −0.060 | 0.011 | 0.225 |
| bio_4 | 0.209 | 0.416 | −0.163 | −0.740 | −0.622 | −0.688 | 0.321 | −0.641 | −0.713 | −0.350 | −0.728 | 0.488 | −0.383 | 1.000 | 0.404 | −0.282 | 0.929 | 0.671 | −0.330 |
| bio_5 | 0.969 | 0.991 | 0.825 | −0.453 | −0.611 | −0.298 | −0.371 | −0.631 | −0.289 | −0.801 | −0.126 | 0.606 | 0.269 | 0.404 | 1.000 | 0.730 | 0.535 | 0.528 | 0.451 |
| bio_6 | 0.857 | 0.739 | 0.976 | 0.049 | −0.185 | 0.174 | −0.584 | −0.194 | 0.201 | −0.612 | 0.401 | 0.090 | 0.361 | −0.282 | 0.730 | 1.000 | −0.188 | 0.044 | 0.733 |
| bio_7 | 0.333 | 0.511 | −0.022 | −0.712 | −0.649 | −0.642 | 0.189 | −0.667 | −0.663 | −0.395 | −0.676 | 0.760 | −0.060 | 0.929 | 0.535 | −0.188 | 1.000 | 0.705 | −0.258 |
| bio_8 | 0.401 | 0.517 | 0.144 | −0.761 | −0.730 | −0.725 | 0.122 | −0.727 | −0.731 | −0.362 | −0.754 | 0.573 | 0.011 | 0.671 | 0.528 | 0.044 | 0.705 | 1.000 | −0.278 |
| bio_9 | 0.580 | 0.471 | 0.719 | 0.241 | 0.104 | 0.288 | −0.405 | 0.091 | 0.332 | −0.458 | 0.573 | −0.062 | 0.225 | −0.330 | 0.451 | 0.733 | −0.258 | −0.278 | 1.000 |
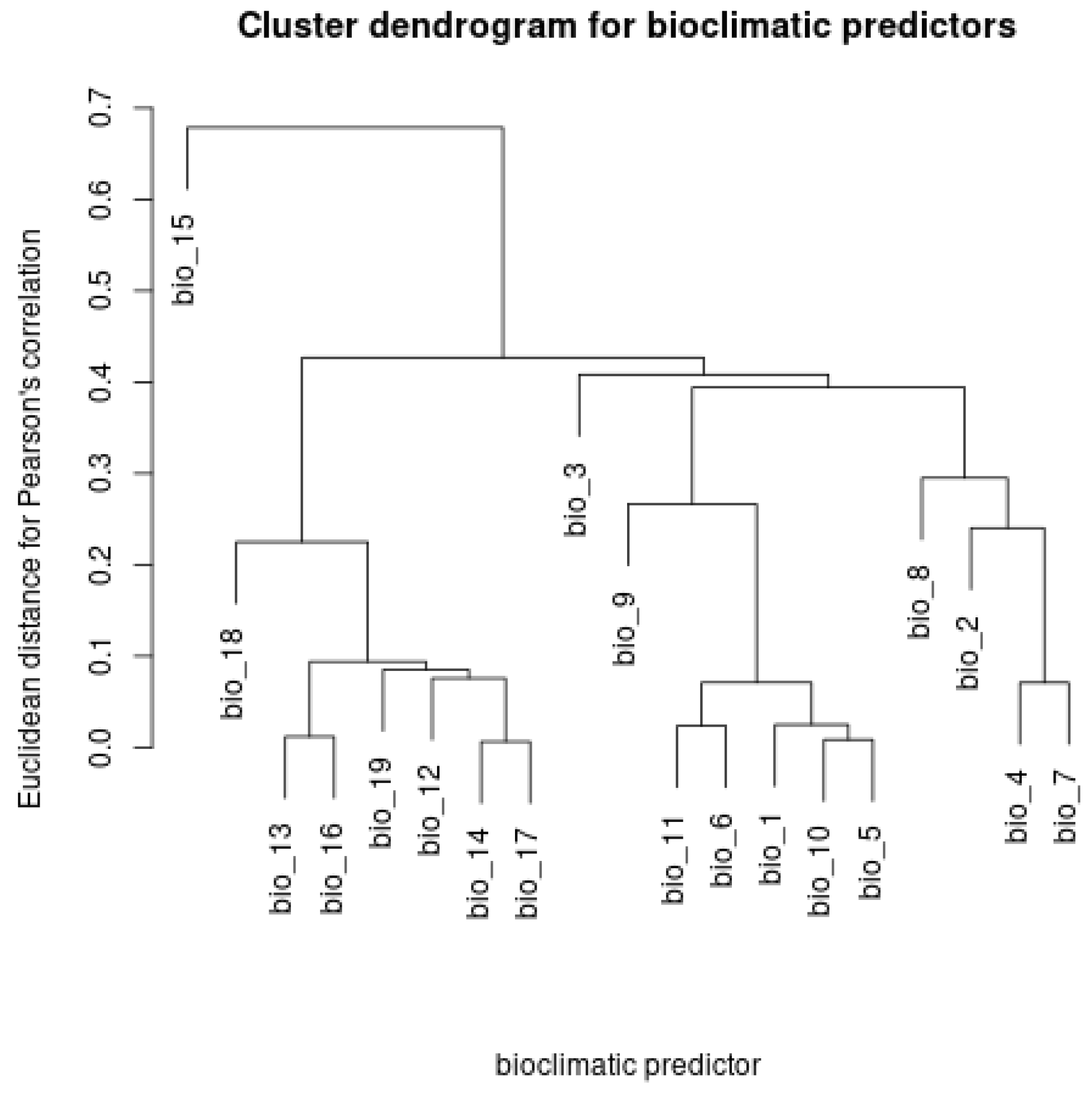
Appendix D. Dendrogram of the Hierarchical Clustering for the Pearson’s Correlation Coefficients among Rasters of Bioclimatic Variables
Appendix E. Response Curves of the Selected Bioclimatic Variables

References
- Cahill, A.E.; Aiello-Lammens, M.E.; Fisher-Reid, M.C.; Hua, X.; Karanewsky, C.J.; Yeong Ryu, H.; Sbeglia, G.C.; Spagnolo, F.; Waldron, J.B.; Warsi, O.; et al. How does climate change cause extinction? Proc. R. Soc. B Biol. Sci. 2013, 280, 20121890. [Google Scholar] [CrossRef] [PubMed]
- Moritz, C.; Agudo, R. The future of species under climate change: Resilience or decline? Science 2013, 341, 504–508. [Google Scholar] [CrossRef] [PubMed]
- Lovejoy, T.E.; Hannah, L.J. (Eds.) Biodiversity and Climate Change; Yale University Press: New Haven, United States, 2019. [Google Scholar]
- Jackson, S.T.; Sax, D.F. Balancing biodiversity in a changing environment: Extinction debt, immigration credit and species turnover. Trends Ecol. Evol. 2019, 2010, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.J.; Smith, A.B.; Dekker, S.C.; Eppinga, M.B.; Leitão, P.J.; Moreno-Mateos, D.; Morueta-Holme, N.; Ruggeri, M. The role of land use and land cover change in climate change vulnerability assessments of biodiversity: A systematic review. Landsc. Ecol. 2021, 36, 3367–3382. [Google Scholar] [CrossRef]
- Dawson, T.P.; Jackson, S.T.; House, J.I.; Prentice, I.C.; Mace, G.M. Beyond Predictions: Biodiversity Conservation in a Changing Climate. Science 2011, 2011, 53–58. [Google Scholar] [CrossRef]
- Santangeli, A.; Lehikoinen, A.; Bock, A.; Peltonen-Sainio, P.; Jauhiainen, L.; Girardello, M.; Valkama, J. Stronger response of farmland birds than farmers to climate change leads to the emergence of an ecological trap. Biol. Conserv. 2018, 217, 166–172. [Google Scholar] [CrossRef]
- Estrada, A.; Delgado, M.P.; Arroyo, B.; Traba, J.; Morales, M.B. Forecasting large-scale habitat suitability of European bustards under climate change: The role of environmental and geographic variables. PLoS ONE 2016, 11, 0149810. [Google Scholar] [CrossRef]
- Kubelka, V.; Šálek, M.; Tomkovich, P.; Végvári, Z.; Freckleton, R.P.; Székely, T. Global pattern of nest predation is disrupted by climate change in shorebirds. Science 2018, 362, 680–683. [Google Scholar] [CrossRef]
- Araújo, M.B.; Pearson, R.G.; Rahbek, C. Equilibrium of species’ distributions with climate. Ecography 2005, 28, 693–695. [Google Scholar] [CrossRef]
- Porfirio, L.L.; Harris, R.M.; Lefroy, E.C.; Hugh, S.; Gould, S.F.; Lee, G.; Bindoff, N.L.; Mackey, B. Improving the use of species distribution models in conservation planning and management under climate change. PLoS ONE 2014, 9, e113749. [Google Scholar] [CrossRef]
- Alonso, J.C.; Palacín, C. The world status and population trends of the Great Bustard (Otis tarda). Chin. Birds 2010, 1, 141–147. [Google Scholar] [CrossRef]
- Alonso, J.C. The Great Bustard: Past, present and future of a globally threatened species. Ornis Hung. 2014, 22, 1–13. [Google Scholar] [CrossRef]
- Mi, C.; Huettmann, F.; Sun, R.; Guo, Y. Combining occurrence and abundance distribution models for the conservation of the Great Bustard. PeerJ 2017, 5, 4160. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, S.; Varga, K.; Kosztyi, B.; Lontay, L.; Déri, E.; Török, P.; Tóthmérész, B. Grassland restoration to conserve landscape-level biodiversity: A synthesis of early results from a large-scale project. Appl. Veg. Sci. 2012, 15, 264–276. [Google Scholar] [CrossRef]
- Deák, B.; Valkó, O.; Török, P.; Tóthmérész, B. Factors threatening grassland specialist plants-A multi-proxy study on the vegetation of isolated grasslands. Biol. Conserv. 2016, 204, 255–262. [Google Scholar] [CrossRef]
- Faragó, S.; Végvári, Z. Túzok. In Magyarország Madáratlasza; Agrárminisztérium; Szép, T., Csörgő, T., Halmos, G., Lovászi, P., Nagy, K., Schmidt, A., Eds.; Magyar Madártani és Természetvédelmi Egyesület: Budapest, Hungary, 2021. [Google Scholar]
- Szabó, K.; Bozsó, M.; Boros, E.; Pénzes, Z. A Túzok Hazai Populációinak Genetikai Változatossága [Genetic Diversity of the Hungarian Great Bustard Populations]. A Kárpát-Medence Állatvilágának Kialakulása [The Evolution of the Fauna in Carpathian Basin]; Magyar Természettudományi Múzeum: Budapest, Hungary, 2007; pp. 297–302. [Google Scholar]
- Larrosa, C.; Carrasco, L.R.; Milner-Gulland, E.J. Unintended feedbacks: Challenges and opportunities for improving conservation effectiveness. Conserv. Lett. 2016, 9, 316–326. [Google Scholar] [CrossRef]
- Loiselle, B.A.; Howell, C.A.; Graham, C.H.; Goerck, J.M.; Brooks, T.; Smith, K.G.; Williams, P.H. Avoiding pitfalls of using species distribution models in conservation planning. Conserv. Biol. 2003, 17, 1591–1600. [Google Scholar] [CrossRef]
- Villero, D.; Pla, M.; Camps, D.; Ruiz-Olmo, J.; Brotons, L. Integrating species distribution modelling into decision-making to inform conservation actions. Biodivers. Conserv. 2017, 26, 251–271. [Google Scholar] [CrossRef]
- Varga, Z. Geographical patterns of biological diversity in the Palaearctic Region and the Carpathian Basin. Acta Zool. Acad. Sci. Hung. 1995, 41, 71–92. [Google Scholar]
- Billerman, B.; Keeney, K.; Rodewald, P.G.; Schulenberg, T.S. (Eds.) Birds of the World; Cornell Laboratory of Ornithology: Ithaca, NY, USA, 2022. [Google Scholar]
- Pinto, M.; Rocha, P.; Moreira, F. Long-term trends in great bustard (Otis tarda) populations in Portugal suggest concentration in single high quality area. Biol. Conserv. 2005, 124, 415–423. [Google Scholar] [CrossRef]
- Kessler, M. Status of the Western Great Bustard Otis tarda tarda in Asia, and its significance to an updated estimate of the global population of Great Bustards. Sandgrouse Great Bustard Spec. Issue 2022, 44, 6. [Google Scholar]
- Alonso, J.C.; Magaña, M.; Palacín, C.; Martín, C.A. Correlates of male mating success in great bustard leks: The effects of age, weight, and display effort. Behav. Ecol. Sociobiol. 2010, 64, 1589–1600. [Google Scholar] [CrossRef]
- Hijmans, R.J. Raster: Geographic Data Analysis and Modeling. 2021, R package Version 3.5-9. Available online: https://CRAN.R-project.org/package=raster (accessed on 17 November 2022).
- Guarino, M.V.; Sime, L.; Schroeder, D.; Ridley, J. The first 250 years of the Heinrich 11 iceberg discharge: Last Interglacial HadGEM3-GC3. 1 simulations for CMIP6-PMIP4. Clim. Past Discuss. 2022, 1–26. [Google Scholar]
- Phillips, S.J.; Dudík, M. Modeling of species distributions with MaxEnt: New extensions and a comprehensive evaluation. Ecography 2008, 31, 161–175. [Google Scholar] [CrossRef]
- Philips, S.; Dudik, M.; Schapire, R. Maxent Software, Version 3.3. 3e; Global Biodiversity Information Facility: Copenhagen, Denmark, 2010. [Google Scholar]
- Kramer-Schadt, S.; Niedballa, J.; Pilgrim, J.D.; Schröder, B.; Lindenborn, J.; Reinfelder, V.; Stillfried, M.; Heckmann, I.; Scharf, A.K.; Augeri, D.M.; et al. The importance of correcting for sampling bias in MaxEnt species distribution models. Divers. Distrib. 2013, 19, 1366–1379. [Google Scholar] [CrossRef]
- Sterbetz, I. A túzok (Otis t.tarda L.) környezete Magyarországon. Aquila 1976, 1976, 53–73. [Google Scholar]
- Mateo-Tomas, P.; Olea, P.P. Anticipating knowledge to inform species management: Predicting spatially explicit habitat suitability of a colonial vulture spreading its range. PLoS ONE 2010, 5, e12374. [Google Scholar] [CrossRef]
- Shcheglovitova, M.; Anderson, R.P. Estimating optimal complexity for ecological niche models: A jackknife approach for species with small sample sizes. Ecol. Model. 2013, 269, 9–17. [Google Scholar] [CrossRef]
- Phillips, S.J.; Elith, J. POC plots: Calibrating species distribution models with presence-only data. Ecology 2010, 91, 2476–2484. [Google Scholar] [CrossRef]
- Fielding, A.H.; Bell, J.F. A review of methods for the assessment of prediction errors in conservation presence/absence models. Environ. Conserv. 1997, 24, 38–49. [Google Scholar] [CrossRef]
- Radosavljevic, A.; Anderson, R.P. Making better Maxent models of species distributions: Complexity, overfitting and evaluation. J. Biogeogr. 2014, 41, 629–643. [Google Scholar] [CrossRef]
- Roberts, D.R.; Bahn, V.; Ciuti, S.; Boyce, M.S.; Elith, J.; Guillera-Arroita, G.; Hauenstein, S.; Lahoz-Monfort, J.J.; Schröder, B.; Thuiller, W.; et al. Cross-validation strategies for data with temporal, spatial, hierarchical, or phylogenetic structure. Ecography 2017, 40, 913–929. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 17 November 2022).
- Wickham, H.; Bryan, J. Readxl: Read Excel Files. R Package Version 1.3.1. 2019. Available online: https://CRAN.R-project.org/package=readxl (accessed on 17 November 2022).
- Bivand, R.; Lewin-Koh, N. Maptools: Tools for Handling Spatial Objects. R Package Version 1.1-1. 2021. Available online: https://CRAN.R-project.org/package=maptools (accessed on 17 November 2022).
- Bivand, R.; Rundel, C. Rgeos: Interface to Geometry Engine—Open Source (‘GEOS’). R Package Version 0.5-5. 2020. Available online: https://CRAN.R-project.org/package=rgeos (accessed on 17 November 2022).
- Roger, B.; Tim, K.; Barry, R. Rgdal: Bindings for the ‘Geospatial’ Data Abstraction Library. R Package Version 1.5-32. 2022. Available online: https://CRAN.R-project.org/package=rgdal (accessed on 17 November 2022).
- Urbanek, S. rJava: Low-Level R to Java Interface. R Package Version 1.0-5. 2021. Available online: https://CRAN.R-project.org/package=rJava (accessed on 17 November 2022).
- Hijmans, R.J.; Phillips, S.; Leathwick, J.; Elith, J. dismo: Species Distribution Modeling. R Package Version 1.3-3. 2020. Available online: https://CRAN.R-project.org/package=dismo (accessed on 17 November 2022).
- Delgado, M.P.; Traba, J.; Morales, M.B. Climate niche constraints in two coexisting steppe birds: The little and the great bustards. Ardeola 2011, 58, 223–238. [Google Scholar] [CrossRef]
- Rakonczay, Z.; Vörös Könyv, A. Magyarországon Kipusztult és Veszélyeztetett Növény- és állat-Fajták; Akadémiai Kiadó: Budapest, Hungary, 1990. [Google Scholar]
- Juhász, L. A magyar puszta ékessége: A túzok. Díszmadár Mag. 2012, 19, 24–29. [Google Scholar]
- Webb, S.L.; Olson, C.V.; Dzialak, M.R.; Harju, S.M.; Winstead, J.B.; Lockman, D. Landscape features and weather influence nest survival of a ground-nesting bird of conservation concern, the greater sage-grouse, in human-altered environments. Ecol. Process. 2012, 1, 1–15. [Google Scholar] [CrossRef]
- Tanner, E.P.; Elmore, R.D.; Fuhlendorf, S.D.; Davis, C.A.; Dahlgren, D.K.; Orange, J.P. Extreme climatic events constrain space use and survival of a ground-nesting bird. Glob. Change Biol. 2017, 23, 1832–1846. [Google Scholar] [CrossRef]
- Vincze, O.; Kosztolányi, A.; Barta, Z.; Küpper, C.; Alrashidi, M.; Amat, J.A.; Argüelles Ticó, A.; Burns, F.; Cavitt, J.; Conway, W.C.; et al. Parental cooperation in a changing climate: Fluctuating environments predict shifts in care division. Glob. Ecol. Biogeogr. 2017, 26, 347–358. [Google Scholar] [CrossRef]
- Marcer, A.; Sáez, L.; Molowny-Horas, R.; Pons, X.; Pino, J. Using species distribution modelling to disentangle realised versus potential distributions for rare species conservation. Biol. Conserv. 2013, 166, 221–230. [Google Scholar] [CrossRef]

| Decade | Number of Locations |
|---|---|
| 1829–1847 | 3 |
| 1850–1859 | 1 |
| 1860–1869 | 2 |
| 1870–1879 | 3 |
| 1880–1889 | 2 |
| 1890–1899 | 8 |
| 1900–1909 | 1 |
| 1910–1919 | 11 |
| 1920–1929 | 31 |
| 1930–1939 | 58 |
| 1940–1949 | 71 |
| 1950–1959 | 45 |
| 1960–1969 | 100 |
| 1970–1979 | 76 |
| 1980–1989 | 73 |
| 1990–1999 | 20 |
| 2000–2009 | 18 |
| 2010–2014 | 3 |
| AUC | Bioclimatic Variable | Contribution |
|---|---|---|
| (a) Complete set | ||
| AUC = 0.9511 | Mean annual temperature | 0.8043 |
| Annual precipitation | 1.1814 | |
| Precipitation seasonality | 0.6187 | |
| Precipitation of warmest quarter | 0.7611 | |
| Mean diurnal range of temperature | 0.4039 | |
| Isothermality | 0.1899 | |
| Temperature seasonality | 0.6646 | |
| Mean temperature of wettest quarter | 0.9848 | |
| Mean temperature of driest quarter | 0.8748 | |
| (b) Selected subset | ||
| AUC = 0.9375 | Mean annual temperature | 0.8186 |
| Annual precipitation | 1.1317 | |
| Mean temperature of wettest quarter | 0.9472 | |
| Mean temperature of driest quarter | 0.8879 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ottó, B.; Végvári, Z. Bioclimatic Preferences of the Great Bustard in a Steppe Region. Diversity 2022, 14, 1138. https://doi.org/10.3390/d14121138
Ottó B, Végvári Z. Bioclimatic Preferences of the Great Bustard in a Steppe Region. Diversity. 2022; 14(12):1138. https://doi.org/10.3390/d14121138
Chicago/Turabian StyleOttó, Beatrix, and Zsolt Végvári. 2022. "Bioclimatic Preferences of the Great Bustard in a Steppe Region" Diversity 14, no. 12: 1138. https://doi.org/10.3390/d14121138
APA StyleOttó, B., & Végvári, Z. (2022). Bioclimatic Preferences of the Great Bustard in a Steppe Region. Diversity, 14(12), 1138. https://doi.org/10.3390/d14121138





