Trait-Based Investigation Reveals Patterns of Community Response to Nutrient Enrichment in Coastal Mesic Grassland
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Plot Establishment and Nutrient Application
2.3. Biomass and Species Composition Sampling
2.4. Functional Trait Selection and Sampling
2.5. Statistical Analysis
3. Results
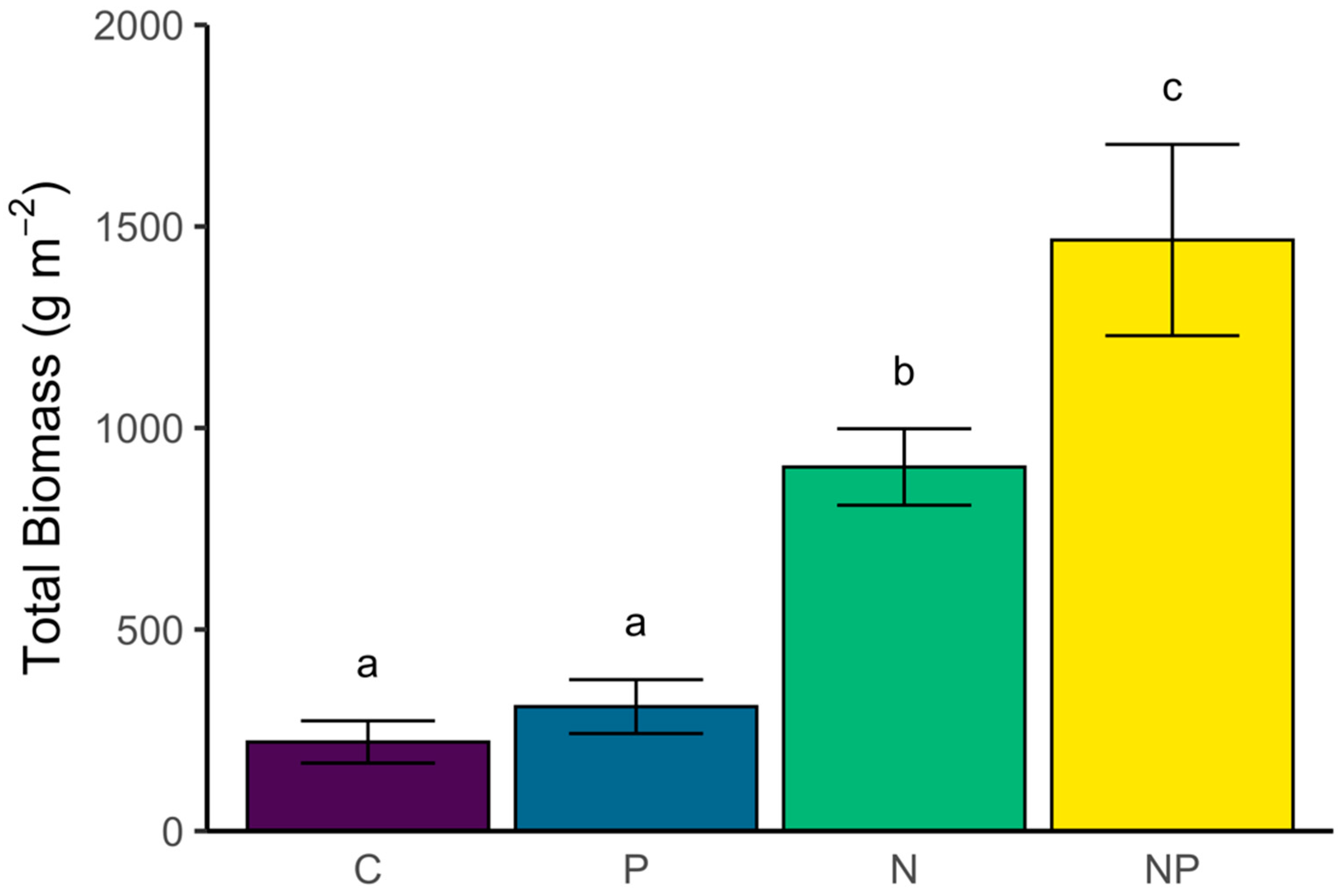
3.1. Biomass Response to Nutrients
3.2. Community-Level Trait Response
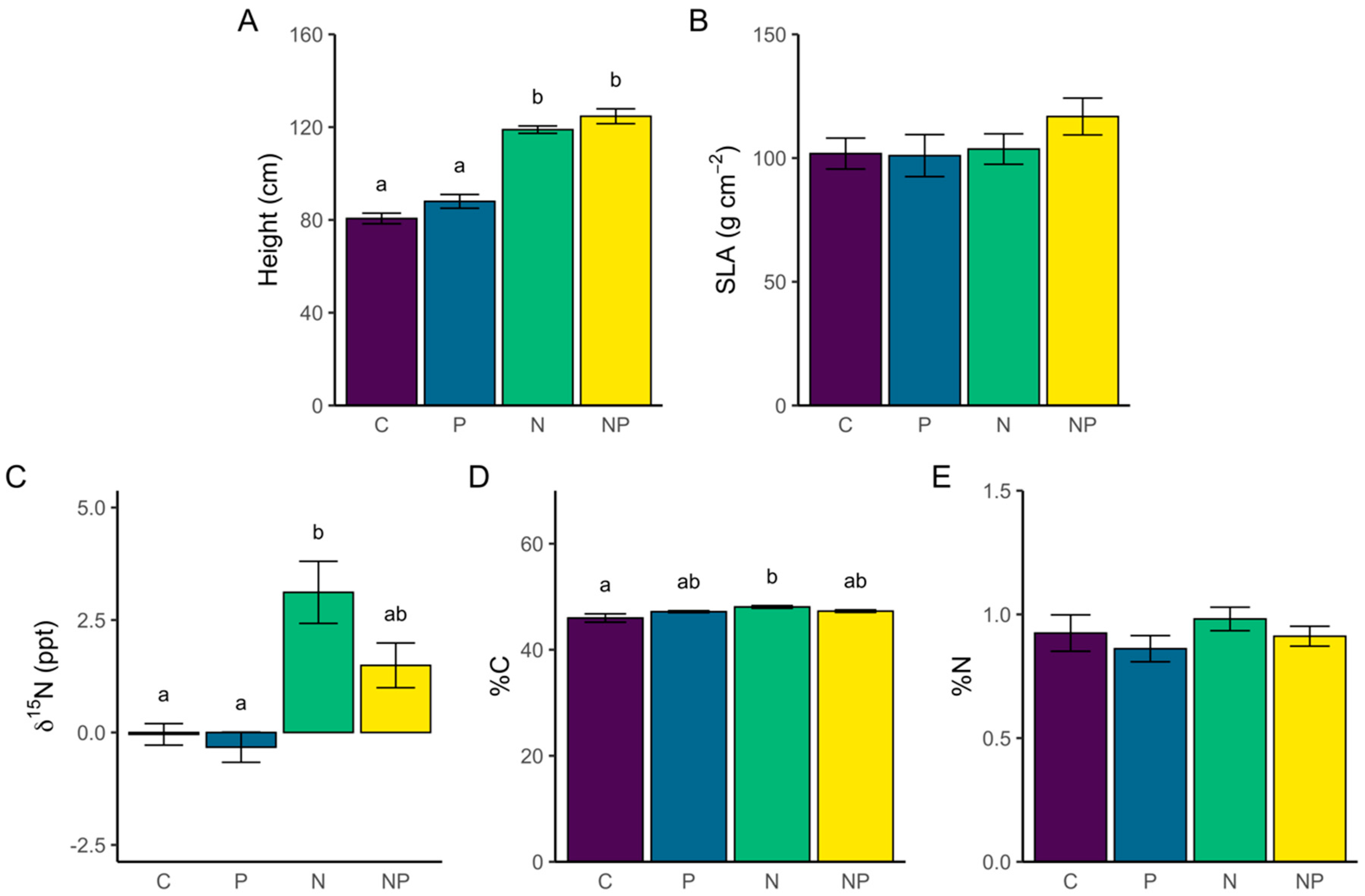
3.2.1. Individual Trait Differences Among Treatments
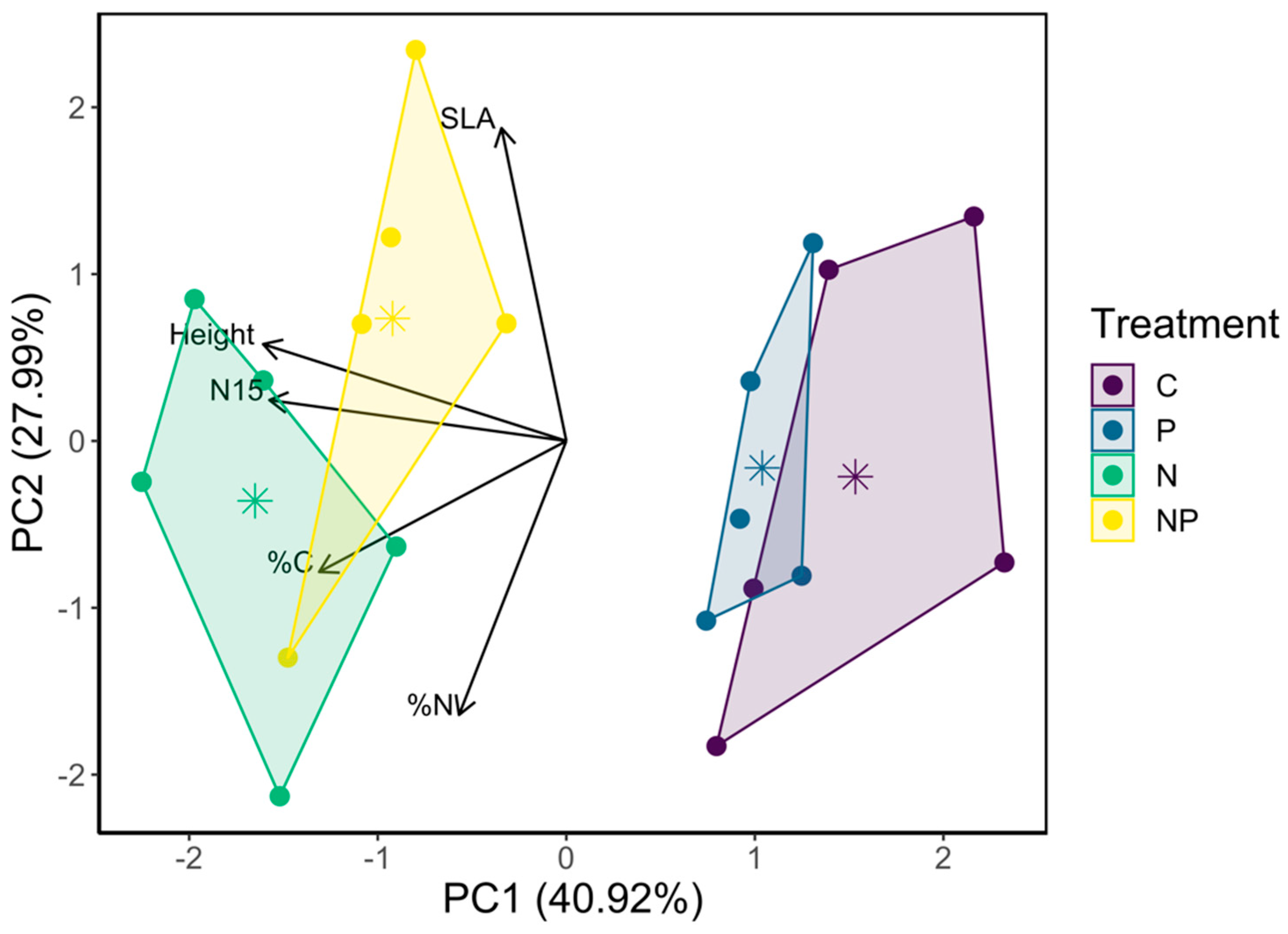
3.2.2. Trait, Species, and Lifeform Composition
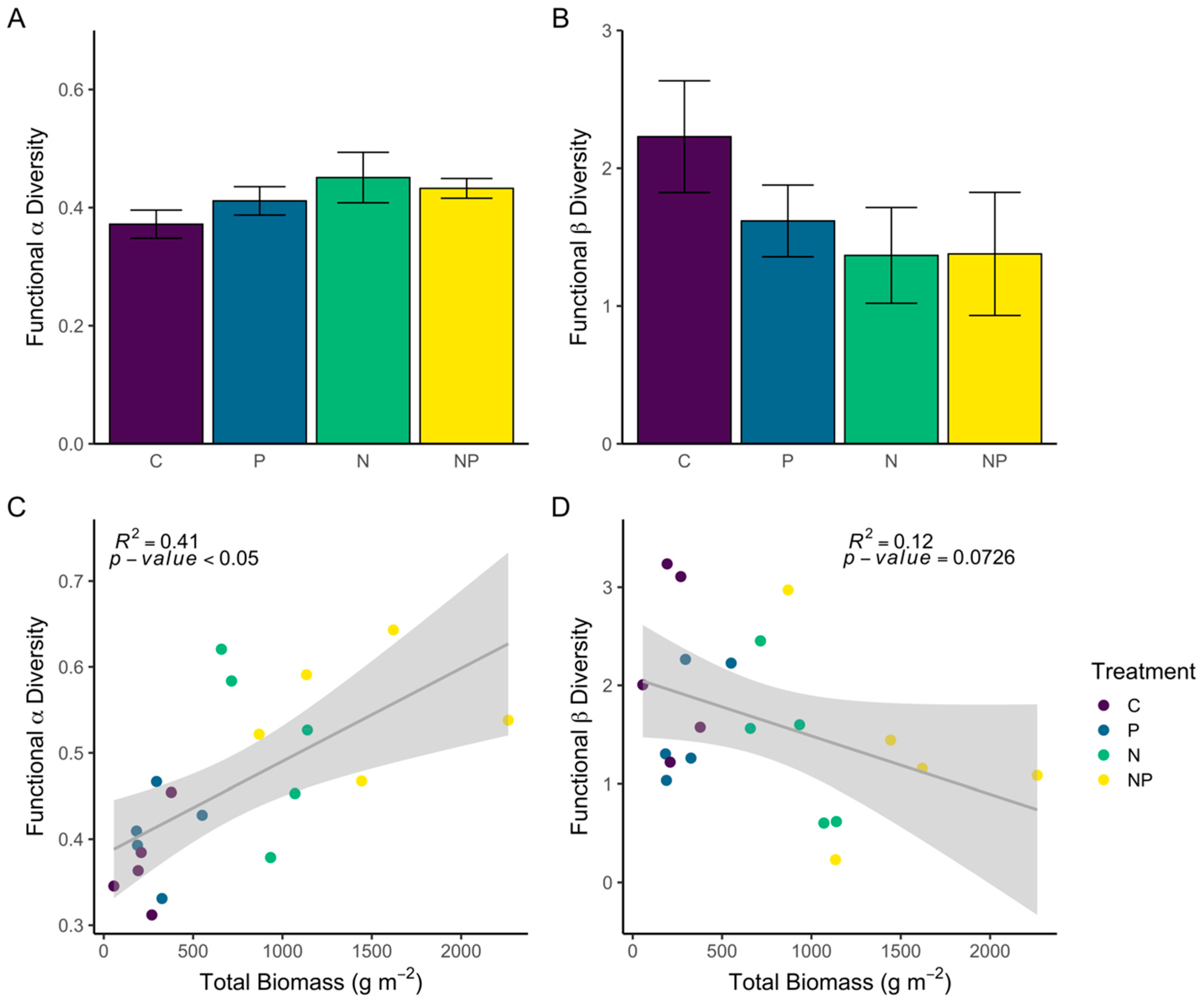
3.2.3. Trait-Based Alpha and Beta Diversity
4. Discussion
Conclusions and Implications for Coastal Systems
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fay, P.A.; Prober, S.M.; Harpole, W.S.; Knops, J.M.H.; Bakker, J.D.; Borer, E.T.; Lind, E.M.; MacDougall, A.S.; Seabloom, E.W.; Wragg, P.D.; et al. Grassland productivity limited by multiple nutrients. Nat. Plants 2015, 1. [Google Scholar] [CrossRef] [PubMed]
- Harpole, W.S.; Sullivan, L.L.; Lind, E.M.; Firn, J.; Adler, P.B.; Borer, E.T.; Chase, J.; Fay, P.A.; Hautier, Y.; Hillebrand, H.; et al. Addition of multiple limiting resources reduces grassland diversity. Nature 2016, 537, 93–96. [Google Scholar] [CrossRef] [PubMed]
- La Pierre, K.J.; Smith, M.D. Functional trait expression of grassland species shift with short- and long-term nutrient additions. Plant. Ecol. 2015, 216, 307–318. [Google Scholar] [CrossRef]
- Borer, E.T.; Seabloom, E.W.; Gruner, D.S.; Harpole, W.S.; Hillebrand, H. Herbivores and nutrients control grassland plant diversity via light limitation. Nature 2014, 508, 517–520. [Google Scholar] [CrossRef]
- Mahowald, N.; Jickells, T.D.; Baker, A.R.; Artaxo, P.; Benitez-Nelson, C.R.; Bergametti, G.; Bond, T.C.; Chen, Y.; Cohen, D.D.; Herut, B.; et al. Global distribution of atmospheric phosphorus sources, concentrations and deposition rates, and anthropogenic impacts. Glob. Biogeochem. Cycles 2008, 22, 1–19. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Aber, J.D.; Howarth, R.H.; Likens, G.E.; Matson, P.A.; Schindler, D.W.; Schlesinger, W.H.; Tilman, D.G. Human alteration of the global nitrogen cycle: Source and consequences. Ecol. Appl. 1997, 7, 737–750. [Google Scholar] [CrossRef]
- Galloway, J.N.; Aber, J.D.; Erisman, J.W.; Seitzinger, S.P.; Howarth, R.W.; Cowling, E.B.; Cosby, B.J. The Nitrogen Cascade. Bioscience 2003, 53, 341–356. [Google Scholar] [CrossRef]
- Rockström, J.; Steffen, W.; Noone, K.; Persson, Å.; Chapin, F.S.; Lambin, E.; Lenton, T.M.; Scheffer, M.; Folke, C.; Schellnuber, H.J.; et al. Planetary Boundaries: Exploring the Safe Operating Space for Humanity. Ecol. Soc. 2009, 14, 32. [Google Scholar] [CrossRef]
- Elser, J.J.; Bracken, M.E.S.; Cleland, E.E.; Gruner, D.S.; Harpole, W.S.; Hillebrand, H.; Ngai, J.T.; Seabloom, E.W.; Shurin, J.B.; Smith, J.E. Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol. Lett. 2007, 10, 1135–1142. [Google Scholar] [CrossRef]
- Harpole, W.S.; Sullivan, L.L.; Lind, E.M.; Firn, J.; Adler, P.B.; Borer, E.T.; Chase, J.; Fay, P.A.; Hautier, Y.; Hillebrand, H.; et al. Out of the shadows: Multiple nutrient limitations drive relationships among biomass, light and plant diversity. Funct. Ecol. 2017, 31, 1839–1846. [Google Scholar] [CrossRef]
- Wilson, S.D.; Tilman, D. Interactive effects of fertilization and disturbance on community structure and resource availability in an old-field plant community. Oecologia 1991, 88, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Clark, C.M.; Cleland, E.E.; Collins, S.L.; Fargione, J.E.; Gough, L.; Gross, K.L.; Pennings, S.C.; Suding, K.N.; Grace, J.B. Environmental and plant community determinants of species loss following nitrogen enrichment. Ecol. Lett. 2007, 10, 596–607. [Google Scholar] [CrossRef] [PubMed]
- Hautier, Y.; Niklaus, P.A.; Hector, A. Competition for light causes plant biodiversity loss after eutrophication. Science 2009, 324, 636–638. [Google Scholar] [CrossRef] [PubMed]
- Demalach, N.; Kadmon, R. Light competition explains diversity decline better than niche dimensionality. Funct. Ecol. 2017, 31, 1834–1838. [Google Scholar] [CrossRef]
- Carson, W.P.; Barrett, G.W. Succession in Old-Field Plant Communities: Effects of Contrasting Types of Nutrient Enrichment. Ecology 1988, 69, 984–994. [Google Scholar] [CrossRef]
- Clark, C.M.; Tilman, D. Loss of plant species after chronic low-level nitrogen deposition to prairie grasslands. Nature 2008, 451, 712–715. [Google Scholar] [CrossRef]
- Koerner, S.E.; Avolio, M.L.; La Pierre, K.J.; Wilcox, K.R.; Smith, M.D.; Collins, S.L. Nutrient additions cause divergence of tallgrass prairie plant communities resulting in loss of ecosystem stability. J. Ecol. 2016, 104, 1478–1487. [Google Scholar] [CrossRef]
- Suding, K.N.; Collins, S.L.; Gough, L.; Clark, C.; Cleland, E.E.; Gross, K.L.; Milchunas, D.G.; Pennings, S. Functional- and abundance-based mechanisms explain diversity loss due to N fertilization. Proc. Natl. Acad. Sci. USA 2005, 102, 4387–4392. [Google Scholar] [CrossRef]
- Firn, J.; McGree, J.M.; Harvey, E.; Flores-Moreno, H.; Schütz, M.; Buckley, Y.M.; Borer, E.T.; Seabloom, E.W.; La Pierre, K.J.; MacDougall, A.M.; et al. Leaf nutrients, not specific leaf area, are consistent indicators of elevated nutrient inputs. Nat. Ecol. Evol. 2019, 3, 400–406. [Google Scholar] [CrossRef]
- Tatarko, A.R.; Knops, J.M.H. Nitrogen addition and ecosystem functioning: Both species abundances and traits alter community structure and function. Ecosphere 2018, 9, e02087. [Google Scholar] [CrossRef]
- Ceulemans, T.; Merckx, R.; Hens, M.; Honnay, O. A trait-based analysis of the role of phosphorus vs. nitrogen enrichment in plant species loss across North-west European grasslands. J. Appl. Ecol. 2011, 48, 1155–1163. [Google Scholar] [CrossRef]
- Zhang, D.; Peng, Y.; Li, F.; Yang, G.; Wang, J.; Yu, J.; Zhou, G.; Yang, Y. Trait identity and functional diversity co-drive response of ecosystem productivity to nitrogen enrichment. J. Ecol. 2019, 107, 2402–2414. [Google Scholar] [CrossRef]
- Hu, G.; Jin, Y.; Liu, J.; Yu, M. Functional diversity versus species diversity: Relationships with habitat heterogeneity at multiple scales in a subtropical evergreen broad-leaved forest. Ecol. Res. 2014. [Google Scholar] [CrossRef]
- Kraft, N.J.B.; Valencia, R.; Ackerly, D.D. Functional traits and niche-based tree community assembly in an Amazonian forest. Science 2008. [Google Scholar] [CrossRef] [PubMed]
- Calow, P. Towards a Definition of Functional Ecology. Funct. Ecol. 1987. [Google Scholar] [CrossRef]
- Laureto, L.M.O.; Cianciaruso, M.V.; Samia, D.S.M. Functional diversity: An overview of its history and applicability. Nat. Conserv. 2015, 13, 112–116. [Google Scholar] [CrossRef]
- Mouillot, D. Niche-assembly vs. dispersal-assembly rules in coastal fish metacommunities: Implications for management of biodiversity in brackish lagoons. J. Appl. Ecol. 2007, 44, 760–767. [Google Scholar] [CrossRef]
- Carmona, C.P.; de Bello, F.; Mason, N.W.H.; Lepš, J. Traits without Borders: Integrating Functional Diversity across Scales. Trends Ecol. Evol. 2016, 31, 382–394. [Google Scholar] [CrossRef]
- Mason, N.W.H.; Richardson, S.J.; Peltzer, D.A.; de Bello, F.; Wardle, D.A.; Allen, R.B. Changes in coexistence mechanisms along a long-term soil chronosequence revealed by functional trait diversity. J. Ecol. 2012, 100, 678–689. [Google Scholar] [CrossRef]
- Niu, K.; Choler, P.; de Bello, F.; Mirotchnick, N.; Du, G.; Sun, S. Fertilization decreases species diversity but increases functional diversity: A three-year experiment in a Tibetan alpine meadow. Agric. Ecosyst. Environ. 2014, 182, 106–112. [Google Scholar] [CrossRef]
- Helsen, K.; Ceulemans, T.; Stevens, C.J.; Honnay, O. Increasing Soil Nutrient Loads of European Semi-natural Grasslands Strongly Alter Plant Functional Diversity Independently of Species Loss. Ecosystems 2014, 17, 169–181. [Google Scholar] [CrossRef]
- Ehrenfeld, J.G. Dynamics and processes of barrier island vegetation. Rev. Aquat. Sci. 1990, 2, 437–480. [Google Scholar]
- Seitzinger, S.P.; Kroeze, C.; Bouwman, A.F.; Caraco, N.; Dentener, F.; Styles, R.V. Global patterns of dissolved inorganic and particulate nitrogen inputs to coastal systems: Recent conditions and future projections. Estuaries 2002, 25, 640–655. [Google Scholar] [CrossRef]
- Aggenbach, C.J.S.; Kooijman, A.M.; Fujita, Y.; van der Hagen, H.; van Til, M.; Cooper, D.; Jones, L. Does atmospheric nitrogen deposition lead to greater nitrogen and. Biol. Conserv. 2017, 212, 416–422. [Google Scholar] [CrossRef]
- Pakeman, R.J.; Alexander, J.; Brooker, R.; Cummins, R.; Fielding, D.; Gore, S.; Hewison, R.; Mitchell, R.; Moore, E.; Orford, K.; et al. Long-term impacts of nitrogen deposition on coastal plant. Environ. Pollut. 2016, 212, 337–347. [Google Scholar] [CrossRef]
- Day, F.P.; Conn, C.; Crawford, E.; Stevenson, M. Long-term effects of nitrogen fertilization on plant community structure on a coastal barrier island dune chronosequence. J. Coast. Res. 2004, 20, 722–730. [Google Scholar] [CrossRef]
- Zinnert, J.C.; Stallins, J.A.; Brantley, S.T.; Young, D.R. Crossing scales: The complexity of barrier-island processes for predicting future change. Bioscience 2017, 67, 39–52. [Google Scholar] [CrossRef]
- Stutz, M.L.; Pilkey, O.H. A Review of Global Barrier Island Distribution. J. Coast. Res. 2001, 34, 15–22. [Google Scholar]
- Sinha, E.; Michalak, A.M.; Balaji, V. Eutrophication will increase during the 21st century as a result of precipitation changes. Science 2017, 357, 1–5. [Google Scholar] [CrossRef]
- Smith, V.H.; Tilman, G.D.; Nekola, J.C. Eutrophication: Impacts of excess nutrient inputs on freshwater, marine, and terrestrial ecosystems. Environ. Pollut. 1999, 100, 179–196. [Google Scholar] [CrossRef]
- Osgood, D.T.; Zieman, J.C. Factors controlling aboveground Spartina alterniflora (Smooth cordgrass) tissue element composition and production in different-age barrier island marshes. Estuaries 1993, 16, 815–826. [Google Scholar] [CrossRef]
- Zinnert, J.C.; Shiflett, S.A.; Via, S.; Bissett, S.; Dows, B.; Manley, P.; Young, D.R. Spatial–Temporal Dynamics in Barrier Island Upland Vegetation: The Overlooked Coastal Landscape. Ecosystems 2016, 19, 685–697. [Google Scholar] [CrossRef]
- Young, D.R.; Brantley, S.T.; Zinnert, J.C.; Vick, J.K. Landscape position and habitat polygons in a dynamic coastal environment. Ecosphere 2011, 2. [Google Scholar] [CrossRef]
- Sinclair, M.N.; Woods, N.N.; Zinnert, J.C. Seasonal facilitative and competitive trade-offs between shrub seedlings and coastal grasses. Ecosphere 2020. [Google Scholar] [CrossRef]
- Moulton, A. Short-Term Effects of Nutrients on a Barrier Island Grassland Community; Virginia Commonwealth University: Richmond, VA, USA, 2017. [Google Scholar]
- Funk, J.L.; Larson, J.E.; Ames, G.M.; Butterfield, B.J.; Cavender-Bares, J.; Firn, J.; Laughlin, D.C.; Sutton-Grier, A.E.; Williams, L.; Wright, J. Revisiting the Holy Grail: Using plant functional traits to understand ecological processes. Biol. Rev. 2016, 92, 1156–1173. [Google Scholar] [CrossRef] [PubMed]
- Laliberté, E.; Norton, D.A.; Scott, D. Contrasting effects of productivity and disturbance on plant functional diversity at local and metacommunity scales. J. Veg. Sci. 2013, 24, 834–842. [Google Scholar] [CrossRef]
- Wright, I.J.; Reich, P.B.; Westoby, M.; Ackerly, D.D.; Baruch, Z.; Bongers, F.; Cavender-Bares, J.; Chapin, T.; Cornellssen, J.H.C.; Diemer, M.; et al. The worldwide leaf economics spectrum. Nature 2004, 428, 821–827. [Google Scholar] [CrossRef]
- Poorter, H.; De Jong, R. A comparison of specific leaf area, chemical composition and leaf construction costs of field plants from 15 habitats differing in productivity. New Phytol. 1999, 143, 163–176. [Google Scholar] [CrossRef]
- Díaz, S.; Hodgson, J.G.; Thompson, K.; Cabido, M.; Cornelissen, J.H.C.; Jalili, A.; Montserrat-Martí, G.; Grime, J.P.; Zarrinkamar, F.; Asri, Y.; et al. The plant traits that drive ecosystems: Evidence from three continents. J. Veg. Sci. 2004, 15, 295–304. [Google Scholar] [CrossRef]
- Liu, X.; Xu, G.; Wu, Y.; Ma, L.; Gao, J.; Zhang, Y.; Liu, P. Leaf characters of Ulmus elongata in fragmented habitats: Implications for conservation. Acta Ecol. Sin. 2017, 37, 346–353. [Google Scholar] [CrossRef]
- Hobbie, E.A.; Högberg, P. Nitrogen isotopes link mycorrhizal fungi and plants to nitrogen dynamics. New Phytol. 2012, 196, 367–382. [Google Scholar] [CrossRef] [PubMed]
- Kleinebecker, T.; Hölzel, N.; Prati, D.; Schmitt, B.; Fischer, M.; Klaus, V.H. Evidence from the real world:15N natural abundances reveal enhanced nitrogen use at high plant diversity in Central European grasslands. J. Ecol. 2014, 102, 456–465. [Google Scholar] [CrossRef]
- Garnier, E.; Cortez, J.; Billès, G.; Navas, M.L.; Roumet, C.; Debussche, M.; Laurent, G.; Blanchard, A.; Aubry, D.; Bellmann, A.; et al. Plant functional markers capture ecosystem properties during secondary succession. Ecology 2004, 85, 2630–2637. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; Mcglinn, D.; Minchin, P.R.; O’hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R Package Version 2.5-4. 2019. Available online: https://cran.r-project.org/web/packages/vegan/index.html (accessed on 15 April 2020).
- Anderson, M.J. Permutational Multivariate Analysis of Variance (PERMANOVA). In Wiley StatsRef: Statistics Reference Online; 2017; Available online: https://onlinelibrary.wiley.com/doi/full/10.1002/9781118445112.stat07841 (accessed on 19 September 2020).
- Botta-Dukát, Z. Rao’s quadratic entropy as a measure of functional diversity based on multiple traits. J. Veg. Sci. 2005, 16, 533–540. [Google Scholar] [CrossRef]
- Laliberte, E.; Legendre, P. A distance-based framework for measuring functional diversity from multiple traits. Ecology 2010, 91, 299–305. [Google Scholar] [CrossRef]
- Laliberté, E.; Legendre, P.; Shipley, B. FD: Measuring Functional Diversity from Multiple Traits, and other Tools for Functional Ecology. R Package. 2015. Available online: https://cran.r-project.org/web/packages/FD/FD.pdf (accessed on 15 April 2020).
- Anderson, M.J.; Ellingsen, K.E.; McArdle, B.H. Multivariate dispersion as a measure of beta diversity. Ecol. Lett. 2006, 9, 683–693. [Google Scholar] [CrossRef]
- Anderson, M.J. Distance-based tests for homogeneity of multivariate dispersions. Biometrics 2006, 62, 245–253. [Google Scholar] [CrossRef]
- Siefert, A.; Ritchie, M.E. Intraspecific trait variation drives functional responses of old-field plant communities to nutrient enrichment. Oecologia 2016, 181, 245–255. [Google Scholar] [CrossRef]
- Hobbie, E.A.; Macko, S.A.; Williams, M. Correlations between foliar δ15N and nitrogen concentrations may indicate-plant-mycorrhizal interactions. Oecologia 2000, 122, 273–283. [Google Scholar] [CrossRef]
- Craine, J.M.; Brookshire, E.N.J.; Cramer, M.D.; Hasselquist, N.J.; Koba, K.; Marin-Spiotta, E.; Wang, L. Ecological interpretations of nitrogen isotope ratios of terrestrial plants and soils. Plant. Soil 2015, 396, 1–26. [Google Scholar] [CrossRef]
- Jung, K.; Gebauer, G.; Gehre, M.; Hofmann, D.; Weißflog, L.; Schüürmann, G. Anthropogenic impacts on natural nitrogen isotope variations in Pinus sylvestris stands in an industrially polluted area. Environ. Pollut. 1997, 97, 175–181. [Google Scholar] [CrossRef]
- Michelsen, A.; Quarmby, C.; Sleep, D.; Jonasson, S. Vascular plant 15N natural abundance in heath and forest tundra ecosystems is closely correlated with presence and type of mycorrhizal fungi in roots. Oecologia 1998, 115, 406–418. [Google Scholar] [CrossRef] [PubMed]
- Högberg, P.; Johannisson, C.; Yarwood, S.; Callesen, I.; Näsholm, T.; Myrold, D.D.; Högberg, M.N. Recovery of ectomycorrhiza after “nitrogen saturation” of a conifer forest. New Phytol. 2011, 189, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D. δ15N as an integrator of the nitrogen cycle. Trends Ecol. Evol. 2001, 16, 153–162. [Google Scholar] [CrossRef]
- Bond, G. Fixation of Nitrogen by Higher Plants Other than Legumes. Annu. Rev. Plant. Physiol. 1967. [Google Scholar] [CrossRef]
- Young, D.R.; Sande, E.; Peters, G.A. Spatial relationships of Frankia and Myrica cerifera on a Virginia, USA Barrier Island. Symbiosis 1992, 12, 209–220. [Google Scholar]
- Craine, J.M.; Elmore, A.J.; Aidar, M.P.M.; Bustamante, M.; Dawson, T.E.; Hobbie, E.A.; Kahmen, A.; MacK, M.C.; McLauchlan, K.K.; Michelsen, A.; et al. Global patterns of foliar nitrogen isotopes and their relationships with climate, mycorrhizal fungi, foliar nutrient concentrations, and nitrogen availability. New Phytol. 2009, 183, 980–992. [Google Scholar] [CrossRef]
- Johnson, N.C.; Rowland, D.L.; Corkidi, L.; Egerton-Warburton, L.M.; Allen, E.B. Nitrogen enrichment alters mycorrhizal allocation at five mesic to semiarid grasslands. Ecology 2003, 84, 1895–1908. [Google Scholar] [CrossRef]
- Grman, E.; Robinson, T.M.P. Resource availability and imbalance affect plant-mycorrhizal interactions: A field test of three hypotheses. Ecology 2013, 94, 62–71. [Google Scholar] [CrossRef]
- Chen, W.; Xu, R.; Wu, Y.; Chen, J.; Zhang, Y.; Hu, T.; Yuan, X.; Zhou, L.; Tan, T.; Fan, J. Plant diversity is coupled with beta not alpha diversity of soil fungal communities following N enrichment in a semi-arid grassland. Soil Biol. Biochem. 2018, 116, 388–398. [Google Scholar] [CrossRef]
- Zhang, N.; Wan, S.; Li, L.; Bi, J.; Zhao, M.; Ma, K. Impacts of urea N addition on soil microbial community in a semi-arid temperate steppe in northern China. Plant. Soil 2008, 311, 19–28. [Google Scholar] [CrossRef]
- Avolio, M.L.; Carroll, I.T.; Collins, S.L.; Houseman, G.R.; Hallett, L.M.; Isbell, F.; Koerner, S.E.; Komatsu, K.J.; Smith, M.D.; Wilcox, K.R. A comprehensive approach to analyzing community dynamics using rank abundance curves. Ecosphere 2019. [Google Scholar] [CrossRef]
- Swenson, N.G.; Erickson, D.L.; Mi, X.; Bourg, N.A.; Forero-Montana, J.; Ge, X.; Howe, R.; Lake, J.K.; Liu, X.; Ma, K.; et al. Phylogenetic and functional alpha and beta diversity in temperate and tropical tree communities. Ecology 2012, 93. [Google Scholar] [CrossRef] [PubMed]
- Schwilk, D.W.; Ackerly, D.D. Limiting similarity and functional diversity along environmental gradients. Ecol. Lett. 2005, 8, 272–281. [Google Scholar] [CrossRef]
- Li, W.; Cheng, J.M.; Yu, K.L.; Epstein, H.E.; Guo, L.; Jing, G.H.; Zhao, J.; Du, G.Z. Plant functional diversity can be independent of species diversity: Observations based on the impact of 4-yrs of nitrogen and phosphorus additions in an alpine meadow. PLoS ONE 2015, 10, e0136040. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, C.; Nunes, A.; Bugalho, M.N.; Branquinho, C.; McCulley, R.L.; Caldeira, M.C. Nutrient addition and drought interact to change the structure and decrease the functional diversity of a Mediterranean grassland. Front. Ecol. Evol. 2018, 6. [Google Scholar] [CrossRef]
- DeMalach, N.; Zaady, E.; Kadmon, R. Light asymmetry explains the effect of nutrient enrichment on grassland diversity. Ecol. Lett. 2017, 20, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Storm, C.; Süss, K. Are low-productive plant communities responsive to nutrient addition? Evidence from sand pioneer grassland. J. Veg. Sci. 2008, 19, 343–354. [Google Scholar] [CrossRef]
| Comparison | F-Value | p-Value |
|---|---|---|
| C vs. P | 0.9 | 0.5260 |
| C vs. N | 6.8 | 0.0135 |
| C vs. NP | 5.2 | 0.0135 |
| P vs. N | 7.4 | 0.0135 |
| P vs. NP | 5.4 | 0.0135 |
| N vs. NP | 2.4 | 0.1236 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brown, J.K.; Zinnert, J.C. Trait-Based Investigation Reveals Patterns of Community Response to Nutrient Enrichment in Coastal Mesic Grassland. Diversity 2021, 13, 19. https://doi.org/10.3390/d13010019
Brown JK, Zinnert JC. Trait-Based Investigation Reveals Patterns of Community Response to Nutrient Enrichment in Coastal Mesic Grassland. Diversity. 2021; 13(1):19. https://doi.org/10.3390/d13010019
Chicago/Turabian StyleBrown, Joseph K., and Julie C. Zinnert. 2021. "Trait-Based Investigation Reveals Patterns of Community Response to Nutrient Enrichment in Coastal Mesic Grassland" Diversity 13, no. 1: 19. https://doi.org/10.3390/d13010019
APA StyleBrown, J. K., & Zinnert, J. C. (2021). Trait-Based Investigation Reveals Patterns of Community Response to Nutrient Enrichment in Coastal Mesic Grassland. Diversity, 13(1), 19. https://doi.org/10.3390/d13010019





