Indo-Pacific Phylogeography of the Lemon Sponge Leucetta chagosensis
Abstract
1. Introduction
2. Materials and Methods
2.1. Sequence Generation
2.2. Phylogenetic Reconstructions
2.3. Population Structure and Genetic Diversity
3. Results
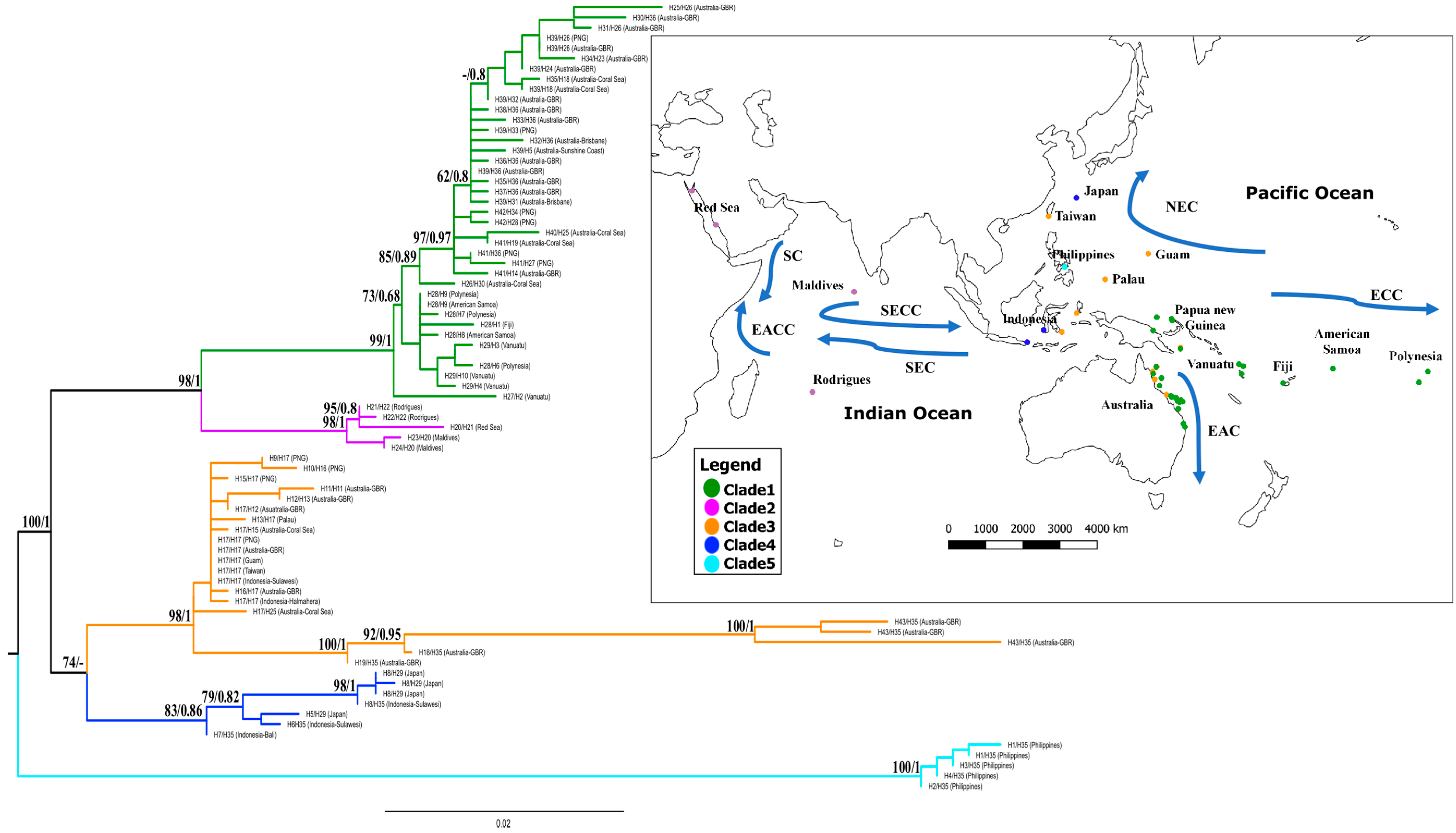
3.1. Phylogenetic Reconstruction
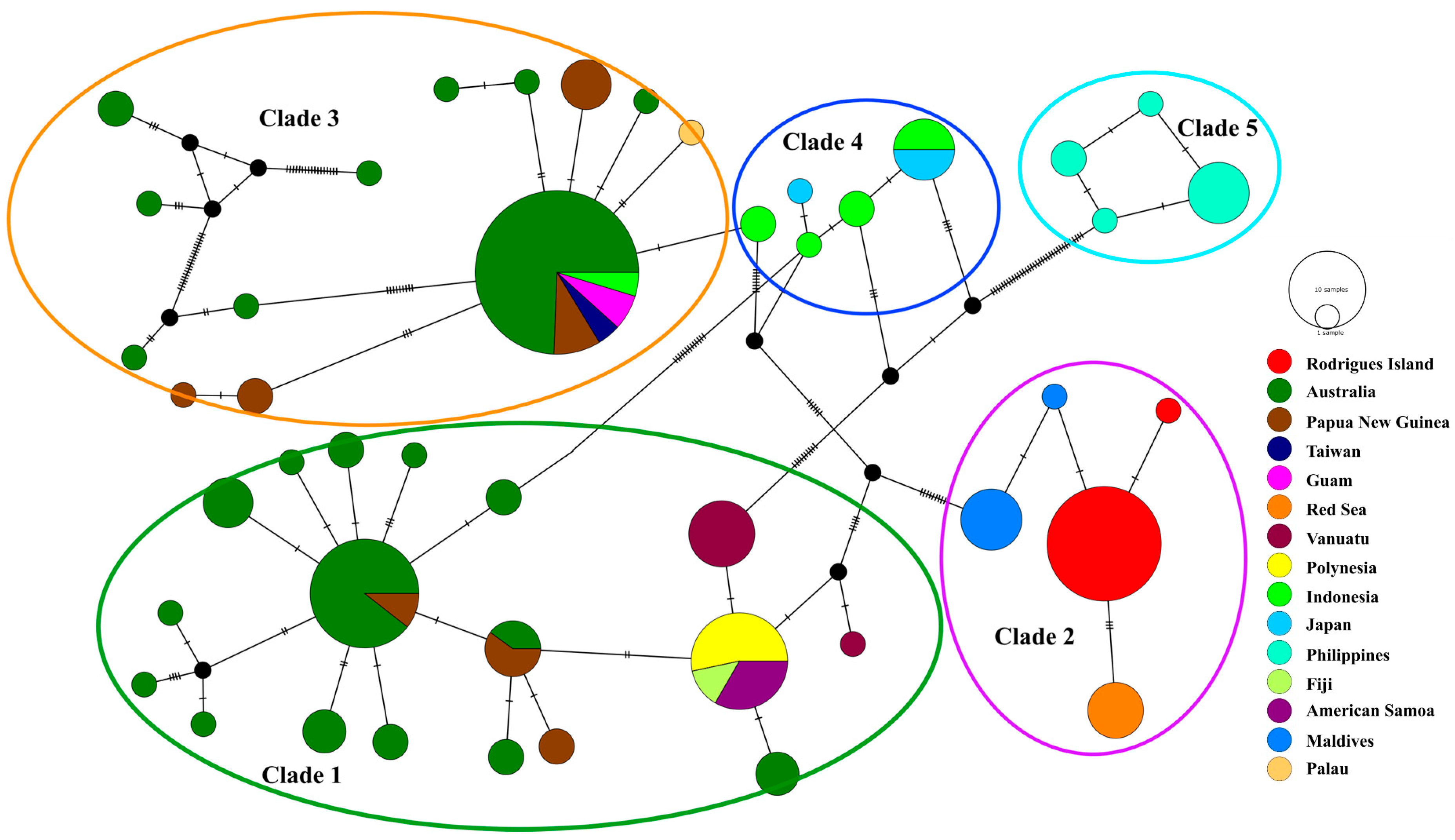
3.2. Population Structure and Genetic Diversity
4. Discussion
4.1. Deep Divergence and Biogeographical Overlap in Cryptic Lineages
4.1.1. Western and Central PO (Clade 1)
4.1.2. Indian Ocean (Clade 2)
4.1.3. Western PO (Clades 3, 4, and 5)
4.2. Genetic Structure and Diversity of L. chagosensis Across Its Range
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hsu, T.-H.; Gwo, J.-C. Genetic diversity and stock identification of small abalone (Haliotis diversicolor) in Taiwan and Japan. PLoS ONE 2017, 12, e0179818. [Google Scholar] [CrossRef]
- Sun, Y.; Wong, E.; Ahyong, S.T.; Williamson, J.; Hutchings, P.; Kupriyanova, E.K. Barcoding and multi-locus phylogeography of the globally distributed calcareous tubeworm genus Hydroides Gunnerus, 1768 (Annelida, Polychaeta, Serpulidae). Mol. Phylogenet. Evol. 2018, 127, 732–745. [Google Scholar] [CrossRef]
- Mouillot, D.; Parravicini, V.; Bellwood, D.R.; Leprieur, F.; Huang, D.; Cowman, P.F.; Albouy, C.; Hughes, T.P.; Thuiller, W.; Guilhaumon, F. Global marine protected areas do not secure the evolutionary history of tropical corals and fishes. Nat. Commun. 2016, 7, 10359. [Google Scholar] [CrossRef] [PubMed]
- Huveneers, C.; Maes, G.E.; Green, M.E.; Simpfendorfer, C.A.; Hoyos-Padilla, E.M.; Duffy, C.J.A.; Meyer, C.G.; Kerwath, S.E.; Salinas-De-León, P.; Van Herwerden, L. Strong trans-Pacific break and local conservation units in the Galapagos shark (Carcharhinus galapagensis) revealed by genome-wide cytonuclear markers. Heredity 2018, 120, 407–421. [Google Scholar] [CrossRef]
- von der Heyden, S. Making evolutionary history count: Biodiversity planning for coral reef fishes and the conservation of evolutionary processes. Coral Reefs 2016, 36, 183–194. [Google Scholar] [CrossRef]
- Doney, S.C.; Ruckelshaus, M.; Duffy, J.E.; Barry, J.P.; Chan, F.; English, C.A.; Galindo, H.M.; Grebmeier, J.M.; Hollowed, A.B.; Knowlton, N.; et al. Climate Change Impacts on Marine Ecosystems. Annu. Rev. Mar. Sci. 2012, 4, 11–37. [Google Scholar] [CrossRef]
- McClanahan, T.R.; Muthiga, N.A. Similar impacts of fishing and environmental stress on calcifying organisms in Indian Ocean coral reefs. Mar. Ecol. Prog. Ser. 2016, 560, 87–103. [Google Scholar] [CrossRef]
- Fricke, R. Fishes of the Mascarene Islands (Reunion, Mauritius, Rodriguez). In An Annotated Checklist with Descriptions of New Species; Koeltz: Koenigstein, Germany, 1999. [Google Scholar]
- Roberts, C.M.; McClean, C.J.; Veron, J.E.N.; Hawkins, J.P.; Allen, G.R.; McAllister, D.E.; Mittermeier, C.G.; Schueler, F.W.; Spalding, M.; Wells, F.; et al. Marine Biodiversity Hotspots and Conservation Priorities for Tropical Reefs. Science 2002, 295, 1280–1284. [Google Scholar] [CrossRef] [PubMed]
- DiBattista, J.D.; Choat, J.H.; Gaither, M.R.; Hobbs, J.-P.A.; Lozano-Cortés, D.F.; Myers, R.F.; Paulay, G.; Rocha, L.A.; Toonen, R.J.; Westneat, M.W.; et al. On the origin of endemic species in the Red Sea. J. Biogeogr. 2015, 43, 13–30. [Google Scholar] [CrossRef]
- Wilson, N.G.; Kirkendale, L.A. Putting the ‘Indo’ back into the Indo-Pacific: Resolving marine phylogeographic gaps. Invertebr. Syst. 2016, 30, 86–94. [Google Scholar] [CrossRef]
- Chambers, D.P.; Tapley, B.D.; Stewart, R.H. Anomalous warming in the Indian Ocean coincident with El Niño. J. Geophys. Res. Space Phys. 1999, 104, 3035–3047. [Google Scholar] [CrossRef]
- Rao, S.A.; Dhakate, A.R.; Saha, S.K.; Mahapatra, S.; Chaudhari, H.S.; Pokhrel, S.; Sahu, S.K. Why is Indian Ocean warming consistently? Clim. Chang. 2012, 110, 709–719. [Google Scholar] [CrossRef]
- Rydbeck, A.V.; Jensen, T.G.; Nyadjro, E.S. Intraseasonal sea surface warming in the western Indian Ocean by oceanic equatorial Rossby waves. Geophys. Res. Lett. 2017, 44, 4224–4232. [Google Scholar] [CrossRef]
- Zhang, Y.; Feng, M.; Du, Y.; Phillips, H.E.; Bindoff, N.L.; McPhaden, M.J. Strengthened Indonesian Throughflow Drives Decadal Warming in the Southern Indian Ocean. Geophys. Res. Lett. 2018, 45, 6167–6175. [Google Scholar] [CrossRef]
- van der Elst, R.; Everett, B.; Jiddawi, N.; Mwatha, G.; Afonso, P.S.; Boulle, D. Fish, fishers and fisheries of the Western Indian Ocean: Their diversity and status. A preliminary assessment. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2005, 363, 263–284. [Google Scholar] [CrossRef]
- Fonteneau, A.; Lucas, V.; Tewkai, E.; Delgado, A.; Demarcq, H. Mesoscale exploitation of a major tuna concentration in the Indian Ocean. Aquat. Living Resour. 2008, 21, 109–121. [Google Scholar] [CrossRef]
- Sharma, R.; Pons, M.; Martin, S.; Kell, L.; Walter, J.; Lauretta, M.; Schirripa, M. Factors related to the decline and rebuilding of billfish stocks in the Atlantic and Indian oceans. ICES J. Mar. Sci. 2017, 75, 880–891. [Google Scholar] [CrossRef]
- Ahti, P.A.; Coleman, R.R.; DiBattista, J.D.; Berumen, M.L.; Rocha, L.A.; Bowen, B.W. Phylogeography of Indo-Pacific reef fishes: Sister wrasses Coris gaimard and C. cuvieri in the Red Sea, Indian Ocean and Pacific Ocean. J. Biogeogr. 2016, 43, 1103–1115. [Google Scholar] [CrossRef]
- Borsa, P.; Durand, J.-D.; Chen, W.-J.; Hubert, N.; Muths, D.; Mou-Tham, G.; Kulbicki, M. Comparative phylogeography of the western Indian Ocean reef fauna. Acta Oecologica 2016, 72, 72–86. [Google Scholar] [CrossRef]
- Benzie, J.A.H. Major genetic differences between crown-of-thorns starfish (Acanthaster planci) populations in the Indian and Pacific oceans. Evolution 1999, 53, 1782–1795. [Google Scholar] [CrossRef]
- Vogler, C.; Benzie, J.; Lessios, H.; Barber, P.; Wörheide, G. A threat to coral reefs multiplied? Four species of crown-of-thorns starfish. Biol. Lett. 2008, 4, 696–699. [Google Scholar] [CrossRef] [PubMed]
- Otwoma, L.M.; Kochzius, M. Genetic Population Structure of the Coral Reef Sea Star Linckia laevigata in the Western Indian Ocean and Indo-West Pacific. PLoS ONE 2016, 11, e0165552. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.K.A.; Bell, J.J. Coastal sponge communities of the West Indian Ocean: Taxonomic affinities, richness and diversity. Afr. J. Ecol. 2002, 40, 337–349. [Google Scholar] [CrossRef]
- van Soest, R.; De Voogd, N.J. Calcareous sponges of the Western Indian Ocean and Red Sea. Zootaxa 2018, 4426, 1–160. [Google Scholar] [CrossRef] [PubMed]
- Dendy, A. No. I.-The Percy Sladen Trust Expedition to the Indian Ocean in 1905, under the Leadership of Mr. J. Stanley Gardiner, M.A. Volume V. No. I.-Report on the Calcareous Sponges collected by H.M.S. “Sealark” in the Indian Ocean. Trans. Linn. Soc. Lond. 1913, 16, 1–29. [Google Scholar] [CrossRef]
- Trentin, F.; Massé, L. Première Approche de la Biodiversité des éponges à la Réunion. 2018. Available online: https://www.researchgate.net/publication/327039827_Premiere_approche_de_la_diversite_des_eponges_a_La_Reunion_Bilan_pour_la_DEAL_Reunion (accessed on 7 December 2020).
- Wörheide, G.; Hooper, J.N.A.; Degnan, B.M. Phylogeography of western Pacific Leucetta ’chagosensis’ (Porifera: Calcarea) from ribosomal DNA sequences: Implications for population history and conservation of the Great Barrier Reef World Heritage Area (Australia). Mol. Ecol. 2002, 11, 1753–1768. [Google Scholar] [CrossRef] [PubMed]
- Wöerheide, G.; Epp, L.S.; Macis, L. Deep genetic divergences among Indo-Pacific populations of the coral reef sponge Leucetta chagosensis (Leucettidae): Founder effects, vicariance, or both? BMC Evol. Biol. 2008, 8, 24. [Google Scholar] [CrossRef]
- Borojevic, R.; Boury-Esnault, N.; Vacelet, J. A revision of the supraspecific classification of the subclass Calcaronea (Porifera, class Calcarea). Zoosystema 2000, 22, 203–263. [Google Scholar]
- Vogler, C.; Benzie, J.A.H.; Barber, P.H.; Erdmann, M.V.; Ambariyanto; Sheppard, C.; Tenggardjaja, K.; Gérard, K.; Wörheide, G. Phylogeography of the Crown-of-Thorns Starfish in the Indian Ocean. PLoS ONE 2012, 7, e43499. [Google Scholar] [CrossRef]
- Lavrov, D.V.; Pett, W.; Voigt, O.; Wörheide, G.; Forget, L.; Lang, B.F.; Kayal, E. Mitochondrial DNA of Clathrina clathrus (Calcarea, Calcinea): Six Linear Chromosomes, Fragmented rRNAs, tRNA Editing, and a Novel Genetic Code. Mol. Biol. Evol. 2012, 30, 865–880. [Google Scholar] [CrossRef]
- Becking, L.E.; Erpenbeck, D.; Peijnenburg, K.T.C.A.; de Voogd, N.J. Phylogeography of the Sponge Suberites diversicolor in Indonesia: Insights into the Evolution of Marine Lake Populations. PLoS ONE 2013, 8, e75996. [Google Scholar] [CrossRef] [PubMed]
- DeBiasse, M.B.; Richards, V.P.; Shivji, M.; Hellberg, M.E. Shared phylogeographical breaks in a Caribbean coral reef sponge and its invertebrate commensals. J. Biogeogr. 2016, 43, 2136–2146. [Google Scholar] [CrossRef]
- Swierts, T.; Peijnenburg, K.T.C.A.; De Leeuw, C.A.; Breeuwer, J.A.J.; Cleary, D.F.R.; de Voogd, N.J. Globally intertwined evolutionary history of giant barrel sponges. Coral Reefs 2017, 36, 933–945. [Google Scholar] [CrossRef]
- Wörheide, G.; Solé-Cava, A.M.; Hooper, J.N.A. Biodiversity, molecular ecology and phylogeography of marine sponges: Patterns, implications and outlooks. Integr. Comp. Biol. 2005, 45, 377–385. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Voigt, O.; Eichmann, V.; Wöerheide, G. First evaluation of mitochondrial DNA as a marker for phylogeographic studies of Calcarea: A case study from Leucetta chagosensis. Hydrobiologia 2011, 687, 101–106. [Google Scholar] [CrossRef]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Chernomor, O.; von Haeseler, A.; Minh, B.Q. Terrace Aware Data Structure for Phylogenomic Inference from Supermatrices. Syst. Biol. 2016, 65, 997–1008. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; Von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the ultrafast bootstrap approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef]
- Huelsenbeck, J.P.; Ronquist, F. MRBAYES: Bayesian inference of phylogeny. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Huelsenbeck, J.P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar] [CrossRef] [PubMed]
- Clement, M.J.; Snell, Q.; Walker, P.; Posada, D.; Crandall, K.A. TCS: Estimating gene genealogies. In Proceedings of the 16th International Parallel and Distributed Processing Symposium, Lauderdale, FL, USA, 15–19 April 2002. [Google Scholar]
- Rozas, J.; Ferrer-Mata, A.; Sánchez-DelBarrio, J.C.; Guirao-Rico, S.; Librado, P.; Ramos-Onsins, S.E.; Sánchez-Gracia, A. DnaSP 6: DNA Sequence Polymorphism Analysis of Large Data Sets. Mol. Biol. Evol. 2017, 34, 3299–3302. [Google Scholar] [CrossRef] [PubMed]
- Excoffier, L.; Lischer, H.E.L. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, M.; Young, C. Effects of physical factors on larval behavior, settlement and recruitment of four tropical demosponges. Mar. Ecol. Prog. Ser. 1996, 138, 169–180. [Google Scholar] [CrossRef]
- Uriz, M.J.; Maldonado, M.; Turon, X.; Martí, R. How do reproductive output, larval behaviour, and recruitment contribute to adult spatial patterns in Mediterranean encrusting sponges? Mar. Ecol. Prog. Ser. 1998, 167, 137–148. [Google Scholar] [CrossRef]
- Crandall, E.D.; Riginos, C.; Bird, C.E.; Liggins, L.; Treml, E.A.; Beger, M.; Barber, P.H.; Connolly, S.R.; Cowman, P.F.; DiBattista, J.D.; et al. The molecular biogeography of the Indo-Pacific: Testing hypotheses with multispecies genetic patterns. Glob. Ecol. Biogeogr. 2019, 28, 943–960. [Google Scholar] [CrossRef]
- Kennett, J.P.; Keller, G.; Srinivasan, M.S. Miocene planktonic foraminiferal biogeography and paleoceanographic development of the Indo-Pacific region. Geol. Soc. Am. Mem. 1985, 163, 197–236. [Google Scholar] [CrossRef]
- Voris, H.K. Maps of Pleistocene sea levels in Southeast Asia: Shorelines, river systems and time durations. J. Biogeogr. 2000, 27, 1153–1167. [Google Scholar] [CrossRef]
- Bowen, B.W.; Gaither, M.R.; DiBattista, J.D.; Iacchei, M.; Andrews, K.R.; Grant, W.S.; Toonen, R.J.; Briggs, J.C. Comparative phylogeography of the ocean planet. Proc. Natl. Acad. Sci. USA 2016, 113, 7962–7969. [Google Scholar] [CrossRef]
- Gaither, M.R.; Bowen, B.W.; Bordenave, T.-R.; Rocha, L.A.; Newman, S.J.; Gomez, J.A.; van Herwerden, L.; Craig, M.T. Phylogeography of the reef fish Cephalopholis argus (Epinephelidae) indicates Pleistocene isolation across the Indo-Pacific barrier with contemporary overlap in the coral triangle. BMC Evol. Biol. 2011, 11, 189. [Google Scholar] [CrossRef] [PubMed]
- Siddall, M.E.; Rohling, E.J.; Almogilabin, A.; Hemleben, C.; Meischner, D.; Schmelzer, I.; Smeed, D.A. Sea-level fluctuations during the last glacial cycle. Nat. Cell Biol. 2003, 423, 853–858. [Google Scholar] [CrossRef] [PubMed]
- DiBattista, J.D.; Berumen, M.L.; Gaither, M.R.; Rocha, L.A.; Eble, J.A.; Choat, J.H.; Craig, M.T.; Skillings, D.J.; Bowen, B.W. After continents divide: Comparative phylogeography of reef fishes from the Red Sea and Indian Ocean. J. Biogeogr. 2013, 40, 1170–1181. [Google Scholar] [CrossRef]
- Fernandez-Silva, I.; Randall, J.E.; Coleman, R.R.; DiBattista, J.D.; Rocha, L.A.; Reimer, J.D.; Meyer, C.G.; Bowen, B.W. Yellow tails in the Red Sea: Phylogeography of the Indo-Pacific goatfish Mulloidichthys flavolineatus reveals isolation in peripheral provinces and cryptic evolutionary lineages. J. Biogeogr. 2015, 42, 2402–2413. [Google Scholar] [CrossRef]
- Obura, D. The Diversity and Biogeography of Western Indian Ocean Reef-Building Corals. PLoS ONE 2012, 7, e45013. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.W.; Cheang, C.C.; Yeung, C.W.; Chirapart, A.; Gerung, G.; Ang, P. Recent expansion led to the lack of genetic structure of Sargassum aquifolium populations in Southeast Asia. Mar. Biol. 2014, 161, 785–795. [Google Scholar] [CrossRef]
- Woodruff, D.S. Biogeography and conservation in Southeast Asia: How 2.7 million years of repeated environmental fluctuations affect today’s patterns and the future of the remaining refugial-phase biodiversity. Biodivers. Conserv. 2010, 19, 919–941. [Google Scholar] [CrossRef]
- Guo, W.; Ng, W.L.; Wu, H.; Li, W.; Zhang, L.; Qiao, S.; Yang, X.; Shi, X.; Huang, Y.-L. Chloroplast phylogeography of a widely distributed mangrove species, Excoecaria agallocha, in the Indo-West Pacific region. Hydrobiologia 2017, 807, 333–347. [Google Scholar] [CrossRef]
- Sorenson, L.; Allen, G.R.; Erdmann, M.V.; Dai, C.-F.; Liu, S.Y.V. Pleistocene diversification of the Pomacentrus coelestis species complex (Pisces: Pomacentridae): Historical biogeography and species boundaries. Mar. Biol. 2014, 161, 2495–2507. [Google Scholar] [CrossRef]
- Payo, D.A.; Leliaert, F.; Verbruggen, H.; D’Hondt, S.; Calumpong, H.P.; De Clerck, O. Extensive cryptic species diversity and fine-scale endemism in the marine red alga Portieria in the Philippines. Proc. R. Soc. B Boil. Sci. 2013, 280, 20122660. [Google Scholar] [CrossRef]
- Grant, W. Shallow population histories in deep evolutionary lineages of marine fishes: Insights from sardines and anchovies and lessons for conservation. J. Hered. 1998, 89, 415–426. [Google Scholar] [CrossRef]
- Bellwood, D.R.; Renema, W.; Rosen, B.R. Biodiversity hotspots, evolution and coral reef biogeography: A review. In Biotic Evolution and Environmental Change in Southeast Asia: Systematics Association Special; Gower, D.J., Johnson, K.G., Richardson, J.E., Rosen, B.R., Rüber, L., Williams, S.T., Eds.; Cambridge University Press (CUP): Cambridge, UK, 2012; pp. 216–245. [Google Scholar]
- Briggs, J.C.; Bowen, B.W. Marine shelf habitat: Biogeography and evolution. J. Biogeogr. 2013, 40, 1023–1035. [Google Scholar] [CrossRef]
- Gaither, M.R.; Rocha, L.A. Origins of species richness in the Indo-Malay-Philippine biodiversity hotspot: Evidence for the centre of overlap hypothesis. J. Biogeogr. 2013, 40, 1638–1648. [Google Scholar] [CrossRef]
- Briggs, J.C. Centrifugal speciation and centres of origin. J. Biogeogr. 2000, 27, 1183–1188. [Google Scholar] [CrossRef]
- Bellwood, D.; Meyer, C.P. Searching for heat in a marine biodiversity hotspot. J. Biogeogr. 2009, 36, 569–576. [Google Scholar] [CrossRef]
- Evans, S.M.; McKenna, C.; Simpson, S.D.; Tournois, J.; Genner, M.J. Patterns of species range evolution in Indo-Pacific reef assemblages reveal the Coral Triangle as a net source of transoceanic diversity. Biol. Lett. 2016, 12, 20160090. [Google Scholar] [CrossRef]
| Sampling Region | Number of Specimens |
|---|---|
| Indian Ocean | |
| Rodrigues | 25 |
| Red Sea | 7 |
| Maldives | 9 |
| Western Pacific Ocean | |
| Australia | 90 |
| Guam | 4 |
| Indonesia | 14 |
| Papua New Guinea | 19 |
| Palau | 2 |
| Taiwan | 2 |
| Japan | 4 |
| Philippines | 10 |
| Central Pacific Ocean | |
| American Samoa | 5 |
| Fiji | 2 |
| Polynesia | 8 |
| Vanuatu | 8 |
| AMOVA Results for cox3 | |||||
| Source of variation | d.f. | s.s. | v.c. | % var | Fst |
| Among populations | 4 | 1456.36 | 10.92 | 87.54 | 0.88 |
| Within populations | 183 | 284.456 | 1.55 | 12.46 | |
| AMOVA Results for 28S | |||||
| Source of variation | d.f. | s.s. | v.c. | % var | Fst |
| Among populations | 4 | 85.08 | 0.62 | 46.23 | 0.46 |
| Within populations | 183 | 131.76 | 0.72 | 53.77 | |
| Grouping | # seq | # Haplotypes | # Variable Sites | Nucleotide Diversity | Haplotype Diversity | ||||
|---|---|---|---|---|---|---|---|---|---|
| cox3 | 28S | cox3 | 28S | cox3 | 28S | cox3 | 28S | ||
| Global | 188 | 45 | 36 | 102 | 26 | 0.046 | 0.006 | 0.914 | 0.881 |
| Clade 1 | 72 | 19 | 25 | 30 | 23 | 0.008 | 0.006 | 0.875 | 0.856 |
| Clade 2 | 34 | 5 | 3 | 8 | 2 | 0.005 | 0.001 | 0.581 | 0.533 |
| Clade 3 | 62 | 14 | 8 | 54 | 9 | 0.012 | 0.001 | 0.517 | 0.344 |
| Clade 4 | 10 | 4 | 2 | 3 | 1 | 0.002 | 0.001 | 0.644 | 0.533 |
| Clade 5 | 10 | 4 | 1 | 2 | 0 | 0.002 | 0.000 | 0.644 | 0.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasnin, O.; Voigt, O.; Wörheide, G.; Murillo Rincón, A.P.; von der Heyden, S. Indo-Pacific Phylogeography of the Lemon Sponge Leucetta chagosensis. Diversity 2020, 12, 466. https://doi.org/10.3390/d12120466
Pasnin O, Voigt O, Wörheide G, Murillo Rincón AP, von der Heyden S. Indo-Pacific Phylogeography of the Lemon Sponge Leucetta chagosensis. Diversity. 2020; 12(12):466. https://doi.org/10.3390/d12120466
Chicago/Turabian StylePasnin, Olivier, Oliver Voigt, Gert Wörheide, Andrea P. Murillo Rincón, and Sophie von der Heyden. 2020. "Indo-Pacific Phylogeography of the Lemon Sponge Leucetta chagosensis" Diversity 12, no. 12: 466. https://doi.org/10.3390/d12120466
APA StylePasnin, O., Voigt, O., Wörheide, G., Murillo Rincón, A. P., & von der Heyden, S. (2020). Indo-Pacific Phylogeography of the Lemon Sponge Leucetta chagosensis. Diversity, 12(12), 466. https://doi.org/10.3390/d12120466






