Seawater Acidification Affects Beta-Diversity of Benthic Communities at a Shallow Hydrothermal Vent in a Mediterranean Marine Protected Area (Underwater Archaeological Park of Baia, Naples, Italy)
Abstract
:1. Introduction
2. Material and Methods
2.1. Study Area
2.2. Sampling Campaign and Data Analysis
3. Results
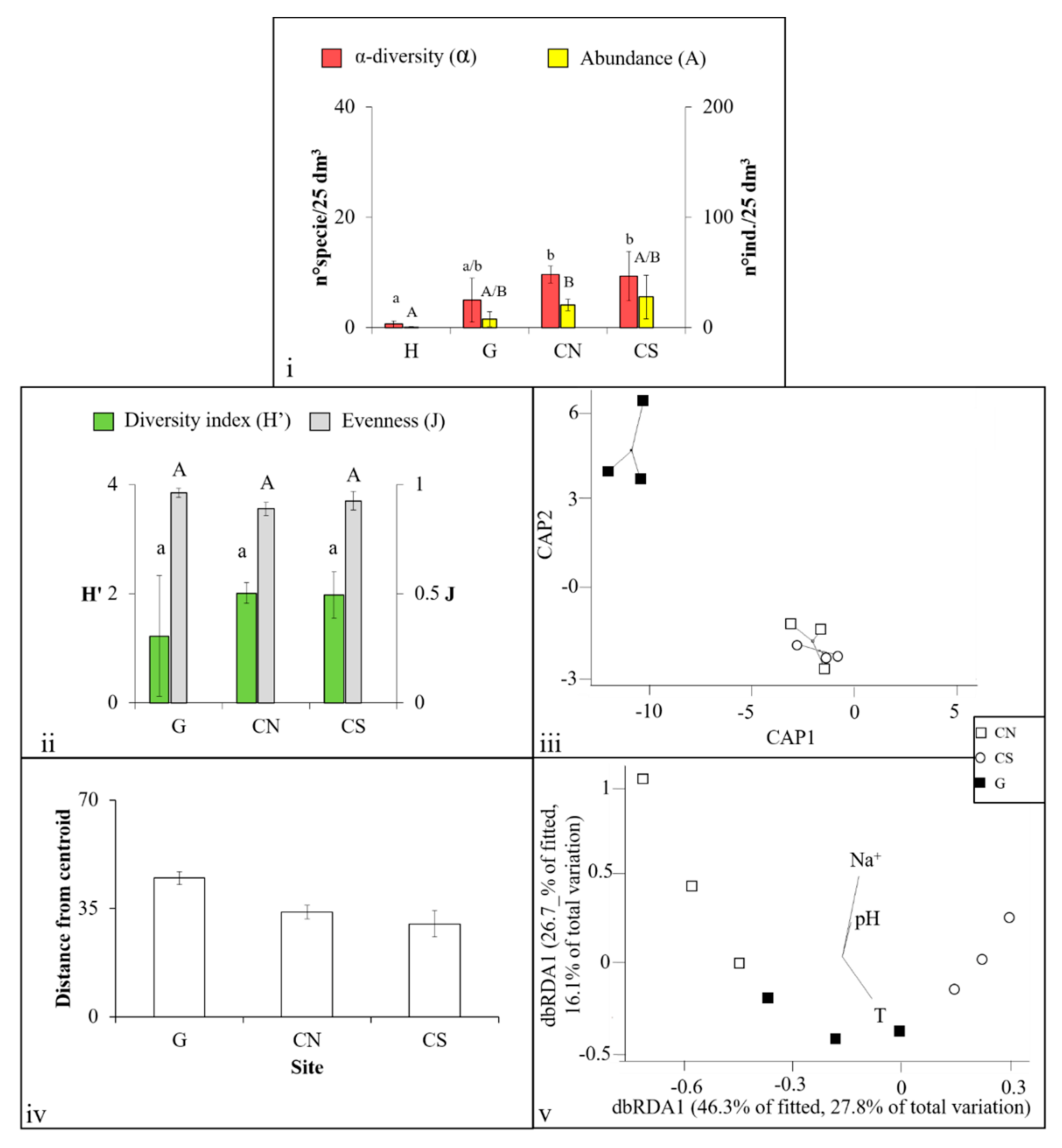
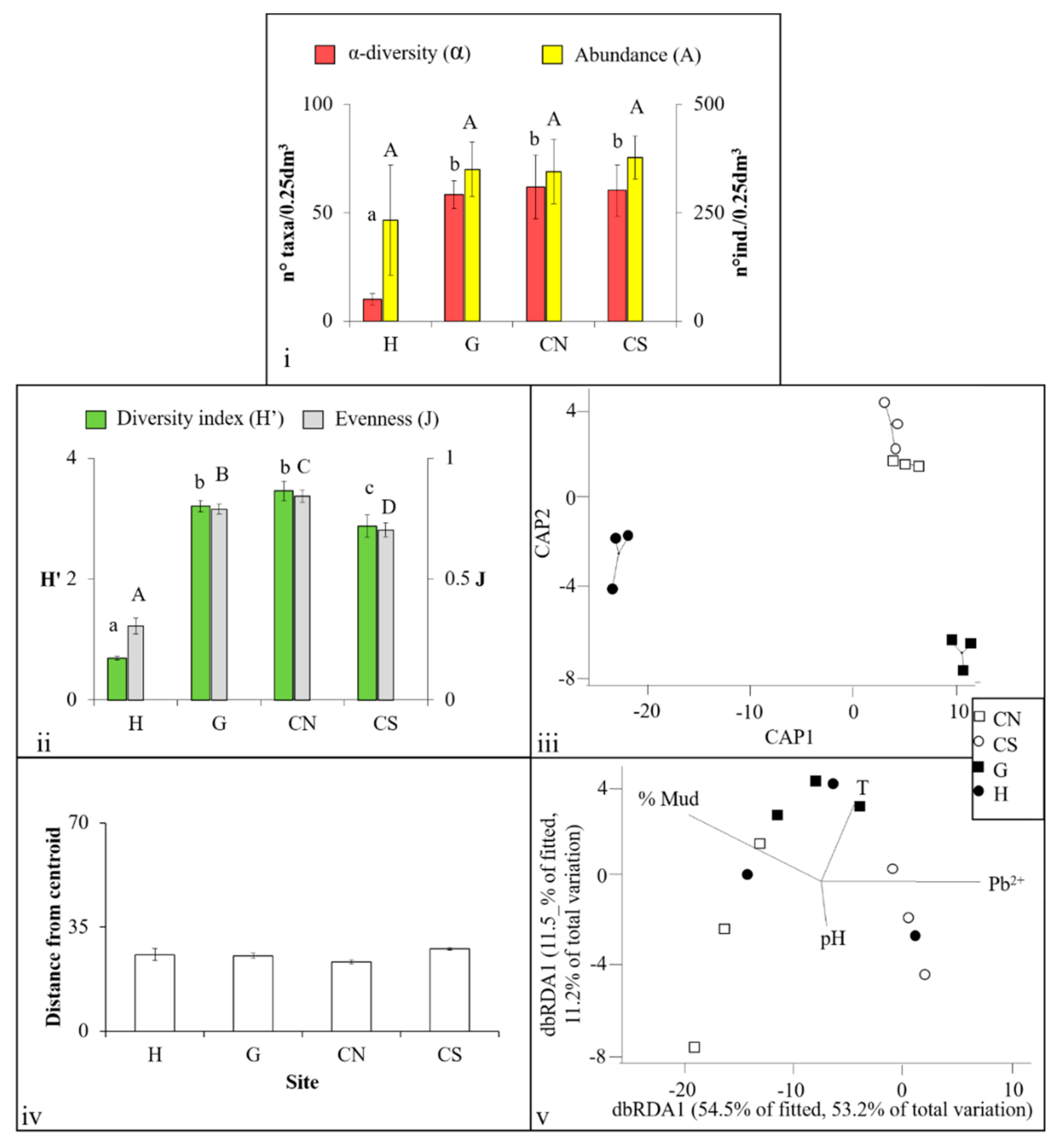
3.1. Mollusca Assemblages
3.2. Polychaeta Assemblages
3.3. Crustacea Assemblages
3.4. Nematoda Assemblages
4. Discussions
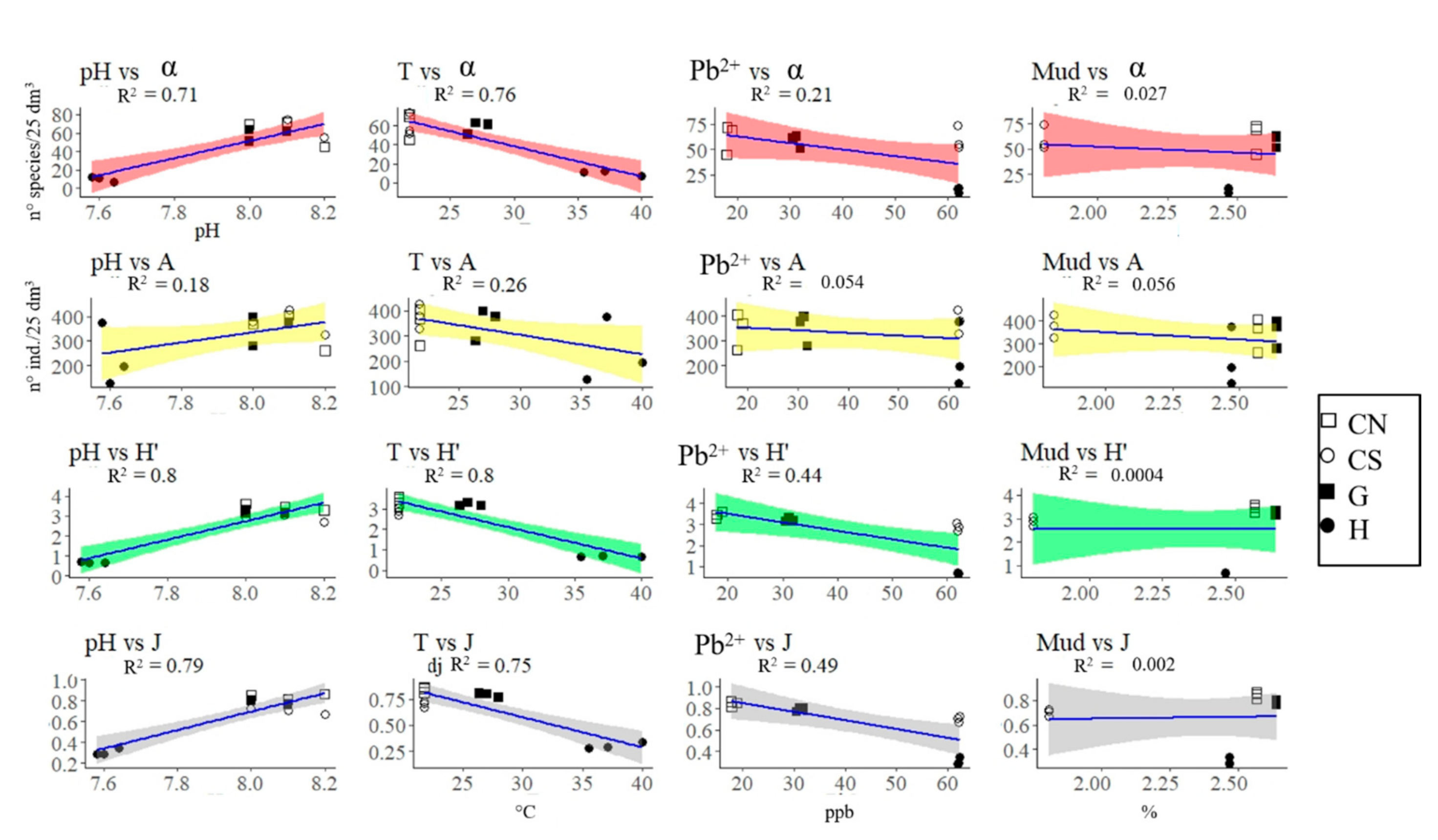
4.1. Effects of Acidification on β-Diversity
4.2. Assemblages’ Strategies to Mitigate Acidification Effects
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Donnarumma, L.; Sandulli, R.; Appolloni, L.; Sánchez-Lizaso, J.L.; Russo, G.F. Assessment of structural and functional diversity of mollusc assemblages within vermetid bioconstructions. Diversity 2018, 10, 96. [Google Scholar] [CrossRef] [Green Version]
- Donnarumma, L.; Sandulli, R.; Appolloni, L.; Russo, G.F. Assessing molluscs functional diversity within different coastal habitats of Mediterranean marine protected areas. Ecol. Quest. 2018, 29, 35–51. [Google Scholar]
- Gerlach, S.A. On the importance of marine meiofauna for benthos communities. Oecologia 1971, 6, 176–190. [Google Scholar] [CrossRef]
- Castelli, A.; Lardicci, C.; Tagliapietra, D. Soft-bottom macrobenthos. In Mediterranean Marine Benthos: A Manual of Methods for Its Sampling and Study; Gambi, M.C., Dappiano, M., Eds.; S.I.B.M—Società Italiana di Biologia Marina: Genova, Italy, 2003; pp. 99–132. [Google Scholar]
- Shmida, A.V.I.; Wilson, M. V Biological determinants of species diversity. J. Biogeogr. 1985, 12, 1–20. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Bruno, J.F. The impact of climate change on the world’s marine ecosystems. Science 2010, 328, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Bruno, J.F.; Bertness, M. Habitat modification and facilitation in benthic marine communities. In Marine Community Ecology; Bertness, M., Gaines, S., Hay, M., Eds.; Sinauer Associates: Sunderland, MA, USA, 2001; pp. 201–218. [Google Scholar]
- Buonocore, E.; Donnarumma, L.; Appolloni, L.; Miccio, A.; Russo, G.F.; Franzese, P.P. Marine natural capital and ecosystem services: An environmental accounting model. Ecol. Modell. 2020, 424, 109029. [Google Scholar] [CrossRef]
- Buonocore, E.; Appolloni, L.; Russo, G.F.; Franzese, P.P. Assessing natural capital value in marine ecosystems through an environmental accounting model: A case study in Southern Italy. Ecol. Modell. 2020, 419, 108958. [Google Scholar] [CrossRef]
- Rendina, F.; Bouchet, P.J.; Appolloni, L.; Russo, G.F.; Sandulli, R.; Kolzenburg, R.; Putra, A.; Ragazzola, F. Physiological response of the coralline alga Corallina officinalis L. to both predicted long-term increases in temperature and short-term heatwave events. Mar. Environ. Res. 2019, 150, 104764. [Google Scholar] [CrossRef]
- O’Leary, J.K.; Micheli, F.; Airoldi, L.; Boch, C.; De Leo, G.; Elahi, R.; Ferretti, F.; Graham, N.A.J.; Litvin, S.Y.; Low, N.H.; et al. The resilience of marine ecosystems to climatic disturbances. Bioscience 2017, 67, 208–220. [Google Scholar] [CrossRef]
- Sanda, T.; Hamasaki, K.; Dan, S.; Kitada, S. Expansion of the Northern Geographical Distribution of Land Hermit Crab Populations: Colonization and Overwintering Success of Coenobita purpureus on the Coast of the Boso. Zool. Stud. 2019, 58, e25. [Google Scholar]
- Kao, K.; Keshavmurthy, S.; Tsao, C.; Wang, J.; Chen, A. Repeated and Prolonged Temperature Anomalies Negate Symbiodiniaceae Genera Shuffling in the Coral Platygyra verweyi (Scleractinia; Merulinidae). Zool. Stud. 2018, 57, 1–14. [Google Scholar]
- Anderson, M.J.; Crist, T.O.; Chase, J.M.; Vellend, M.; Inouye, B.D.; Freestone, A.L.; Sanders, N.J.; Cornell, H.V.; Comita, L.S.; Davies, K.F.; et al. Navigating the multiple meanings of β diversity: A roadmap for the practicing ecologist. Ecol. Lett. 2011, 14, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Baeten, L.; Vangansbeke, P.; Hermy, M.; Peterken, G.; Vanhuyse, K.; Verheyen, K. Distinguishing between turnover and nestedness in the quantification of biotic homogenization. Biodivers. Conserv. 2012, 21, 1399–1409. [Google Scholar] [CrossRef] [Green Version]
- Bevilacqua, S.; Plicanti, A.; Sandulli, R.; Terlizzi, A. Measuring more of Beta-diversity: Quantifying patterns of variation in assemblage heterogeneity. An insight from marine benthic assemblages. Ecol. Indic. 2012, 18, 140–148. [Google Scholar] [CrossRef]
- Appolloni, L.; Bevilacqua, S.; Sbrescia, L.; Sandulli, R.; Terlizzi, A.; Russo, G.F. Does full protection count for the maintenance of β-diversity patterns in marine communities? Evidence from Mediterranean fish assemblages. Aquat. Conserv. Mar. Freshw. Ecosyst. 2017, 27, 828–838. [Google Scholar] [CrossRef] [Green Version]
- Thrush, S.F.; Hewitt, J.E.; Cummings, V.J.; Norkko, A.; Chiantore, M. β-diversity and species accumulation in Antarctic coastal benthos: Influence of habitat, distance and productivity on ecological connectivity. PLoS ONE 2010, 5, e11899. [Google Scholar] [CrossRef]
- Thrush, S.F.; Hewitt, J.E.; Lohrer, A.M. Interaction networks in coastal soft-sediments highlight the potential for change in ecological resilience. Ecol. Appl. 2012, 22, 1213–1223. [Google Scholar] [CrossRef]
- Fritz, K.M.; Dodds, W.K. Resistance and resilience of macroinvertebrate assemblages to drying and flood in a tallgrass prairie stream system. Hydrobiologia 2004, 527, 99–112. [Google Scholar] [CrossRef]
- Townsend, C.R.; Hildrew, A.G. Species traits in relation to a habitat templet for river systems. Freshw. Biol. 1994, 31, 265–275. [Google Scholar] [CrossRef]
- Peterson, G.; Allen, C.R.; Holling, C.S. Ecological resilience, biodiversity, and scale. Ecosystems 1998, 1, 6–18. [Google Scholar] [CrossRef]
- Holling, C.S.; Schindler, D.W.; Walker, B.W.; Roughgarden, J. Biodiversity in the functioning of ecosystems: An ecological synthesis. In Biodiversity Loss: Economic and Ecological Issues; Perrings, C., Karl-Goran, M., Folke, C., Holling, C.S., Bengt-Owe, J., Eds.; Cambridge University Press: Cambridge, UK, 1995; pp. 44–83. [Google Scholar]
- Montefalcone, M.; Parravicini, V.; Bianchi, C.N. Quantification of coastal ecosystem resilience. Treatise Estuar. Coast. Sci. 2011, 10, 49–70. [Google Scholar]
- Hall-Spencer, J.M.; Rodolfo-Metalpa, R.; Martin, S.; Ransome, E.; Fine, M.; Turner, S.M.; Rowley, S.J.; Tedesco, D.; Buia, M.C. Volcanic carbon dioxide vents show ecosystem effects of ocean acidification. Nature 2008, 454, 96–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gattuso, J.-P.; Hansson, L. Ocean Acidification; Oxford University Press: Oxford, UK, 2011; ISBN 0199591091. [Google Scholar]
- Wang, T.; Chan, T.; Chan, B.K.K. Diversity and community structure of decapod crustaceans at hydrothermal vents and nearby deep-water fishING grounds off Kueishan Island, Taiwan: A high biodiversity deep-sea area in the NW Pacific. Bull. Mar. Sci. 2013, 89, 505–528. [Google Scholar] [CrossRef]
- Chan, B.K.K.; Wang, T.; Chen, P.; Lin, C. Community Structure of Macrobiota and Environmental Parameters in Shallow Water Hydrothermal Vents off Kueishan Island, Taiwan. PLoS ONE 2016, 11, e0148675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donnarumma, L.; Lombardi, C.; Cocito, S.; Gambi, M.C. Settlement pattern of Posidonia oceanica epibionts along a gradient of ocean acidification: An approach with mimics. Mediterr. Mar. Sci. 2014, 15, 498–509. [Google Scholar] [CrossRef] [Green Version]
- Berge, J.A.; Bjerkeng, B.; Pettersen, O.; Schaanning, M.T.; Øxnevad, S. Effects of increased sea water concentrations of CO2 on growth of the bivalve Mytilus edulis L. Chemosphere 2006, 62, 681–687. [Google Scholar] [CrossRef]
- Gazeau, F.; Quiblier, C.; Jansen, J.M.; Gattuso, J.P.; Middelburg, J.J.; Heip, C.H.R. Impact of elevated CO2 on shellfish calcification. Geophys. Res. Lett. 2007, 34, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Linares, C.; Vidal, M.; Canals, M.; Kersting, D.K.; Amblas, D.; Aspillaga, E.; Cebrián, E.; Delgado-Huertas, A.; Díaz, D.; Garrabou, J.; et al. Persistent natural acidification drives major distribution shifts in marine benthic ecosystems. Proc. R. Soc. B Biol. Sci. 2015, 282, 20150587. [Google Scholar] [CrossRef]
- Tedesco, D. I fluidi fumarolici sottomarini dell’era vulcanica napoletana: Cambiamenti globali, dinamiche vulcaniche e genesi dei fluidi, mitigazione del rischio vulcanico. In Proceedings of the Il Fuoco Dal Mare—Vulcanismo e Ambienti Sottomarini; Coiro, P., Russo, G.F., Eds.; Giannini Editore: Napoli, Italy, 2008; pp. 99–112. [Google Scholar]
- Appolloni, L.; Sandulli, R.; Vetrano, G.; Russo, G.F. Assessing the effects of habitat patches ensuring propagule supply and different costs inclusion in marine spatial planning through multivariate analyses. J. Environ. Manag. 2018, 214, 45–55. [Google Scholar] [CrossRef] [Green Version]
- Appolloni, L.; Sandulli, R.; Bianchi, C.N.; Russo, G.F. Spatial analyses of an integrated landscape-seascape territorial system: The case of the overcrowded Gulf of Naples, Southern Italy. J. Environ. Account. Manag. 2018, 6, 365–380. [Google Scholar] [CrossRef]
- Donnarumma, L.; Appolloni, L.; Chianese, E.; Bruno, R.; Baldrighi, E.; Guglielmo, R.; Russo, G.F.; Zeppilli, D.; Sandulli, R. Environmental and Benthic Community Patterns of the Shallow Hydrothermal Area of Secca Delle Fumose (Baia, Naples, Italy). Front. Mar. Sci. 2019, 6, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Baldrighi, E.; Zeppilli, D.; Appolloni, L.; Donnarumma, L.; Chianese, E.; Russo, G.F.; Sandulli, R. Meiofaunal communities and nematode diversity characterizing the Secca delle Fumose shallow vent area (Gulf of Naples, Italy). PeerJ 2020, 8, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Kenny, A.J.; Sotheran, I. Characterising the Physical Properties of Seabed Habitats. In Methods for the Study of Marine Benthos; Eleftheriou, A., Ed.; John Wiley & Sons: Chichester, West Sussex, UK, 2013; ISBN 1118542371. [Google Scholar]
- Schumacher, B.A. Methods for the Determination of Total Organic Carbon (TOC) in Soils and Sediments, NCEA-C-1282, 1st ed.; Schumacher, B.A., Ed.; US Environmental Protection Agency, Office of Research and Development: Washington, DC, USA, 2002.
- Chianese, E.; Tirimberio, G.; Riccio, A. PM2.5 and PM10 in the urban area of Naples: Chemical composition, chemical properties and influence of air masses origin. J. Atmos. Chem. 2019, 76, 151–169. [Google Scholar] [CrossRef]
- Anderson, M.J. A new method for non parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar]
- Terlizzi, A.; Anderson, M.J.; Fraschetti, S.; Benedetti-Cecchi, L. Scales of spatial variation in Mediterranean subtidal sessile assemblages at different depths. Mar. Ecol. Prog. Ser. 2007, 332, 25–39. [Google Scholar] [CrossRef] [Green Version]
- Anderson, M.J. Permutation tests for univariate or multivariate analysis of variance and regression. Can. J. Fish. Aquat. Sci. 2001, 58, 626–639. [Google Scholar] [CrossRef]
- Anderson, M.; Braak, C. Ter Permutation tests for multi-factorial analysis of variance. J. Stat. Comput. Simul. 2003, 73, 85–113. [Google Scholar] [CrossRef]
- Anderson, M.J.; Willis, T.J. Canonical Analysis of Principal Coordinates: A Useful Method of Constrained Ordination for Ecology. Ecology 2003, 84, 511–525. [Google Scholar] [CrossRef]
- Anderson, M.J.; Ellingsen, K.E.; McArdle, B.H. Multivariate dispersion as a measure of beta diversity. Ecol. Lett. 2006, 9, 683–693. [Google Scholar] [CrossRef]
- R Development Core Team, R. R: A Language and Environment for Statistical Computing. R Found. Stat. Comput. 2011, 1, 409. [Google Scholar]
- Kindt, R.; Coe, R. Tree Diversity Analysis: A Manual and Software for Common Statistical Methods for Ecological and Biodiversity Studie; World Agroforestry Centre (ICRAF): Nairobi, Kenya, 2005; ISBN 92 9059 179 X. [Google Scholar]
- Anderson, M.J. DISTLM v. 5: A FORTRAN computer program to calculate a distance-based multivariate analysis for a linear model. Dep. Stat. Univ. Auckl. N. Z. 2004, 10, 2016. [Google Scholar]
- Legendre, P.; Anderson, M.J. Distance-based redundancy analysis: Testing multispecies responses in multicfacorial ecological experiments. Ecol. Monogr. 1999, 69, 1–24. [Google Scholar] [CrossRef]
- Mastrototaro, F.; D’Onghia, G.; Tursi, A. Spatial and seasonal distribution of ascidians in a semi-enclosed basin of the Mediterranean Sea. J. Mar. Biol. Assoc. UK 2008, 88, 1053–1061. [Google Scholar] [CrossRef]
- Gambi, M.C.; Giangrande, A. Distribution of soft-bottom polychaetes in two coastal areas of the Tyrrhenian Sea (Italy): Structural analysis. Estuar. Coast. Shelf Sci. 1986, 23, 847–862. [Google Scholar] [CrossRef]
- Fanelli, E.; Lattanzi, L.; Nicoletti, L.; Tomassetti, P. Decapod crustaceans of Tyrrhenian Sea soft bottoms (central Mediterranean). Crustac. J. Crustac. Res. 2005, 78, 641. [Google Scholar]
- Gambi, M.C.; Conti, G.; Bremec, C.S. Polycliaete distribution, diversity and seasonality related to seagrass cover in shallow soft bottoms of the Tyrrhenian Sea (Italy). Sci. Mar. 1998, 62, 1–17. [Google Scholar]
- Cantone, G.; Fassari, G. Polychaetous annelids of soft-bottoms around the Gulf of Catania (Sicily). Comm. Int. Explor. Mer Mediterr. 1983, 28, 251–252. [Google Scholar]
- Smith, E.P. BACI Design. Wiley StatsRef Stat. Ref. Online 2014, 1, 141–148. [Google Scholar]
- Magurran, A.E.; Dornelas, M.; Moyes, F.; Gotelli, N.J.; McGill, B. Rapid biotic homogenization of marine fish assemblages. Nat. Commun. 2015, 6, 8405. [Google Scholar] [CrossRef] [Green Version]
- Séguin, A.; Gravel, D.; Archambault, P. Effect of disturbance regime on alpha and beta diversity of rock pools. Diversity 2014, 6, 1–17. [Google Scholar] [CrossRef]
- Thrush, S.F.; Hewitt, J.E.; Dayton, P.K.; Coco, G.; Lohrer, A.M.; Norkko, A.; Norkko, J.; Chiantore, M. Forecasting the limits of resilience: Integrating empirical research with theory. Proc. R. Soc. B Biol. Sci. 2009, 276, 3209–3217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Appolloni, L.; Ferrigno, F.; Russo, G.F.; Sandulli, R. β-Diversity of morphological groups as indicator of coralligenous community quality status. Ecol. Indic. 2020, 109, 105840. [Google Scholar] [CrossRef]
- Martin, J.; Bastida, R. Life history and production of Capitella Capitata (Capitellidae: Polychaeta) in Río de La Plata Estuary (Argentina). Thalassas 2006, 22, 25–38. [Google Scholar]
- Nandan, S.B. Retting of coconut husk-a unique case of water pollution on the South West coast of India. Int. J. Environ. Stud. 1997, 52, 335–355. [Google Scholar] [CrossRef]
- Thiermann, F.; Vismann, B.; Giere, O. Sulphide tolerance of the marine nematode Oncholaimus campylocercoides—A result of internal sulphur formation? Mar. Ecol. Prog. Ser. 2000, 193, 251–259. [Google Scholar] [CrossRef]
- Hale, R.; Calosi, P.; Mcneill, L.; Mieszkowska, N.; Widdicombe, S. Predicted levels of future ocean acidifi cation and temperature rise could alter community structure and biodiversity in marine benthic communities. Oikos 2011, 120, 661–674. [Google Scholar] [CrossRef]
- Whiteley, N.M. Physiological and ecological responses of crustaceans to ocean acidification. Mar. Ecol. Prog. Ser. 2011, 430, 257–271. [Google Scholar] [CrossRef] [Green Version]
- Kerrison, P.; Hall-Spencer, J.M.; Suggett, D.J.; Hepburn, L.J.; Steinke, M. Assessment of pH variability at a coastal CO2 vent for ocean acidification studies. Estuar. Coast. Shelf Sci. 2011, 94, 129–137. [Google Scholar] [CrossRef] [Green Version]
- Vizzini, S.; Di Leonardo, R.; Costa, V.; Tramati, C.D.; Luzzu, F.; Mazzola, A. Trace element bias in the use of CO2 vents as analogues for low pH environments: Implications for contamination levels in acidified oceans. Estuar. Coast. Shelf Sci. 2013, 134, 19–30. [Google Scholar] [CrossRef]
- Widdicombe, S.; Dashfield, S.L.; McNeill, C.L.; Needham, H.R.; Beesley, A.; McEvoy, A.; Øxnevad, S.; Clarke, K.R.; Berge, J.A. Effects of CO2 induced seawater acidification on infaunal diversity and sediment nutrient fluxes. Mar. Ecol. Prog. Ser. 2009, 379, 59–75. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.; Chan, T.Y.; Chan, B.K.K. Molluscan diversity in shallow water hydrothermal vents off Kueishan Island, Taiwan. Mar. Biodivers. 2018, 48, 709–714. [Google Scholar] [CrossRef]
- Seibel, B.A.; Walsh, P.J. Biological impacts of deep-sea carbon dioxide injection inferred from indices of physiological performance. J. Exp. Biol. 2003, 206, 641–650. [Google Scholar] [CrossRef] [Green Version]
- Fabricius, K.E.; Langdon, C.; Uthicke, S.; Humphrey, C.; Noonan, S.; De’ath, G.; Okazaki, R.; Muehllehner, N.; Glas, M.S.; Lough, J.M. Losers and winners in coral reefs acclimatized to elevated carbon dioxide concentrations. Nat. Clim. Chang. 2011, 1, 165. [Google Scholar] [CrossRef]
- Somerfield, P.J.; Dashfield, S.L.; Warwick, R.M. Three-dimensional spatial structure: Nematodes in a sandy tidal flat. Mar. Ecol. Prog. Ser. 2007, 336, 177–186. [Google Scholar] [CrossRef] [Green Version]
- Dashfield, S.L.; Somerfield, P.J.; Widdicombe, S.; Austen, M.C.; Nimmo, M. Impacts of ocean acidification and burrowing urchins on within-sediment pH profiles and subtidal nematode communities. J. Exp. Mar. Bio. Ecol. 2008, 365, 46–52. [Google Scholar] [CrossRef]
- Bevilacqua, S.; Sandulli, R.; Plicanti, A.; Terlizzi, A. Taxonomic distinctness in Mediterranean marine nematodes and its relevance for environmental impact assessment. Mar. Pollut. Bull. 2012, 64, 1409–1416. [Google Scholar] [CrossRef]
- Semprucci, F.; Balsamo, M.; Appolloni, L.; Sandulli, R. Assessment of ecological quality status along the Apulian coasts (eastern Mediterranean Sea) based on meiobenthic and nematode assemblages. Mar. Biodivers. 2018, 48, 105–115. [Google Scholar] [CrossRef]

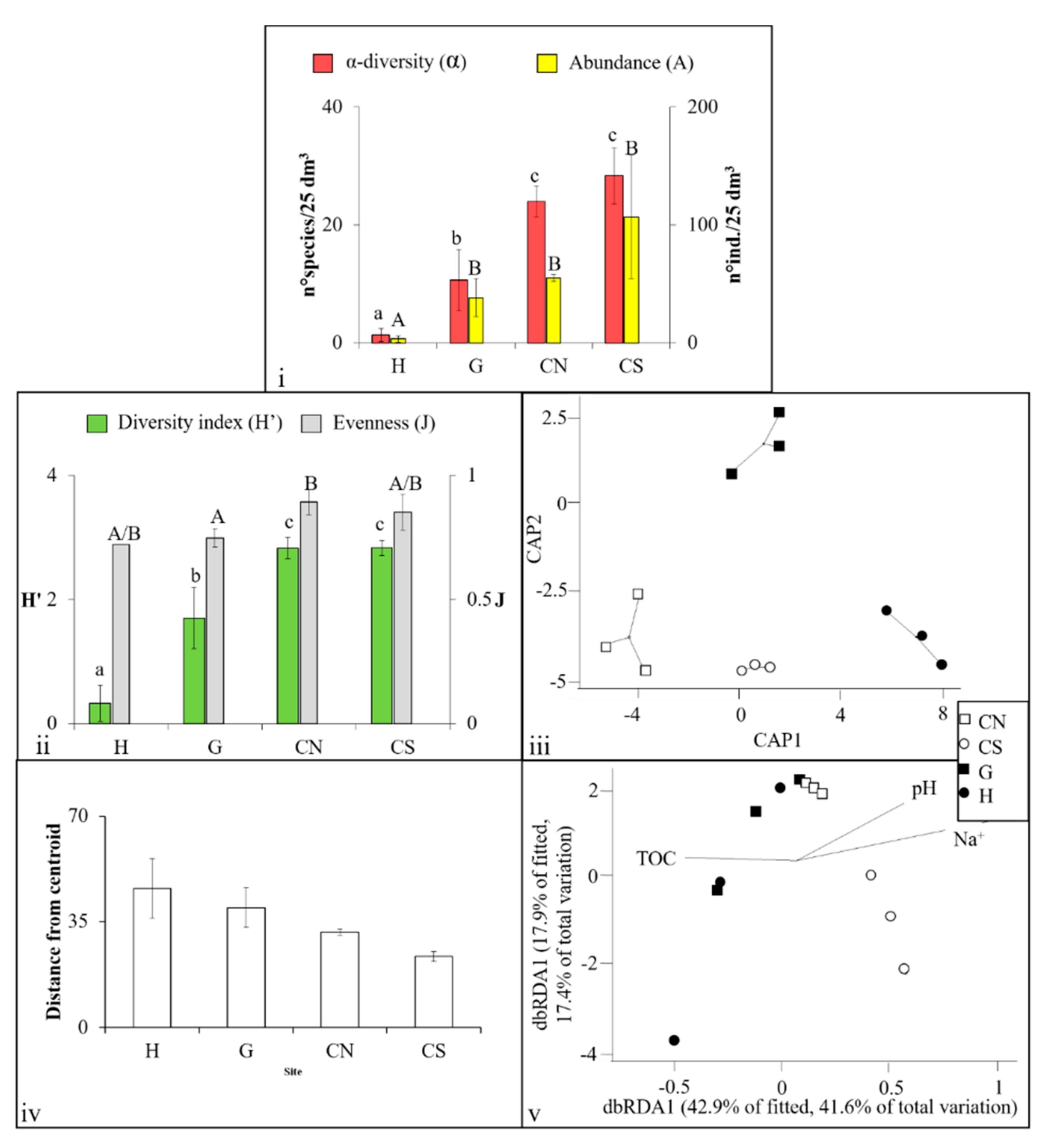
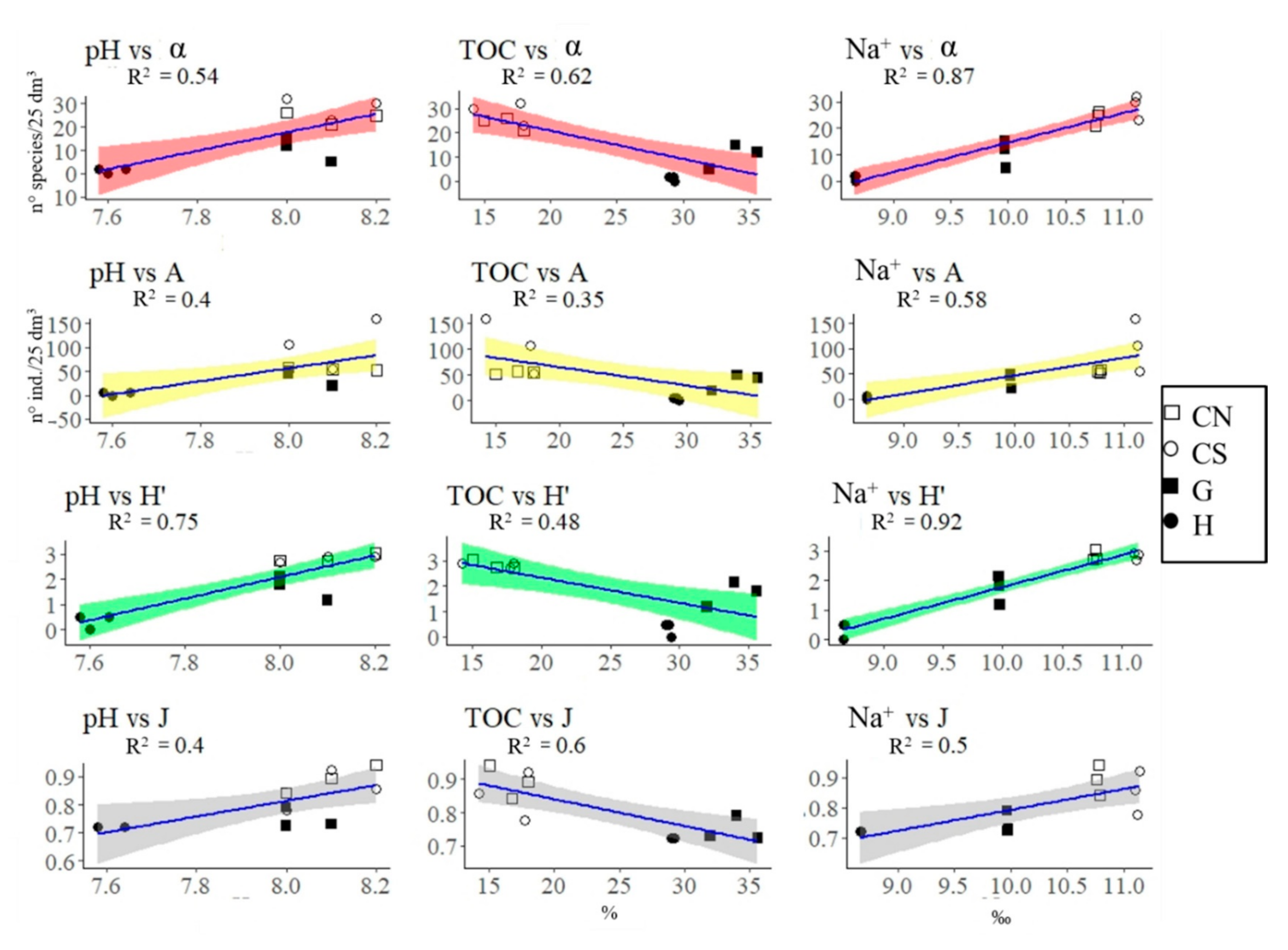
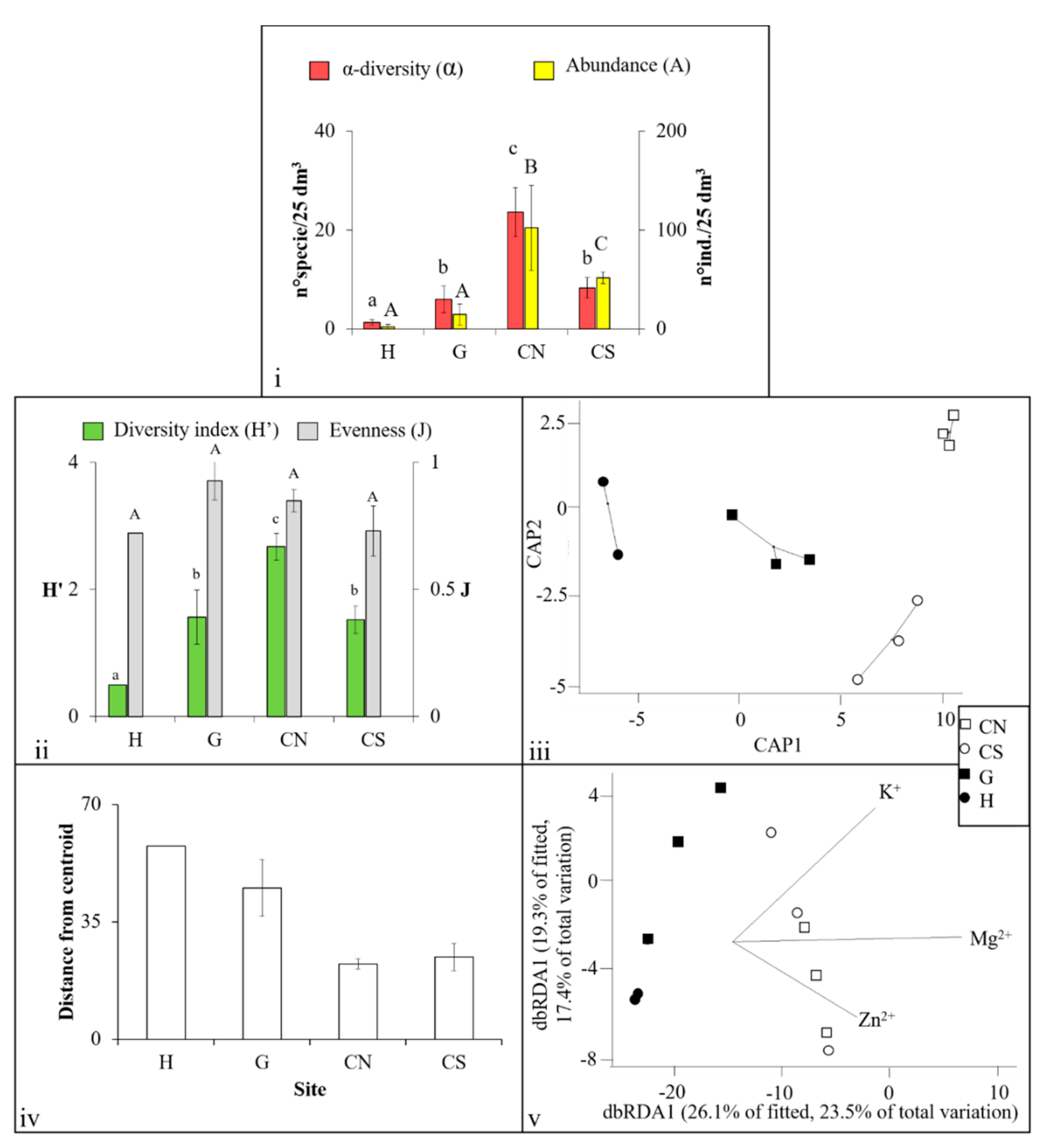
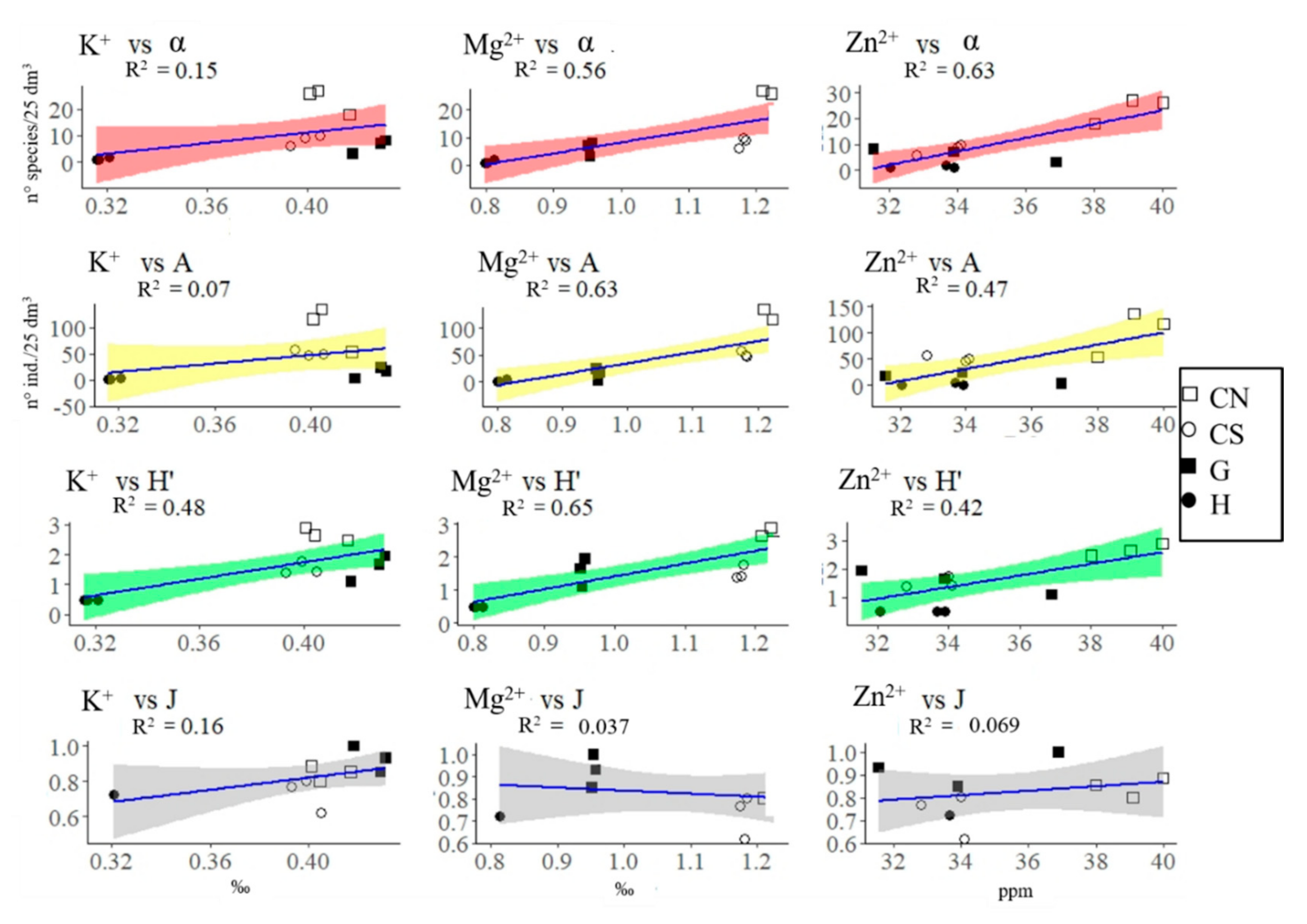

| Variables | H | G | CN | CS | Measurement Methods (for Further Information on Methodology See [36]) | |
|---|---|---|---|---|---|---|
| Depth | 12.7 | 9.7 | 8.9 | 11.9 | Scuba computer | |
| Sediment variables | ||||||
| Temperatures (°C) | 37.53 ± 2.28 | 29.1 ± 2.81 | 21.8 ± 0.2 | 21.8 ± 0.2 | In situ by underwater thermometer | |
| pH | 7.56 ± 0.05 | 8 ± 0.01 | 8.1 ± 0.03 | 8.1 ± 0.01 | pH evaluation (pH/ORP Meter, HI98171, and probe HI 1230, Hanna Instruments) | |
| Gravel (%) | 7.41 | 3.96 | 17.84 | 21.52 | Sediment was sieved over a series of 11 sieves with mesh size ranging from 1 cm to 63 mm [38]. Fractions were dried in an oven at 60 °C for 48 h and weighed; TOC was determined according to [39] and expressed as % of sediment. | |
| Sand (%) | 90.12 | 93.4 | 79.6 | 76.67 | ||
| Mud (%) | 2.47 | 2.64 | 2.57 | 1.81 | ||
| TOC (%) | 34.78 | 30.03 | 17.05 | 18.14 | ||
| Interstitial water variables | ||||||
| Ions (‰) | Na+ | 8.66826 ± 0.0045 | 9.97321 ± 0.0105 | 10.776925 ± 0.0125 | 11.120825 ± 0.0187 | ICS1100 ion chromatographic system [40]; anions were detected with an AS22 column working with a cell volume of 100 mL and a solution of 3.5 mM of sodium carbonate/bicarbonate as eluent, while cations were determined with a CS12A column working with a cell volume of 25 mL and 20 mM methanesulfonic acid solution as eluent. |
| K+ | 0.317855 ± 0.0076 | 0.42634 ± 0.0102 | 0.407405 ± 0.0085 | 0.399125 ± 0.0062 | ||
| Mg2+ | 0.805 ± 0.0073 | 0.954705 ± 0.0038 | 1.219475 ± 0.0088 | 1.1795 ± 0.0052 | ||
| Ca2+ | 0.3275 ± 0.0083 | 0.4722 ± 0.0095 | 0.50329 ± 0.0092 | 0.385725 ± 0.0073 | ||
| Cl− | 19.512965 ± 0.0205 | 23.726385 ± 0.0157 | 26.0395 ± 0.0175 | 25.97656 ± 0.0186 | ||
| NO3− | 0.02877 ± 0.00006 | 0.0255 ± 0.00006 | 0.02607 ± 0.00003 | 0.02568 ± 0.00032 | ||
| SO42− | 3.1523 ± 0.0036 | 2.6585 ± 0.0065 | 3.36988 ± 0.0031 | 3.8885 ± 0.0059 | ||
| S2− | n.d | 130.58 | n.d | n.d | ||
| Metals (ppb) | Zn2+ | 33.66 ± 0.51 | 34.56 ± 3.86 | 39.09 ± 0.50 | 33.65 ± 0.50 | |
| Pb2+ | 62.02 ± 0.16 | 31.29 ± 0.52 | 18.31 ± 0.60 | 62.02 ± 0.16 | ||
| Cd2+ | 4.42 ± 0.19 | n.d. | n.d. | 4.42 ± 0.18 | ||
| Cu2+ | n.d. | 8.88 ± 0.21 | 5.25 ± 0.21 | n.d. | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Appolloni, L.; Zeppilli, D.; Donnarumma, L.; Baldrighi, E.; Chianese, E.; Russo, G.F.; Sandulli, R. Seawater Acidification Affects Beta-Diversity of Benthic Communities at a Shallow Hydrothermal Vent in a Mediterranean Marine Protected Area (Underwater Archaeological Park of Baia, Naples, Italy). Diversity 2020, 12, 464. https://doi.org/10.3390/d12120464
Appolloni L, Zeppilli D, Donnarumma L, Baldrighi E, Chianese E, Russo GF, Sandulli R. Seawater Acidification Affects Beta-Diversity of Benthic Communities at a Shallow Hydrothermal Vent in a Mediterranean Marine Protected Area (Underwater Archaeological Park of Baia, Naples, Italy). Diversity. 2020; 12(12):464. https://doi.org/10.3390/d12120464
Chicago/Turabian StyleAppolloni, Luca, Daniela Zeppilli, Luigia Donnarumma, Elisa Baldrighi, Elena Chianese, Giovanni Fulvio Russo, and Roberto Sandulli. 2020. "Seawater Acidification Affects Beta-Diversity of Benthic Communities at a Shallow Hydrothermal Vent in a Mediterranean Marine Protected Area (Underwater Archaeological Park of Baia, Naples, Italy)" Diversity 12, no. 12: 464. https://doi.org/10.3390/d12120464
APA StyleAppolloni, L., Zeppilli, D., Donnarumma, L., Baldrighi, E., Chianese, E., Russo, G. F., & Sandulli, R. (2020). Seawater Acidification Affects Beta-Diversity of Benthic Communities at a Shallow Hydrothermal Vent in a Mediterranean Marine Protected Area (Underwater Archaeological Park of Baia, Naples, Italy). Diversity, 12(12), 464. https://doi.org/10.3390/d12120464








