A Survey from 2015 to 2019 to Investigate the Occurrence of Pesticide Residues in Dead Honeybees and Other Matrices Related to Honeybee Mortality Incidents in Italy
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Chemicals
2.3. Sample Preparation
2.4. LC-MS/MS Analysis
2.5. GC-MS/MS Analysis
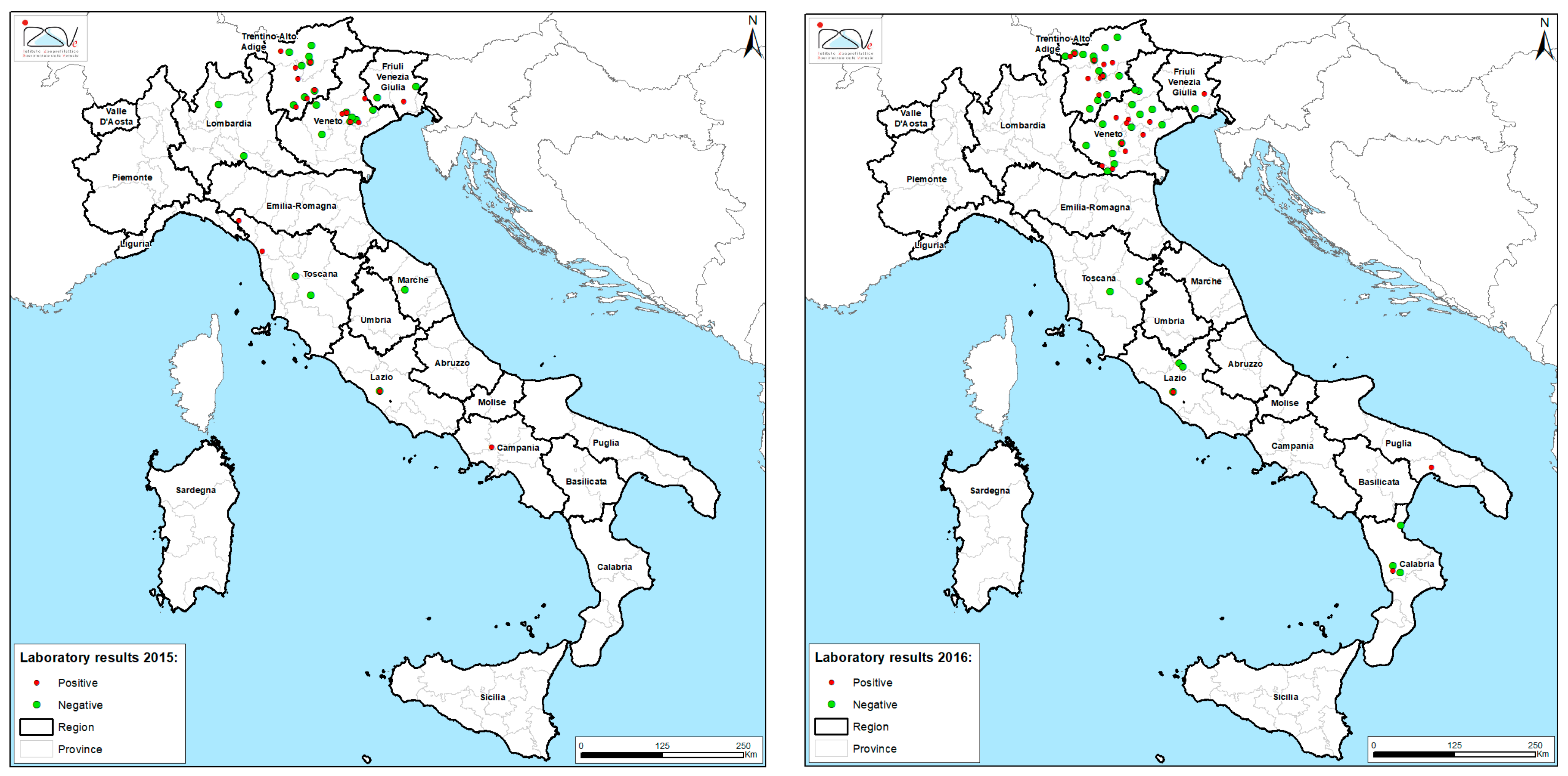
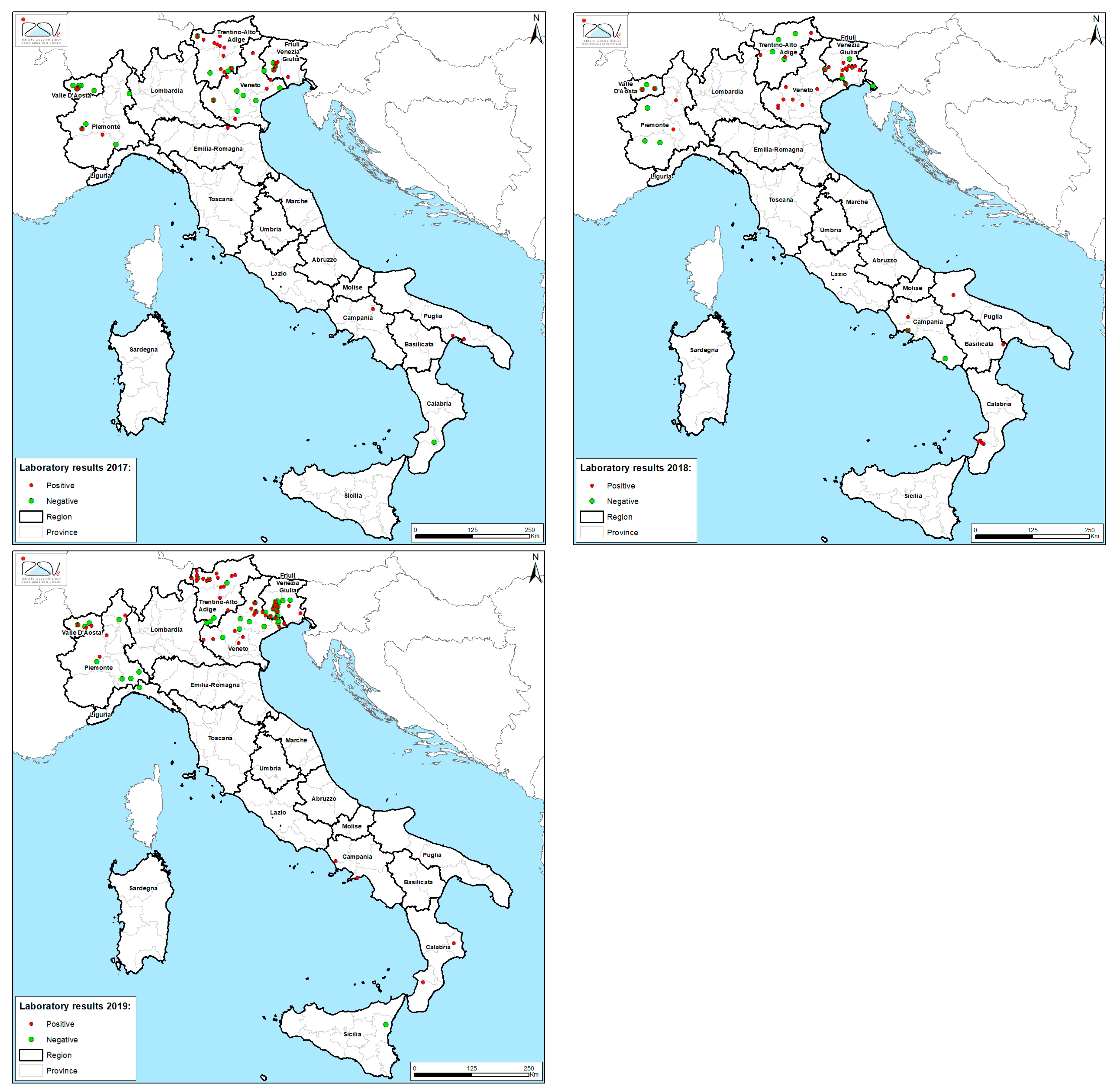
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Active Substances | Substance Group | Pesticide Type a | Detection Method |
|---|---|---|---|
| Acetamiprid | neonicotinoid | IN | HPLC-MS |
| Acrinathrin | pyrethroid | IN-AC | GC-ECD |
| Aldicarb * | carbamate | IN-AC-NE | HPLC-MS |
| Aldicarb-sulfone (Aldicarb metabolite) * | carbamate | IN-AC-NE | HPLC-MS |
| Aldicarb-sulfoxide (Aldicarb metabolite) * | carbamate | IN-AC-NE | HPLC-MS |
| Alpha-Endosulfan * | organochlorine | IN-AC | GC-ECD |
| Azoxystrobin | strobilurin | FU | HPLC-MS |
| Benalaxyl | Acylalanine | FU | HPLC-MS |
| Beta-Endosulfan * | organochlorine | IN-AC | GC-ECD |
| Bifenthrin | pyrethroid | IN-AC | GC-ECD |
| Bitertanol * | triazole | FU | HPLC-MS |
| Boscalid | carboxamide | FU | HPLC-MS |
| Bromopropylate * | benzilate | AC | HPLC-MS |
| Bupirimate | pyrimidinol | FU | HPLC-MS |
| Captan | phtalimide | FU-BA | GC-ECD |
| Carbaryl * | carbamate | IN-PG | HPLC-MS |
| Carbendazim | benzimidazole | FU | HPLC-MS |
| Carbofuran * | carbamate | IN-AC-NE | HPLC-MS |
| Carbofuran-3-hydroxy (Carbofuran metabolite) * | carbamate | IN-AC-NE | HPLC-MS |
| Carbofuran-3-keto (Carbofuran metabolite) * | carbamate | IN-AC-NE | HPLC-MS |
| Carboxine | oxathiin | FU | HPLC-MS |
| Chlorfenvinphos * | organophosphate | IN-AC | GC-ECD |
| Chloridazon | pyridazinone | HB | HPLC-MS |
| Chlormequat chloride | quaternary ammonium | PG | HPLC-MS |
| Chlorothalonil | chloronitrile | FU | GC-ECD |
| Chlorpropham | carbamate | HB-PG | GC-ECD |
| Chlorpyrifos | organophosphate | IN | GC-ECD |
| Chlorpyrifos-methyl | organophosphate | IN-AC | HPLC-MS |
| Clomazone | isoxazolidinone | HB | HPLC-MS |
| Clothianidin | neonicotinoid | IN | HPLC-MS |
| Coumaphos * | organophosphate | IN-AC | GC-ECD |
| Cyfluthrin-beta | pyretroid | IN | GC-ECD |
| Cyhalothrin * | pyretroid | IN | GC-ECD |
| Cypermethrin | pyretroid | IN | GC-ECD |
| Cyproconazole | triazole | FU | HPLC-MS |
| Cyprodinil | anilinopyrimidine | FU | HPLC-MS |
| Deltamethrin | pyretroid | IN | GC-ECD |
| Diflubenzuron | benzoylurea | IN | HPLC-MS |
| Dimethoate | organophosphate | IN-AC | HPLC-MS |
| Dimethomorph | morpholine | FU | HPLC-MS |
| Dinotefuran | neonicotinoid | IN | HPLC-MS |
| Dithianon | quinone | FU | HPLC-MS |
| Dodemorph | morpholine (isomer mix) | FU | HPLC-MS |
| Dodine | guanidine | FU | HPLC-MS |
| Endosulfan sulfate | organochlorine | IN-AC | GC-ECD |
| Esfenvalerate | pyretroid | IN | GC-ECD |
| Ethoprophos | organophosphate | IN-NE | GC-ECD |
| Etofenprox | pyretroid | IN | HPLC-MS |
| Etoxazole | diphenyl oxazoline | AC | HPLC-MS |
| Etridiazole | aromatic hydrocarbon | FU | GC-ECD |
| Fenamidone | imidazole | FU | HPLC-MS |
| Fenamiphos | organophosphate | NE | HPLC-MS |
| Fenamiphos sulfone (Fenamiphos metabolite) | organophosphate | NE | HPLC-MS |
| Fenamiphos sulfoxide (Fenamiphos metabolite) | organophosphate | NE | HPLC-MS |
| Fenarimol * | pyrimidine | FU | HPLC-MS |
| Fenazaquin | quinazoline | IN-AC | HPLC-MS |
| Fenbuconazole | triazole | FU | HPLC-MS |
| Fenothiocarb * | thiocarbamate | AC | HPLC-MS |
| Fenoxycarb | carbamate | IN | HPLC-MS |
| Fenpropidin | piperidine | FU | HPLC-MS |
| Fenpropimorph | morpholine | FU | HPLC-MS |
| Fenpyroximate | pyrazole | IN-AC | HPLC-MS |
| Fenvalerate * | pyretroid | IN-AC | GC-ECD |
| Fipronil | phenylpyrazole | IN | GC-ECD |
| Flazasulfuron | sulfonylurea | HB | HPLC-MS |
| Fluazifop-P-butyl | aryloxyphenoxypropionate | HB | HPLC-MS |
| Flufenacet | oxyacetamide | HB | HPLC-MS |
| Flufenoxuron * | benzoylurea | IN-AC | HPLC-MS |
| Fludioxonil | phenylpyrrole | FU | HPLC-MS |
| Fluopicolide | benzamide | FU | GC-ECD |
| Fluopyram | benzamide, pyramide | FU-NE | GC-ECD |
| Fluquinconazole | triazole | FU | GC-ECD |
| Flusilazole | triazole | FU | HPLC-MS |
| Flutriafol | triazole | FU | HPLC-MS |
| Folpet | phtalimide | FU | GC-ECD |
| Forchlorfenuron | phenylurea | PG | HPLC-MS |
| Formetanate | formamidine | IN-AC | HPLC-MS |
| Fosthiazate | organophosphate | IN-NE | HPLC-MS |
| Imazalil | imidazole | FU | HPLC-MS |
| Imazosulfuron | sulfonylurea | HB | HPLC-MS |
| Imidacloprid | neonicotinoid | IN | HPLC-MS |
| Indoxacarb | oxadiazine | IN | GC-ECD |
| Iprodione | dicarboximide | FU | GC-ECD |
| Iprovalicarb | carbamate | FU | HPLC-MS |
| Isopyrazam | pyrazole | FU | GC-ECD |
| Kresoxim-methyl | strobilurin | FU | GC-ECD |
| Lambda-cyhalothrin | pyrethroid | IN | GC-ECD |
| Linuron | urea | HB | HPLC-MS |
| Malaoxon (Malathion metabolite) ° | organophosphate | IN-AC | HPLC-MS |
| Malathion | organophosphate | IN-AC | HPLC-MS |
| Mepanipyrim | anilinopyrimidine | FU | HPLC-MS |
| Metalaxyl | phenylamide | FU | HPLC-MS |
| Metalaxyl-M | phenylamide | FU | HPLC-MS |
| Metamitron | triazinone | HB | HPLC-MS |
| Metazachlor | chloroacetamide | HB | HPLC-MS |
| Methiocarb | carbamate | IN-MO-RE | HPLC-MS |
| Methiocarb sulfoxide (Methiocarb metabolite) | carbamate | IN-MO-RE | HPLC-MS |
| Methomyl | carbamate | IN-AC | HPLC-MS |
| Metolachor | chloroacetanilide | HB | HPLC-MS |
| Metribuzin | triazinone | HB | HPLC-MS |
| Myclobutanil | triazole | FU | GC-ECD |
| Nytempiram ° | neonicotinoid | IN | HPLC-MS |
| Omethoate (Dimethoate metabolite) * | organophosphate | IN-AC | HPLC-MS |
| Oxamyl | carbamate | IN-AC-NE | HPLC-MS |
| Penconazole | triazole | FU | GC-ECD |
| Pencycuron | phenylurea | FU | HPLC-MS |
| Pendomethalin | dinitroaniline | HB | GC-ECD |
| Permethrin * | pyretroid | IN | GC-ECD |
| Phosmet | organophosphate | IN-AC | GC-ECD |
| Piperonyl butoxide ° | safrole | SY | HPLC-MS |
| Pirimicarb | carbamate | IN | HPLC-MS |
| Pirimicarb-desmethyl (Pirimicarb metabolite) | carbamate | IN | HPLC-MS |
| Pirimiphos-methyl | organophosphate | IN-AC | HPLC-MS |
| Prochloraz | imidazole | FU | HPLC-MS |
| Procymidone * | dicarboximide | FU | GC-ECD |
| Propamocarb | carbamate | FU | HPLC-MS |
| Propiconazole * | triazole | FU | HPLC-MS |
| Propyzamide | benzamide | HB | HPLC-MS |
| Pyraclostrobin | strobilurin | FU | HPLC-MS |
| Pyridaben | pyridazinone | IN-AC | HPLC-MS |
| Pyrimethanil | anilinopyrimidine | FU | HPLC-MS |
| Pyriproxyfen | unclassified | IN | HPLC-MS |
| Quinoxyfen * | quinoline | FU | GC-ECD |
| Rimsulfuron | sulfonylurea | HB | HPLC-MS |
| Rotenone * | isoflavones | IN | HPLC-MS |
| Spirodiclofen | tetronic acid | IN-AC | HPLC-MS |
| Spirotetramat | tetramic acid | IN | HPLC-MS |
| Tau-Fluvalinate | pyretroid | IN-AC | GC-ECD |
| Tebuconazole | triazole | FU | HPLC-MS |
| Tebufenozide | diacylhydrazine | IN | HPLC-MS |
| Tebufenpyrad | pyrazole | AC | HPLC-MS |
| Teflubenzuron | benzoylurea | IN | HPLC-MS |
| Tefluthrin | pyrethroid | IN | HPLC-MS |
| Tepraloxydim | cyclohexanedione | HB | HPLC-MS |
| Terbuthylazine | triazine | HB | HPLC-MS |
| Tetraconazole | triazole | FU | HPLC-MS |
| Tetramethrin * | pyrethroid | IN | GC-ECD |
| Thiabendazole | benzimidazole | FU | HPLC-MS |
| Thiacloprid | neonicotinoid | IN | HPLC-MS |
| Thiamethoxam | neonicotinoid | IN | HPLC-MS |
| Thifensulfuron-methyl | solfonylurea | HB | HPLC-MS |
| Thiobencarb * | thiocarbamate | HB | HPLC-MS |
| Thiodicarb * | carbamate | IN | HPLC-MS |
| Thiram | dithiocarbamate | FU | HPLC-MS |
| Thiophanate-methyl | benzimidazole | FU | HPLC-MS |
| Tolclofos-methyl | chlorophenyl | FU | GC-ECD |
| Tribenuron-methyl | solfonylurea | HB | HPLC-MS |
| Trifloxystrobin | strobulirin | FU | GC-ECD |
| Triflumuron | benzoylurea | IN | HPLC-MS |
| Triticonazole | triazole | FU | HPLC-MS |
References
- Goulson, D.; Nicholls, E.; Botías, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 2015, 347, 1255957. [Google Scholar] [CrossRef] [PubMed]
- Porrini, C.; Mutinelli, F.; Bortolotti, L.; Granato, A.; Laurenson, L.; Roberts, K.; Gallina, A.; Silvester, N.; Medrzycki, P.; Renzi, T.; et al. The status of honey bee health in Italy: Results from the nationwide bee monitoring network. PLoS ONE 2016, 11, e0155411. [Google Scholar] [CrossRef] [PubMed]
- Gallai, N.; Salles, J.; Settele, J.; Vaissière, B.E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 2009, 68, 810–821. [Google Scholar] [CrossRef]
- Garibaldi, L.A.; Aizen, M.A.; Klein, A.M.; Cunningham, S.A.; Harder, L.D. Global growth and stability of agricultural yield decrease with pollinator dependence. Proc. Natl. Acad. Sci. USA 2011, 108, 5909–5914. [Google Scholar] [CrossRef]
- Jacques, A.; Laurent, M.; EPILOBEE Consortium; Ribière-Chabert, M.; Saussac, M.; Bougeard, S.; Budge, G.E.; Hendrikx, P.; Chauzat, M.P. A pan-European epidemiological study reveals honey bee colony survival depends on beekeeper education and disease control. PLoS ONE 2017, 12, e0172591. [Google Scholar] [CrossRef]
- Pham-Delègue, M.H. Abeilles; Editions de La Martinière: Paris, France, 1998; p. 47. [Google Scholar]
- Lambert, O.; Piroux, M.; Puyo, S.; Thorin, C.; Larhantec, M.; Delbac, F.; Poliquen, H. Bees, honey and pollen as sentinels for lead environmental contamination. Environ. Pollut. 2012, 170, 33–43. [Google Scholar] [CrossRef]
- Bonmatin, J.M.; Giorio, C.; Girolami, V.; Goulson, D.; Kreutzweiser, D.P.; Krupke, C.; Liess, M.; Long, E.; Marzaro, M.; Mitchell, E.A.; et al. Environmental fate and exposure; neonicotinoids and fipronil. Environ. Sci. Poll. Res. Int. 2015, 22, 35–67. [Google Scholar] [CrossRef]
- Greatti, M.; Barbattini, R.; Stravisi, A.; Sabatini, A.G.; Rossi, S. Presence of the a.i. imidacloprid on vegetation near corn fields sown with Gaucho® dressed seeds. Bull. Insectol. 2006, 59, 99–103. [Google Scholar]
- Rete Rurale. Available online: https://www.reterurale.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/1369 (accessed on 31 October 2019).
- Porrini, C.; Sgolastra, F.; Renzi, T. Bee Emergency Service Team (BEST): Bee losses and mortality reports in Italy (2012–2014). Bull. Insectol. 2014, 67, 294. [Google Scholar]
- Mutinelli, F.; Sgolastra, F.; Porrini, C.; Medrzycki, P.; Bortolotti, L.; Granato, A.; Gallina, A.; Lodesani, M. The status of honey bee health in Italy: Results from the nationwide monitoring network. In Proceedings of the XI European Congress of Entomology, Naples, Italy, 2–6 July 2018; p. 318. [Google Scholar]
- EC. Regulation (EC) No 1107/2009 of the European Parliament and of the Council of 21 October 2009 Concerning the Placing of Plant Protection Products on the Market and Repealing Council Directives 79/117/EEC and 91/414/EEC. OJ EU 2009, L 309; EC: Brussels, Belgium, 2009; pp. 1–50. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32009R1107 (accessed on 31 October 2019).
- EC. Directive 2009/128/EC of the European Parliament and of the Council of 21 October 2009 Establishing a Framework for Community Action to Achieve the Sustainable Use of Pesticides. OJ EU 2009, L 309; EC: Brussels, Belgium, 2009; pp. 71–86. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=celex%3A32009L0128 (accessed on 31 October 2019).
- EC. Commission Directive 2010/21/EU of 12 March 2010 Amending Annex I to Council Directive 91/414/EEC as Regards the Specific Provisions Relating to Clothianidin, Thiamethoxam, Fipronil and Imidacloprid. OJ EU 2010, L 65; EC: Brussels, Belgium, 2010; pp. 27–30. Available online: https://eur-lex.europa.eu/eli/dir/2010/21/oj (accessed on 31 October 2019).
- Italian Ministry of Health. Linee Guida Per la Gestione Delle Segnalazioni di Moria o Spopolamento Degli Alveari Connesse All’utilizzo di fitoFarmaci [Guideline for the Management of Reporting of Mortality or Dwindling of Beehives Linked to the Use of Pesticides] 0016168-31/07/2014-DGSAF-COD_UO-P; Italian Ministry of Health: Rome, Italy, 2014. Available online: http://www.izsvenezie.it/linee-guida-per-la-gestione-delle-segnalazioni-di-moriao-spopolamento-degli-alveari-connesse-allutilizzo-di-fitofarmaci (accessed on 31 October 2019).
- Anastassiades, M.; Lehotay, S.J.; Stajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and ‘dispersive SPE’ for the determination of pesticide residues in produce. JAOAC Int. 2003, 86, 412–431. Available online: http://www.ingentaconnect.com/contentone/aoac/jaoac/2003/00000086/00000002/art00023 (accessed on 31 October 2019).
- Lambert, O.; Piroux, M.; Puyo, S.; Thorin, C.; L’Hostis, M.; Wiest, L.; Bulete, A.; Delbac, F.; Pouliquen, H. Widespread occurrence of chemical residues in beehive matrices from apiaries located in different landscapes of western France. PLoS ONE 2013, 8, e67007. [Google Scholar] [CrossRef] [PubMed]
- Calatayud-Vernich, P.; Calatayud, F.; Simò, E.; Picò, Y. Pesticide residues in honey bees, pollen and beeswax: Assessing beehive exposure. Environ. Pollut. 2018, 241, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Kiljanek, T.; Niewiadowska, A.; Gaweł, M.; Semeniuk, S.; Borzecka, M.; Posyniak, A.; Pohorecka, K. Multiple pesticide residues in live and poisoned honeybees—Preliminary exposure assessment. Chemosphere 2017, 175, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Wiest, L.; Bulete, A.; Giroud, B.; Fratta, C.; Amic, S.; Lambert, O.; Pouliquen, H.; Arnaudguilhem, C. Multi-residue analysis of 80 environmental contaminants in honeys, honeybees and pollens by one extraction procedure followed by liquid and gas chromatography coupled with mass spectrometric detection. J. Chromatogr. A 2011, 1218, 5743–5756. [Google Scholar] [CrossRef] [PubMed]
- Mullin, C.A.; Frazier, M.; Frazier, J.L.; Ashcraft, S.; Simonds, R.; van Engelsdorp, D.; Pettis, J.S. High levels of miticides and agrochemicals in North American apiaries: Implications for honey bee health. PLoS ONE 2010, 5, e9754. [Google Scholar] [CrossRef] [PubMed]
- EU. Commission Implementing Regulation (EU) No. 485/2013 of 24 May 2013 Amending Implementing Regulation (EU) No 540/2011, as Regards the Conditions of Approval of the Active Substances Clothianidin, Thiamethoxam and Imidacloprid, and Prohibiting the Use and Sale of Seeds Treated with Plant Protection Products Containing Those Active Substances. OJ EU, 2013, L 139; EU: Brussels, Belgium, 2013; pp. 12–26. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2013:139:0012:0026:EN:PDF (accessed on 31 October 2019).
- EU. Commission Implementing Regulation (EU) No. 781/2013 of 14 August 2013 Amending Implementing Regulation (EU) No 540/2011, as Regards the Conditions of Approval of the Active Substance Fipronil, and Prohibiting the Use and Sale of Seeds Treated with Plant Protection Products Containing this Active Substance. OJ EU 2013, L 219; EU: Brussels, Belgium, 2013; pp. 22–25. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2013:219:0022:0025:EN:PDF (accessed on 31 October 2019).
- Mitchell, E.A.D.; Mulhauser, B.; Mulot, M.; Mutabazi, A.; Glauser, G.; Aebi, A. A worldwide survey of neonicotinoids in honey. Science 2017, 358, 109–111. [Google Scholar] [CrossRef]
- Calatayud-Vernich, P.; Calatayud, F.; Simó, E.; Pascual Aguilar, J.A.; Picó, Y. A two-year monitoring of pesticide hazard in-hive: High honey bee mortality rates during insecticide poisoning episodes in apiaries located near agricultural settings. Chemosphere 2019, 232, 471–480. [Google Scholar] [CrossRef]
- Gaweł, M.; Kiljanek, T.; Niewiadowska, A.; Semeniuk, S.; Goliszek, M.; Burek, O.; Posyniak, A. Determination of neonicotinoids and 199 other pesticide residues in honey by liquid and gas chromatography coupled with tandem mass spectrometry. Food Chem. 2019, 282, 36–47. [Google Scholar] [CrossRef]
- Simon-Delso, N.; Martin, G.S.; Bruneau, E.; Minsart, L.-A.; Mouret, C.; Hautier, L. Honeybee colony disorder in crop areas: The role of pesticides and viruses. PLoS ONE 2014, 9, e103073. [Google Scholar] [CrossRef]
- Christen, V.; Krebs, J.; Fent, K. Fungicides chlorothanolin, azoxystrobin and folpet induce transcriptional alterations in genes encoding enzymes involved in oxidative phosphorylation and metabolism in honey bees (Apis mellifera) at sublethal concentrations. J. Hazard. Mater. 2019, 377, 215–226. [Google Scholar] [CrossRef]
- Johnson, R.M.; Dahlgren, L.; Siegfried, B.D.; Ellis, M.D. Acaricide, fungicide and drug interactions in honey bees (Apis mellifera). PLoS ONE 2013, 8, e54092. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Bayo, F.; Goulson, D.; Pennacchio, F.; Nazzi, F.; Goka, K.; Desneux, N. Are bee diseases linked to pesticides?—A brief review. Environ. Int. 2016, 89, 7–11. [Google Scholar]
- Sgolastra, F.; Medrzycki, P.; Bortolotti, L.; Renzi, M.T.; Tosi, S.; Bogo, G.; Teper, D.; Porrini, C.; Molowny-Horas, R.; Bosch, J. Synergistic mortality between a neonicotinoid insecticide and an ergosterol-biosynthesis-inhibiting fungicide in three bee species. Pest Manag. Sci. 2017, 73, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
- Martinello, M.; Baratto, C.; Manzinello, C.; Piva, E.; Borin, A.; Toson, M.; Granato, A.; Boniotti, M.B.; Gallina, A.; Mutinelli, F. Spring mortality in honey bees in northeastern Italy: Detection of pesticides and viruses in dead honey bees and other matrices. J. Apic. Res. 2017, 56, 239–254. [Google Scholar] [CrossRef]
- Di Prisco, G.; Cavaliere, V.; Annoscia, D.; Varricchio, P.; Caprio, E.; Nazzi, F.; Gargiulo, G.; Pennacchio, F. Neonicotinoid clothianidin adversely affects insect immunity and promotes replication of a viral pathogen in honey bees. Proc. Nat. Acad. Sci. USA 2013, 110, 18466–18471. [Google Scholar] [CrossRef]
- Chauzat, M.; Faucon, J. Pesticide residues in beeswax samples collected from honey bee colonies (Apis mellifera L.) in France. Pest Manag. Sci. 2007, 63, 1100–1106. [Google Scholar] [CrossRef]
- EU Pesticides Database. Available online: https://ec.europa.eu/food/plant/pesticides/eu-pesticides-database/public/?event=homepage&language=EN (accessed on 19 December 2019).
- Kochansky, J.; Wilzer, K.; Feldlaufer, M. Comparison of the transfer of coumaphos from beeswax into syrup and honey. Apidologie 2001, 32, 119–125. [Google Scholar] [CrossRef]
- Mutinelli, F. Veterinary medicinal products to control Varroa destructor in honey bee colonies (Apis mellifera) and related EU legislation—An update. J. Apic. Res. 2016, 55, 78–88. [Google Scholar] [CrossRef]
- EU. Regulation (EU) No 528/2012 of the European Parliament and of the Council of 22 May 2012 Concerning the Making Available on the Market and Use of Biocidal Products. OJ EU, 2012, L167; EU: Brussels, Belgium, 2012; pp. 1–123. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32012R0528 (accessed on 31 October 2019).
- PPDB. Pesticide Properties Database. University of Hertfordshire. 2016. Available online: http://sitem.herts.ac.uk/aeru/ppdb/en/index.htm (accessed on 31 October 2019).
- Johnson, R.M.; Pollock, H.S.; Berenbaum, M.R. Synergistic interactions between in-hive miticides in Apis mellifera. J. Econ. Entomol. 2009, 102, 474–479. [Google Scholar] [CrossRef]
- Pilling, E.D.; Jepson, P.C. Synergism between EBI fungicides and a pyrethroid insecticide in the honeybee (Apis mellifera). Pestic. Sci. 1993, 39, 293–297. [Google Scholar] [CrossRef]
| Active Ingredient | Pesticide Type a | Substance Group | Honeybees | Other Matrices b | ||
|---|---|---|---|---|---|---|
| Prevalence (%) | Range (ng per Bee) | Prevalence (%) | Range (mg kg−1) | |||
| 3-ketocarbofuran | metabolite | carbamate | - | - | 0.7 | 0.04 |
| acetamiprid | I | neonicotinoid | 0.8 | 1.9–6.6 | 1.7 | 0.03–0.06 |
| acrinathrin | I-A | pyrethroid | 2.0 | 42.7–473 | 1.7 | 0.2–0.8 |
| azoxystrobin | F | strobilurin | 1.2 | 1.0–5.4 | 2.5 | 0.01–0.02 |
| boscalid | F | carboxamide | 1.2 | <LOQ -25.3 | 0.8 | 0.02 |
| bromopropylate | A | benzilate | 1.2 | 5.7–14.5 | - | - |
| bupirimate | F | pyrimidinol | 0.8 | 1.6–1.8 | - | - |
| carbendazim | F | benzimidazole | 0.4 | 12.9 | 0.8 | 0.04 |
| chlormequat chloride | PGR | quarternary ammonium compound | 0.4 | 1.9 | 0.8 | 0.02 |
| chlorothalonil | F | chloronitrile | 0.4 | 84.8 | - | - |
| chlorfenvinphos | I-A | organophosphate | 2.8 | <LOQ -162.0 | 7.6 | 0.01–0.4 |
| chlorpyrifos | I | organophosphate | 12.9 | 1.0–1688.8 | 8.5 | 0.01–0.5 |
| chlorpyrifos-mehyl | I-A | organophosphate | 6.8 | 1.1–316.0 | 1.7 | 0.01 |
| chlorpropham | I-PGR | carbamate | 0.8 | 7.2 | - | - |
| clomazone | H | isoxazolidinone | 0.4 | 1.1 | - | - |
| clothianidin | I | neonicotinoid | 3.6 | 1.0–17.0 | - | - |
| coumaphos | I | organothiophosphate | 1.6 | 1.5–50.0 | 10.2 | 0.01–0.7 |
| cymoxanil | F | cyanoacetamide oxime | - | - | 0.8 | 0.03 |
| cypermethrin | I | pyrethroid | 2.8 | 1.1–7602.0 | - | - |
| cyprodinil | F | anilinopyrimidine | 2.8 | <LOQ -484.5 | 0.8 | 1.1 |
| deltamethrin | I-M | pyrethroid | - | - | 3.4 | 0.05–0.1 |
| desmethyl-pirimicarb | I | carbamate | 0.4 | 1.7 | 2.5 | 0.03–0.1 |
| dimethoate | I-A | organophosphate | 3.2 | 23.3–1647.4 | 1.7 | |
| dimethomorph | F | morpholine | 3.2 | <LOQ -4.3 | 9.3 | 0.02–47.0 |
| dodine | F | guanidine | - | - | 1.7 | 0.01–0.2 |
| ethoprophos | I-N | organophosphate | 0.4 | 1.7 | 1.7 | 0.2 |
| etofenprox | I | pyrethroid | 6.0 | <LOQ -17.5 | 2.5 | 0.01–0.03 |
| fipronil | I | phenylpyrazole | 0.4 | 139.6 | 0.8 | 1.8 |
| fludioxonil | F | phenylpyrrole | 1.2 | 1.0–6.5 | 0.8 | 0.01 |
| fluopicolide | F | benzamide | 0.8 | 3.1 | - | - |
| fluopyram | F | benzamide, pyramide | 2.0 | 4.0–6.7 | - | - |
| flutriafol | F | triazol | 0.4 | 2.0 | - | - |
| folpet | F | phthalimide | 2.4 | 9.1–46.0 | 4.2 | 0.3–511.1 |
| imidacloprid | I | neonicotinoid | 11.6 | 1.2–402.6 | 3.4 | 0.02–4.1 |
| indoxacarb | I | oxadiazine | 0.8 | 2 | - | - |
| iprodione | F | dicarboximide | 1.2 | 3.0–96.3 | 0.8 | 0.03 |
| kresoxim-methyl | F-B | strobilurin | 0.8 | 2.0–4.2 | 0.8 | 0.01 |
| metalaxil | F | phenylamide | 2.0 | 1.0–9.0 | 5.1 | 0.04–0.4 |
| metalaxyl-M | F | phenylamide | - | - | 0.8 | 0.03 |
| methiocarb | I | carbamate | 4.8 | <LOQ -46.5 | 11.0 | 0.01–70.6 |
| methiocarb sulfoxide | metabolite | carbamate | 2.4 | 2.5–5.4 | 2.5 | 0.01–7.6 |
| methomyl | I-A | carbamate | 3.6 | 1.0–765.0 | 2.5 | 0.2–0.3 |
| metrafenone | F | benzophenone | 0.8 | 1.4–2.8 | 0.8 | 0.03 |
| metribuzin | H | triazinone | - | - | 0.8 | 0.08 |
| omethoate | I-A | organophosphate | 3.2 | 4.7–102.2 | - | - |
| penconazole | F | triazole | 6.4 | 1.0–90.0 | 2.5 | 0.01–0.02 |
| pendimethalin | H | dinitroaniline | 2.0 | 1.2–4.3 | 2.5 | 0.01–0.03 |
| permethrin | I | pyrethroid | 13.3 | 1.6–134,665.0 | 5.1 | 0.02–2.0 |
| phosmet | I-A | organophosphate | 3.6 | 1.0–280.8 | 1.7 | 0.2–0.7 |
| piperonil butoxide | - | cyclic aromatic | 11.2 | 1.0–66,827.0 | 11 | 0.01–2.3 |
| propamocarb | F | carbamate | 2.8 | 1.1–8.5 | 2.5 | 0.02–3.6 |
| propiconazole | F | triazole | 2.4 | 1.4–6.0 | - | - |
| pyraclostrobin | F | strobilurin | 0.4 | 6.7 | 1.7 | 0.01–0.3 |
| pyrimethanil | F | anilinopyrimidine | 4.4 | 1.0–89.0 | 6.8 | 0.02–0.8 |
| pyriproxyfen | I | unclassified | 0.8 | 3.7–5.9 | - | - |
| quinoxyfen | F | quinoline | 1.6 | 2.8–13.5 | 1.7 | 0.01 |
| rotenone | I-A | isoflavone | 0.8 | 1.5–2.4 | 0.8 | 0.04 |
| s-metolachlor | H | chloroacetamide | - | - | 0.8 | 0.6 |
| tau-fluvalinate | I-A | synthetic pyrethroid | 38.2 | 1.0–1018.0 | 53.4 | 0.02–95.9 |
| tefluthrin | I | pyrethroid | 3.6 | 1.1–33.1 | 4.2 | 0.07–1.6 |
| terbuthylazine | H | triazine | - | - | 4.2 | 0.02–1.0 |
| tetraconazole | F | triazole | - | - | 4.2 | 0.01–1.1 |
| tetramethrin | I | pyrethroid | 8.4 | 1.0–71,096.0 | 2.5 | 0.03–0.9 |
| thiacloprid | I | neonicotinoid | 4.0 | 1.1–9.5 | 1.7 | 0.01–0.04 |
| thiamethoxam | I | neonicotinoid | 1.6 | 1.8–33.6 | - | - |
| thifensulfuron-methyl | H | sulfonylurea | 0.4 | 2.1 | - | - |
| thiodicarb | I | carbamate | 0.8 | 2.0–5.0 | - | - |
| thiophanate-methyl | F | benzimidazole | 0.8 | 5.8–78.6 | 0.8 | 0.2 |
| tribenuron-methyl | H | sulfonylurea | 0.4 | 1.0 | - | - |
| trifloxystrobin | F | strobilurin | 0.8 | 8.1–19.9 | 0.8 | 1.4 |
| Year | n. of Samples | n. of Positive Samples (%) | n. of Pesticides Detected | Most Frequent | Prevalence (%) |
|---|---|---|---|---|---|
| 2015 | 68 | 28 (41) | 20 | fluvalinate | 50.0 |
| piperonyl butoxide | 25.0 | ||||
| cypermethrin | 14.3 | ||||
| chlorpyrifos | 14.3 | ||||
| 2016 | 112 | 47 (42) | 22 | chlorpyrifos | 25.5 |
| permethrin | 25.5 | ||||
| tetramethrin | 25.5 | ||||
| piperonil butoxide | 23.4 | ||||
| fluvalinate | 21.3 | ||||
| 2017 | 95 | 48 (50) | 22 | fluvalinate | 41.7 |
| chlorpyrifos | 37.5 | ||||
| methomyl | 16.7 | ||||
| 2018 | 85 | 63 (74) | 36 | fluvalinate | 34.9 |
| imidacloprid | 14.3 | ||||
| etofenprox | 12.7 | ||||
| methiocarb | 12.7 | ||||
| 2019 | 147 | 63 (43) | 34 | fluvalinate | 46.0 |
| chlorpyrifos | 15.9 | ||||
| imidacloprid | 14.3 | ||||
| Sum | 507 | 249 (50) | 63 | fluvalinate | 38.2 |
| permethrin | 13.3 | ||||
| chlorpyrifos | 12.9 |
| Year | n. of Samples | Matrix | n. of Positive Samples (%) | n. of Pesticides Detected | Most Frequent | Prevalence (%) |
|---|---|---|---|---|---|---|
| 2015 | 18 | 12 (67) | 12 | tau-fluvalinate | 83.3 | |
| tetraconazole | 25.0 | |||||
| 15 | comb | 10 (67) | 10 | tau-fluvalinate | 100.0 | |
| 1 | honey | 1 (100) | 2 | |||
| 2 | vegetable | 1 (50) | 3 | |||
| 2016 | 39 | 22 (56) | 15 | tau-fluvalinate | 45.5 | |
| metalaxyl | 13.6 | |||||
| permethrin | 13.6 | |||||
| 28 | comb | 15 (54) | 7 | tau-fluvalinate | 67 | |
| 3 | pollen | 2 (67) | 1 | metalaxyl | 100 | |
| 3 | honey | 0 | ||||
| 5 | vegetable | 3 (60) | 6 | |||
| 2017 | 23 | 13 (57) | 10 | tau-fluvalinate | 30.8 | |
| chlorpyrifos | 23.1 | |||||
| desmethyl-pirimicarb | 15.4 | |||||
| 11 | comb | 10 (91) | 9 | tau-fluvalinate | 60.0 | |
| 7 | pollen | 3 (43) | 3 | tau-fluvalinate | 66.7 | |
| 5 | honey | 0 | ||||
| 2018 | 36 | 30 (83) | 26 | tau-fluvalinate | 50 | |
| methiocarb | 50 | |||||
| coumaphos | 26.7 | |||||
| 11 | comb | 10 (91) | 10 | tau-fluvalinate | 100.0 | |
| 10 | pollen | 9 (90) | 14 | tau-fluvalinate | 66.7 | |
| 7 | honey | 5 (71) | 1 | methiocarb | 100.0 | |
| 8 | vegetable | 6 (75) | 12 | deltamethrin | 66.7 | |
| 2019 | 73 | 41(56) | 27 | tau-fluvalinate | 58.5 | |
| pyrimethanil | 19.5 | |||||
| dimetomorph | 17.1 | |||||
| 32 | comb | 25 (78) | 16 | tau-fluvalinate | 68.0 | |
| 14 | pollen | 11 (78) | 11 | tau-fluvalinate-pyrimethanil | 27.3 | |
| 25 | honey | 3 (12) | 2 | tau-fluvalinate | 66.7 | |
| 2 | vegetable | 2 (100) | 6 | |||
| Sum | 189 | 118 (67) | 51 | tau-fluvalinate | 53.4 | |
| methiocarb | 13.6 | |||||
| piperonyl butoxide | 11.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinello, M.; Manzinello, C.; Borin, A.; Avram, L.E.; Dainese, N.; Giuliato, I.; Gallina, A.; Mutinelli, F. A Survey from 2015 to 2019 to Investigate the Occurrence of Pesticide Residues in Dead Honeybees and Other Matrices Related to Honeybee Mortality Incidents in Italy. Diversity 2020, 12, 15. https://doi.org/10.3390/d12010015
Martinello M, Manzinello C, Borin A, Avram LE, Dainese N, Giuliato I, Gallina A, Mutinelli F. A Survey from 2015 to 2019 to Investigate the Occurrence of Pesticide Residues in Dead Honeybees and Other Matrices Related to Honeybee Mortality Incidents in Italy. Diversity. 2020; 12(1):15. https://doi.org/10.3390/d12010015
Chicago/Turabian StyleMartinello, Marianna, Chiara Manzinello, Alice Borin, Larisa Elena Avram, Nicoletta Dainese, Ilenia Giuliato, Albino Gallina, and Franco Mutinelli. 2020. "A Survey from 2015 to 2019 to Investigate the Occurrence of Pesticide Residues in Dead Honeybees and Other Matrices Related to Honeybee Mortality Incidents in Italy" Diversity 12, no. 1: 15. https://doi.org/10.3390/d12010015
APA StyleMartinello, M., Manzinello, C., Borin, A., Avram, L. E., Dainese, N., Giuliato, I., Gallina, A., & Mutinelli, F. (2020). A Survey from 2015 to 2019 to Investigate the Occurrence of Pesticide Residues in Dead Honeybees and Other Matrices Related to Honeybee Mortality Incidents in Italy. Diversity, 12(1), 15. https://doi.org/10.3390/d12010015






