The Importance of Isotopic Turnover for Understanding Key Aspects of Animal Ecology and Nutrition
Abstract
1. Introduction
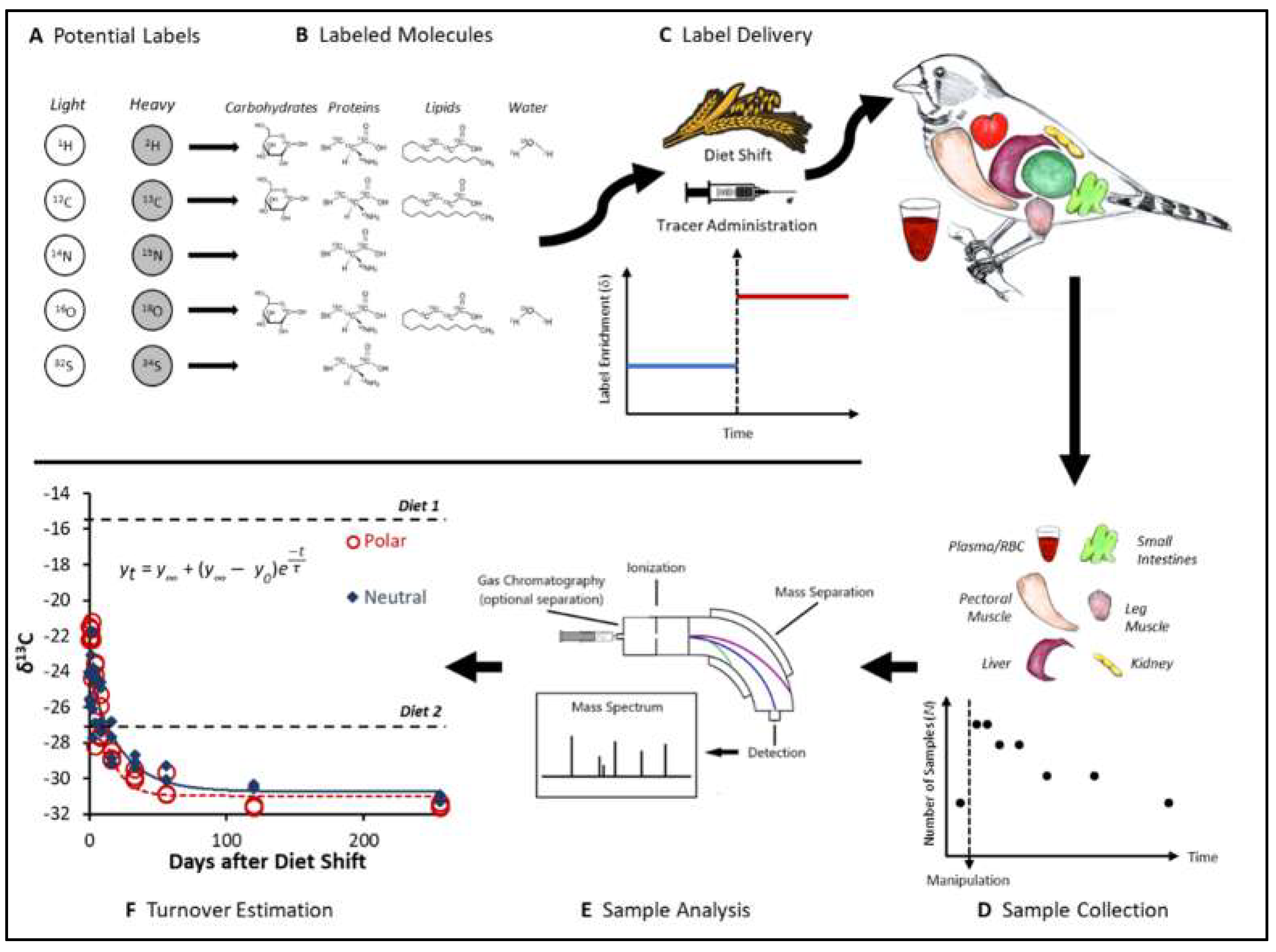
2. How to Measure Turnover Rates with Stable Isotopes: from Elements to Molecules
3. The Dynamics of Stable Isotopes in Organisms: A Physiological Foundation of Ecological Applications of Stable Isotopes
3.1. Considerations for Isotopic Diet Reconstructions: Variation in Turnover Among Individuals
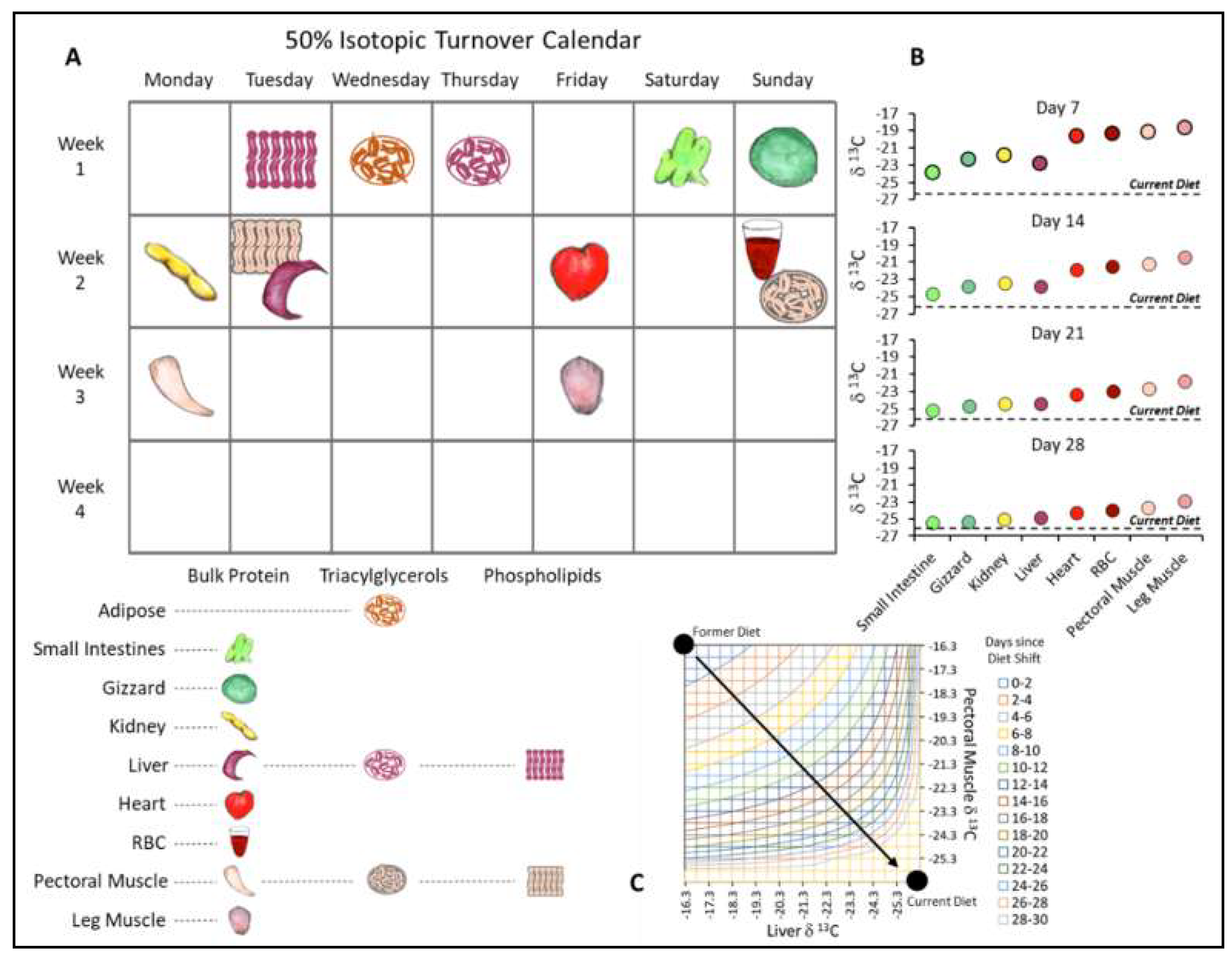
3.2. The Physiological Basis of Isotopic Clocks: Variation in Turnover among Tissues
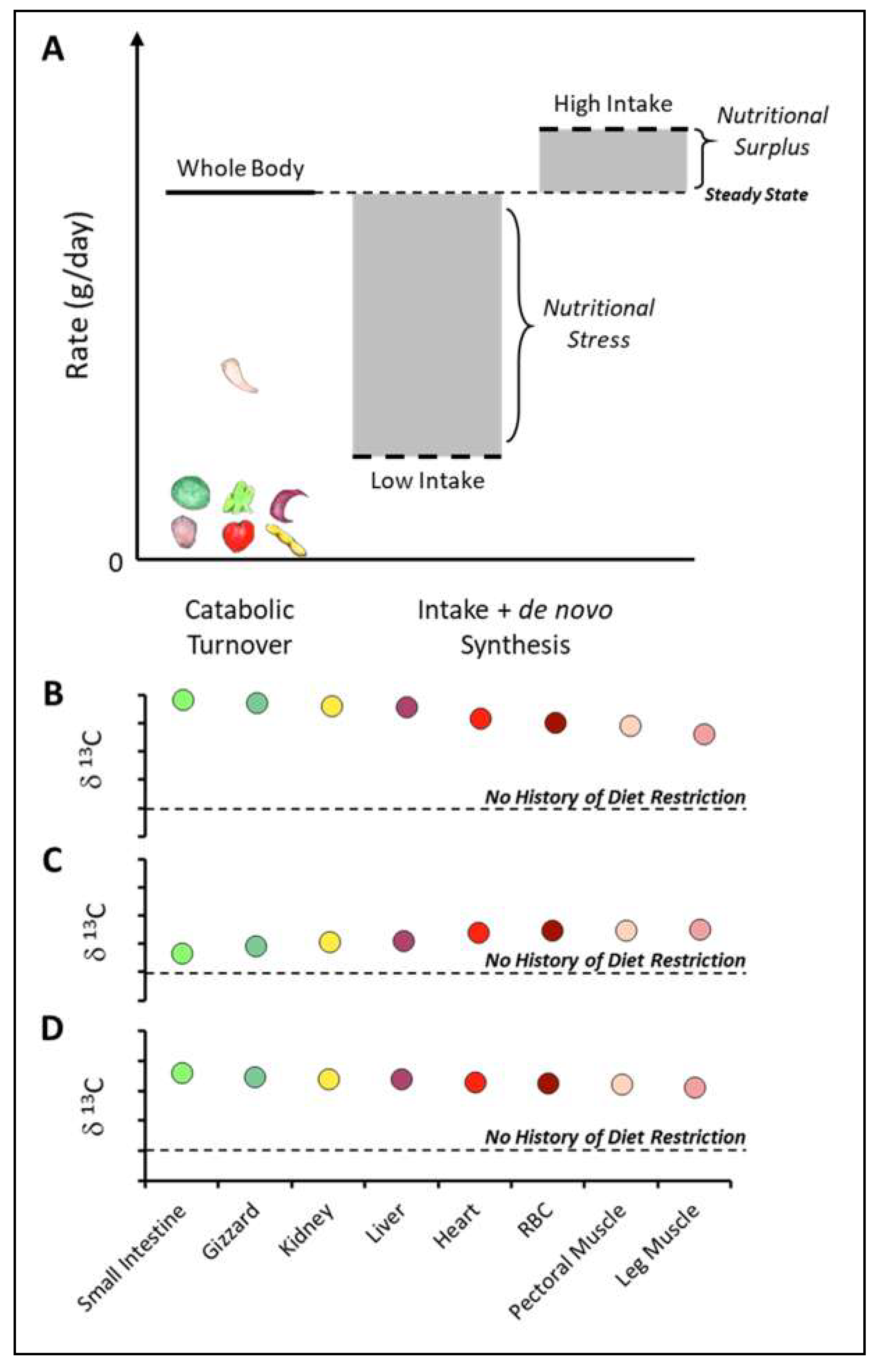
3.3. Turnover and Diet Requirements
4. The Use and Importance of Turnover Data for Isotopic Studies of Ecology and Nutrition
4.1. Reconstructing Diets from Isotopic Signatures
4.2. Pinpointing Dietary Shifts with Isotopic Clocks
4.3. Inferring Past and Present Nutritional Status from Turnover and Isotopic Signatures
5. The Future of Isotopic Turnover
5.1. What Mechanisms Drive the Turnover of Isotopes?
5.2. How Can Turnover Be Integrated with Tracking Technologies?
5.3. How Can Turnover Inform Wildlife Nutrition Studies?
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Newsome, S.D.; Rio Martinez del, C.; Bearhop, S.; Phillips, D.L. A Niche for Isotope Ecology. Front. Ecol. Environ. 2007, 5, 429–436. [Google Scholar] [CrossRef]
- Inger, R.; Bearhop, S. Applications of stable isotope analyses to avian ecology. IBIS 2008, 150, 447–461. [Google Scholar] [CrossRef]
- Ben-David, M.; Newsome, S.D.; Whiteman, J.P. Lipid and amino acid composition influence incorporation and discrimination of 13 C and 15 N in mink. J. Mammal. 2012, 93, 399–412. [Google Scholar] [CrossRef]
- Gannes, L.Z.; O’Brien, D.M.; Martinez del Rio, C. Stable isotopes in animal ecology: Assumptions, caveats, and a call for more laboratory experiments. Ecology 1997, 78, 1271–1276. [Google Scholar] [CrossRef]
- Post, D.M. Using stable isotopes to estimate trophic position: Models, methods, and assumptions. Ecology 2002, 83, 703–718. [Google Scholar] [CrossRef]
- Martínez Del Rio, C.; Wolf, N.; Carleton, S.A.; Gannes, L.Z. Isotopic ecology ten years after a call for more laboratory experiments. Biol. Rev. 2009, 84, 91–111. [Google Scholar] [CrossRef] [PubMed]
- Wolf, N.; Carleton, S.A.; Martinez del Rio, C. Ten years of experimental animal isotopic ecology. Funct. Ecol. 2009, 23, 17–26. [Google Scholar] [CrossRef]
- Bearhop, S.; Waldron, S.; Votier, S.C.; Furness, R.W. Factors that influence assimilation rates and fractionation of nitrogen and carbon stable isotopes in avian blood and feathers. Physiol. Biochem. Zool. 2002, 75, 451–458. [Google Scholar] [CrossRef]
- Gannes, L.Z.; Martinez del Rio, C.; Koch, P. Natural abundance variations in stable isotopes and their potential uses in animal physiological ecology. Comp. Biochem. Physiol. 1998, 119, 725–737. [Google Scholar] [CrossRef]
- Podlesak, D.W.; McWilliams, S.R. Metabolic routing of dietary nutrients in birds: Effects of diet quality and macronutrient composition revealed using stable isotopes. Physiol. Biochem. Zool. 2006, 79, 534–549. [Google Scholar] [CrossRef] [PubMed]
- Newsome, S.D.; Wolf, N.; Peters, J.; Fogel, M.L. Amino acid δ13C analysis shows flexibility in the routing of dietary protein and lipids to the tissue of an omnivore. Integr. Comp. Biol. 2014, 54, 890–902. [Google Scholar] [CrossRef] [PubMed]
- Hobson, K.A.; Clark, R.G. Assessing avian diets using stable isotopes I: Turnover of 13C in tissues. Condor 1992, 94, 181–188. [Google Scholar] [CrossRef]
- Inger, R.; Ruxton, G.D.; Newton, J.; Colhoun, K.; Robinson, J.A.; Jackson, A.L.; Bearhop, S. Temporal and intrapopulation variation in prey choice of wintering geese determined by stable isotope analysis. J. Anim. Ecol. 2006, 75, 1190–1200. [Google Scholar] [CrossRef] [PubMed]
- Martínez del Rio, C.; Carleton, S.A. How fast and how faithful: The dynamics of isotopic incorporation into animal tissues. J. Mammal. 2012, 93, 353–359. [Google Scholar] [CrossRef]
- Davis, M.; Munoz, S.P. The temporal scale of diet and dietary proxies. Ecol. Evol. 2016, 6, 1883–1897. [Google Scholar] [CrossRef]
- Thomas, S.M.; Crowther, T.W. Predicting rates of isotopic turnover across the animal kingdom: A synthesis of existing data. J. Anim. Ecol. 2015, 84, 861–870. [Google Scholar] [CrossRef]
- Vander Zanden, M.J.; Clayton, M.K.; Moody, E.K.; Solomon, C.T.; Weidel, B.C. Stable isotope turnover and half-life in animal tissues: A literature synthesis. PLoS ONE 2015, 10, e0116182. [Google Scholar] [CrossRef]
- Hesslein, R.H.; Hallard, K.A.; Ramlal, P. Replacement of Sulfur, Carbon, and Nitrogen in Tissue of Growing Broad Whitefish (Coregonus nasus) in Response to a Change in Diet Traced by d34S, d13C, and d15N. Can. J. Fish. Aquat. Sci. 1993, 50, 2071–2076. [Google Scholar] [CrossRef]
- Cerling, T.E.; Ayliffe, L.K.; Dearing, M.D.; Ehleringer, J.R.; Passey, B.H.; Podlesak, D.W.; Torregrossa, A.M.; West, A.G. Determining biological tissue turnover using stable isotopes: The reaction progress variable. Oecologia 2007, 151, 175–189. [Google Scholar] [CrossRef]
- Lifson, N.; McClintock, R. Theory of use of the turnover rates of body water for measuring enery and material balance. J. Theor. Biol. 1966, 12, 46–74. [Google Scholar] [CrossRef]
- Visser, G.H.; Boon, P.E.; Meijer, H.A. Validation of the doubly labeled water method in Japanese Quail Coturnix c. japonica chicks: Is there an effect of growth rate? J. Comp. Physiol. B. 2000, 170, 365–372. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Speakman, J. Doubly Labeled Water: Theory and Practice; 1st ed.; Chapman and Hall: London, UK, 1997. [Google Scholar]
- Mitchell, G.W.; Guglielmo, C.G.; Hobson, K.A. Measurement of whole-body CO2 production in birds using real-time laser-derived measurements of hydrogen (δ2H) and oxygen (δ18O) isotope concentrations in water vapor from breath. Physiol. Biochem. Zool. 2015, 88, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Karasov, W.H.; Pinshow, B. Changes in lean mass and in organs of nutrient assimilation in a long-distance passerine migrant at a springtime stopover site. Physiol. Zool. 1998, 71, 435–438. [Google Scholar] [CrossRef]
- McWilliams, S.R.; Whitman, M. Non-destructive techniques to assess body composition of birds: A review and validation study. J. Ornithol. 2013, 154, 597–618. [Google Scholar] [CrossRef]
- Carter, W.A.; Cooper-Mullin, C.; McWilliams, S.R. Turnover of muscle lipids and response to exercise differs between neutral and polar fractions in a model songbird, the Zebra Finch. J. Exp. Biol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Post, D.M.; Layman, C.A.; Arrington, D.A.; Takimoto, G.; Quattrochi, J.; Montaña, C.G. Getting to the fat of the matter: Models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia 2007, 152, 179–189. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Palmer, J.K.; Brandes, W.B. Improvement in the reproducibility of the enzymatic analysis of sucrose in stored golden delicious apples. J. Agric. Food Chem. 1974, 22, 709–712. [Google Scholar] [CrossRef] [PubMed]
- Thiele, C.; Ganzle, M.G.; Vogel, R.F. Sample preparation for amino acid determination by integrated pulsed amperometric detection in foods. Anal. Biochem. 2002, 310, 171–178. [Google Scholar] [CrossRef]
- Yoo, H.; Stephanopoulos, G.; Kelleher, J.K. Quantifying carbon sources for de novo lipogenesis in wild-type and IRS-1 knockout brown adipocytes. J. Lipid Res. 2004, 45, 1324–1332. [Google Scholar] [CrossRef] [PubMed]
- Meier-Augenstein, W. Stable isotope analysis of fatty acids by gas chromatography–isotope ratio mass spectrometry. Anal. Chim. Acta 2002, 465, 63–79. [Google Scholar] [CrossRef]
- Budge, S.M.; Wooller, M.J.; Springer, A.M.; Iverson, S.J.; McRoy, C.P.; Divoky, G.J. Tracing carbon flow in an arctic marine food web using fatty acid-stable isotope analysis. Oecologia 2008, 157, 117–129. [Google Scholar] [CrossRef]
- Ishikawa, N.F. Use of compound-specific nitrogen isotope analysis of amino acids in trophic ecology: Assumptions, applications, and implications. Ecol. Res. 2018, 33, 1–13. [Google Scholar] [CrossRef]
- Levin, E.; Fane, B.; Davidowitz, G. Hawkmoths use nectar sugar to reduce oxidative damage from flight. Science 2017, 355, 733–735. [Google Scholar] [CrossRef] [PubMed]
- Jin, E.S.; Sherry, A.D.; Malloy, C.R. Interaction between the pentose phosphate pathway and gluconeogenesis from glycerol in the liver. J. Biol. Chem. 2014, 289, 32593–32603. [Google Scholar] [CrossRef]
- McCue, M.D. Tracking the oxidative and nonoxidative fates of isotopically labeled nutrients in animals. Bioscience 2011, 61, 217–230. [Google Scholar] [CrossRef]
- Tieszen, L.L.; Boutton, T.W.; Tesdahl, K.G.; Slade, N.A. Fractionation and turnover of stable carbon isotopes in animal tissues: Implications for d13C analysis of diet. Oecologia 1983, 57, 32–37. [Google Scholar] [CrossRef]
- Kelly, J.F. Stable isotopes of carbon and nitrogen in the study of avian and mammalian trophic ecology. Can. J. Zool. 2000, 78, 1–27. [Google Scholar] [CrossRef]
- Cloern, J.E.; Canuel, E.A.; Harris, D. Stable carbon and nitrogen isotope composition of aquatic and terrestrial plants of the San Francisco Bay estuarine system. Limnol. Oceanogr. 2002, 47, 713–729. [Google Scholar] [CrossRef]
- Chisholm, B.S.; Nelson, D.E.; Schwarcz, H.P. Stable-carbon isotope ratios as a measure of marine versus terrestrial protein in ancient diets. Science 1982, 216, 1131–1132. [Google Scholar] [CrossRef]
- DeNiro, M.J.; Epstein, S. Influence of diet on the distribution of carbon isotopes in animals. Geochim. Cosmochim. Acta 1978, 42, 495–506. [Google Scholar] [CrossRef]
- DeNiro, M.J.; Epstein, S. Influence of diet on the distribution of nitrogen isotopes in animals. Geochim. Cosmochim. Acta 1981, 45, 341–351. [Google Scholar] [CrossRef]
- Bowen, G.J.; West, J.B. Isotope landscapes for terrestrial migration research. In Terrestrial Ecology; Hobson, K.A., Wassenaar, L.I., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 79–105. ISBN 9780123738677. [Google Scholar]
- Bowen, G.J.; Wassenaar, L.I.; Hobson, K.A. Global application of stable hydrogen and oxygen isotopes to wildlife forensics. Oecologia 2005, 143, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Martínez Del Rio, C.; Anderson-Sprecher, R. Beyond the reaction progress variable: The meaning and significance of isotopic incorporation data. Oecologia 2008, 156, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Cerling, T.E.; Bowen, G.J.; Ehleringer, J.R.; Sponheimer, M. The reaction progress variable and isotope turnover in biological systems. In Stable Isotopes as Indicators of Ecological Change; Dawson, T.E., Siegwolf, R.T.W., Eds.; Academic Press: New York, NY, USA, 2007; pp. 163–171. [Google Scholar]
- Carleton, S.A.; del Rio, C.M. Growth and catabolism in isotopic incorporation: A new formulation and experimental data. Funct. Ecol. 2010, 24, 805–812. [Google Scholar] [CrossRef]
- Carleton, S.A.; Kelly, L.; Anderson-Sprecher, R.; del Rio, C.M. Should we use one-, or multi-compartment models to describe 13C incorporation into animal tissues? Rapid Commun. Mass Spectrom. 2008, 22, 3008–3014. [Google Scholar] [CrossRef] [PubMed]
- Wolfrum, C.; Spener, F. Fatty acids as regulators of lipid metabolism. Eur. J. Lipid Sci. Technol. 2000, 102, 746–762. [Google Scholar] [CrossRef]
- Twining, C.W.; Lawrence, P.; Winkler, D.W.; Flecker, A.S.; Brenna, J.T. Conversion efficiency of α-linolenic acid to omega-3 highly unsaturated fatty acids in aerial insectivore chicks. J. Exp. Biol. 2018, 221, 1–8. [Google Scholar] [CrossRef]
- MacAvoy, S.E.; Arneson, L.S.; Bassett, E. Correlation of metabolism with tissue carbon and nitrogen turnover rate in small mammals. Oecologia 2006, 150, 190–201. [Google Scholar] [CrossRef]
- Carleton, S.A.; Martínez del Rio, C. The effect of cold-induced increased metabolic rate on the rate of 13C and 15N incorporation in house sparrows (Passer domesticus). Oecologia 2005, 144, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Bauchinger, U.; Keil, J.; McKinney, R.A.; Starck, J.M.; McWilliams, S.R. Exposure to cold but not exercise increases carbon turnover rates in specific tissues of a passerine. J. Exp. Biol. 2010, 213, 526–534. [Google Scholar] [CrossRef][Green Version]
- Hobson, K.A.; Yohannes, E. Establishing elemental turnover in exercising birds using a wind tunnel: Implications for stable isotope tracking of migrants. Can. J. Zool. 2007, 85, 703–708. [Google Scholar] [CrossRef]
- Tsahar, E.; Wolf, N.; Izhaki, I.; Arad, Z.; del Martínez, C. Dietary protein influences the rate of 15N incorporation in blood cells and plasma of yellow-vented bulbuls (Pycnonotus xanthopygos). J. Exp. Biol. 2008, 211, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Braun, A.; Auerswald, K.; Vikari, A.; Schnyder, H. Dietary protein content affects isotopic carbon and nitrogen turnover. Rapid Commun. Mass Spectrom. 2013, 27, 2676–2684. [Google Scholar] [CrossRef] [PubMed]
- Storm-Suke, A.; Norris, D.R.; Wassenaar, L.I.; Chin, E.; Nol, E. Factors influencing the turnover and net isotopic discrimination of hydrogen isotopes in proteinaceous tissue: Experimental results using Japanese Quail. Physiol. Biochem. Zool. 2012, 85, 376–384. [Google Scholar] [CrossRef]
- Colborne, S.F.; Fisk, A.T.; Johnson, T.B. Tissue-specific turnover and diet-tissue discrimination factors of carbon and nitrogen isotopes of a common forage fish held at two temperatures. Rapid Commun. Mass Spectrom. 2017, 31, 1405–1414. [Google Scholar] [CrossRef]
- Kim, S.L.; del Rio, C.M.; Casper, D.; Koch, P.L. Isotopic incorporation rates for shark tissues from a long-term captive feeding study. J. Exp. Biol. 2012, 215, 2495–2500. [Google Scholar] [CrossRef]
- Murray, I.W.; Wolf, B.O. Tissue carbon incorporation rates and diet-to-tissue discrimination in ectotherms: Tortoises are really slow. Physiol. Biochem. Zool. 2012, 8500, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Cloyed, C.S.; Newsome, S.D.; Eason, P.K. Trophic discrimination factors and incorporation rates of carbon- and nitrogen-stable isotopes in adult green frogs, Lithobates clamitans. Physiol. Biochem. Zool. 2015, 88, 576–585. [Google Scholar] [CrossRef][Green Version]
- Foglia, T.A.; Cartwright, A.L.; Gyurik, R.J.; Philips, J.G. Fatty acid turnover rates in the adipose tissues of the growing chicken (Gallus domesticus). Lipids 1994, 29, 497–502. [Google Scholar] [CrossRef]
- Carter, W.A.; Whiteman, J.P.; Cooper-mullin, C.; Newsome, S.D.; Mcwilliams, S.R. Dynamics of individual fatty acids in muscle fat stores and membranes of a songbird and its functional and ecological importance. Physiol. Biochem. Zool. 2019, 92, 239–251. [Google Scholar] [CrossRef]
- Jennings, S.; Maxwell, T.A.D.; Schratzberger, M.; Milligan, S.P. Body-size dependent temporal variations in nitrogen stable isotope ratios in food webs. Mar. Ecol. Prog. Ser. 2008, 370, 199–206. [Google Scholar] [CrossRef]
- Glazier, D.S. Metabolic scaling in complex living systems. Systems 2014, 2, 451–540. [Google Scholar] [CrossRef]
- Glazier, D.S. Is metabolic rate a universal ‘pacemaker’ for biological processes? Biol. Rev. 2015, 90, 377–407. [Google Scholar] [CrossRef]
- Bauchinger, U.; McWilliams, S. Carbon turnover in tissues of a passerine bird: Allometry, isotopic clocks, and phenotypic flexibility in organ size. Physiol. Biochem. Zool. 2009, 82, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.L.; Eldridge, P.M. Estimating the timing of diet shifts using stable isotopes. Oecologia 2006, 147, 195–203. [Google Scholar] [CrossRef]
- Podlesak, D.W.; McWilliams, S.R.; Hatch, K.A. Stable isotopes in breath, blood, feces and feathers can indicate intra-individual changes in the diet of migratory songbirds. Oecologia 2005, 142, 501–510. [Google Scholar] [CrossRef]
- Bauchinger, U.; McWilliams, S.R. Extent of phenotypic flexibility during long-distance flight is determined by tissue-specific turnover rates: A new hypothesis. J. Avian Biol. 2010, 41, 603–608. [Google Scholar] [CrossRef]
- Bauchinger, U.; McWilliams, S.R. Tissue-specific mass changes during fasting: The protein turnover hypothesis. In Comparative Physiology of Fasting, Starvation, and Food Limitation; McCue, M.D., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; p. 430. [Google Scholar]
- Onodera, J.; Ohsumi, Y. Autophagy is required for maintenance of amino acid levels and protein synthesis under nitrogen starvation. J. Biol. Chem. 2005, 280, 31582–31586. [Google Scholar] [CrossRef]
- Demeyer, D.; Fievez, V. Is the synthesis of rumen bacterial protein limited by the availability of pre-formed amino acids and/or peptides? Br. J. Nutr. 2004, 91, 175–176. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mizrahy, O.; Bauchinger, U.; Aamidor, S.E.; McWilliams, S.R.; Pinshow, B. Availability of water affects renewal of tissues in migratory blackcaps during stopover. Integr. Comp. Biol. 2011, 51, 374–384. [Google Scholar] [CrossRef]
- Klasing, K.C. Comparative Avian Nutrition; CAB International: Wallingford, UK, 1998; ISBN 9780851992198. [Google Scholar]
- Stevens, L. Avian Biochemistry and Molecular Biology, 1st ed.; Cambridge University Press: Cambridge, UK, 2004; ISBN 9780521612111. [Google Scholar]
- Twining, C.W.; Brenna, J.T.; Hairston, N.G.; Flecker, A.S. Highly unsaturated fatty acids in nature: What we know and what we need to learn. Oikos 2016, 125, 749–760. [Google Scholar] [CrossRef]
- Salini, M.J.; Poppi, D.; Turchini, G.M.; Glencross, B.D. Defining the allometric relationship between size and individual fatty acid turnover in barramundi Lates calcarifer. Comp. Biochem. Physiol.-Part A Mol. Integr. Physiol. 2016, 201, 79–86. [Google Scholar] [CrossRef] [PubMed]
- DeNiro, M.J. Stable Isotopy and Archaeology. Am. Sci. 1987, 75, 182–191. [Google Scholar]
- Hobson, K.A.; Clark, R.G. Assessing avian diets using stable isotopes II: Factors influencing diet-tissue frationation. Condor 1992, 94, 189–197. [Google Scholar] [CrossRef]
- Ben-David, M.; Flaherty, E.A. Stable isotopes in mammalian research: A beginner’s guide. J. Mammal. 2012, 93, 312–328. [Google Scholar] [CrossRef]
- Nielsen, J.M.; Clare, E.L.; Hayden, B.; Brett, M.T.; Kratina, P. Diet tracing in ecology: Method comparison and selection. Methods Ecol. Evol. 2018, 9, 278–291. [Google Scholar] [CrossRef]
- Phillips, D.L.; Inger, R.; Bearhop, S.; Jackson, A.L.; Moore, J.W.; Parnell, A.C.; Semmens, B.X.; Ward, E.J. Best practices for use of stable isotope mixing models in food-web studies. Can. J. Zool. 2014, 835, 823–835. [Google Scholar] [CrossRef]
- Phillips, D.L. Converting isotope values to diet composition: The use of mixing models. J. Mammal. 2012, 93, 342–352. [Google Scholar] [CrossRef]
- Layman, C.A.; Araujo, M.S.; Boucek, R.; Hammerschlag-peyer, C.M.; Harrison, E.; Jud, Z.R.; Matich, P.; Rosenblatt, A.E.; Vaudo, J.J.; Yeager, L.A.; et al. Applying stable isotopes to examine food-web structure: An overview of analytical tools. Biol. Rev. 2012, 87, 545–562. [Google Scholar] [CrossRef] [PubMed]
- Rubenstein, D.R.; Hobson, K.A. From birds to butterflies: Animal movement patterns and stable isotopes. Trends Ecol. Evol. 2004, 19, 256–263. [Google Scholar] [CrossRef]
- Hammer, B.T.; Fogel, M.L.; Hoering, T.C. Stable carbon isotope ratios of fatty acids in seagrass and redhead ducks. Chem. Geol. 1998, 152, 29–41. [Google Scholar] [CrossRef]
- Renfrew, R.B.; Hill, J.M.; Kim, D.H.; Romanek, C.; Perlut, N.G. Winter diet of Bobolink, a long-distance migratory grassland bird, inferred from feather isotopes. Condor 2017, 119, 439–448. [Google Scholar] [CrossRef]
- Asante, C.K.; Hobson, K.A.; Bond, A.L.; Jardine, T.D. Resource partitioning among five species of waterfowl (Anas spp.) at an autumn migratory stopover: Combining stable isotope and mercury biomarkers. Can. J. Zool. 2017, 95, 279–286. [Google Scholar] [CrossRef]
- Rubenstein, D.R.; Chamberlain, C.P.; Holmes, R.T.; Ayres, M.P.; Waldbauer, J.R.; Graves, R.; Tuross, N.C. Linking ranges wintering breeding and a migratory of songbird using stable isotopes. Science 2002, 295, 1062–1065. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.M.; Renfrew, R.B. Migratory patterns and connectivity of two North American grassland bird species. Ecol. Evol. 2018, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.A.; Whitman, E.R.; Garcia-Rojas, M.I.; Bellgrove, A.; Ekins, M.; Hays, G.C.; Heithaus, M.R. Individual specialization in a migratory grazer reflects long-term diet selectivity on a foraging ground: Implications for isotope-based tracking. Oecologia 2018, 188, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Steenweg, R.J.; Crossin, G.T.; Kyser, T.K.; Merkel, F.R.; Gilchrist, H.G.; Hennin, H.L.; Robertson, G.J.; Provencher, J.F.; Mills Flemming, J.; Love, O.P. Stable isotopes can be used to infer the overwintering locations of prebreeding marine birds in the Canadian Arctic. Ecol. Evol. 2017, 1–11. [Google Scholar] [CrossRef]
- Wang, S.W.; Budge, S.M.; Gradinger, R.R.; Iken, K.; Wooller, M.J. Fatty acid and stable isotope characteristics of sea ice and pelagic particulate organic matter in the Bering Sea: Tools for estimating sea ice algal contribution to Arctic food web production. Oecologia 2014, 174, 699–712. [Google Scholar] [CrossRef]
- Graham, C.; Oxtoby, L.; Wang, S.W.; Budge, S.M.; Wooller, M.J. Sourcing fatty acids to juvenile polar cod (Boreogadus saida) in the Beaufort Sea using compound-specific stable carbon isotope analyses. Polar Biol. 2014, 37, 697–705. [Google Scholar] [CrossRef]
- Wang, S.W.; Budge, S.M.; Iken, K.; Gradinger, R.R.; Springer, A.M.; Wooller, M.J. Importance of sympagic production to Bering Sea zooplankton as revealed from fatty acid-carbon stable isotope analyses. Mar. Ecol. Prog. Ser. 2015, 518, 31–50. [Google Scholar] [CrossRef]
- Careddu, G.; Letizia, M.; Calizza, E.; Carlino, P.; Bentivoglio, F.; Orlandi, L.; Rossi, L. Effects of terrestrial input on macrobenthic food webs of coastal sea are detected by stable isotope analysis in Gaeta Gulf. Estuar. Coast. Shelf Sci. 2015, 154, 158–168. [Google Scholar] [CrossRef]
- Larsen, T.; Ventura, M.; Andersen, N.; Brien, D.M.O.; Piatkowski, U.; Mccarthy, M.D. Tracing carbon sources through aquatic and terrestrial food webs using amino acid stable isotope fingerprinting. PLoS ONE 2013, 8, e73441. [Google Scholar] [CrossRef]
- Dalerum, F.; Angerbjorn, A. Resolving temporal variation in vertebrate diets using naturally occurring stable isotopes. Oecologia 2005, 144, 647–658. [Google Scholar] [CrossRef]
- Mizutani, H.; Fukuda, M.; Kabaya, Y. 13C and 15 N Enrichment factors of feathers of 11 species of adult birds. Ecology 1992, 73, 1391–1395. [Google Scholar] [CrossRef]
- Marques, F.P.; Cardoso, L.G.; Haimovici, M.; Bugoni, L. Trophic ecology of Magellanic penguins (Spheniscus magellanicus) during the non-breeding period. Estuar. Coast. Shelf Sci. 2018, 210, 109–122. [Google Scholar] [CrossRef]
- Gómez, C.; Larsen, T.; Popp, B.; Hobson, K.A.; Cadena, C.D. Assessing seasonal changes in animal diets with stable—isotope analysis of amino acids: A migratory boreal songbird switches diet over its annual cycle. Oecologia 2018, 187, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Guidotti, S.; Jansen, H.G. Doubly Labelled Water analysis: Preparation, memory correction, calibration and quality assurance for d2H and d18O measurements over four orders of magnitudes. Rapid Commun. Mass Spectrom. 2013, 27, 1055–1066. [Google Scholar] [CrossRef]
- Gretebeck, R.J.; Schoeller, D.A.; Socki, R.A.; Davis-street, J.; Gibson, E.K.; Schulz, L.O.; Lane, H.W.; Randall, J.; Schoeller, D.A.; Rick, A.; et al. Adaptation of the doubly labeled water method for subjects consuming isotopically enriched water. J. Appl. Physiol. 1997, 82, 563–570. [Google Scholar] [CrossRef]
- Milakovic, B.; Parker, K.L. Quantifying carnivory by grizzly bears in a multi-ungulate system. J. Wildl. Manag. 2013, 77, 39–47. [Google Scholar] [CrossRef]
- Halley, D.J.; Minagawa, M.; Nieminen, M.; Gaare, E. Diet: Tissue stable isotope fractionation of carbon and nitrogen in blood plasma and whole blood of male reindeer Rangifer tarandus. Polar Biol. 2010, 33, 1303–1309. [Google Scholar] [CrossRef]
- Heady, W.N.; Moore, J.W. Tissue turnover and stable isotope clocks to quantify resource shifts in anadromous rainbow trout. Oecologia 2013, 172, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Oppel, S.; Powell, A.N. Carbon isotope turnover in blood as a measure of arrival time in migratory birds using isotopically distinct environments. J. Ornithol. 2010, 151, 123–131. [Google Scholar] [CrossRef]
- Mohan, J.A.; Smith, S.D.; Connelly, T.L.; Attwood, E.T.; McClelland, J.W.; Herzka, S.Z.; Walther, B.D. Tissue-specific isotope turnover and discrimination factors are affected by diet quality and lipid content in an omnivorous consumer. J. Exp. Mar. Bio. Ecol. 2016, 479, 35–45. [Google Scholar] [CrossRef]
- Moore, J.W.; Gordon, J.; Carr-Harris, C.; Gottesfeld, A.S.; Wilson, S.M.; Russell, J.H. Assessing estuaries as stopover habitats for juvenile pacific salmon. Mar. Ecol. Prog. Ser. 2016, 559, 201–215. [Google Scholar] [CrossRef]
- Catry, T.; Lourenço, P.M.; Granadeiro, J.P. Quantifying population size of migrant birds at stopover sites: Combining count data with stopover length estimated from stable isotope analysis. Methods Ecol. Evol. 2018, 9, 502–512. [Google Scholar] [CrossRef]
- Boggie, M.A.; Carleton, S.A.; Collins, D.P.; Vradenburg, J.; Sroka, C.J. Using stable isotopes to estimate reliance on agricultural food subsidies and migration timing for a migratory bird. Ecosphere 2018, 9. [Google Scholar] [CrossRef]
- Rennie, M.J. An introduction to the use of tracers in nutrition and metabolism. Proc. Nutr. Soc. 1999, 58, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Millward, D.J.; Smith, K. The application of stable-isotope tracers to study human musculoskeletal protein turnover: A tale of bag filling and bag enlargement. J. Physiol. 2019, 597, 1235–1249. [Google Scholar] [CrossRef]
- Hobson, K.A.; Alisauskas, R.T.; Clark, R.G. Stable-nitrogen isotope enrichment in avian tissues due to fasting and nutritional stress: Implications for isotopic analyses of diet. Condor 1993, 95, 388–394. [Google Scholar] [CrossRef]
- Deschner, T.; Fuller, B.T.; Oelze, V.M.; Boesch, C.; Hublin, J.; Mundry, R.; Richards, M.P.; Ortmann, S.; Hohmann, G. Identification of energy consumption and nutritional stress by isotopic and elemental analysis of urine in bonobos (Pan paniscus). Rapid Commun. Mass Spectrom. 2012, 26, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Neuberger, F.M.; Jopp, E.; Graw, M.; Puschel, K.; Grupe, G. Signs of malnutrition and starvation—Reconstruction of nutritional life histories by serial isotopic analyses of hair. Forensic Sci. Int. 2013, 226, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Reitsema, L.J. Beyond diet reconstruction: Stable isotope applications to human physiology, health, and nutrition. Am. J. Hum. Biol. 2013, 25, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Reitsema, L.J. Laboratory and field methods for stable isotope analysis in human biology. Am. J. Hum. Biol. 2015, 27, 593–604. [Google Scholar] [CrossRef] [PubMed]
- Hatch, K.A.; Crawford, M.A.; Kunz, A.W.; Thomsen, S.R.; Eggett, D.L.; Nelson, S.T.; Roeder, B.L. An objective means of diagnosing anorexia nervosa and bulimia nervosa using 15N/14N and 13C/12C ratios in hair. Rapid Commun. Mass Spectrom. 2006, 20, 3367–3373. [Google Scholar] [CrossRef] [PubMed]
- Hatch, K.A.; Sacksteder, K.A.; Treichel, I.W.; Cook, M.E.; Porter, W.P. Early detection of catabolic state via change in 13C/12C ratios of blood proteins. Biochem. Biophys. Res. Commun. 1995, 212, 719–726. [Google Scholar] [CrossRef]
- Gorokhova, E. Individual growth as a non-dietary determinant of the isotopic niche metrics. Methods Ecol. Evol. 2018, 9, 269–277. [Google Scholar] [CrossRef]
- Karlson, A.M.L.; Reutgard, M.; Garbaras, A.; Gorokhova, E. Isotopic niche reflects stress-induced variability in physiological status. R. Soc. Open Sci. 2018, 5, 1–18. [Google Scholar] [CrossRef]
- Guilford, T.; Åkesson, S.; Gagliardo, A.; Holland, R.A.; Mouritsen, H.; Muheim, R.; Wiltschko, R.; Wiltschko, W.; Bingman, V.P. Migratory navigation in birds: New opportunities in an era of fast-developing tracking technology. J. Exp. Biol. 2011, 214, 3705–3712. [Google Scholar] [CrossRef]
- Alerstam, T. Optimal bird migration revisited. J. Ornithol. 2011, 152. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carter, W.A.; Bauchinger, U.; McWilliams, S.R. The Importance of Isotopic Turnover for Understanding Key Aspects of Animal Ecology and Nutrition. Diversity 2019, 11, 84. https://doi.org/10.3390/d11050084
Carter WA, Bauchinger U, McWilliams SR. The Importance of Isotopic Turnover for Understanding Key Aspects of Animal Ecology and Nutrition. Diversity. 2019; 11(5):84. https://doi.org/10.3390/d11050084
Chicago/Turabian StyleCarter, Wales A., Ulf Bauchinger, and Scott R. McWilliams. 2019. "The Importance of Isotopic Turnover for Understanding Key Aspects of Animal Ecology and Nutrition" Diversity 11, no. 5: 84. https://doi.org/10.3390/d11050084
APA StyleCarter, W. A., Bauchinger, U., & McWilliams, S. R. (2019). The Importance of Isotopic Turnover for Understanding Key Aspects of Animal Ecology and Nutrition. Diversity, 11(5), 84. https://doi.org/10.3390/d11050084




