Diversity Patterns of Different Life Forms of Plants along an Elevational Gradient in Crete, Greece
Abstract
1. Introduction
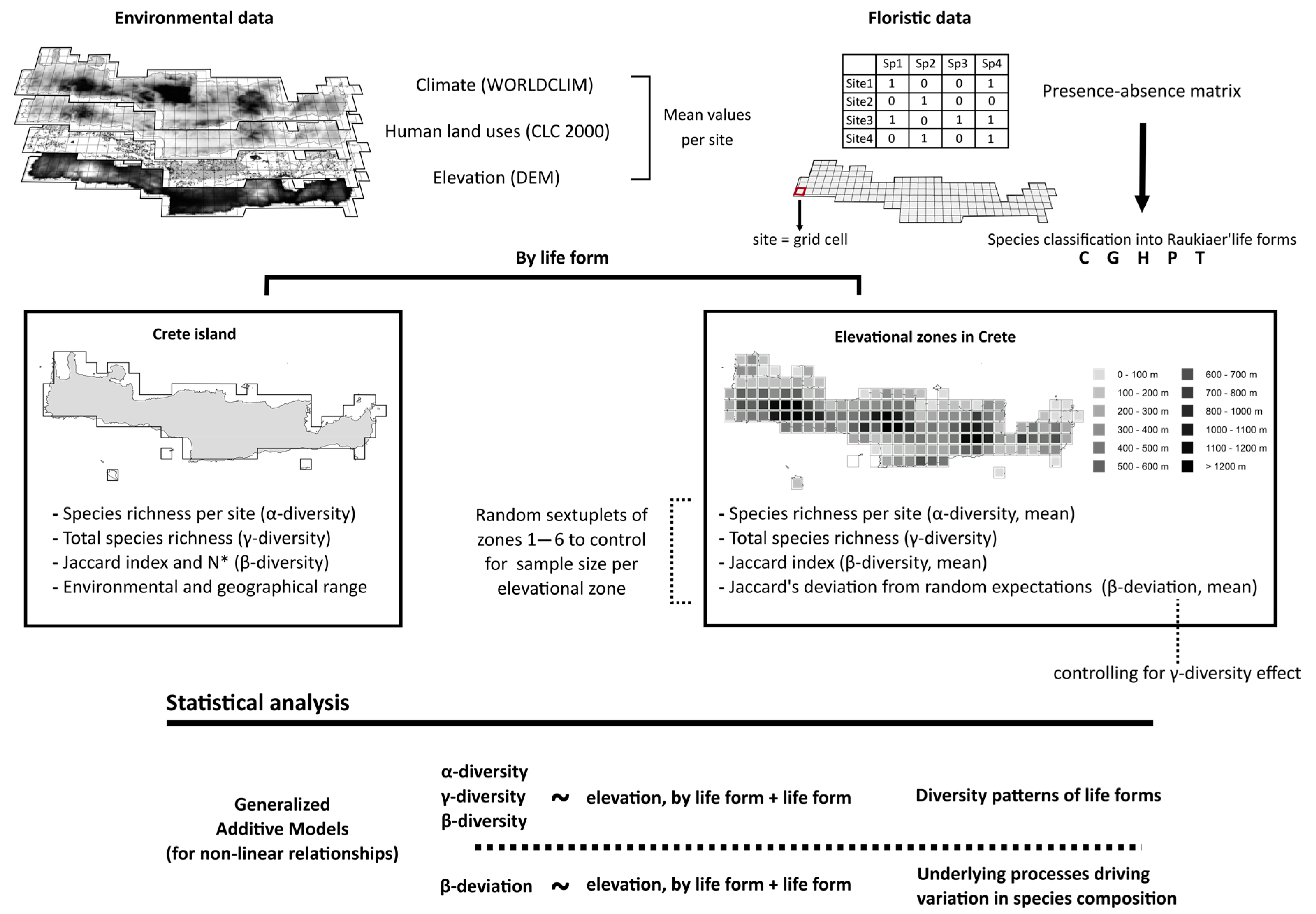
2. Materials and Methods
2.1. Study Area
2.2. Floristic Data
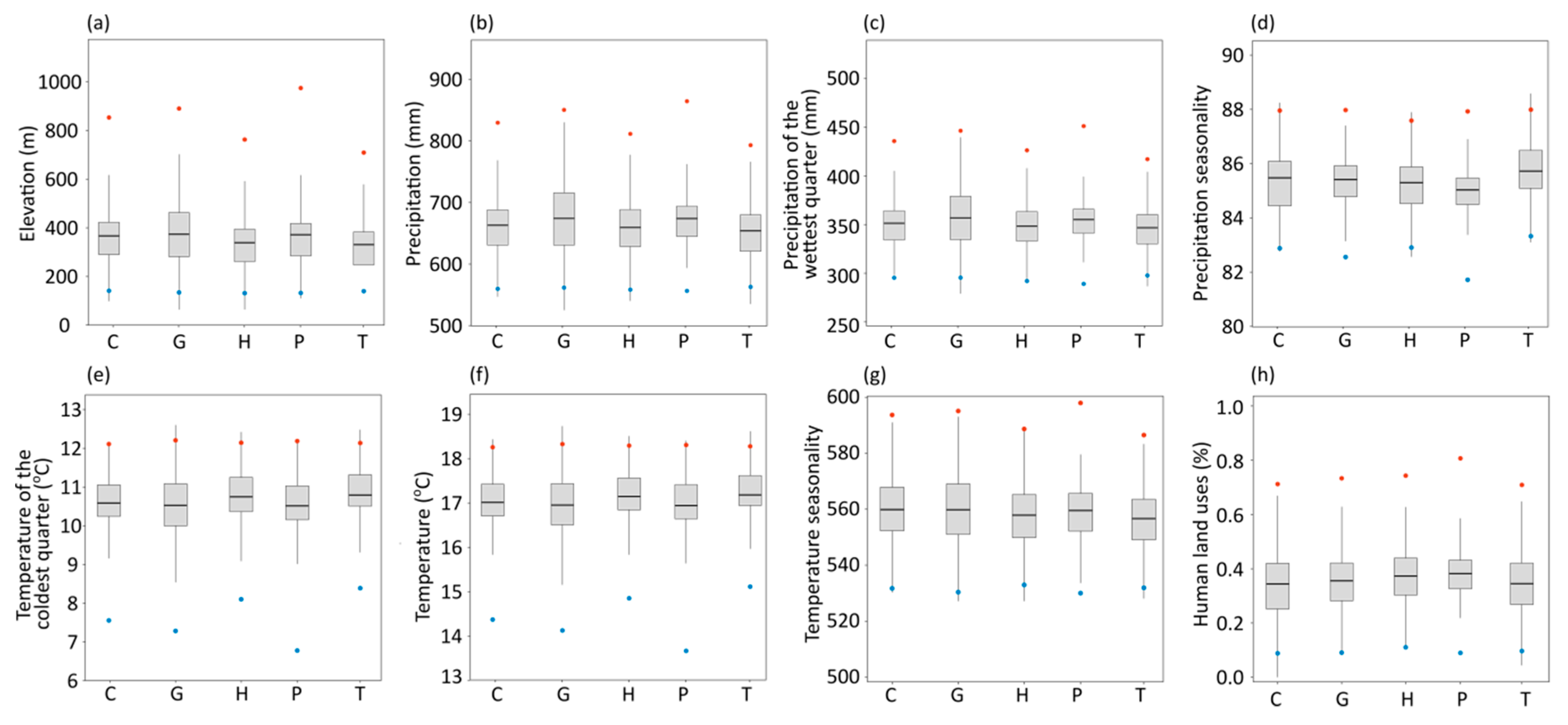
2.3. Elevation and Environmental Variables
2.4. Statistical Analysis
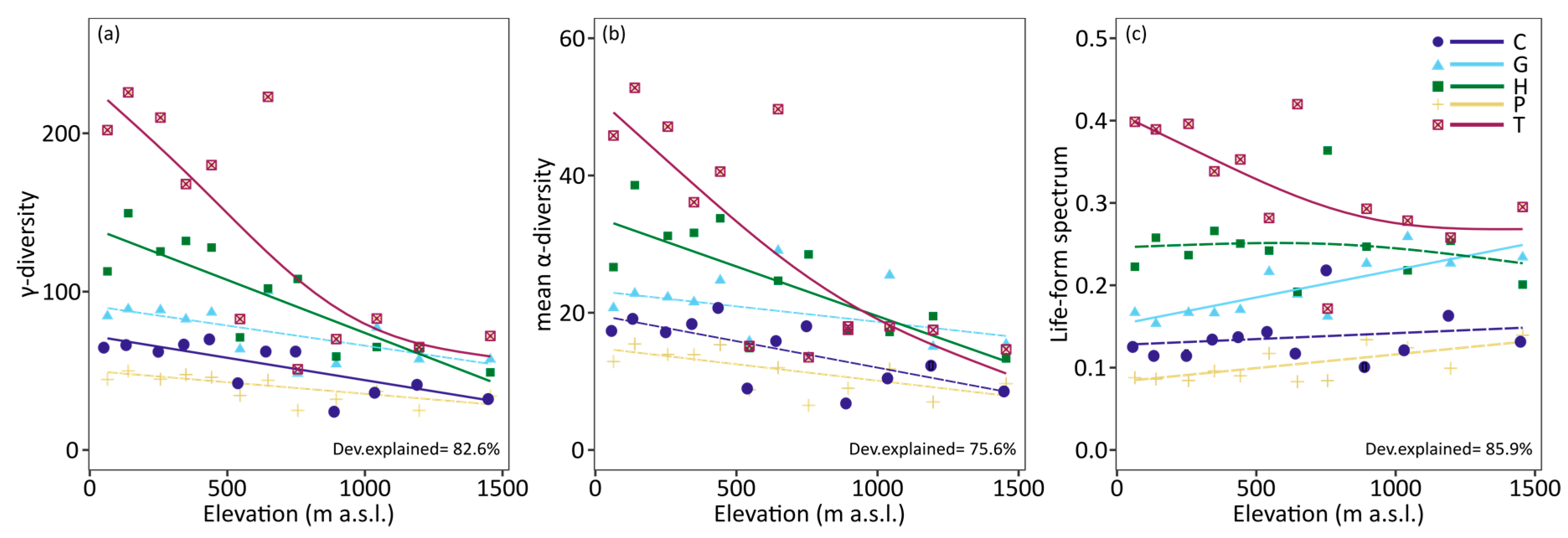
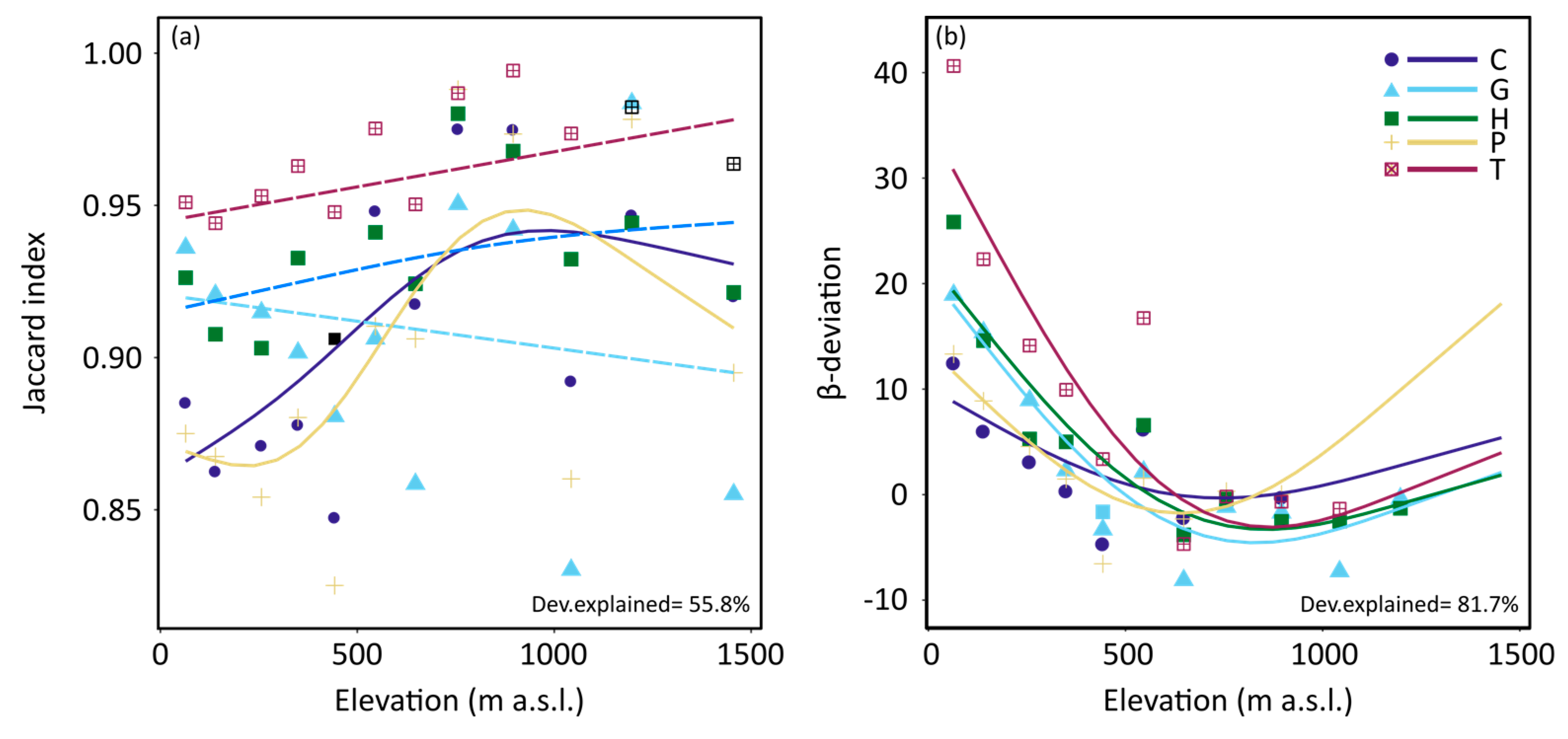
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tershy, B.R.; Shen, K.-W.; Newton, K.M.; Holmes, N.D.; Croll, D.A. The importance of islands for the protection of biological and linguistic diversity. Bioscience 2015, 65, 592–597. [Google Scholar] [CrossRef]
- Whittaker, R.J.; Fernández-Palacios, J.M.; Matthews, T.J.; Borregaard, M.K.; Triantis, K.A. Island biogeography: Taking the long view of nature’s laboratories. Science 2017, 357, eaam8326. [Google Scholar] [CrossRef] [PubMed]
- MacArthur, R.H.; Wilson, E.O. An equilibrium theory of insular zoogeography. Evolution 1963, 17, 373–387. [Google Scholar] [CrossRef]
- Hortal, J.; Triantis, K.A.; Meiri, S.; Thébault, E.; Sfenthourakis, S. Island species richness increases with habitat diversity. Am. Nat. 2009, 174, E205–E217. [Google Scholar] [CrossRef]
- Kallimanis, A.; Bergmeier, E.; Panitsa, M.; Georghiou, K.; Delipetrou, P.; Dimopoulos, P. Biogeographical determinants for total and endemic species richness in a continental archipelago. Biodivers. Conserv. 2010, 19, 1225–1235. [Google Scholar] [CrossRef]
- Triantis, K.A.; Guilhaumon, F.; Whittaker, R.J. The island species–area relationship: Biology and statistics. J. Biogeogr. 2012, 39, 215–231. [Google Scholar] [CrossRef]
- Whittaker, R.H. Gradient analysis of vegetation. Biol. Rev. 1967, 42, 207–264. [Google Scholar] [CrossRef]
- Körner, C. The use of ‘altitude’in ecological research. Trends Ecol. Evol. 2007, 22, 569–574. [Google Scholar] [CrossRef]
- Sundqvist, M.K.; Sanders, N.J.; Wardle, D.A. Community and ecosystem responses to elevational gradients: Processes, mechanisms, and insights for global change. Annu. Rev. Ecol. Evol. Syst. 2013, 44, 261–280. [Google Scholar] [CrossRef]
- McGill, B.J.; Enquist, B.J.; Weiher, E.; Westoby, M. Rebuilding community ecology from functional traits. Trends Ecol. Evol. 2006, 21, 178–185. [Google Scholar] [CrossRef]
- Thuiller, W.; Lavorel, S.; Araújo, M.B. Niche properties and geographical extent as predictors of species sensitivity to climate change. Glob. Ecol. Biogeogr. 2005, 14, 347–357. [Google Scholar] [CrossRef]
- Newbold, T.; Scharlemann, J.P.; Butchart, S.H.; Şekercioğlu, Ç.H.; Alkemade, R.; Booth, H.; Purves, D.W. Ecological traits affect the response of tropical forest bird species to land-use intensity. Proc. R. Soc. B Biol. Sci. 2013, 280, 20122131. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, J.; Lavorel, S.; Garnier, E.; Diaz, S.; Buchmann, N.; Gurvich, D.; Reich, P.B.; Ter Steege, H.; Morgan, H.; Van Der Heijden, M. A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust. J. Bot. 2003, 51, 335–380. [Google Scholar] [CrossRef]
- Klimes, L. Life-forms and clonality of vascular plants along an altitudinal gradient in E Ladakh (NW Himalayas). Basic Appl. Ecol. 2003, 4, 317–328. [Google Scholar] [CrossRef]
- Castro, H.; Lehsten, V.; Lavorel, S.; Freitas, H. Functional response traits in relation to land use change in the Montado. Agric. Ecosyst. Environ. 2010, 137, 183–191. [Google Scholar] [CrossRef]
- Boulangeat, I.; Lavergne, S.; Van Es, J.; Garraud, L.; Thuiller, W. Niche breadth, rarity and ecological characteristics within a regional flora spanning large environmental gradients. J. Biogeogr. 2012, 39, 204–214. [Google Scholar] [CrossRef]
- Broennimann, O.; Vittoz, P.; Moser, D.; Guisan, A. Rarity types among plant species with high conservation priority in Switzerland. Bot. Helv. 2005, 115, 95–108. [Google Scholar] [CrossRef]
- Chiarucci, A.; Fattorini, S.; Foggi, B.; Landi, S.; Lazzaro, L.; Podani, J.; Simberloff, D. Plant recording across two centuries reveals dramatic changes in species diversity of a Mediterranean archipelago. Sci. Rep. 2017, 7, 5415. [Google Scholar] [CrossRef]
- Ebeling, A.; Pompe, S.; Baade, J.; Eisenhauer, N.; Hillebrand, H.; Proulx, R.; Roscher, C.; Schmid, B.; Wirth, C.; Weisser, W.W. A trait-based experimental approach to understand the mechanisms underlying biodiversity–ecosystem functioning relationships. Basic Appl. Ecol. 2014, 15, 229–240. [Google Scholar] [CrossRef]
- De Bello, F.; Lepš, J.; Sebastià, M.T. Variations in species and functional plant diversity along climatic and grazing gradients. Ecography 2006, 29, 801–810. [Google Scholar] [CrossRef]
- Marini, L.; Battisti, A.; Bona, E.; Federici, G.; Martini, F.; Pautasso, M.; Hulme, P.E. Alien and native plant life-forms respond differently to human and climate pressures. Glob. Ecol. Biogeogr. 2012, 21, 534–544. [Google Scholar] [CrossRef]
- Lomolino, M.V. Elevation gradients of species-density: Historical and prospective views. Glob. Ecol. Biogeogr. 2001, 10, 3–13. [Google Scholar] [CrossRef]
- Rahbek, C. The role of spatial scale and the perception of large-scale species-richness patterns. Ecol. Lett. 2005, 8, 224–239. [Google Scholar] [CrossRef]
- Rahbek, C. The elevational gradient of species richness: A uniform pattern? Ecography 1995, 18, 200–205. [Google Scholar] [CrossRef]
- McCain, C.M.; Grytnes, J.A. Elevational gradients in species richness. In Encyclopedia of Life Sciences; Wiley: Chichester, UK, 2010. [Google Scholar]
- Nogués-Bravo, D.; Araújo, M.; Romdal, T.; Rahbek, C. Scale effects and human impact on the elevational species richness gradients. Nature 2008, 453, 216. [Google Scholar] [CrossRef]
- Wang, G.; Zhou, G.; Yang, L.; Li, Z. Distribution, species diversity and life-form spectra of plant communities along an altitudinal gradient in the northern slopes of Qilianshan Mountains, Gansu, China. Plant Ecol. 2003, 165, 169–181. [Google Scholar] [CrossRef]
- Bhattarai, K.R.; Vetaas, O.R. Variation in plant species richness of different life forms along a subtropical elevation gradient in the Himalayas, east Nepal. Glob. Ecol. Biogeogr. 2003, 12, 327–340. [Google Scholar] [CrossRef]
- Watkins Jr, J.E.; Cardelús, C.; Colwell, R.K.; Moran, R.C. Species richness and distribution of ferns along an elevational gradient in Costa Rica. Am. J. Bot. 2006, 93, 73–83. [Google Scholar] [CrossRef]
- Desalegn, W.; Beierkuhnlein, C. Plant species and growth form richness along altitudinal gradients in the southwest Ethiopian highlands. J. Veg. Sci. 2010, 21, 617–626. [Google Scholar] [CrossRef]
- Sharma, N.; Behera, M.D.; Das, A.P.; Panda, R.M. Plant richness pattern in an elevation gradient in the Eastern Himalaya. Biodivers. Conserv. 2019, 28, 1–20. [Google Scholar] [CrossRef]
- Anderson, M.J.; Crist, T.O.; Chase, J.M.; Vellend, M.; Inouye, B.D.; Freestone, A.L.; Sanders, N.J.; Cornell, H.V.; Comita, L.S.; Davies, K.F. Navigating the multiple meanings of β diversity: A roadmap for the practicing ecologist. Ecol. Lett. 2011, 14, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Panitsa, M.; Tzanoudakis, D.; Sfenthourakis, S. Turnover of plants on small islets of the eastern Aegean Sea within two decades. J. Biogeogr. 2008, 35, 1049–1061. [Google Scholar] [CrossRef]
- Sang, W. Plant diversity patterns and their relationships with soil and climatic factors along an altitudinal gradient in the middle Tianshan Mountain area, Xinjiang, China. Ecol. Res. 2009, 24, 303–314. [Google Scholar] [CrossRef]
- Kraft, N.J.; Comita, L.S.; Chase, J.M.; Sanders, N.J.; Swenson, N.G.; Crist, T.O.; Stegen, J.C.; Vellend, M.; Boyle, B.; Anderson, M.J. Disentangling the drivers of β diversity along latitudinal and elevational gradients. Science 2011, 333, 1755–1758. [Google Scholar] [CrossRef] [PubMed]
- Tello, J.S.; Myers, J.A.; Macía, M.J.; Fuentes, A.F.; Cayola, L.; Arellano, G.; Loza, M.I.; Torrez, V.; Cornejo, M.; Miranda, T.B. Elevational gradients in β-diversity reflect variation in the strength of local community assembly mechanisms across spatial scales. PLoS ONE 2015, 10, e0121458. [Google Scholar] [CrossRef] [PubMed]
- Sabatini, F.M.; Jiménez-Alfaro, B.; Burrascano, S.; Lora, A.; Chytrý, M. Beta-diversity of central European forests decreases along an elevational gradient due to the variation in local community assembly processes. Ecography 2018, 41, 1038–1048. [Google Scholar] [CrossRef]
- Hoffmann, S.; Schmitt, T.M.; Chiarucci, A.; Irl, S.D.; Rocchini, D.; Vetaas, O.R.; Tanase, M.A.; Mermoz, S.; Bouvet, A.; Beierkuhnlein, C. Remote sensing of β-diversity: Evidence from plant communities in a semi-natural system. Appl. Veg. Sci. 2019, 22, 13–26. [Google Scholar] [CrossRef]
- Tang, Z.; Fang, J.; Chi, X.; Feng, J.; Liu, Y.; Shen, Z.; Wang, X.; Wang, Z.; Wu, X.; Zheng, C. Patterns of plant beta-diversity along elevational and latitudinal gradients in mountain forests of China. Ecography 2012, 35, 1083–1091. [Google Scholar] [CrossRef]
- Mena, J.L.; Vázquez-Domínguez, E. Species turnover on elevational gradients in small rodents. Glob. Ecol. Biogeogr. 2005, 14, 539–547. [Google Scholar] [CrossRef]
- Ah-Peng, C.; Wilding, N.; Kluge, J.; Descamps-Julien, B.; Bardat, J.; Chuah-Petiot, M.; Strasberg, D.; Hedderson, T.A. Bryophyte diversity and range size distribution along two altitudinal gradients: Continent vs. island. Acta Oecologica 2012, 42, 58–65. [Google Scholar] [CrossRef]
- Henriques, D.S.; Borges, P.A.; Ah-Peng, C.; Gabriel, R. Mosses and liverworts show contrasting elevational distribution patterns in an oceanic island (Terceira, Azores): The influence of climate and space. J. Bryol. 2016, 38, 183–194. [Google Scholar] [CrossRef]
- Chase, J.M.; Leibold, M.A. Ecological Niches: Linking Classical and Contemporary Approaches; University of Chicago Press: Chicago, IL, USA, 2003. [Google Scholar]
- Guerin, G.R.; Andersen, A.N.; Rossetto, M.; van Leeuwen, S.; Byrne, M.; Sparrow, B.; Rodrigo, M.; Lowe, A.J. Consistent sorting but contrasting transition zones in plant communities along bioclimatic gradients. Acta Oecologica 2019, 95, 74–85. [Google Scholar] [CrossRef]
- Hubbell, S.P. The Unified Neutral Theory of Biodiversity and Biogeography (MPB-32); Princeton University Press: Princeton, NJ, USA, 2001. [Google Scholar]
- Chase, J.M.; Myers, J.A. Disentangling the importance of ecological niches from stochastic processes across scales. Philos. Trans. R. Soc. B Biol. Sci. 2011, 366, 2351–2363. [Google Scholar] [CrossRef] [PubMed]
- Chase, J.M. Drought mediates the importance of stochastic community assembly. Proc. Natl. Acad. Sci. USA 2007, 104, 17430–17434. [Google Scholar] [CrossRef] [PubMed]
- Legendre, P.; Mi, X.; Ren, H.; Ma, K.; Yu, M.; Sun, I.F.; He, F. Partitioning beta diversity in a subtropical broad-leaved forest of China. Ecology 2009, 90, 663–674. [Google Scholar] [CrossRef]
- Stegen, J.C.; Freestone, A.L.; Crist, T.O.; Anderson, M.J.; Chase, J.M.; Comita, L.S.; Cornell, H.V.; Davies, K.F.; Harrison, S.P.; Hurlbert, A.H.; et al. Stochastic and deterministic drivers of spatial and temporal turnover in breeding bird communities. Glob. Ecol. Biogeogr. 2013, 22, 202–212. [Google Scholar] [CrossRef]
- Heino, J.; Melo, A.S.; Bini, L.M.; Altermatt, F.; Al-Shami, S.A.; Angeler, D.G.; Bonada, N.; Brand, C.; Callisto, M.; Cottenie, K. A comparative analysis reveals weak relationships between ecological factors and beta diversity of stream insect metacommunities at two spatial levels. Ecol. Evol. 2015, 5, 1235–1248. [Google Scholar] [CrossRef]
- Madon, O.; Médail, F. The ecological significance of annuals on a Mediterranean grassland (Mt Ventoux, France). Plant Ecol. 1997, 129, 189–199. [Google Scholar] [CrossRef]
- Turland, N.J.; Chilton, L.; Press, J.R. Flora of the Cretan Area: Annotated Checklist and Atlas; HMSO: London, UK, 1993. [Google Scholar]
- Médail, F.; Quézel, P. Biodiversity hotspots in the Mediterranean Basin: Setting global conservation priorities. Conserv. Biol. 1999, 13, 1510–1513. [Google Scholar] [CrossRef]
- Davis, S.; Heywood, V.; Hamilton, A. Centres of plant diversity. Nat. Hist. 1994, 111, 1. [Google Scholar]
- Trigas, P.; Panitsa, M.; Tsiftsis, S. Elevational gradient of vascular plant species richness and endemism in Crete–the effect of post-isolation mountain uplift on a continental island system. PLoS ONE 2013, 8, e59425. [Google Scholar] [CrossRef] [PubMed]
- Raunkiaer, C. The Life Forms of Plants and Statistical Plant Geography; Oxford University Press: Oxford, UK, 1934. [Google Scholar]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. A J. R. Meteorol. Soc. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Wheeler, B.; Torchiano, M. lmPerm: Permutation Tests for Linear Models. R package version 1.1-2. 2010. Available online: http://CRAN.R-project.org/package=lmPerm (accessed on 25 August 2013).
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’hara, R.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Package ‘vegan’. Commun. Ecol. PackageVersion 2013, 2, 1–295. [Google Scholar]
- Lazarina, M.; Sgardeli, V.; Kallimanis, A.S.; Sgardelis, S.P. An effort-based index of beta diversity. Methods Ecol. Evol. 2013, 4, 217–225. [Google Scholar] [CrossRef]
- Ulrich, W.; Gotelli, N.J. A null model algorithm for presence–absence matrices based on proportional resampling. Ecol. Model. 2012, 244, 20–27. [Google Scholar] [CrossRef]
- Hastie, T.J. Generalized additive models. In Statistical Models in S; Routledge: Abingdon, UK, 2017; pp. 249–307. [Google Scholar]
- Wood, S.N. mgcv: GAMs and generalized ridge regression for R. R News 2001, 1, 20–25. [Google Scholar]
- RC Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Steinbauer, M.J.; Irl, S.D.; Beierkuhnlein, C. Elevation-driven ecological isolation promotes diversification on Mediterranean islands. Acta Oecologica 2013, 47, 52–56. [Google Scholar] [CrossRef]
- Keddy, P.A. Assembly and response rules: Two goals for predictive community ecology. J. Veg. Sci. 1992, 3, 157–164. [Google Scholar] [CrossRef]
- Diaz, S.; Cabido, M.; Casanoves, F. Plant functional traits and environmental filters at a regional scale. J. Veg. Sci. 1998, 9, 113–122. [Google Scholar] [CrossRef]
- Irl, S.D.; Schweiger, A.H.; Hoffmann, S.; Beierkuhnlein, C.; Hartmann, H.; Pickel, T.; Jentsch, A. Spatiotemporal dynamics of plant diversity and endemism during primary succession on an oceanic-volcanic island. J. Veg. Sci. 2019, 30, 587–598. [Google Scholar] [CrossRef]
- Körner, C. Alpine plant life. In Functional Plant Ecology of High Mountain Ecosystems; with 47 tables; Springer Science & Business Media: Berlin, Germany, 2003. [Google Scholar]
- Blumler, M.A. What is the ‘True’Mediterranean-type vegetation? In Geographical Changes in Vegetation and Plant Functional Types; Springer: Cham, Switzerland, 2018; pp. 117–139. [Google Scholar]
- Arianoutsou, M.; Delipetrou, P.; Celesti-Grapow, L.; Basnou, C.; Bazos, I.; Kokkoris, Y.; Blasi, C.; Vilà, M. Comparing naturalized alien plants and recipient habitats across an east–west gradient in the Mediterranean Basin. J. Biogeogr. 2010, 37, 1811–1823. [Google Scholar] [CrossRef]
- Carlsson, B.A.; Karlsson, P.S.; Svensson, B.M. Alpine and subalpine vegetation. Acta Phytogeogr. Suec. 1999, 84, 75–90. [Google Scholar]
- Herben, T.; Krahulec, F.; Hadincová, V.; Kováf Ova, M. Small-scale spatial dynamics of plant species in a grassland community over six years. J. Veg. Sci. 1993, 4, 171–178. [Google Scholar] [CrossRef]
- Pellissier, L.; Fournier, B.; Guisan, A.; Vittoz, P. Plant traits co-vary with altitude in grasslands and forests in the European Alps. Plant Ecol. 2010, 211, 351–365. [Google Scholar] [CrossRef]
- Vogiatzakis, I.; Griffiths, G.H.; Mannion, A.M. Environmental factors and vegetation composition, Lefka Ori massif, Crete, S. Aegean. Glob. Ecol. Biogeogr. 2003, 12, 131–146. [Google Scholar] [CrossRef]
- Debussche, M.; Escarré, J.; Lepart, J.; Houssard, C.; Lavorel, S. Changes in Mediterranean plant succession: Old-fields revisited. J. Veg. Sci. 1996, 7, 519–526. [Google Scholar] [CrossRef]
- Procheş, Ş.; Cowling, R.M.; Goldblatt, P.; Manning, J.C.; Snijman, D.A. An overview of the Cape geophytes. Biol. J. Linn. Soc. 2006, 87, 27–43. [Google Scholar] [CrossRef]
- Danin, A.; Orshan, G. The distribution of Raunkiaer life forms in Israel in relation to the environment. J. Veg. Sci. 1990, 1, 41–48. [Google Scholar] [CrossRef]
- Kazakis, G.; Ghosn, D.; Vogiatzakis, I.; Papanastasis, V. Vascular plant diversity and climate change in the alpine zone of the Lefka Ori, Crete. Biodivers. Conserv. 2007, 16, 1603–1615. [Google Scholar] [CrossRef]
- Sánchez-González, A.; López-Mata, L. Plant species richness and diversity along an altitudinal gradient in the Sierra Nevada, Mexico. Divers. Distrib. 2005, 11, 567–575. [Google Scholar] [CrossRef]
- Descombes, P.; Vittoz, P.; Guisan, A.; Pellissier, L. Uneven rate of plant turnover along elevation in grasslands. Alp. Bot. 2017, 127, 53–63. [Google Scholar] [CrossRef]
- McCain, C.M.; Beck, J. Species turnover in vertebrate communities along elevational gradients is idiosyncratic and unrelated to species richness. Glob. Ecol. Biogeogr. 2016, 25, 299–310. [Google Scholar] [CrossRef]
- La Sorte, F.A.; McKinney, M.L.; Pyšek, P.; Klotz, S.; Rapson, G.; Celesti-Grapow, L.; Thompson, K. Distance decay of similarity among European urban floras: The impact of anthropogenic activities on β diversity. Glob. Ecol. Biogeogr. 2008, 17, 363–371. [Google Scholar] [CrossRef]
- La Sorte, F.A.; McKinney, M.L.; Pyšek, P. Compositional similarity among urban floras within and across continents: Biogeographical consequences of human-mediated biotic interchange. Glob. Chang. Biol. 2007, 13, 913–921. [Google Scholar] [CrossRef]
- Socolar, J.B.; Gilroy, J.J.; Kunin, W.E.; Edwards, D.P. How should beta-diversity inform biodiversity conservation? Trends Ecol. Evol. 2016, 31, 67–80. [Google Scholar] [CrossRef]
| Chamaephytes | Geophytes | Hemicryptophytes | Phanerophytes | Therophytes | |
|---|---|---|---|---|---|
| Species Distribution Related Variables | |||||
| geographical range | 0.09 ± 0.11 a | 0.05 ± 0.04 b | 0.02 ± 0.01 c | 0.13 ± 0.14 d | 0.01 ± 0.00 c |
| % Single- & doubletons | 0.24 | 0.23 | 0.20 | 0.16 | 0.25 |
| Diversity Indices | |||||
| γ-diversity | 176 (12%) * | 227 (15%) * | 379 (25%) * | 100 (7%) * | 636 (42%) * |
| mean α-diversity | 16.53 ± 12.99 ab | 21.38 ± 17.14 a | 28.99 ± 26.97 c | 13.05 ± 10.88 d | 38.21 ± 44.88 bc |
| Ν* index | 9.53 | 10.25 | 12.65 | 7.54 | 17.54 |
| Jaccard index | 0.89 ± 0.11 | 0.91 ± 0.10 | 0.92 ± 0.09 | 0.88 ± 0.13 | 0.96 ± 0.06 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lazarina, M.; Charalampopoulos, A.; Psaralexi, M.; Krigas, N.; Michailidou, D.-E.; Kallimanis, A.S.; Sgardelis, S.P. Diversity Patterns of Different Life Forms of Plants along an Elevational Gradient in Crete, Greece. Diversity 2019, 11, 200. https://doi.org/10.3390/d11100200
Lazarina M, Charalampopoulos A, Psaralexi M, Krigas N, Michailidou D-E, Kallimanis AS, Sgardelis SP. Diversity Patterns of Different Life Forms of Plants along an Elevational Gradient in Crete, Greece. Diversity. 2019; 11(10):200. https://doi.org/10.3390/d11100200
Chicago/Turabian StyleLazarina, Maria, Athanasios Charalampopoulos, Maria Psaralexi, Nikos Krigas, Danai-Eleni Michailidou, Athanasios S. Kallimanis, and Stefanos P. Sgardelis. 2019. "Diversity Patterns of Different Life Forms of Plants along an Elevational Gradient in Crete, Greece" Diversity 11, no. 10: 200. https://doi.org/10.3390/d11100200
APA StyleLazarina, M., Charalampopoulos, A., Psaralexi, M., Krigas, N., Michailidou, D.-E., Kallimanis, A. S., & Sgardelis, S. P. (2019). Diversity Patterns of Different Life Forms of Plants along an Elevational Gradient in Crete, Greece. Diversity, 11(10), 200. https://doi.org/10.3390/d11100200






