Forest Diversity and Structure in the Amazonian Mountain Ranges of Southeastern Ecuador
Abstract
:1. Introduction
2. Methods
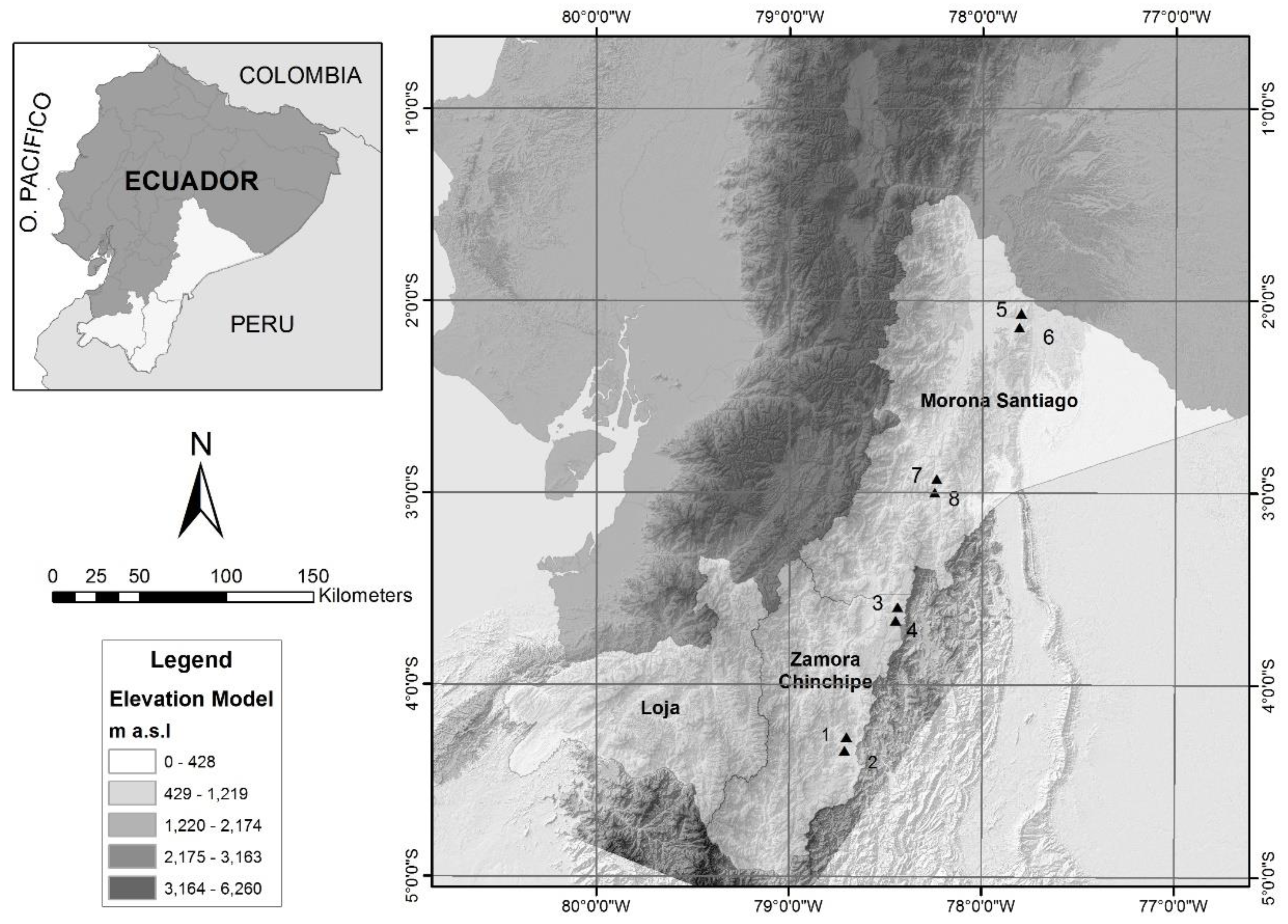
2.1. Study Area
2.2. Plot Installation
2.3. Climatic and Edaphic Variables
2.4. Statistical Analysis
3. Results
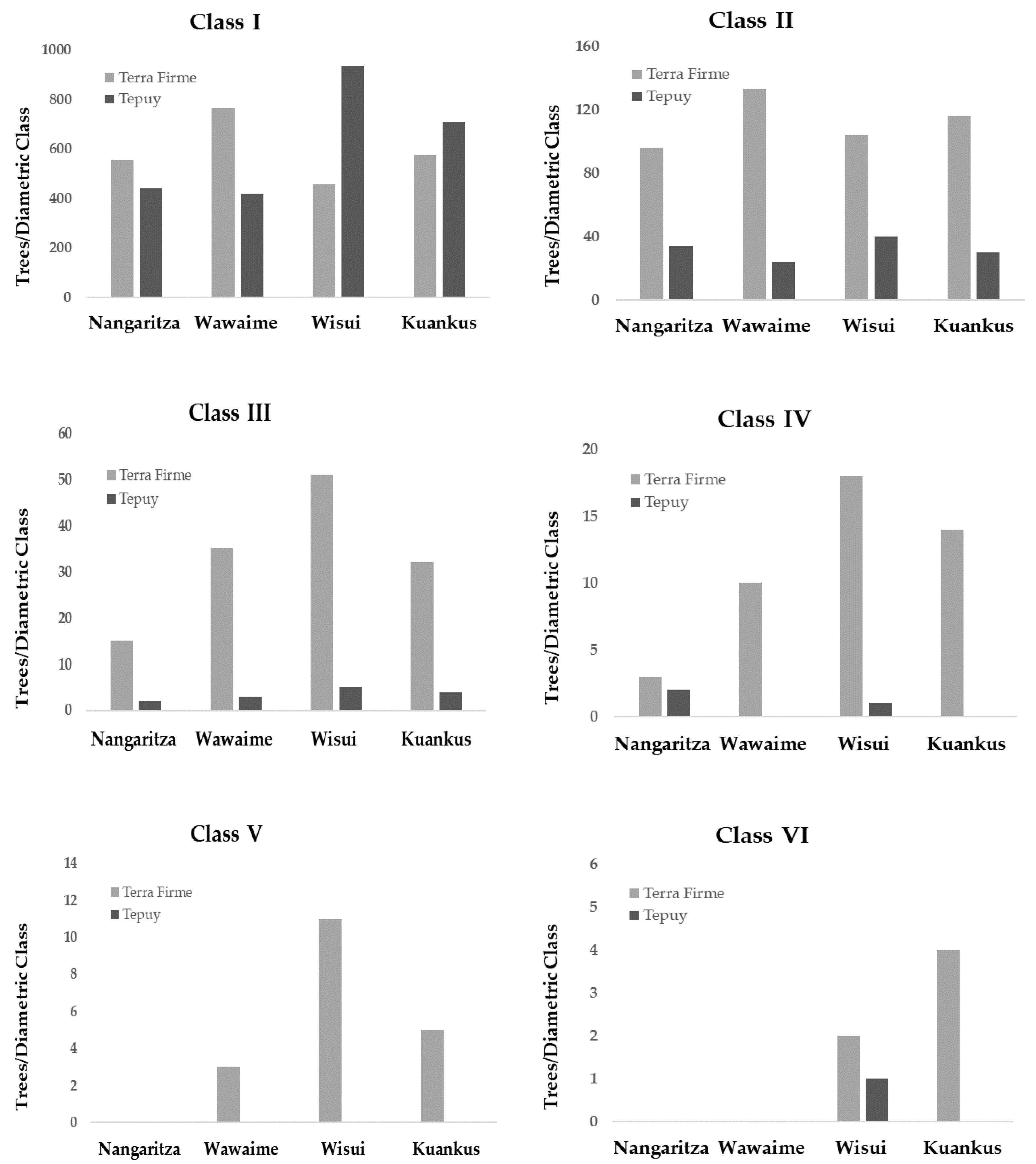
3.1. Alpha Diversity and Forest Structure
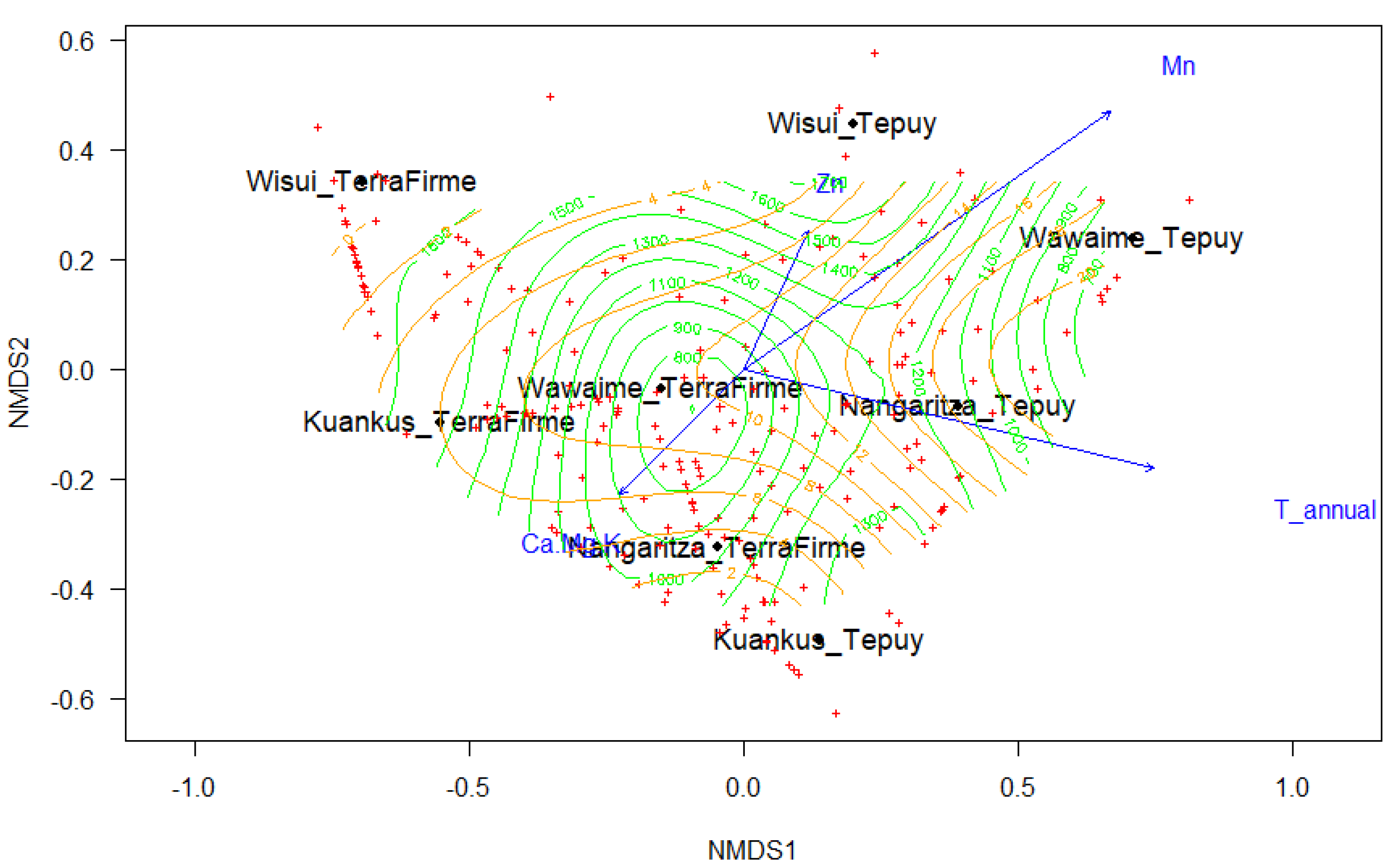
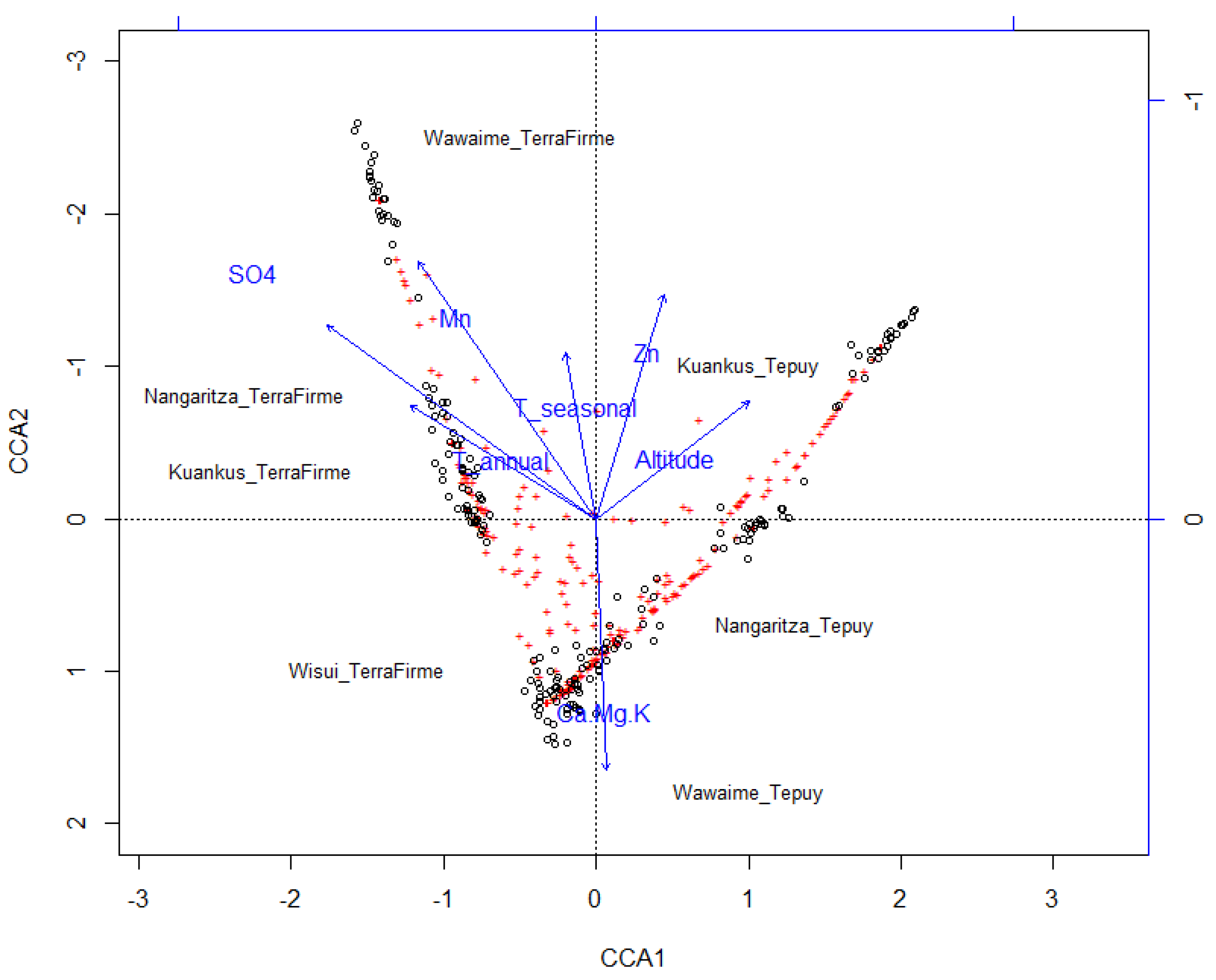
3.2. Species Turnover
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shi, H.; Singh, A.; Kant, S.; Zhu, Z.; Waller, E. Integrating Habitat Status, Human Population Pressure, and Protection Status into Biodiversity Conservation Priority Setting. Conserv. Biol. 2005, 19, 1273–1285. [Google Scholar] [CrossRef]
- Guevara Andino, J.E.; Pitman, N.C.A.; ter Steege, H.; Mogollón, H.; Ceron, C.; Palacios, W.; Oleas, N.; Fine, P.V.A. Incorporating Phylogenetic Information for the Definition of Floristic Districts in Hyperdiverse Amazon Forests: Implications for Conservation. Ecol. Evol. 2017, 7, 9639–9650. [Google Scholar] [CrossRef] [PubMed]
- Valencia, R.; Balsley, H.; Paz y Miño, G. High Tree Alpha Diversity in Amazonian Ecuador. Biodivers. Conserv. 1994, 3, 21–28. [Google Scholar] [CrossRef]
- Añazco, M.; Morales, M.; Palacios, W.; Vega, E.; Cuesta, A. Sector Forestal Ecuatoriano: Propuestas para una gestión forestal sostenible; Serie Investigación y Sistematización No. 8; Programa Regional ECOBONA-INTERCOOPERATION: Quito, Ecuador, 2010. [Google Scholar]
- Malhi, Y.; Phillips, O.L.; Lloyd, J.; Baker, T.; Wright, J.; Almeida, S.; Arroyo, L.; Frederiksen, T.; Grace, G.; Higuchi, N.; et al. An international Network to Monitor the Structure, Composition and Dynamics of Amazonian Forests (RAINFOR). J. Veg. Sci. 2002, 13, 439–450. [Google Scholar] [CrossRef]
- Dinerstein, E.; Olson, D.M.; Graham, D.J.; Webster, A.L.; Pimm, S.A.; Bookbinder, M.P.; Ledec, G.C. A Conservation Assessment of the Terrestrial Ecoregions of Latin America and the Caribbean; The World Bank/World Wildlife Fund: Washington, DC, USA, 1995. [Google Scholar]
- Duque, A.; Cárdenas, D.; Rodríguez, N. Dominancia Florística y Variabilidad Estructural en Bosques de Tierra Firme en el Noroccidente de la Amazonía Colombiana. Caldasia 2003, 25, 139–152. [Google Scholar]
- Neill, D.A. Cordillera del Cóndor: Botanical Treasures between the Andes and the Amazon. Plant Talk 2005, 41, 17–21. [Google Scholar]
- Silva, J.; Montoya, A.; López, D.; Hurtado, H. Variación florística de especies arbóreas a escala local en un bosque de tierra firme en la Amazonia colombiana. Acta Amaz. 2010, 40, 179–188. [Google Scholar] [CrossRef]
- Gentry, H.A. Tree Species Richness of Upper Amazonian Forests. Proc. Natl. Acad. Sci. USA 1988, 85, 156–159. [Google Scholar] [CrossRef]
- Phillips, O.L.; Hall, P.; Gentry, A.H.; Sawyer, S.A.; Vasquez, R. Dynamics and Species Richness of Tropical Rain Forests. Proc. Natl. Acad. Sci. USA 1994, 91, 2805–2809. [Google Scholar] [CrossRef]
- Valencia, R.; Foster, R.B.; Villa, G.; Condit, R.; Svenning, J.C.; Hernández, C.; Romeleroux, K.; Losos, E.; Magard, E.; Basley, H. Tree Species Distributions and Local Habitat Variation in the Amazon: Large Forest Plot in Eastern Ecuador. J. Ecol. 2004, 92, 214–229. [Google Scholar] [CrossRef]
- ter Steege, H.; Sabatier, D.; Casterllanos, H.; Van Andel, T.; Duivenvoorden, J.; De Oliveira, A.; Ek, R.; Lilwak, R.; Maas, P.; Mori, S. An Analysis of the Floristic Composition and Diversity of Amazonian Forests Including Those of the Guiana Shield. J. Trop. Ecol. 2000, 16, 801–828. [Google Scholar] [CrossRef]
- Fabregas, G. The Cordillera del Cóndor region of Ecuador and Peru: A Biological Assessment; Schulenberg, T.S., Awbrey, K., Eds.; RAP Working Papers 7; Consernation International: Washington, DC, USA, 1997; pp. 1–231. [Google Scholar]
- Vásquez, R.; Phillips, O. Allpahuayo: Floristics, Structure, and Dynamics of a High.Diversity Forest in Amazonian Perú. Ann. Mo. Bot. Gard. 2000, 87, 499–527. [Google Scholar] [CrossRef]
- Pitman, N.C.A.; Terborgh, J.W.; Silman, M.R.; Percy-Núñez, V.; Neill, D.A.; Ceron, C.E.; Palacios, W.A.; Aulestia, M. Dominance and Distribution of Tree Species in Upper Amazonian Terra Firme Forests. Ecology 2001, 82, 2101–2117. [Google Scholar] [CrossRef]
- Ulloa, C.; Neill, D. Phainantha Shuariorum (Melastomataceae), una especie nueva de la Cordillera del Cóndor, Ecuador, disyunta de un género guayanés. Novon 2006, 16, 281–285. [Google Scholar] [CrossRef]
- Guayasamin, J.M.; Bonaccorso, E. (Eds.) Evaluación Ecológica Rápida de la Biodiversidad de los Tepuyes de la Cuenca Alta del Río Nangaritza, Cordillera del Cóndor, Ecuador; Conservación Internacional: Quito, Ecuador, 2011. [Google Scholar]
- Neill, D.; Asanza, M. Lozania nunkui (Lacistemataceae), A New Species from the Sandstone Plateaus of the Cordillera del Cóndor in Ecuador and Peru. Novon 2012, 22, 207–211. [Google Scholar] [CrossRef]
- Dallmeier, F. Long-Term Monitoring of Biological Diversity in Tropical Forest Areas-Methods for Establishment and Inventory of Permanent Plots; United Nations Educational, Scientific and Cultural Organization: Paris, France, 1992. [Google Scholar]
- Campbell, P.; Comiskey, J.; Alonso, A.; Dallmeier, F.; Nuñez, P.; Beltran, H.; Baldeon, S.; Nauray, W.; Colina, R.D.L.; Acurio, L.; et al. Modified Whittaker Plots as An Assessment and Monitoring Tool for Vegetation in A Lowland Tropical Rainforest. Environ. Monit. Assess. 2002, 76, 19–41. [Google Scholar] [CrossRef]
- Condit, R.S.; Ashton, M.S.; Balslev, H.; Brokaw, N.V.L.; Bunyavejchewin, S.; Chuyong, G.B.; Co, L.; Dattaraja, H.S.; Davies, S.J.; Esufali, S.; et al. Tropical Tree A-Diversity: Results from A Worldwide Network of Large Plots. Biologiske Skri. 2005, 55, 565–582. [Google Scholar]
- Brako, L.; Zarucchi, J.L. Catálogo de las Angiospermas y Gimnospermas del Perú. Monogr. Syst. Bot. Mo. Bot. Gard. 1993, 45, 1–1286. [Google Scholar]
- Jørgensen, P.M.; León-Yánez, S. (Eds.) Catalogue of the Vascular Plants of Ecuador. Monogr. Syst. Bot. Mo. Bot. Gard. 1999, 75, 1–1181. [Google Scholar]
- León-Yánez, S.; Valencia, R.; Pitman, N.; Endara, L.; Ulloa, C.; Navarrete, H. (Eds.) Libro rojo de las plantas endémicas del Ecuador, 2nd ed.; Pontificia Universidad Católica del Ecuador: Quito, Ecuador, 2011. [Google Scholar]
- Cabrera, O.; Benítez, Á.; Cumbicus, N.; Naranjo, C.; Ramón, P.; Tinitana, F.; Escudero, A. Geomorphology and Altitude Effects on the Diversity and Structure of the Vanishing Montane Forest of Southern Ecuador. Diversity 2019, 11, 32. [Google Scholar] [CrossRef]
- Madsen, J.E.; Øllgaard, B. Floristic Composition, Structure, and Dynamics of An Upper Montane rain Forest iSouthern Ecuador. Nord. J. Bot. 1994, 14, 403–423. [Google Scholar] [CrossRef]
- Magurran, A.E. Why diversity? In Ecological Diversity and Its Measurement; Springer: Dordrecht, The Netherlands, 1988; pp. 1–5. [Google Scholar]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very High Resolution Interpolated Climate Surfaces for Global Land Areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Mccullagh, P.; Nelder, J.A. Generalized Linear Models, 2nd ed.; CRC Press/Chapman and Hall: Boca Raton, FL, USA, 1989. [Google Scholar]
- R Development Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; ISBN 3-900051-07-0. [Google Scholar]
- Hill, M.O.; Gauch, H.G. Detrended Correspondence Analysis: An Improved Ordination Technique. Vegetation 1980, 42, 47–58. [Google Scholar] [CrossRef]
- ter Braak, C.J.F. Canonical Correspondence Analysis, A New Eigenvector Technique for Multivariate Direct Gradient Analysis. Ecology 1986, 67, 1167–1179. [Google Scholar] [CrossRef]
- Legendre, P.; Anderson, M.J. Distance-Based Redundancy Analysis: Testing Multispecies Responses in Multifactorial Ecological Experiments. Ecol Monogr 1999, 69, 1–24. [Google Scholar] [CrossRef]
- Oksanen, J. Constrained Ordination: Tutorial with R and Vegan. 2012. Available online: http://cc.oulu.fi/~jarioksa/opetus/metodi/sessio2.pdf (accessed on 31 May 2019).
- Cayuela, L.; Golicher, D.J.; Rey Benayas, J.M.; González-Espinosa, M.; Ramírez-Marcial, N. Fragmentation, Disturbance and Tree Diversity Conservation in Tropical Montane Forests. J. Appl. Ecol. 2006, 43, 1172–1181. [Google Scholar] [CrossRef]
- Legendre, P.; Gallagher, E.D. Ecologically Meaningful Transformations for Ordination of Species Data. Oecologia 2001, 129, 271–280. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Oksanen, M.J. Package ‘Vegan’. Community Ecology Package; Version, 2(9). 2013. Available online: https://cran.r-project.org/web/packages/vegan/vegan.pdf (accessed on 12 October 2018).
- Condit, R.; Hubbell, S.P.; Lafrankie, J.V.; Sukumar, R.; Manokaran, N.; Foster, R.B.; Ashton, P.S. Species-Area and Species-Individual Relationships for Tropical Trees: A Comparison of Three 50-ha Plots. J. Ecol. 1996, 84, 549–562. [Google Scholar] [CrossRef]
- Gentry, A.H. Floristic Similarities and Differences between Southern Central America and Upper and Central Amazonia. In Four Neotropical Rainforests; Gentry, A.H., Ed.; Yale University Press: New Haven, CT, USA, 1990; pp. 141–157. [Google Scholar]
- Huamantupa, I. Análisis de la composición arbórea en los bosques amazónicos de tierra firme en la base de los Andes. Tesis de Maestría, Universidad Internacional Menéndez Pelayo, Madrid, Spain, 2009. [Google Scholar]
- Condit, R.; Pitman, N.; Leigh, E.G.; Chave, J.; Terborgh, J.; Foster, R.B.; Nuñez, P.; Aguilar, S.; Valencia, R.; Villa, G.; et al. Betadiversity in Tropical Forest Trees. Science 2002, 295, 666–669. [Google Scholar] [CrossRef]
- Fine, P.V.A.; Villacorta, R.; Pitman, N.; Mesones, I.; Kembel, W. A Floristic Study of the White-Sand Forests of Peru. Ann. Mo. Bot. Gard. 2010, 97, 283–305. [Google Scholar] [CrossRef]
- Lieberman, D.; Lieberman, M.; Peralta, R.; Hartshorn, G. Tropical Forest Structure and Composition on A Large-Scale Altitudinal Gradient in Costa Rica. J. Ecol. 1996, 84, 137–152. [Google Scholar] [CrossRef]
- Aiba, S.; Kitayama, K. Structure, Composition and Species Diversity in An Altitude-Substrate Matrix of Rain Forest Tree Communities on Mount Kinabalu, Borneo. Plant. Ecol. 1999, 140, 139–157. [Google Scholar] [CrossRef]
- Kessler, M. The Elevational Gradient of Andean Plant Endemism: Varying Influences of Taxon-Specific Traits and Topography at Different Taxonomic Levels. J. Biogeogr. 2002, 29, 1159–1165. [Google Scholar] [CrossRef]
- Homeier, J.; Breckle, S.; Gunter, S.; Rollembeck, R.; Leuschner, C. Tree Diversity, Forest Structure and Productivity along Altitudinal and Topographical Gradients in a Species-Rich Ecuadorian Montane Rain Forest. Biotropica 2010, 42, 140–148. [Google Scholar] [CrossRef]
- Clinebell, R.R.; Phillips, O.L.; Gentry, A.H.; Stark, N.; Zuuring, H. Prediction of Neotropical Tree and Liana Species Richness from Soil and Climatic Data. Biodivers. Conserv. 1995, 4, 56–90. [Google Scholar] [CrossRef]
- Givnish, T.J. On the Causes of Gradients in Tropical Tree Diversity. J. Ecol. 1999, 87, 193–210. [Google Scholar] [CrossRef]
- Tilman, D.; Pacala, S. The Maintenance of Species Diversity in Plant Communities. Species Diversity in Ecological Communities; University of Chicago Press: Chicago, IL, USA, 1993. [Google Scholar]
- Rajaniemi, T.K. Explaining Productivity-Diversity Relationships in Plants. Oikos 2003, 101, 449–457. [Google Scholar] [CrossRef]
- Slik, J.F.; Raes, N.; Aiba, S.I.; Brearley, F.Q.; Cannon, C.H.; Meijaard, E.; Nagamasu, H.; Nilus, R.; Paoli, G.; Poulsen, A.; et al. Environmental Correlates for Tropical Tree Diversity and Distribution Patterns in Borneo. Divers. Distrib. 2009, 15, 523–532. [Google Scholar] [CrossRef]
- Escudero, A. Community Patterns on Exposed Cliffs in a Mediterranean Calcareous Mountain. Vegetatio 1996, 125, 99–110. [Google Scholar] [CrossRef]
- Espinosa, C.I.; Cabrera, O.; Luzuriaga, A.L.; Escudero, A. What Factors Affect Diversity and Species Composition of Endangered Tumbesian Dry Forests in Southern Ecuador? Biotropica 2011, 43, 15–22. [Google Scholar] [CrossRef]
- Poulsen, A.D.; Tuomisto, H.; Baslev, H. Edaphic and Floristic Variation a 1-ha Plot of Lowland Amazonian Rain Forest. Biotropica 2006, 38, 468–478. [Google Scholar] [CrossRef]
- Duivenvoorden, J.F.; Lips, J.M. A Land-Ecological Study of Soils, Vegetation, and Plant Diversity in Colombian Amazonia; Tropenbos Foundation: Wageningen, The Netherlands, 1995. [Google Scholar]
- Toumisto, H.; Ruokolainen, K.; Kalliola, R.; Linna, A.; Danjoy, W.; Rodriguez, M. Dissecting Amazonian Biodiversity. Science 1995, 269, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Tuomisto, H.; Ruokolainen, K.; Yli-Halla, M. Dispersal, Environment, and Floristic Variation of Western Amazonian Forests. Science 2003, 299, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Duque, A.; Sanchez, M.; Cavelier, J.; Duivenvoorden, J.F. Different Floristic Patterns of Woody Understorey and Canopy Plants in Colombian Amazonia. J. Trop. Ecol. 2002, 18, 499–525. [Google Scholar] [CrossRef]
- Phillips, O.L.; Nuñez, P.; Vargas, A.; Monteagudo, A.; Peñacruz, A.; Chuspezans, M.E.; Galiano, W.; Sanchez, M.; Yli-Halla, M.; Rose, S. Habitat Association among Amazonian Tree Species: A Landscape Scale Approach. J. Ecol. 2003, 91, 757–775. [Google Scholar] [CrossRef]
- ter Steege, H.; Pitman, N.C.A.; Phillips, O.L.; Chave, J.; Sabatier, D.; Duque, A.; Molino, J.F.; Prevost, M.-F.; Spichiger, R.; Castellanos, H.; et al. Continental-Scale Patterns of Canopy Tree Composition and Function across Amazonia. Nature 2006, 443, 444–447. [Google Scholar] [CrossRef]
- Chytry, M.; Danihelka, J.; Kubesova, S.; Lustyk, P.; Ermakov, N.; Hajek, M.; Hajkova, P.; Koci, M.; Otypkova, Z.; Rolecek, J.; et al. Diversity of Forest Vegetation across a Strong Gradient of Climatic Continentally: Western Sayan Mountains, Southern Siberia. Plant Ecol. 2008, 196, 61–83. [Google Scholar] [CrossRef]
- Sollins, P. Factors Influencing Species Composition in Tropical Lowland Rain Forest: Does Soil Matter? Ecology 1998, 79, 23–30. [Google Scholar] [CrossRef]
- Duque, A.; Sanchez, M.; Cavelier, J.; Duivenvoorden, J.F.; Mirana, P.; Mirana, J.; Mtapi, A. 2001 Relación Bosque-Ambiente en el Medio Caquetá, Amazonia colombiana. In Evaluación de recursos vegetales no maderables en la Amazonia noroccidental; Duivenvoorden, J., Balslev, H., Cavelier, J., Grandez, C., Tuomisto, H., Valencia, R., Eds.; Fluid Phase Equilibria 2001; Caracterización, Ambiental; IBED, Universiteit van Amsterdam: Amsterdam, The Netherlands, 2001; pp. 23–30. [Google Scholar]
- Jones, M.M.; Tuomisto, H.; Olivas, P.C. Differences in the Degree of Environmental Control on Large and Small Tropical Plants: Just a Sampling Effect? J. Ecol. 2008, 96, 367–377. [Google Scholar] [CrossRef]
| MODEL | Coef. | SD | t-Value | p |
|---|---|---|---|---|
| Intercept | 16.355 | 0.3695 | 44.257 | <0.001 |
| PC1 | 0.3693 | 0.1277 | 2.892 | 0.0042 |
| PC2 | −0.8885 | 0.1489 | −5.967 | <0.001 |
| Vectors | NMDS1 | NMDS2 | r2 | p-Value |
|---|---|---|---|---|
| Soil | ||||
| Potassium | −0.3582 | −0.9336 | 0.13 | 0.001 *** |
| Boron | 0.2160 | 0.9764 | 0.13 | 0.001 *** |
| Acidity | −0.2344 | −0.9721 | 0.10 | 0.001 *** |
| Magnesium | −0.2496 | −0.9684 | 0.09 | 0.002 ** |
| Manganese | −0.1163 | −0.9932 | 0.08 | 0.001 *** |
| Ammonium | 0.1225 | 0.9925 | 0.07 | 0.003 ** |
| Calcium | −0.3771 | −0.9262 | 0.07 | 0.003 ** |
| Cooper | −0.1009 | −0.9949 | 0.07 | 0.004 ** |
| Zinc | −0.2935 | −0.9560 | 0.06 | 0.003 ** |
| Organic Material | 0.1225 | 0.9925 | 0.06 | 0.05 |
| Sodium | −0.1816 | −0.9834 | 0.04 | 0.023 * |
| Climatic | NMDS1 | NMDS2 | r2 | p-Value |
| Mean Annual Temperature | 0.4554 | −0.8903 | 0.05 | 0.009 ** |
| Seasonal Precipitation | 0.9169 | −0.3990 | 0.03 | 0.048 * |
| Terrain | NMDS1 | NMDS2 | r2 | p-Value |
| Altitude | 0.1486 | 0.9889 | 0.11 | 0.001 *** |
| Forest Type | Families | Genera | Species | Trees/ha | Shanon | Basal Area/ha |
|---|---|---|---|---|---|---|
| Nangaritza Terra Firme I | 39 | 81 | 121 | 670 | 3.99 | 21.35 |
| Wawaime Terra Firme II | 41 | 90 | 135 | 948 | 4.02 | 34.65 |
| Wisui Terra Firme III | 45 | 100 | 155 | 643 | 4.3 | 36.02 |
| Kuankos Terra Firme IV | 43 | 106 | 191 | 748 | 3.93 | 36.01 |
| Summary | 42 ± 2.6 | 94.3 ± 11 | 150.5 ± 30.4 | 752.3 ± 137.9 | 4.1 ± 0.16 | 32 ± 7.1 |
| Nangaritza Tepuy I | 39 | 61 | 80 | 478 | 3.92 | 22.74 |
| Wawaime Tepuy II | 24 | 33 | 50 | 445 | 2.8 | 9.12 |
| Wisui Tepuy III | 35 | 68 | 87 | 985 | 3.5 | 21.06 |
| Kuankos Tepuy IV | 38 | 74 | 138 | 743 | 3.5 | 12.79 |
| Summary | 34 ± 6.9 | 59 ± 18.1 | 88.8 ± 36.5 | 662 ± 252.3 | 3.4 ± 0.5 | 16.4 ± 6.5 |
| Site/Plot | CLASS I | CLASS II | CLASS III | CLASS IV | CLASS V | CLASS VI | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Df | Chi-sq | p | Chi-sq | p | Chi-sq | p | Chi-sq | p | Chi-sq | p | Chi-sq | p | |
| Nangaritza | 1 | 3.8 | 0.05 * | 17.8 | <0.001 *** | 7.2 | 0.007 ** | 0.94 | 0.33 ns | n/a | n/a | n/a | n/a |
| Nangaritza | |||||||||||||
| Wawaime | 1 | 31.5 | <0.001 *** | 29.7 | <0.001 *** | 25.9 | <0.001 *** | 10.7 | 0.001 *** | 3.12 | 0.08 ns | n/a | n/a |
| Wawaime | |||||||||||||
| Wisui | 1 | 30.7 | <0.001 *** | 10.3 | 0.001 *** | 22.6 | <0.001 *** | 8.2 | 0.004 ** | 9.3 | 0.003 ** | 0.35 | 0.55 ns |
| Wisui | |||||||||||||
| Kuankos | 1 | 3.7 | 0.05 * | 13.6 | <0.001 *** | 12.9 | <0.001 *** | 13.7 | <0.001 *** | 3.1 | 0.07 ns | 3.1 | 0.07 ns |
| Kuankos | |||||||||||||
| PLOTS | Average Dissimilarity |
|---|---|
| Tepuy I–Terra Firme III | 99.87 |
| Terra Firme III–Tepuy IV | 99.86 |
| Tepuy I–Terra Firme IV | 99.75 |
| Terra Firme III–Tepuy III | 99.75 |
| Terra Firme I–Terra Firme III | 99.3 |
| Terra Firme IV–Tepuy IV | 99.06 |
| Tepuy I–Tepuy IV | 99.02 |
| Terra Firme I–Tepuy III | 98.94 |
| Tepuy I–Tepuy III | 98.81 |
| Terra Firme I–Terra Firme IV | 98.55 |
| Tepuy I–Terra Firme II | 98.29 |
| Terra Firme I–Tepuy II | 97.53 |
| Terra Firme I–Tepuy I | 97.4 |
| Terra Firme II–TY III | 97.24 |
| Terra Firme II–Terra Firme III | 96.65 |
| Terra Firme III–Terra Firme IV | 96.58 |
| Terra Firme II–Tepuy II | 96.49 |
| Terra Firme II–Terra Firme IV | 95.5 |
| Terra Firme II–Tepuy IV | 95.3 |
| Tepuy I–UY II | 93.92 |
| Terra Firme I–Tepuy IV | 93.34 |
| Terra Firme I–Terra Firme II | 87.54 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quizhpe, W.; Benítez, Á.; Cuenca, K.; Uvidia, H.; Huamantupa, I.; Muñoz, J.; Cabrera, O. Forest Diversity and Structure in the Amazonian Mountain Ranges of Southeastern Ecuador. Diversity 2019, 11, 196. https://doi.org/10.3390/d11100196
Quizhpe W, Benítez Á, Cuenca K, Uvidia H, Huamantupa I, Muñoz J, Cabrera O. Forest Diversity and Structure in the Amazonian Mountain Ranges of Southeastern Ecuador. Diversity. 2019; 11(10):196. https://doi.org/10.3390/d11100196
Chicago/Turabian StyleQuizhpe, Wilson, Ángel Benítez, Klever Cuenca, Hernán Uvidia, Isau Huamantupa, Jesús Muñoz, and Omar Cabrera. 2019. "Forest Diversity and Structure in the Amazonian Mountain Ranges of Southeastern Ecuador" Diversity 11, no. 10: 196. https://doi.org/10.3390/d11100196
APA StyleQuizhpe, W., Benítez, Á., Cuenca, K., Uvidia, H., Huamantupa, I., Muñoz, J., & Cabrera, O. (2019). Forest Diversity and Structure in the Amazonian Mountain Ranges of Southeastern Ecuador. Diversity, 11(10), 196. https://doi.org/10.3390/d11100196






