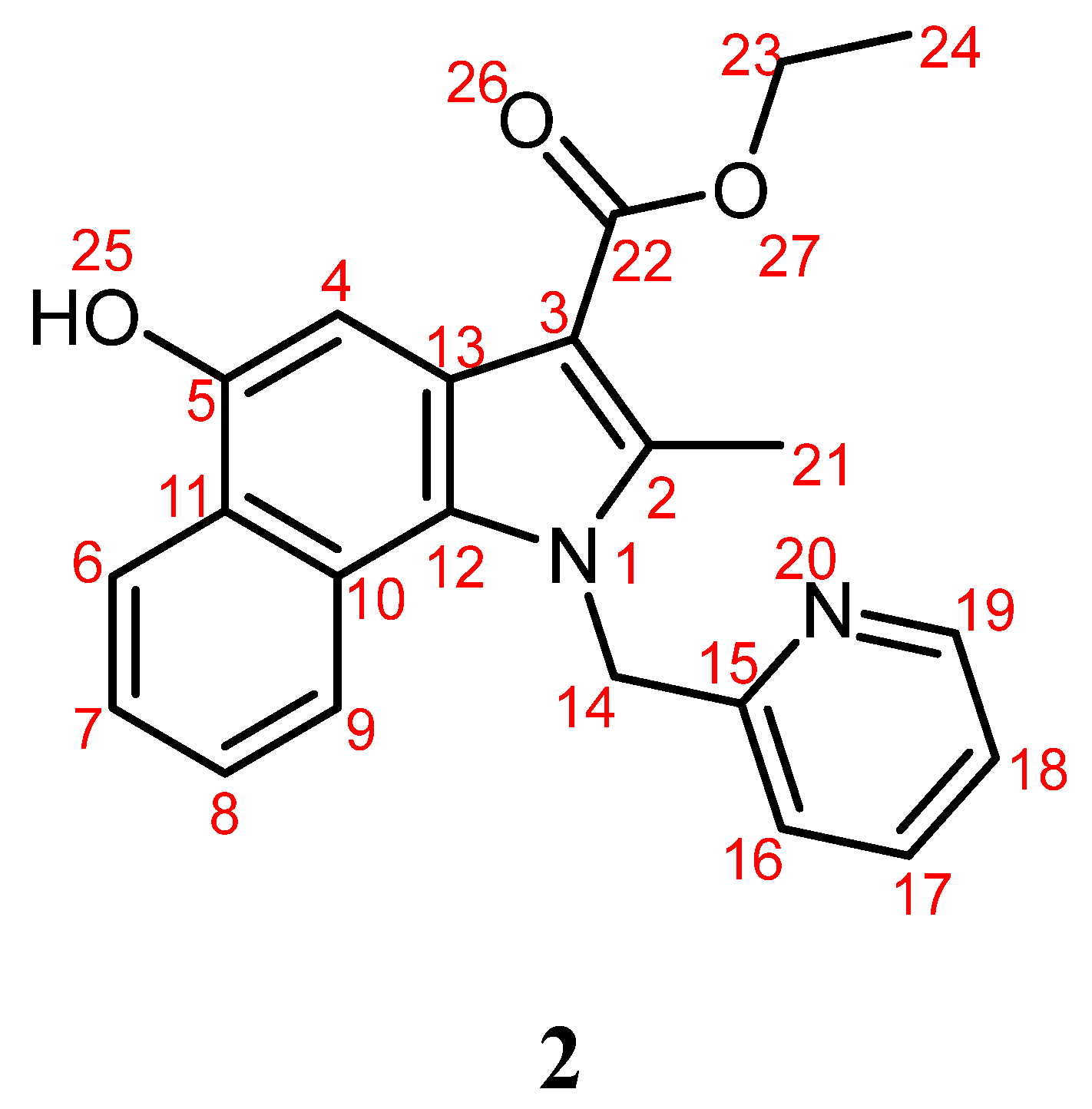
Ethyl 5-Hydroxy-2-methyl-1-(pyridin-2-ylmethyl)benzo[g]indole-3-carboxylate
Abstract
1. Introduction
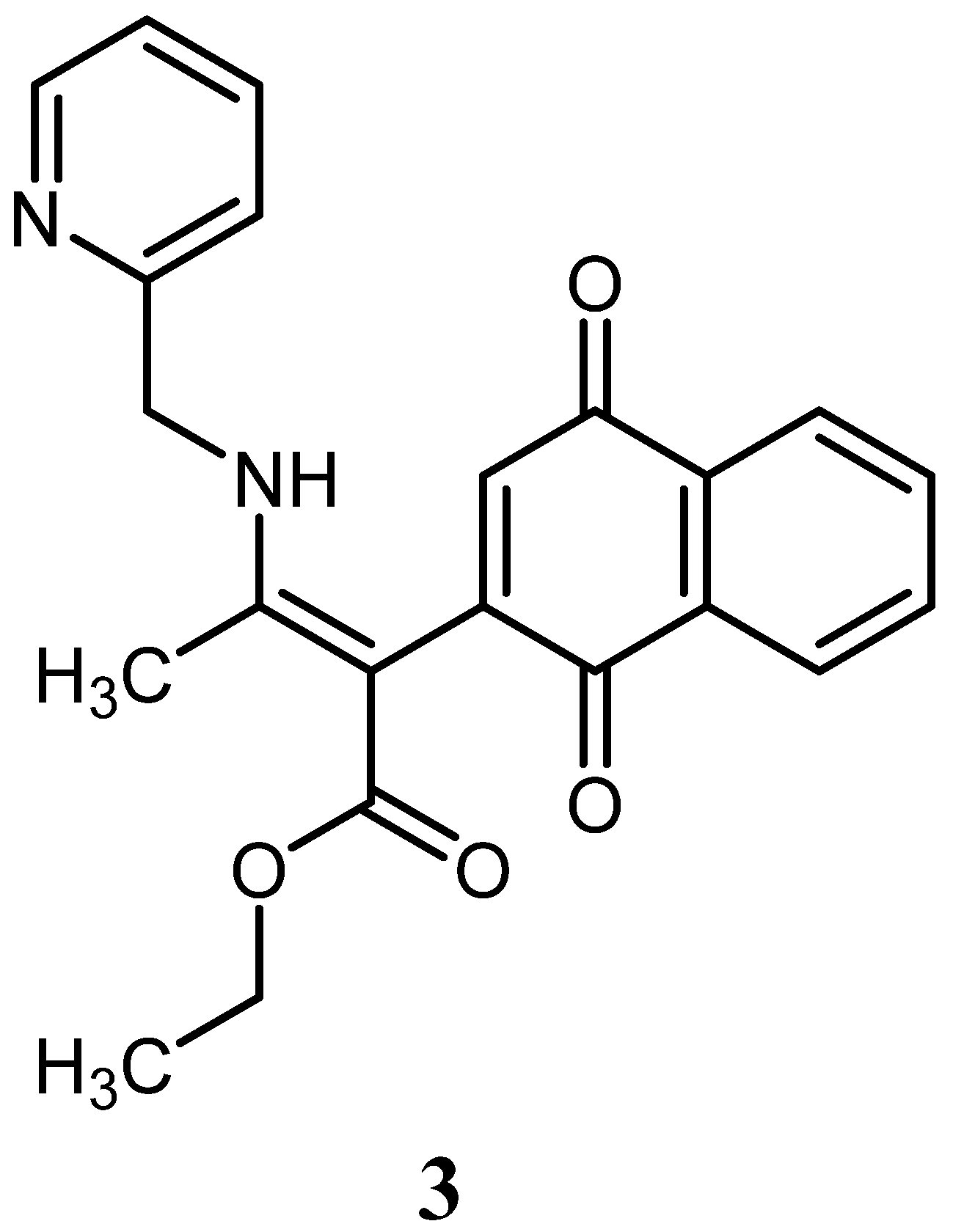
2. Results
3. Discussion
4. Materials and Methods
4.1. Materials
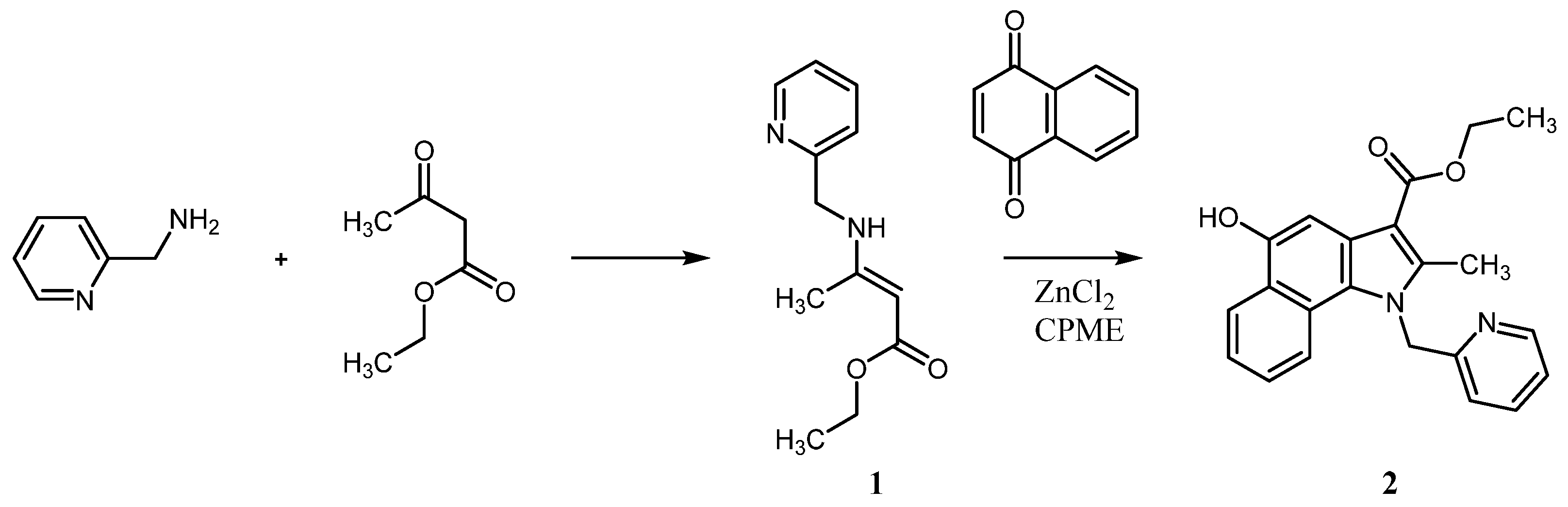
4.2. Synthesis of Ethyl 3-[(Pyridin-2-ylmethyl)amino]but-2-enoate 1
4.3. Synthesis of Ethyl 5-Hydroxy-2-methyl-1-(pyridin-2-ylmethyl)benzo[g]indole-3-carboxylate 2
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Umer, S.M.; Solangi, M.; Khan, K.M.; Saleem, R.S.Z. Indole-Containing Natural Products 2019–2022: Isolations, Reappraisals, Syntheses, and Biological Activities. Molecules 2022, 27, 7586. [Google Scholar] [CrossRef] [PubMed]
- McKinney, J.; Teigen, K.; Frøystein, N.Å.; Salaün, C.; Knappskog, P.M.; Haavik, J.; Martínez, A. Conformation of the Substrate and Pterin Cofactor Bound to Human Tryptophan Hydroxylase. Important Role of Phe313 in Substrate Specificity. Biochemistry 2001, 40, 15591–15601. [Google Scholar] [CrossRef] [PubMed]
- Schwenker, G.; Stiefvater, K. Prodrugs Nichtsteroidaler Antirheumatika, 3. Mitt.: Synthese Und Hydrolyse von Acyloxy-2-Oxopropanen. Arch. Pharm. 1991, 324, 547–550. [Google Scholar] [CrossRef]
- Haviernik, J.; Štefánik, M.; Fojtíková, M.; Kali, S.; Tordo, N.; Rudolf, I.; Hubálek, Z.; Eyer, L.; Ruzek, D. Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses. Viruses 2018, 10, 184. [Google Scholar] [CrossRef] [PubMed]
- Syed, Y.Y. Anlotinib: First Global Approval. Drugs 2018, 78, 1057–1062. [Google Scholar] [CrossRef] [PubMed]
- Romero, D.L.; Morge, R.A.; Biles, C.; Berrios-Pena, N.; May, P.D.; Palmer, J.R.; Johnson, P.D.; Smith, H.W.; Busso, M. Discovery, Synthesis, and Bioactivity of Bis(Heteroaryl)Piperazines. 1. A Novel Class of Non-Nucleoside HIV-1 Reverse Transcriptase Inhibitors. J. Med. Chem. 1994, 37, 999–1014. [Google Scholar] [CrossRef] [PubMed]
- Dillard, R.D.; Bach, N.J.; Draheim, S.E.; Berry, D.R.; Carlson, D.G.; Chirgadze, N.Y.; Clawson, D.K.; Hartley, L.W.; Johnson, L.M.; Jones, N.D.; et al. Indole Inhibitors of Human Nonpancreatic Secretory Phospholipase A2. 2. Indole-3-Acetamides with Additional Functionality. J. Med. Chem. 1996, 39, 5137–5158. [Google Scholar] [CrossRef]
- Mukai, C.; Takahashi, Y. A New Entry to the Synthesis of 2,3-Disubstituted Indoles. Org. Lett. 2005, 7, 5793–5796. [Google Scholar] [CrossRef] [PubMed]
- Saito, A.; Kanno, A.; Hanzawa, Y. Synthesis of 2,3-Disubstituted Indoles by a Rhodium-Catalyzed Aromatic Amino-Claisen Rearrangement of N-Propargyl Anilines. Angew. Chem. Int. Ed. 2007, 46, 3931–3933. [Google Scholar] [CrossRef] [PubMed]
- Nenitzescu, C.D. Derivatives of 2-methyl-5-hydroxyindole. Bull. Soc. Chim. Rom. 1929, 11, 37. [Google Scholar]
- Patrick, J.B.; Saunders, E.K. Studies on the Nenitzescu Synthesis of 5-Hydroxyindoles. Tetrahedron Lett. 1979, 20, 4009–4012. [Google Scholar] [CrossRef]
- Suryavanshi, P.A.; Sridharan, V.; Menéndez, J.C. Expedient, One-Pot Preparation of Fused Indoles via CAN-Catalyzed Three-Component Domino Sequences and Their Transformation into Polyheterocyclic Compounds Containing Pyrrolo [1,2-a]Azepine Fragments. Org. Biomol. Chem. 2010, 8, 3426–3436. [Google Scholar] [CrossRef] [PubMed]
- Teymori, A.; Sedaghat, A.; Kobarfard, F. Ca-Mediated Nenitzescu Synthesis of 5-Hydroxyindoles. Chem. Pap. 2023, 77, 1791–1795. [Google Scholar] [CrossRef]
- Thönnißen, V.; Atodiresei, I.L.; Patureau, F.W. Atroposelective Nenitzescu Indole Synthesis. Chem.—Eur. J. 2023, 29, e202300279. [Google Scholar] [CrossRef] [PubMed]
- Satta, G.; Usala, E.; Solinas, A.; Römer, M.; Livesi, M.; Pira, G.M.; Beccu, A.; Carboni, S.; Gaspa, S.; De Luca, L.; et al. Nenitzescu Synthesis of 5-Hydroxyindoles with Zinc, Iron and Magnesium Salts in Cyclopentyl Methyl Ether. Eur. J. Org. Chem. 2021, 2021, 5835–5842. [Google Scholar] [CrossRef]
- Defant, A.; Mancini, I. A Study on the Regioselectivity in N,C-Acylation of β-Enamino-Esters. Tetrahedron 2013, 69, 4586–4590. [Google Scholar] [CrossRef]
- Xu, P.; Huang, K.; Liu, Z.; Zhou, M.; Zeng, W. An Efficient and Convenient Synthesis of 1,2,3-Trisubstituted Pyrroles via Iodocyclization from Ethyl Acetoacetate. Tetrahedron Lett. 2013, 54, 2929–2933. [Google Scholar] [CrossRef]
- Muramatsu, Y.; Hans Wedepohl, K. The Distribution of Iodine in the Earth’s Crust. Chem. Geol. 1998, 147, 201–216. [Google Scholar] [CrossRef]
- Pennington, J.A. A Review of Iodine Toxicity Reports. J. Am. Diet. Assoc. 1990, 90, 1571–1581. [Google Scholar] [CrossRef] [PubMed]
- Southern, A.P.; Anastasopoulou, C.; Jwayyed, S. Iodine Toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Capello, C.; Fischer, U.; Hungerbühler, K. What Is a Green Solvent? A Comprehensive Framework for the Environmental Assessment of Solvents. Green Chem. 2007, 9, 927–934. [Google Scholar] [CrossRef]
- Azzena, U.; Carraro, M.; Pisano, L.; Monticelli, S.; Bartolotta, R.; Pace, V. Cyclopentyl Methyl Ether: An Elective Ecofriendly Ethereal Solvent in Classical and Modern Organic Chemistry. ChemSusChem 2019, 12, 40–70. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Satta, G.; Gaspa, S.; Luca, L.D.; Pisano, L.; Carraro, M. Ethyl 5-Hydroxy-2-methyl-1-(pyridin-2-ylmethyl)benzo[g]indole-3-carboxylate. Molbank 2024, 2024, M1840. https://doi.org/10.3390/M1840
Satta G, Gaspa S, Luca LD, Pisano L, Carraro M. Ethyl 5-Hydroxy-2-methyl-1-(pyridin-2-ylmethyl)benzo[g]indole-3-carboxylate. Molbank. 2024; 2024(3):M1840. https://doi.org/10.3390/M1840
Chicago/Turabian StyleSatta, Giuseppe, Silvia Gaspa, Lidia De Luca, Luisa Pisano, and Massimo Carraro. 2024. "Ethyl 5-Hydroxy-2-methyl-1-(pyridin-2-ylmethyl)benzo[g]indole-3-carboxylate" Molbank 2024, no. 3: M1840. https://doi.org/10.3390/M1840
APA StyleSatta, G., Gaspa, S., Luca, L. D., Pisano, L., & Carraro, M. (2024). Ethyl 5-Hydroxy-2-methyl-1-(pyridin-2-ylmethyl)benzo[g]indole-3-carboxylate. Molbank, 2024(3), M1840. https://doi.org/10.3390/M1840




