2-((5-(3-(2-Fluorophenyl)acryloyl)-4-methylthiazol-2-yl)amino)isoindoline-1,3-dione
Abstract
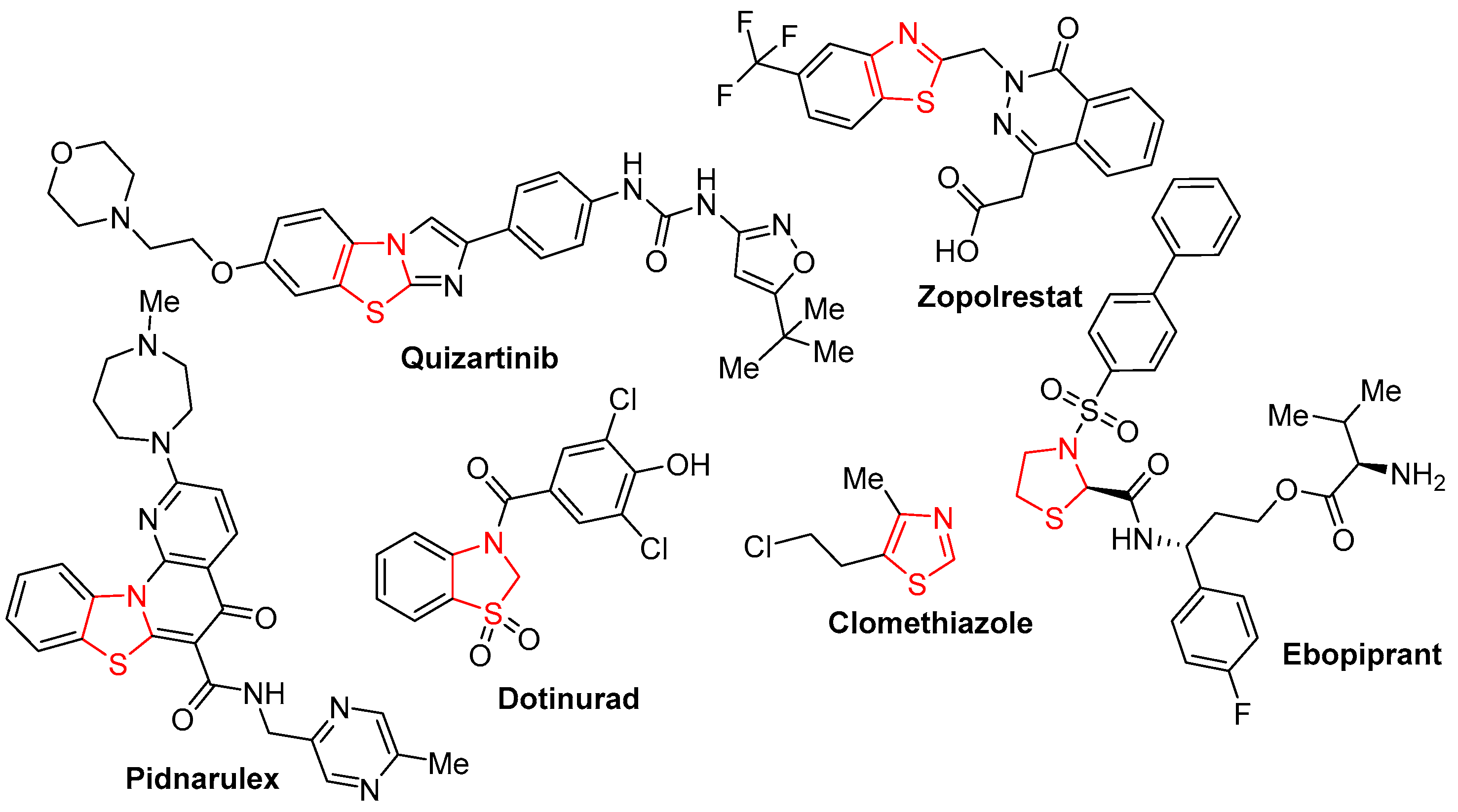
1. Introduction
2. Results and Discussion
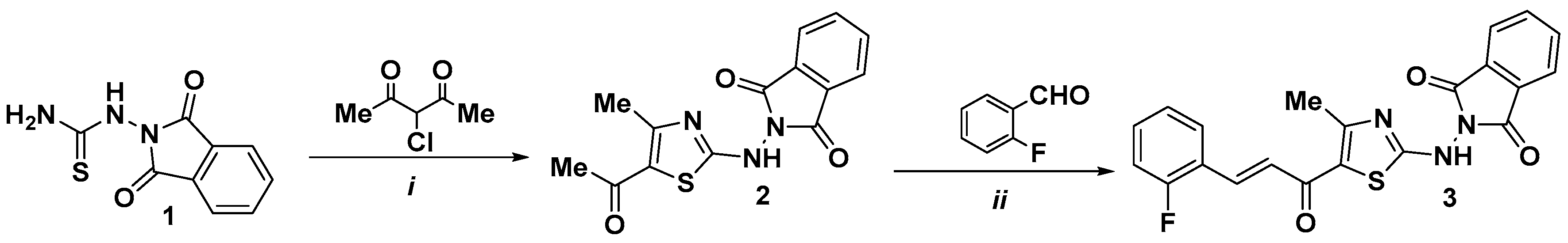
2.1. Synthesis of the Title Compound 3
2.2. In Vitro Evaluation of the Anticancer Activity of Compound 3
2.3. Molecular and Pharmacokinetic Properties
3. Materials and Methods
3.1. General Information and Compound 3 Synthesis
3.1.1. 2-((5-Acetyl-4-methylthiazol-2-yl)amino)isoindoline-1,3-dione (2)
3.1.2. (E)-2-((5-(3-(2-Fluorophenyl)acryloyl)-4-methylthiazol-2-yl)amino)isoindoline-1,3-dione (3)
3.2. In Vitro Anticancer Assay
3.3. Molecular and Pharmacokinetic Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Petrou, A.; Fesatidou, M.; Geronikaki, A. Thiazole ring—A biologically active scaffold. Molecules 2021, 26, 3166. [Google Scholar] [CrossRef] [PubMed]
- Roszczenko, P.; Holota, S.; Szewczyk, O.K.; Dudchak, R.; Bielawski, K.; Bielawska, A.; Lesyk, R. Thiazolidinone-bearing hybrid molecules in anticancer drug design. Int. J. Mol. Sci. 2022, 23, 13135. [Google Scholar] [CrossRef] [PubMed]
- Niu, Z.X.; Wang, Y.T.; Zhang, S.N.; Li, Y.; Chen, X.B.; Wang, S.Q.; Liu, H.M. Application and synthesis of thiazole ring in clinically approved drugs. Eur. J. Med. Chem. 2023, 250, 115172. [Google Scholar] [CrossRef] [PubMed]
- Shepeta, Y.; Lozynskyi, A.; Sulyma, M.; Nektegayev, I.; Grellier, P.; Lesyk, R. Synthesis and biological activity evaluation of new thiazolidinone-diclofenac hybrid molecules. Phosphorus Sulfur Silicon Relat. Elem. 2020, 195, 836–841. [Google Scholar] [CrossRef]
- Gupta, A.; Mishra, P.; Kashaw, S.K.; Jatav, V.; Stables, J.P. Synthesis and anticonvulsant activity of some novel 3-aryl amino/amino-4-aryl-5-imino-Δ2-1,2,4-thiadiazoline. Eur. J. Med. Chem. 2008, 43, 749–754. [Google Scholar] [CrossRef] [PubMed]
- Lozynskyi, A.V.; Yushyn, I.M.; Konechnyi, Y.T.; Roman, O.M.; Matiykiv, O.V.; Smaliukh, O.V.; Mosula, L.M.; Polovkovych, S.V.; Lesyk, R.B. Synthesis and evaluation of biological activity of 1-[2-amino-4-methylthiazol-5-yl]-3-arylpropenones. Biopolym. Cell 2021, 37, 389–399. [Google Scholar] [CrossRef]
- Monks, A.; Scudiero, D.; Skehan, P.; Shoemaker, R.; Paull, K.; Vistica, D.; Hose, C.; Langley, J.; Cronise, P.; Vaigro-Wolff, A.; et al. Feasibility of a high-flux anticancer drug screen using a diverse panel of cultured human tumor cell lines. J. Natl. Cancer Inst. 1991, 83, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Boyd, M.R.; Paull, K.D. Some practical considerations and applications of the National Cancer Institute in vitro anticancer drug discovery screen. Drug Dev. Res. 1995, 34, 91–109. [Google Scholar] [CrossRef]
- Boyd, M.R. Cancer Drug Discovery and Development; Teicher, B.A., Ed.; Humana Press: Totowa, NJ, USA, 1997; Volume 2, pp. 23–43. [Google Scholar]
- Shoemaker, R.H. The NCI60 human tumour cell line anticancer drug screen. Nat. Rev. Cancer 2006, 6, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Rostom, S.A.F. Synthesis and in vitro antitumor evaluation of some indeno[1,2-c]pyrazol(in)es substituted with sulfonamide, sulfonylurea(-thiourea) pharmacophores, and some derived thiazole ring systems. Bioorganic Med. Chem. 2006, 14, 6475–6485. [Google Scholar] [CrossRef] [PubMed]
- SwissADME. Available online: http://www.swissadme.ch/ (accessed on 20 July 2023).
| Compound | 60 Cell Lines Assay in One Dose, 10 μM | ||
|---|---|---|---|
| Mean Growth, % | Range of Growth, % | Most Sensitive Cell Line(s) Growth Inhibition Percent 1/Line/Panel 2 | |
| 3 | 12.53 | −49.97 to 62.87 | −3.50/RPMI-8226/L −17.47/HOP-62/NSCLC 3.87/KM12/CC −49.97/SF-539/CNSC 4.88/SNB-19/CNSC 3.98/U251/CNSC −22.59/MDA-MB-435/M 4.94/SK-MEL-5/M 0.65/UACC-62/M −5.19/OVCAR-3/OC −27.71/OVCAR-8/OC −3.97/NCI/ADR-RES/OC 1.87/RXF 393/RC −2.72/SN12C/RC −44.35/DU-145/PC −10.35/T-47D/BC −15.65/MDA-MB-468/BC |
| Disease | Cell Line | GI50, μM 1 | SI (GI50) | TGI, μM | SI (TGI) | LC50, μM |
|---|---|---|---|---|---|---|
| NSC lung cancer | HOP-62 NCI-H460 | 3.18 6.25 | 4.94 2.51 | >100.0 28.1 | - 1.80 | >100.0 >100.0 |
| Colon cancer | COLO 205 | 1.70 | 9.24 | No data | No data | No data |
| Ovarian cancer | SK-OV-3 | 3.28 | 4.79 | >100.0 | - | >100.0 |
| Breast cancer | T-47D | 2.78 | 5.65 | >100.0 | - | >100.0 |
| Physicochemical Properties | ||
|---|---|---|
| 1 | Molecular weight | 407.42 |
| 2 | Num. of heavy atoms | 29 |
| 3 | Num. of arom. heavy atoms | 17 |
| 4 | Num. of rotatable bonds | 5 |
| 5 | Num. of H-bond acceptors | 5 |
| 6 | Num. of H-bond donors | 1 |
| 7 | Molar refractivity | 111.14 |
| 8 | TPSA Å2 | 107.61 |
| 9 | Consensus log Po/w | 3.82 |
| 10 | LipinskiˈRule | Yes |
| Pharmacokinetics | ||
| 11 | GI absorption | High |
| 12 | BBB permeant | No |
| 13 | P-gp substrate | No |
| 14 | CYP1A2 inhibitor | Yes |
| 15 | CYP2C19 inhibitor | Yes |
| 16 | CYP2C9 inhibitor | Yes |
| 17 | CYP2D6 inhibitor | No |
| 18 | CYP3A4 inhibitor | Yes |
| 19 | Log Kp (SP) (cm/s) (skin permeation) | −5.42 |
| 20 | Bioavailability score | 0.55 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fedusevych, O.-M.; Lozynskyi, A.; Sulyma, M.; Lesyk, R. 2-((5-(3-(2-Fluorophenyl)acryloyl)-4-methylthiazol-2-yl)amino)isoindoline-1,3-dione. Molbank 2024, 2024, M1785. https://doi.org/10.3390/M1785
Fedusevych O-M, Lozynskyi A, Sulyma M, Lesyk R. 2-((5-(3-(2-Fluorophenyl)acryloyl)-4-methylthiazol-2-yl)amino)isoindoline-1,3-dione. Molbank. 2024; 2024(1):M1785. https://doi.org/10.3390/M1785
Chicago/Turabian StyleFedusevych, Olha-Maria, Andrii Lozynskyi, Marta Sulyma, and Roman Lesyk. 2024. "2-((5-(3-(2-Fluorophenyl)acryloyl)-4-methylthiazol-2-yl)amino)isoindoline-1,3-dione" Molbank 2024, no. 1: M1785. https://doi.org/10.3390/M1785
APA StyleFedusevych, O.-M., Lozynskyi, A., Sulyma, M., & Lesyk, R. (2024). 2-((5-(3-(2-Fluorophenyl)acryloyl)-4-methylthiazol-2-yl)amino)isoindoline-1,3-dione. Molbank, 2024(1), M1785. https://doi.org/10.3390/M1785







