4-Chloro-6-(chloromethyl)-1-methyl-1H-pyrazolo[3,4-d]pyrimidine
Abstract
:1. Introduction
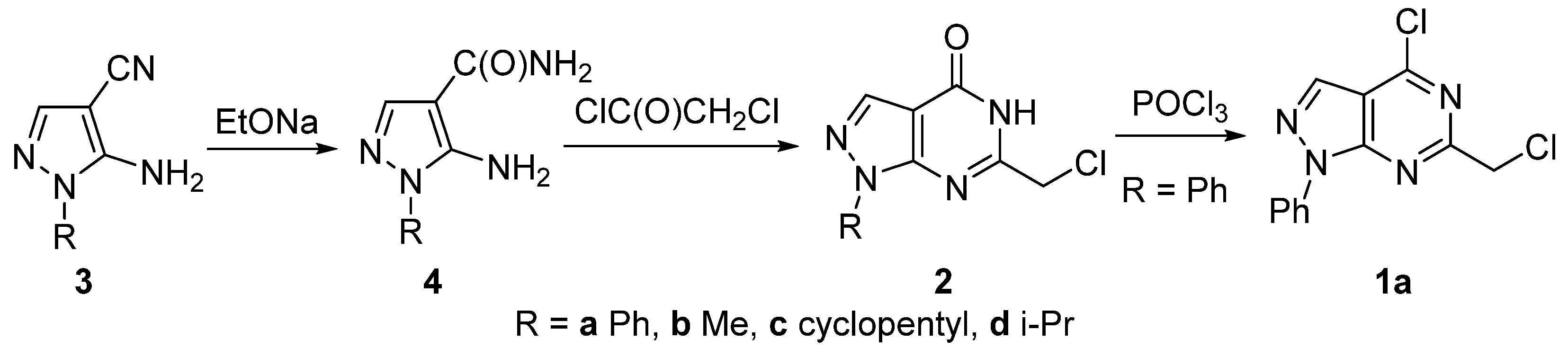
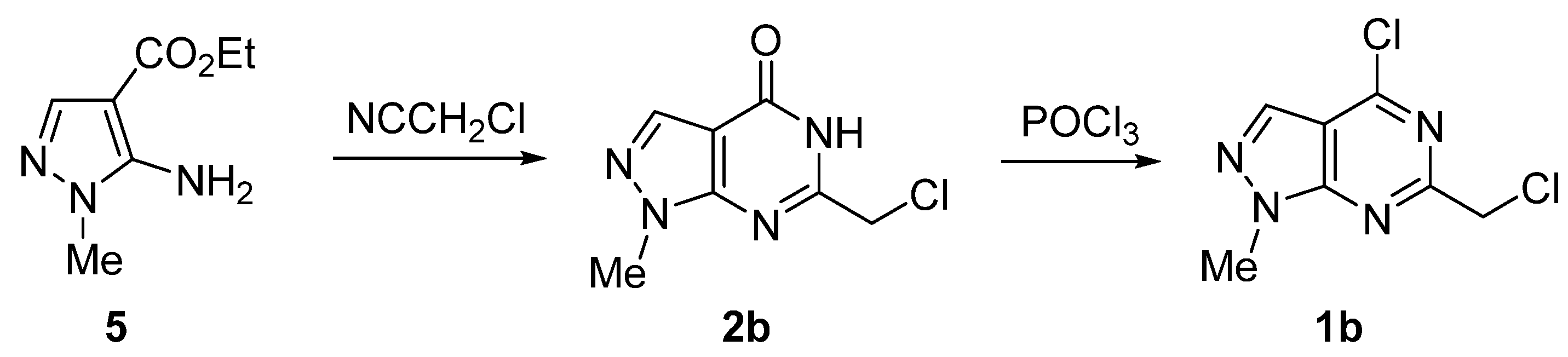
2. Results and Discussion
3. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Elnagdi, M.H.; Al-Awadi, N.; Abdelhamid, I.A. Bicyclic 5-6 Systems: Other Four Heteroatoms 2:2. In Comprehensive Heterocyclic Chemistry III; Katritzky, A.R., Ramsden, C.A., Scriven, E.F.V., Taylor, R.J.K., Eds.; Elsevier: Oxford, UK, 2008; Volume 10, pp. 600–658. [Google Scholar] [CrossRef]
- Kumar, V.; Kaur, K.; Gupte, G.P.; Sharma, A.K. Pyrazole containing natural products: Synthetic preview and biological significance. Eur. J. Med. Chem. 2013, 69, 735–753. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ju, Q.; Sun, J.; Huang, L.; Wu, S.; Wang, S.; Li, Y.; Guan, Z.; Zhu, Q.; Xu, Y. Discovery of novel dual extracellular regulated protein kinases (erk) and phosphoinositide 3-kinase (pi3k) inhibitors as a promising strategy for cancer therapy. Molecules 2020, 25, 5693. [Google Scholar] [CrossRef] [PubMed]
- Janetka, J.W.; Hopper, A.T.; Yang, Z.; Barks, J.; Dhason, M.S.; Wang, Q.; Sibley, L.D. Optimizing pyrazolopyrimidine inhibitors of calcium dependent protein kinase 1 for treatment of acute and chronic toxoplasmosis. J. Med. Chem. 2020, 63, 6144–6163. [Google Scholar] [CrossRef] [PubMed]
- Devarakonda, M.; Doonaboina, R.; Vanga, S.; Vemu, J.; Boni, S.; Mailavaram, R.P. Synthesis of novel 2-alkyl-4-substituted-amino-pyrazolo[3,4-d]pyrimidines as new leads for anti-bacterial and anti-cancer activity. Med. Chem. Res. 2013, 22, 1090–1101. [Google Scholar] [CrossRef]
- Shaaban, M.A.; Elshaier, Y.A.M.M.; Hammad, A.H.; Farag, N.A.; Haredy, H.H.; AbdEl-Ghany, A.A.; Mohamed, K.O. Design and synthesis of pyrazolo[3,4-d]pyrimidinone derivatives: Discovery of selective phosphodiesterase-5 inhibitors. Bioorg. Med. Chem. Lett. 2020, 30, 127337. [Google Scholar] [CrossRef] [PubMed]
- Burdi, D.F.; Tanaka, D. Tricyclic Piperazine Derivative. US Patent 2016/83391, 24 March 2016. [Google Scholar]
- Bol’but, A.V.; Vovk, M.V. Condensed pyrimidine systems. 5.6-Methyl-functionalized in pyrazolo[3,4-d]pyrimidin-4(5H)-ones. Zhurn. Organ. Farm. Khim. 2006, 4, 57–61. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogurtsov, V.A.; Rakitin, O.A. 4-Chloro-6-(chloromethyl)-1-methyl-1H-pyrazolo[3,4-d]pyrimidine. Molbank 2021, 2021, M1253. https://doi.org/10.3390/M1253
Ogurtsov VA, Rakitin OA. 4-Chloro-6-(chloromethyl)-1-methyl-1H-pyrazolo[3,4-d]pyrimidine. Molbank. 2021; 2021(3):M1253. https://doi.org/10.3390/M1253
Chicago/Turabian StyleOgurtsov, Vladimir A., and Oleg A. Rakitin. 2021. "4-Chloro-6-(chloromethyl)-1-methyl-1H-pyrazolo[3,4-d]pyrimidine" Molbank 2021, no. 3: M1253. https://doi.org/10.3390/M1253
APA StyleOgurtsov, V. A., & Rakitin, O. A. (2021). 4-Chloro-6-(chloromethyl)-1-methyl-1H-pyrazolo[3,4-d]pyrimidine. Molbank, 2021(3), M1253. https://doi.org/10.3390/M1253






