(R,S)-2-{[4-(4-Methoxyphenyl)-5-phenyl-4H-1,2,4-triazol-3-yl] thio}-1-phenyl-1-ethanol
Abstract
1. Introduction
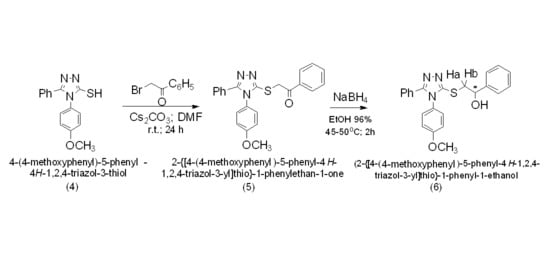
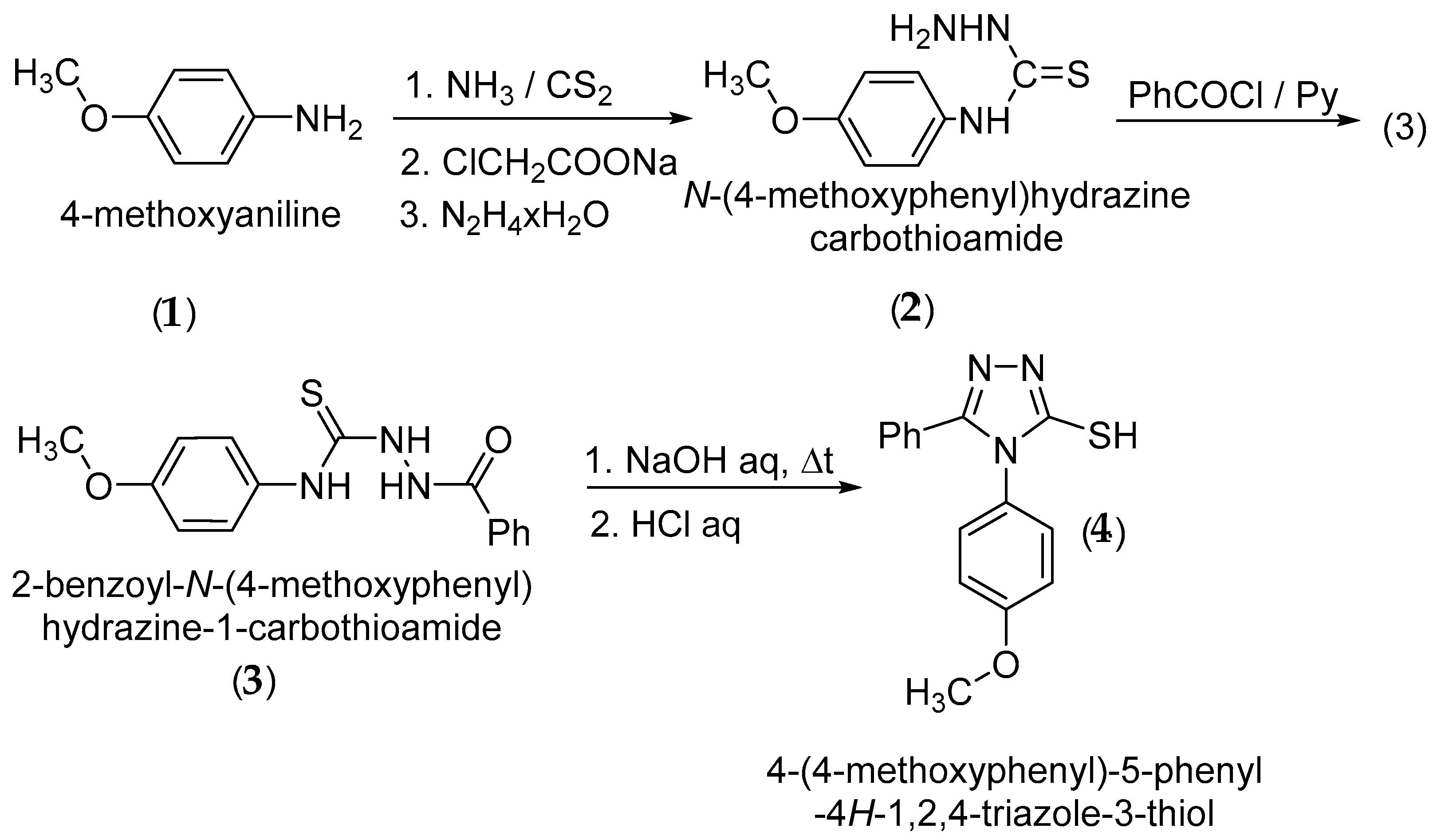
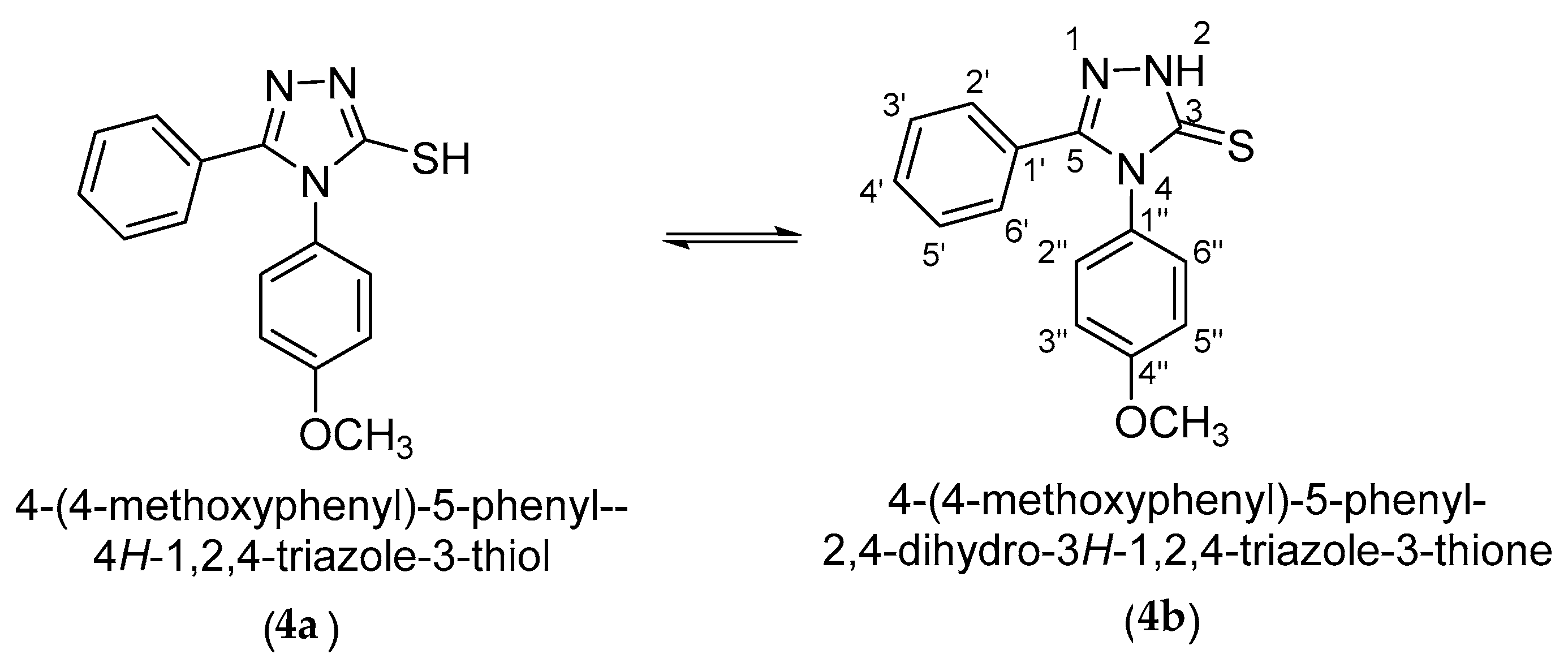
2. Results and Discussion
3. Materials and Methods
3.1. NMR Characterization of 4-(4-methoxyphenyl)-5-phenyl-4H-1,2,4-triazole-3-thiol (4)
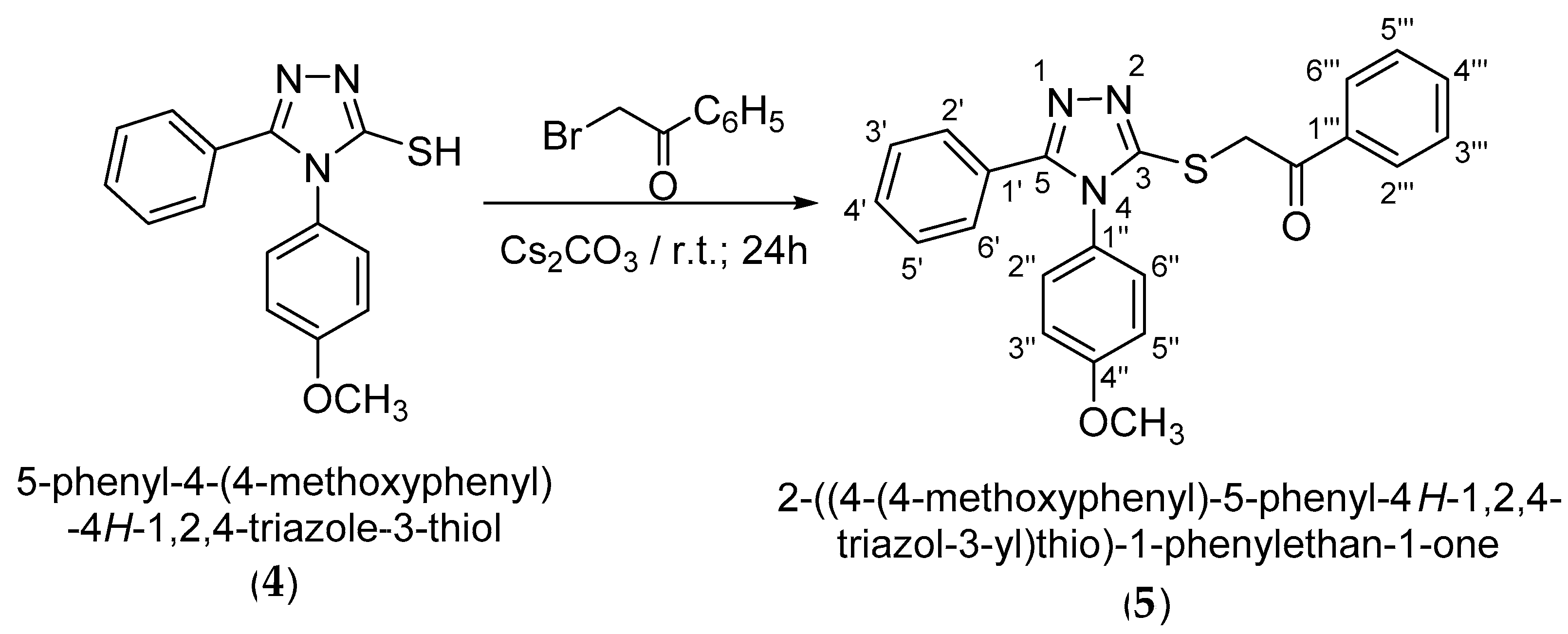
3.2. Synthesis of 2-{[4-(-4-methoxyphenyl)-5-phenyl-4H-1,2,4-triazol-3-yl]thio}-1-phenylethan-1-one (5)
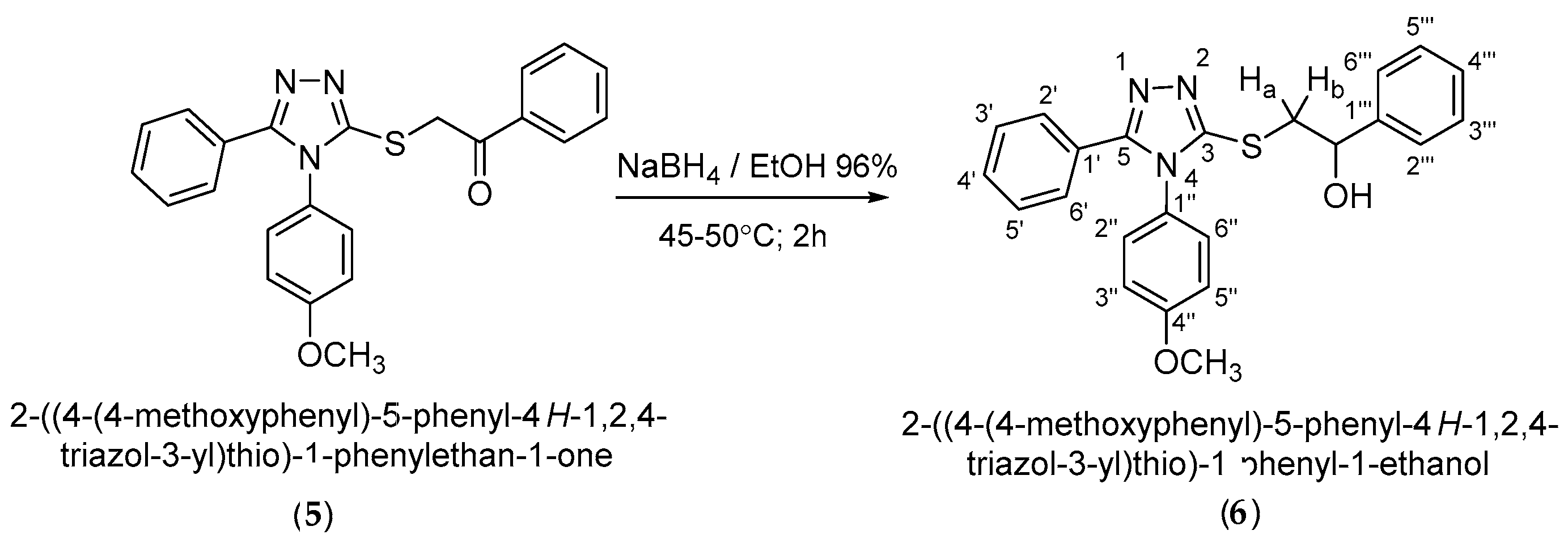
3.3. Synthesis of 2-{[4-(4-methoxyphenyl)-5-phenyl-4H-1,2,4-triazol-3-yl]thio}-1-phenyl-1-ethanol (6)
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Ghanaat, J.; Khalilzadeh, M.A.; Zareyee, D. Molecular docking studies, biological evaluation and synthesis of novel 3-mercapto-1,2,4-triazole derivates. Mol. Div. 2020, 25, 223–232. [Google Scholar] [CrossRef] [PubMed]
- De, S.K.; Stebbins, J.L.; Chen, L.H.; Riel-Mehan, M.; Machleidt, T.; Dahl, R.; Pellecchia, M. Design, Synthesis, and Structure—Activity Relationship of Substrate Competitive, Selective and In Vivo Active Triazole and Thiadiazole Inhibitors of the c-Jun N-Terminal Kinase. J. Med. Chem. 2009, 52, 1943–1952. [Google Scholar] [CrossRef] [PubMed]
- Krishna, K.M.; Inturi, B.; Pujar, G.V.; Purohit, M.N.; Vijaykumar, G.S. Design, synthesis and 3D-QSAR studies of new diphenylamine containing 1,2,4-triazoles as potential antitubercular agents. Eur. J. Med. Chem. 2014, 84, 516–529. [Google Scholar] [CrossRef] [PubMed]
- Goswami, B.N.; Kataky, J.C.S.; Baruah, J.N. Synthesis and antibacterial activity of 1-(2,4-dichlorobenzoyl)-4-substituted thiosemicarbazides, 1,2,4-triazoles and their methyl derivatives. J. Heterocycl. Chem. 1984, 21, 1225–1229. [Google Scholar] [CrossRef]
- Tehranchian, S.; Akbarzadeh, T.; Fazeli, M.; Reza, M.; Jamalifar, H.; Shafiee, A. Synthesis and antibacterial activity of 1-[1,2,4-triazol-3-yl] and 1-[1,3,4-thiadiazol-2-yl]-3-methylthio-6,7-dihydrobenzo[c]thiophen-4(5H)ones. Bioorg. Med. Chem. Lett. 2005, 15, 1023–1025. [Google Scholar] [CrossRef] [PubMed]
- Dimri, A.K.; Parmar, S.S. Synthesis of 3-aryl-4-oxothiazolin-2-yl(4-ethoxy-3-methoxy)phenyl hydrazones as possible anticonvulsants. J. Heterocycl. Chem. 1978, 15, 335–336. [Google Scholar] [CrossRef]
- Radl, S. Preparation of some pyrazole derivatives by extrusion of elemental sulfur from 1,3,4-thiadiazines. Collect. Czech Chem. Commun. 1992, 57, 656–659. [Google Scholar] [CrossRef]
- Nuțiu, M.; Bercean, V.; Birău, M. Synthesis of some 4-aryl-thiosemicarbazides. Ann. West Univ. Timiş. 1996, 5, 7–10. [Google Scholar]
- Golovlyova, S.M.; Moskvichev, Y.A.; Alov, E.M.; Kobylinsky, D.B.; Ermolaeva, V.V. Synthesis of novel five-membered nitrogen-containing heterocyclic compounds from derivatives of arylsulfonyl and arylthioacetic and propionic acids. Chem. Heterocycl. Comp. 2001, 37, 1102–1106. [Google Scholar] [CrossRef]
- Ledeți, I.; Bercean, V.; Alexa, A.; Foica, C.; Șuta, L.-M.; Dehelean, C.; Trandafirescu, C.; Muntean, D.; Licker, M.; Fuliaș, A. Preparation and antibacterial properties of substituted 1,2,4-triazoles. J. Chem. 2015, 2015, 879343. [Google Scholar] [CrossRef]
- Salvatore, R.N.; Smith, R.A.; Nischwitz, A.K.; Gavin, T. A mild and highly convenient chemoselective alkylation of thiols using Cs2CO3–TBAI. Tetrahedron Lett. 2005, 46, 8931–8935. [Google Scholar] [CrossRef]
- Varala, R.; Rao, K.S. Cesium salts in organic synthesis: A Review. Curr. Org. Chem. 2015, 19, 1242–1274. [Google Scholar] [CrossRef]
- Gómez, A.B.; Ahlsten, N.; Platero-Prats, A.E.; Martín-Matute, B. Synthesis of 4,5-disubstituted 2-aminothiazoles from α,β-unsaturated ketones: Preparation of 5-benzyl-4-methyl-2-aminothiazolium hydrochloride salt. Org. Synth. 2014, 91, 185–200. [Google Scholar] [CrossRef]
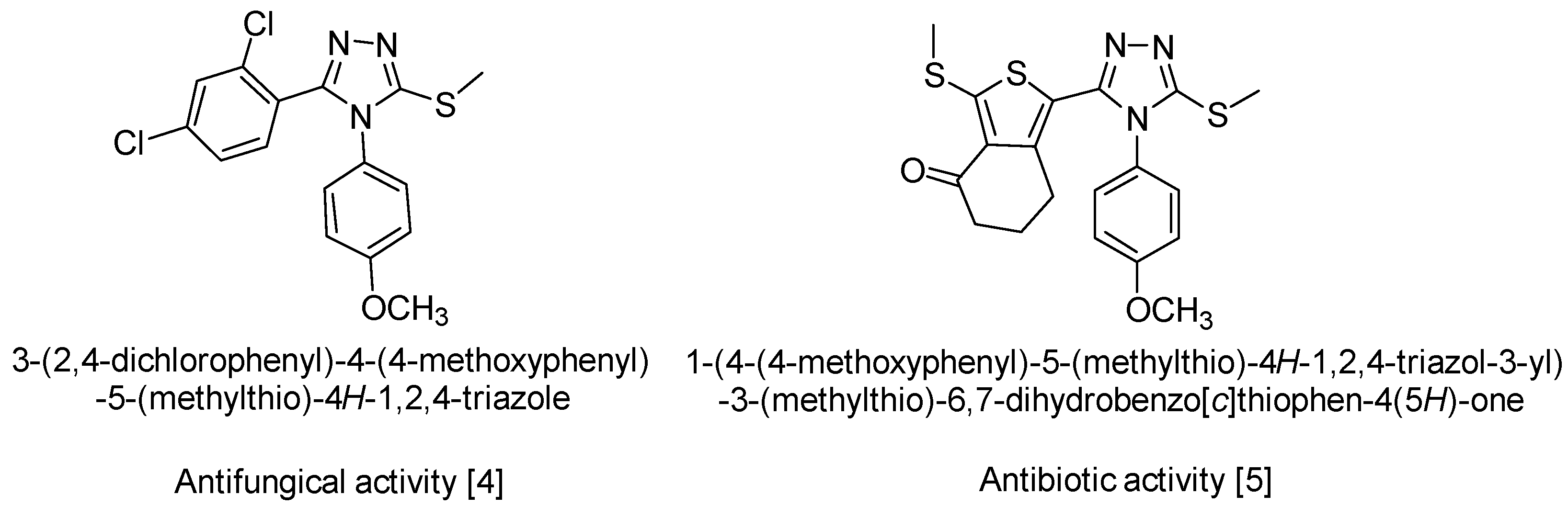
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wurfer, F.-G.; Badea, V. (R,S)-2-{[4-(4-Methoxyphenyl)-5-phenyl-4H-1,2,4-triazol-3-yl] thio}-1-phenyl-1-ethanol. Molbank 2021, 2021, M1231. https://doi.org/10.3390/M1231
Wurfer F-G, Badea V. (R,S)-2-{[4-(4-Methoxyphenyl)-5-phenyl-4H-1,2,4-triazol-3-yl] thio}-1-phenyl-1-ethanol. Molbank. 2021; 2021(2):M1231. https://doi.org/10.3390/M1231
Chicago/Turabian StyleWurfer, Flavius-Gabriel, and Valentin Badea. 2021. "(R,S)-2-{[4-(4-Methoxyphenyl)-5-phenyl-4H-1,2,4-triazol-3-yl] thio}-1-phenyl-1-ethanol" Molbank 2021, no. 2: M1231. https://doi.org/10.3390/M1231
APA StyleWurfer, F.-G., & Badea, V. (2021). (R,S)-2-{[4-(4-Methoxyphenyl)-5-phenyl-4H-1,2,4-triazol-3-yl] thio}-1-phenyl-1-ethanol. Molbank, 2021(2), M1231. https://doi.org/10.3390/M1231