7-Bromo-[1,2,5]selenadiazolo[3,4-d]pyridazin-4(5H)-one
Abstract
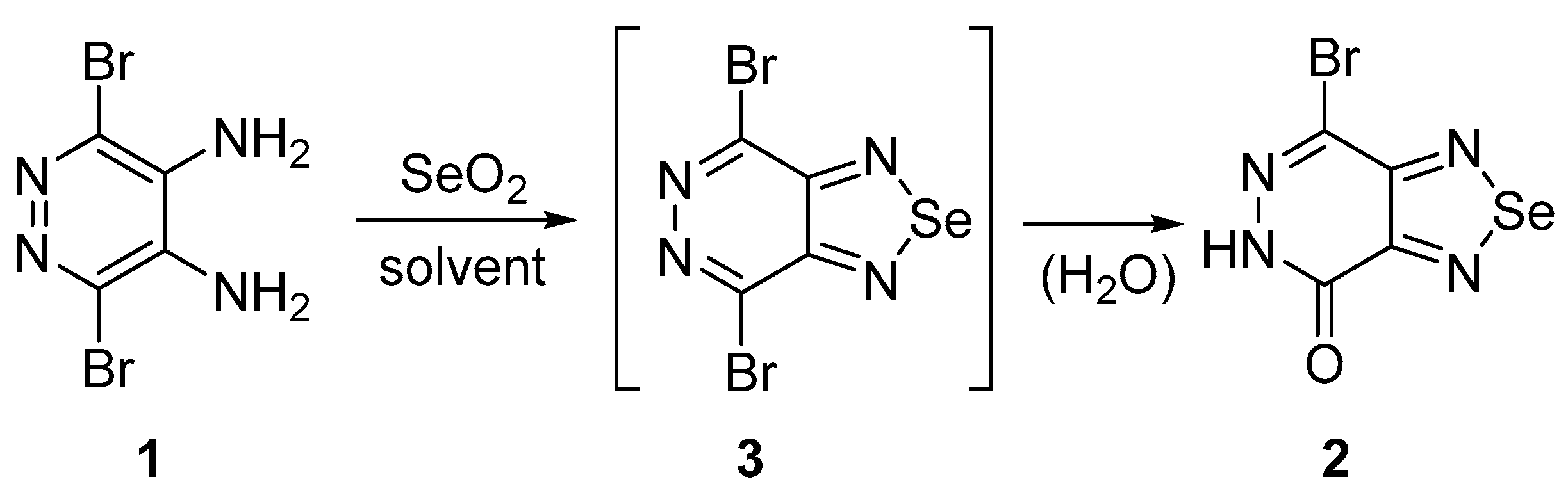
1. Introduction
2. Results and Discussion
3. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Konstantinova, L.S.; Knyazeva, E.A.; Rakitin, O.A. Recent Developments in the Synthesis and Applications of 1,2,5-Thia- and Selenadiazoles. A Review. Org. Prep. Proc. Int. 2014, 46, 475–544. [Google Scholar] [CrossRef]
- Rakitin, O.A.; Zibarev, A.V. Recent Progress in Synthesis and Applications of 5-Membered Chalcogen-Nitrogen π-Heterocycles with Three Heteroatoms. Asian J. Org. Chem. 2018, 7, 2397–2416. [Google Scholar] [CrossRef]
- Rakitin, O.A. Recent developments in the synthesis of 1,2,5-thiadiazoles and 2,1,3-benzothiadiazoles. Synthesis 2019, 51, 4338–4347. [Google Scholar] [CrossRef]
- Rakitin, O.A. Fused 1,2,5-thia- and 1,2,5-selenadiazoles: Synthesis and application in materials chemistry. Tetrahedron Lett. 2020, 61, 152230. [Google Scholar] [CrossRef]
- Knyazeva, E.A.; Rakitin, O.A. Influence of structural factors on the photovoltaic properties of dye-sensitized solar cells. Russ. Chem. Rev. 2016, 85, 1146–1183. [Google Scholar] [CrossRef]
- Lee, C.-P.; Chun-Ting Li, C.-T.; Ho, K.-C. Use of organic materials in dye-sensitized solar cells. Mater. Today 2017, 20, 267–283. [Google Scholar] [CrossRef]
- Carella, A.; Borbone, F.; Centore, R. Research Progress on Photosensitizers for DSSC. Front. Chem. 2018, 6, 481. [Google Scholar] [CrossRef] [PubMed]
- Mikhailov, M.S.; Gudim, N.S.; Knyazeva, E.A.; Tanaka, E.; Zhang, L.; Mikhalchenko, L.V.; Robertson, N.; Rakitin, O.A. 9-(p-Tolyl)-2,3,4,4a,9,9a-hexahydro-1H-carbazole—a new donor building-block in the design of sensitizers for dye-sensitized solar cells. J. Photochem. Photobiol. A 2020, 391, 112333. [Google Scholar] [CrossRef]
- Leventis, A.; Chmovzh, T.N.; Knyazeva, E.A.; Han, Y.; Heeney, M.J.; Rakitin, O.A.; Bronstein, H. A novel low-bandgap pyridazine thiadiazole-based conjugated polymer with deep molecular orbital levels. Polym. Chem. 2020, 11, 581–585. [Google Scholar] [CrossRef]
- Korshunov, V.M.; Chmovzh, T.N.; Golovanov, I.S.; Knyazeva, E.A.; Mikhalchenko, L.V.; Saifutyarov, R.S.; Avetisov, I.C.; Woollins, J.D.; Taydakov, I.V.; Rakitin, O.A. Candle light-style OLEDs with benzochalcogenadiazoles cores. Dyes Pigm. 2021, 185, 108917. [Google Scholar] [CrossRef]
- Knyazeva, E.A.; Rakitin, O.A. 4,7-Dibromo-substituted 2,1,3-benzothia(selena, oxa)diazoles and [1,2,5]thia(selena)diazolo[3,4-c]pyridine as building blocks in solar cells components (microreview). Chem. Heterocycl. Comp. 2017, 53, 855–857. [Google Scholar] [CrossRef]
- Chmovzh, T.N.; Knyazeva, E.A.; Ustimenko, O.O.; Rakitin, O.A. 3,6-Dibromopyridazine-4,5-diamine. Molbank 2019, 2019, M1053. [Google Scholar] [CrossRef]
- Chmovzh, T.N.; Knyazeva, E.A.; Mikhalchenko, L.V.; Golovanov, I.S.; Amelichev, S.A.; Rakitin, O.A. Synthesis of 4,7-dibromo derivative of ultrahigh electron-deficient [1,2,5]thiadiazolo[3,4-d]pyridazine heterocycle and its cross-coupling reactions. Eur. J. Org. Chem. 2018, 41, 5668–5677. [Google Scholar] [CrossRef]
| Entry | Solvent | Dehydrating Agent | Temperature, °C | Time, h | Yield of 2, % |
|---|---|---|---|---|---|
| 1 | EtOH | - | 78 | 1 | 75 |
| 2 | MeСN | - | 81 | 1 | 77 |
| 3 | MeСN | MgSO4 | 81 | 1 | 76 |
| 4 | MeСN | Molecular sieves | 81 | 1 | 75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chmovzh, T.N.; Rakitin, O.A. 7-Bromo-[1,2,5]selenadiazolo[3,4-d]pyridazin-4(5H)-one. Molbank 2021, 2021, M1229. https://doi.org/10.3390/M1229
Chmovzh TN, Rakitin OA. 7-Bromo-[1,2,5]selenadiazolo[3,4-d]pyridazin-4(5H)-one. Molbank. 2021; 2021(2):M1229. https://doi.org/10.3390/M1229
Chicago/Turabian StyleChmovzh, Timofey N., and Oleg A. Rakitin. 2021. "7-Bromo-[1,2,5]selenadiazolo[3,4-d]pyridazin-4(5H)-one" Molbank 2021, no. 2: M1229. https://doi.org/10.3390/M1229
APA StyleChmovzh, T. N., & Rakitin, O. A. (2021). 7-Bromo-[1,2,5]selenadiazolo[3,4-d]pyridazin-4(5H)-one. Molbank, 2021(2), M1229. https://doi.org/10.3390/M1229







