6,6′-{[Ethane-1,2-diylbis(azaneylylidene)]bis(methaneylylidene)}bis[2-(hexyloxy)phenolato] Nickel(II)
Abstract
1. Introduction
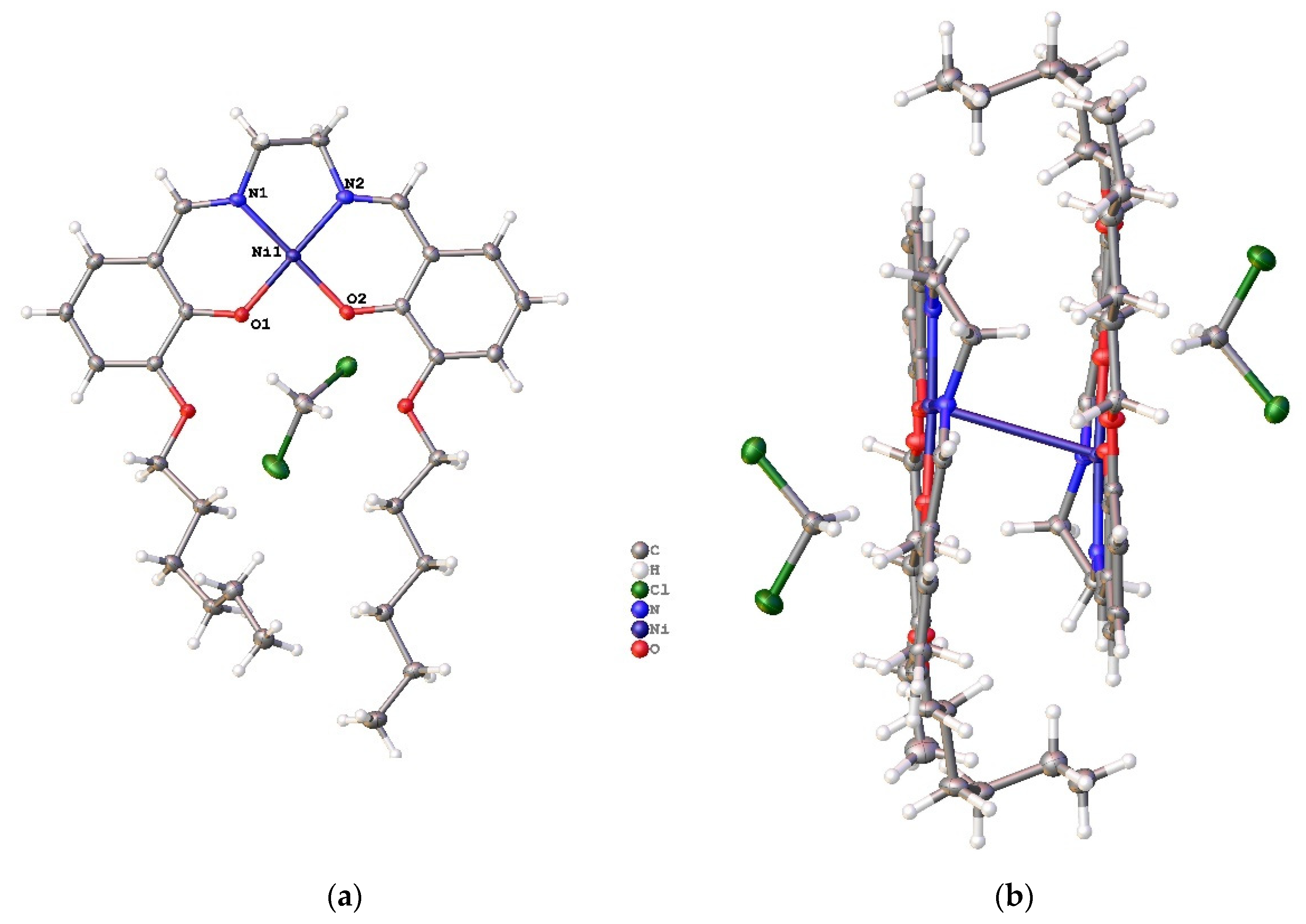
2. Results
3. Materials and Methods
3.1. General Considerations
3.2. Synthesis of 6,6′-{[Ethane-1,2-diylbis(azaneylylidene)]bis(methaneylylidene)}bis[2-(hexyloxy)phenolato] Nickel(II)
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gupta, K.C.; Sutar, A.K. Catalytic activities of Schiff base transition metal complexes. Coord. Chem. Rev. 2008, 252, 1420–1450. [Google Scholar] [CrossRef]
- Chepurnaya, I.A.; Karushev, M.P.; Alekseeva, E.V.; Lukyanov, D.A.; Levin, O.V. Redox-conducting polymers based on metal-salen complexes for energy storage applications. Pure Appl. Chem. 2020. [Google Scholar] [CrossRef]
- Bott-Neto, J.L.; Martins, T.S.; Machado, S.A.S.; Ticianelli, E.A. Electrocatalytic Oxidation of Methanol, Ethanol, and Glycerol on Ni(OH)2 Nanoparticles Encapsulated with Poly[Ni(salen)] Film. ACS Appl. Mater. Interfaces 2019, 11, 30810–30818. [Google Scholar] [CrossRef] [PubMed]
- Dahm, C.E.; Peters, D.G. Catalytic reduction of α, ω-dihaloalkanes with nickel(I) salen as a homogeneous-phase and polymer-bound mediator. J. Electroanal. Chem. 1996, 406, 119–129. [Google Scholar] [CrossRef]
- Konev, A.S.; Kayumov, M.Y.; Karushev, M.P.; Novoselova, Y.V.; Lukyanov, D.A.; Alekseeva, E.V.; Levin, O.V. Polymeric Metal Salen-Type Complexes as Catalysts for Photoelectrocatalytic Hydrogen Peroxide Production. ChemElectroChem 2018, 5, 3138–3142. [Google Scholar] [CrossRef]
- Martins, T.S.; Bott-Neto, J.L.; Raymundo-Pereira, P.A.; Ticianelli, E.A.; Machado, S.A.S. An electrochemical furosemide sensor based on pencil graphite surface modified with polymer film Ni-salen and Ni(OH)2/C nanoparticles. Sens. Actuators B Chem. 2018, 276, 378–387. [Google Scholar] [CrossRef]
- Martin, C.S.; Dadamos, T.R.L.; Teixeira, M.F.S. Development of an electrochemical sensor for determination of dissolved oxygen by nickel–salen polymeric film modified electrode. Sens. Actuators B Chem. 2012, 175, 111–117. [Google Scholar] [CrossRef]
- Pinheiro, C.; Parola, A.J.; Pina, F.; Fonseca, J.; Freire, C. Electrocolorimetry of electrochromic materials on flexible ITO electrodes. Sol. Energy Mater. Sol. Cells 2008, 92, 980–985. [Google Scholar] [CrossRef]
- Nunes, M.; Araújo, M.; Bacsa, R.; Ferreira, R.V.; Castillejos, E.; Serp, P.; Hillman, A.R.; Freire, C. N-doped few-layered graphene-polyNi complex nanocomposite with excellent electrochromic properties. Carbon 2017, 120, 32–43. [Google Scholar] [CrossRef]
- Alekseeva, E.V.; Chepurnaya, I.A.; Malev, V.V.; Timonov, A.M.; Levin, O.V. Polymeric nickel complexes with salen-type ligands for modification of supercapacitor electrodes: Impedance studies of charge transfer and storage properties. Electrochim. Acta 2017, 225, 378–391. [Google Scholar] [CrossRef]
- Gao, F.; Li, J.; Kang, F.; Zhang, Y.; Wang, X.; Ye, F.; Yang, J. Preparation and Characterization of a Poly[Ni(salen)]/Multiwalled Carbon Nanotube Composite by in Situ Electropolymerization as a Capacitive Material. J. Phys. Chem. C 2011, 115, 11822–11829. [Google Scholar] [CrossRef]
- Chen, C.; Zhu, Z.; Li, X.; Li, J. Electropolymerization and energy storage of poly[Ni(salphen)]/MWCNT composite materials for supercapacitors. J. Appl. Polym. Sci. 2017, 134. [Google Scholar] [CrossRef]
- Vereshchagin, A.A.; Lukyanov, D.A.; Kulikov, I.R.; Panjwani, N.A.; Alekseeva, E.A.; Behrends, J.; Levin, O.V. The fast and the capacious: A [Ni(salen)]-TEMPO redox-conducting polymer for organic batteries. Batter. Supercaps 2020. [Google Scholar] [CrossRef]
- Novozhilova, M.V.; Smirnova, E.A.; Karushev, M.P.; Timonov, A.M.; Malev, V.V.; Levin, O.V. Synthesis and study of catalysts of electrochemical oxygen reduction reaction based on polymer complexes of nickel and cobalt with Schiff bases. Russ. J. Electrochem. 2016, 52, 1183–1190. [Google Scholar] [CrossRef]
- Dahm, C.E.; Peters, D.G.; Simonet, J. Electrochemical and spectroscopic characterization of anodically formed nickel salen polymer films on glassy carbon, platinum, and optically transparent tin oxide electrodes in acetonitrile containing tetramethylammonium tetrafluoroborate. J. Electroanal. Chem. 1996, 410, 163–171. [Google Scholar] [CrossRef]
- Eliseeva, S.N.; Alekseeva, E.V.; Vereshchagin, A.A.; Volkov, A.I.; Vlasov, P.S.; Konev, A.S.; Levin, O.V. Nickel-Salen Type Polymers as Cathode Materials for Rechargeable Lithium Batteries. Macromol. Chem. Phys. 2017, 218. [Google Scholar] [CrossRef]
- O’Meara, C.; Karushev, M.P.; Polozhentceva, I.A.; Dharmasena, S.; Cho, H.; Yurkovich, B.J.; Kogan, S.; Kim, J.H. Nickel-Salen-Type Polymer as Conducting Agent and Binder for Carbon-Free Cathodes in Lithium-Ion Batteries. ACS Appl. Mater. Interfaces 2019, 11, 525–533. [Google Scholar] [CrossRef]
- Beletskii, E.V.; Volosatova, Y.A.; Eliseeva, S.N.; Levin, O.V. The Effect of Electrode Potential on the Conductivity of Polymer Complexes of Nickel with Salen Ligands. Russ. J. Electrochem. 2019, 55, 339–345. [Google Scholar] [CrossRef]
- Zakrzewski, J.; Krawczyk, M. Reactions of nitroxides XIV. Analogs of phenoxy carboxylic herbicides based on the piperidine scaffold; unexpected fungicidal activity of the 2-[(1-oxyl-2,2,6,6-tetramethylpiperidin-4-yl)oxy]butanoic acid. Heterocycl. Commun. 2014, 20. [Google Scholar] [CrossRef]
- Ma, Q.; Chakraborty, D.; Faglioni, F.; Muller, R.P.; Goddard, W.A., 3rd; Harris, T.; Campbell, C.; Tang, Y. Alkylation of phenol: A mechanistic view. J. Phys. Chem. A 2006, 110, 2246–2252. [Google Scholar] [CrossRef]
- Casellato, U.; Guerriero, P.; Tamburini, S.; Vigato, P.A.; Benelli, C. Mononuclear, homo- and heteropolynuclear complexes with acyclic compartmental Schiff bases. Inorg. Chim. Acta 1993, 207, 39–58. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. C 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lukyanov, D.A.; Borisova, A.S.; Levin, O.V. 6,6′-{[Ethane-1,2-diylbis(azaneylylidene)]bis(methaneylylidene)}bis[2-(hexyloxy)phenolato] Nickel(II). Molbank 2020, 2020, M1174. https://doi.org/10.3390/M1174
Lukyanov DA, Borisova AS, Levin OV. 6,6′-{[Ethane-1,2-diylbis(azaneylylidene)]bis(methaneylylidene)}bis[2-(hexyloxy)phenolato] Nickel(II). Molbank. 2020; 2020(4):M1174. https://doi.org/10.3390/M1174
Chicago/Turabian StyleLukyanov, Daniil A., Anna S. Borisova, and Oleg V. Levin. 2020. "6,6′-{[Ethane-1,2-diylbis(azaneylylidene)]bis(methaneylylidene)}bis[2-(hexyloxy)phenolato] Nickel(II)" Molbank 2020, no. 4: M1174. https://doi.org/10.3390/M1174
APA StyleLukyanov, D. A., Borisova, A. S., & Levin, O. V. (2020). 6,6′-{[Ethane-1,2-diylbis(azaneylylidene)]bis(methaneylylidene)}bis[2-(hexyloxy)phenolato] Nickel(II). Molbank, 2020(4), M1174. https://doi.org/10.3390/M1174






