4,4-Bis(hydroxymethyl)-2-phenyl-2-oxazoline
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis
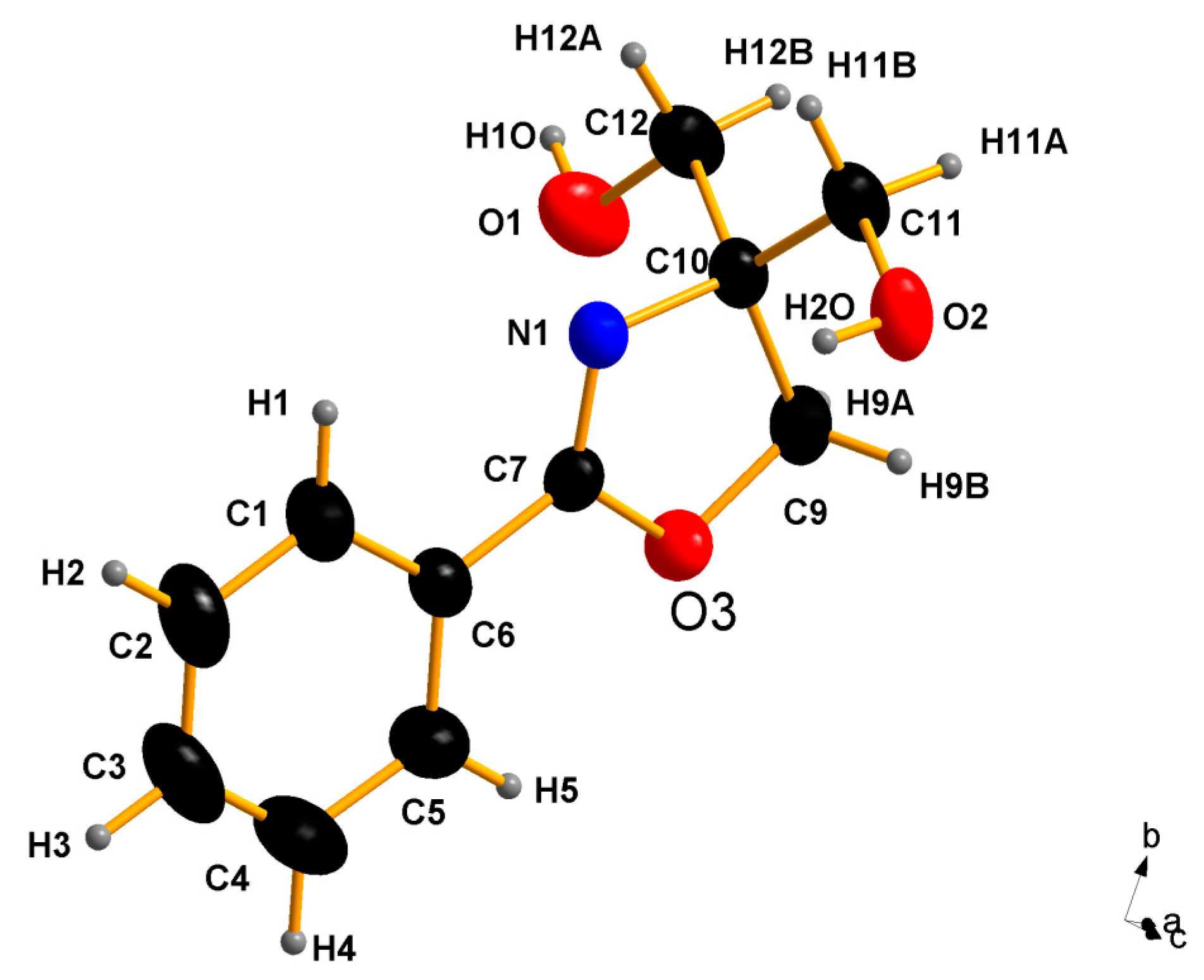
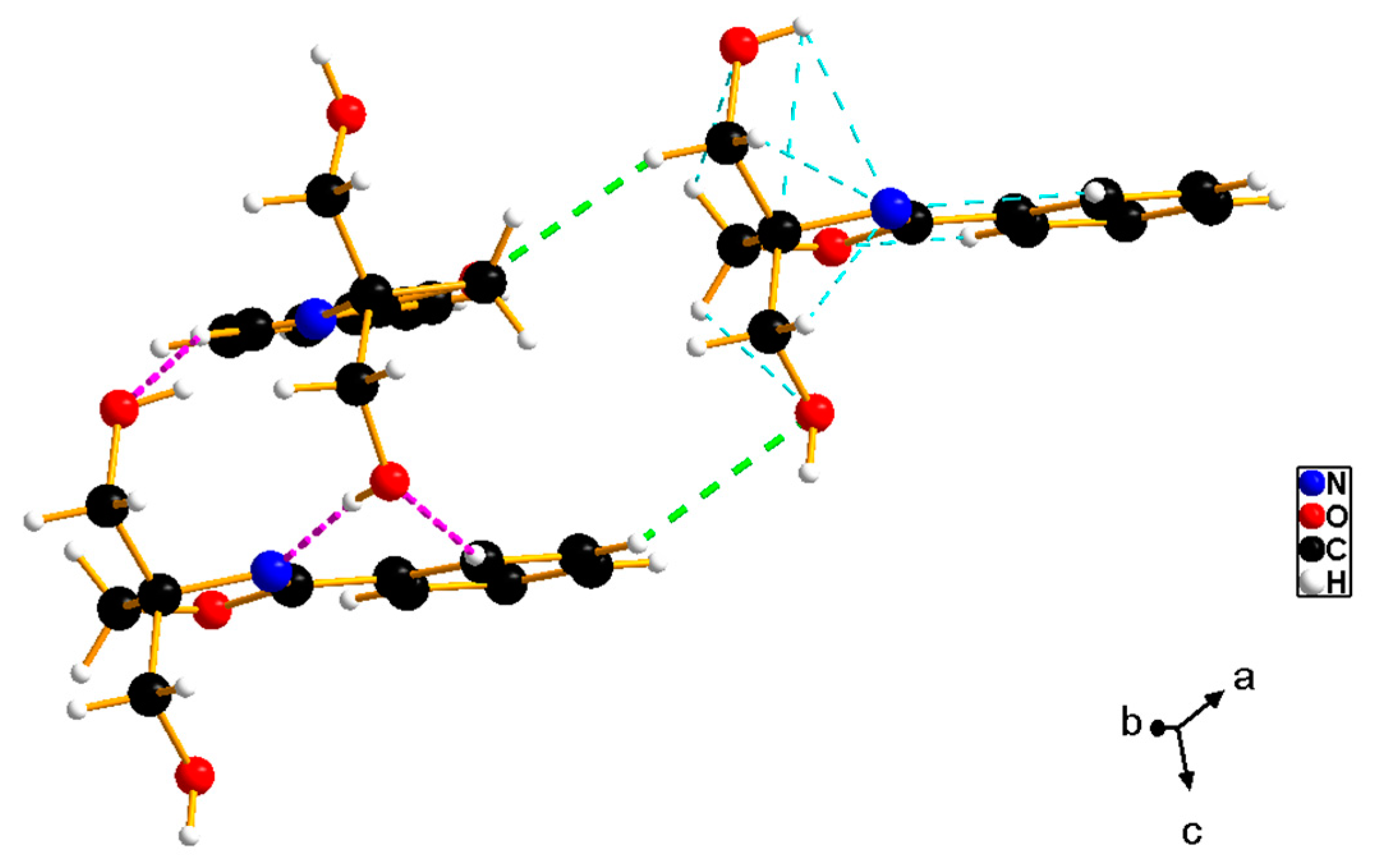
2.2. X-ray Structure Determination of 2
3. Materials and Methods
3.1. General Methods and Physical Measurements
3.2. Resynthesis of 4,4-Bis(hydroxymethyl)-2-phenyl-2-oxazoline
3.3. Crystal Data for the 4,4-Bis(hydroxymethyl)-2-phenyl-2-oxazoline
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sakakura, A.; Umemura, S.; Kondo, R.; Ishihara, K. Dehydrative cyclization catalyzed by the combination of molybdenum (VI) oxides and benzoic acids: First synthesis of the antitumor substance BE-70016. Adv. Synth. Catal. 2007, 349, 551–555. [Google Scholar] [CrossRef]
- Tsuda, M.; Yamakawa, M.; Oka, S.; Tanaka, Y.; Hoshino, Y.; Mikami, Y.; Sato, A.; Fujiwara, H.; Ohizumi, Y.; Kobayashi, J. Brasilibactin A, a cytotoxic compound from actinomycete nocardia brasiliensis. J. Nat. Prod. 2005, 68, 462–464. [Google Scholar] [CrossRef] [PubMed]
- Doi, T. Synthesis of the biologically active natural product cyclodepsipeptides apratoxin A and its analogues. Chem. Pharm. Bull. 2014, 62, 735–743. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, P.; Gong, J.; Wang, B.; Zhang, M.; Wang, J. Solubility of deflazacort in binary solvent mixtures. J. Chem. Eng. Data 2009, 54, 162–164. [Google Scholar] [CrossRef]
- Djurendić, E.; Vujašković, S.D.; Sakač, M.; Ajduković, J.; Gaković, A.; Kojić, V.; Bogdanović, G.; Klisurić, O.; Gaši, K.P. Synthesis and biological evaluation of some new 2-oxazoline and salicylic acid derivatives. Arkivoc 2011, 83–102. [Google Scholar] [CrossRef]
- Meyers, A.I.; Mihelich, E.D. The synthetic utility of 2-oxazolines. Angew. Chem. Int. Ed. Engl. 1976, 15, 270–281. [Google Scholar] [CrossRef]
- Gomez, M.; Muller, G.; Rocamora, M. Coordination chemistry of oxazoline ligands. Coord. Chem. Rev. 1999, 769–835. [Google Scholar] [CrossRef]
- Hargaden, G.C.; Guiry, P.J. Recent applications of oxazoline-containing ligands in asymmetric catalysis. Chem. Rev. 2009, 109, 2505–2550. [Google Scholar] [CrossRef]
- Lambermont-Thijs, H.M.L.; Van Kuringen, H.P.C.; Van der Put, J.P.W.; Schubert, U.S.; Hoogenboom, R. Temperature induced solubility transitions of various poly(2-oxazoline)s in ethanol-water solvent mixtures. Polymers 2010, 2, 188–199. [Google Scholar] [CrossRef]
- Hoogenboom, R. Poly(2-oxazoline)s: A polymer class with numerous potential applications. Angew. Chem. Int. Ed. 2009, 48, 7978–7994. [Google Scholar] [CrossRef]
- Barbeiro, C.S.; Vasconcelos, S.N.S.; De Oliveira, I.M.; Zukerman-Schpector, J.; Caracelli, I.; Maganhi, S.H.; Stefani, H.A. Suzuki-Miyaura cross-coupling reaction catalyzed by palladium complexes of hydroxynaphthalene 2-oxazolines. Chem. Sel. 2017, 2, 8173–8177. [Google Scholar] [CrossRef]
- Caputo, C.A.; Jones, N.D. Developments in asymmetric catalysis by metal complexes of chiral chelating nitrogen-donor ligands. Dalton Trans. 2007, 41, 4627–4640. [Google Scholar] [CrossRef] [PubMed]
- Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Elachqar, A.; El Hajji, S.; Kerbal, A.; Labriti, B.; Martinez, J.; Rolland, V. Synthesis of new triheterocyclic compounds, precursors of biheterocyclic amino acids. J. Mar. Chim. Heterocycl. 2008, 7, 44–49. [Google Scholar]
- Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Elachqar, A.; Kerbal, A. Simple and efficient synthesis of racemic 2-(tert-butoxycarbonylamino)-2-methyl-3-(1H-1,2,4-triazol-1-yl)propanoic acid, a new derivative of β-(1,2,4-triazol-1-yl)alanine. Molecules 2011, 16, 3380–3390. [Google Scholar] [CrossRef] [PubMed]
- Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Elachqar, A.; El Hajji, S.; Labriti, B.; Kerbal, A. Triheterocyclic compounds, oxazolinic precursors of biheterocyclic amino acids, Part II: Phenothiazine derivatives and structural study of regioisomers through 1H-15N 2D NMR HMBC. J. Mar. Chim. Heterocycl. 2014, 13, 39–47. [Google Scholar]
- Aouine, Y.; Alami, A.; El Hallaoui, A. N,N-dibenzyl-1-(1-[(4-methyl-2-phenyl-4,5-dihydrooxazol-4-yl)methyl)]-1H-1,2,3-triazol-4-yl)methanamine. Molbank 2014, 2014, M819. [Google Scholar] [CrossRef]
- Boukhssas, S.; Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Bekkari, H. Synthesis, characterization, and antibacterial activity of diethyl 1-((4-methyl-2-phenyl-4,5-dihydrooxazol-4-yl)methyl)-1H-1,2,3-triazole-4,5-dicarboxylate. J. Chem. 2017, 2017, 4238360. [Google Scholar] [CrossRef]
- Billman, J.H.; Parker, E.E. Amino Acids. IV. Amino acids related to serine. J. Am. Chem. Soc. 1945, 67, 1069–1070. [Google Scholar] [CrossRef]
- Vorbrüggen, H.; Krolikiewicz, K. A simple synthesis of Δ2-oxazolines, Δ2-oxazines, Δ2-thiazolines and 2-substituted benzoxazoles. Tetrahedron 1993, 49, 9353–9372. [Google Scholar] [CrossRef]
- Vorbrüggen, H.; Krolikiewicz, K. A simple synthesis of Δ2-oxazolines, Δ2-oxazines, Δ2-thiazolines and Δ2-imidazolines. Tetrahedron Lett. 1981, 22, 4471–4474. [Google Scholar] [CrossRef]
- Yamamoto, K.; Tsuda, Y.; Kuriyama, M.; Demizu, Y.; Onomura, O. Cu-Catalyzed Enantioselective Synthesis of Oxazolines from Aminotriols via Asymmetric Desymmetrization. Chem. Asian J. 2020, 15, 840–844. [Google Scholar] [CrossRef]
- Korolev, A.M.; Eremenko, L.T.; Meshikhina, L.V.; Nefedov, S.E.; Eremenko, I.L. Synthesis, and study of organic nitrates of the heterofunctional series: Synthesis and structure of tris(hydroxymethyl)aminomethane dinitrate benzoate hydronitrate. Russ. Chem. Bull. 1999, 48, 501–505. [Google Scholar] [CrossRef]
- Korolev, A.M.; Eremenko, L.T.; Meshikhina, L.V. Synthesis and study of organic nitrates of heterofunctional series: Synthesis and some chemical properties of 4,4-bis(nitroxymethyl)-2-phenyl-2-oxazoline and -2-oxazolinium nitrate. Russ. Chem. Bull. 1999, 48, 808–809. [Google Scholar] [CrossRef]
- García-Tellado, F.; Loupy, A.; Petit, A.; Marrero-Terrero, A.L. Solvent-free microwave-assisted efficient synthesis of 4,4-disubstituted 2-oxazolines. Eur. J. Org. Chem. 2003, 22, 4387–4391. [Google Scholar] [CrossRef]
- Boukhssas, S.; Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Zouihri, H. Dibenzyl({1-[(4-methyl-2-phenyl-4,5-dihydro-1,3-oxazol-4-yl)methyl]-1H-1,2,3-triazol-4-yl}methyl)-amine. IUCrData 2017, 2, x170860. [Google Scholar] [CrossRef]
- Boukhssas, S.; Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Zouihri, H. Diethyl 1-[(4-methyl-2-phenyl-4,5-dihydro-1,3-oxazol-4-yl)methyl]-1H-1,2,3-triazole-4,5-dicarboxylate. IUCrData 2017, 2, x171701. [Google Scholar] [CrossRef]
- Dioukhane, K.; Alami, A.; Aouine, Y.; El Omari, M.; El Ammari, L.; Saadi, M.; Assani, A.; Ouarsal, R. Synthesis, crystal structure and IR spectrum studies of 2-(4-Methyl-2-phenyl-4,5-dihydro-oxazol-4-ylmethyl)-isoindole-1,3-dione. Mediterr. J. Chem. 2019, 9, 116–124. [Google Scholar] [CrossRef]
- Hunter, C.A.; Sanders, J.K.M. The nature of π-π interactions. J. Am. Chem. Soc. 1990, 112, 5525–5534. [Google Scholar] [CrossRef]
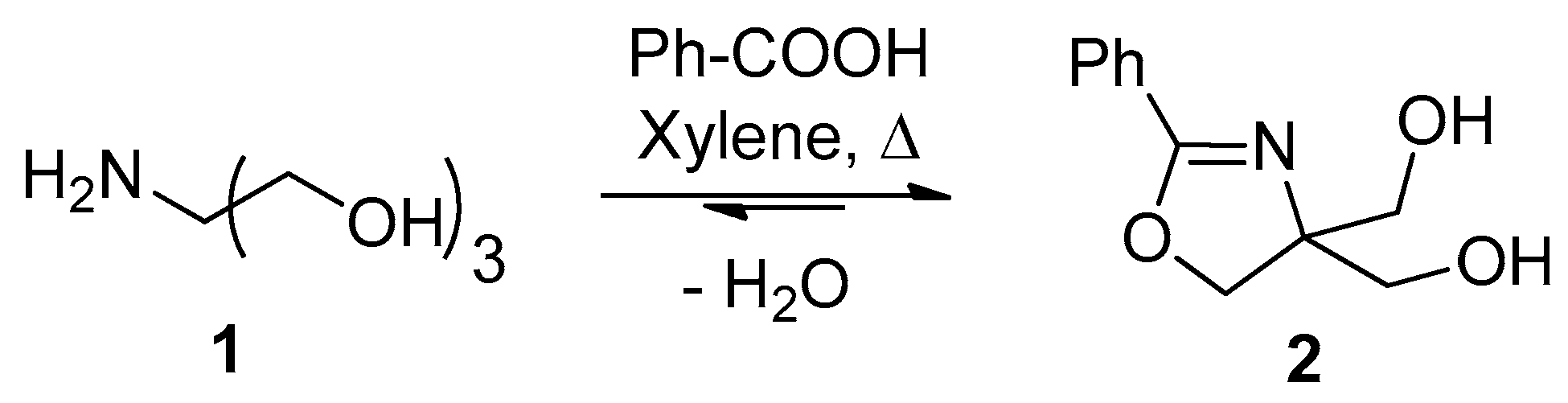

| Position | δH | δC | Correlation 1H-13C |
|---|---|---|---|
| 1 and 1′ | 3.8 (2H, e) | - | - |
| 2 and 2′ | 3.7 (4H, s) | 64.20 | 2H2-C2; 2H2′-C2′ |
| 3 | - | 76.25 | - |
| 4 | 4.45 (2H, s) | 71.09 | 2H4-C4 |
| 6 | - | 165.51 | - |
| 8–13 | 7.15–8.23 (5H, m) | 127.25–131.46 | 5Harom-5Carom |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hajib, S.; Boukhssas, S.; Aouine, Y.; Alami, A.; Faraj, H.; Zouihri, H.; El Bali, B.; Lachkar, M. 4,4-Bis(hydroxymethyl)-2-phenyl-2-oxazoline. Molbank 2020, 2020, M1163. https://doi.org/10.3390/M1163
Hajib S, Boukhssas S, Aouine Y, Alami A, Faraj H, Zouihri H, El Bali B, Lachkar M. 4,4-Bis(hydroxymethyl)-2-phenyl-2-oxazoline. Molbank. 2020; 2020(4):M1163. https://doi.org/10.3390/M1163
Chicago/Turabian StyleHajib, Sara, Salaheddine Boukhssas, Younas Aouine, Anouar Alami, Hassane Faraj, Hafid Zouihri, Brahim El Bali, and Mohammed Lachkar. 2020. "4,4-Bis(hydroxymethyl)-2-phenyl-2-oxazoline" Molbank 2020, no. 4: M1163. https://doi.org/10.3390/M1163
APA StyleHajib, S., Boukhssas, S., Aouine, Y., Alami, A., Faraj, H., Zouihri, H., El Bali, B., & Lachkar, M. (2020). 4,4-Bis(hydroxymethyl)-2-phenyl-2-oxazoline. Molbank, 2020(4), M1163. https://doi.org/10.3390/M1163






