2-(Bicyclo[4.2.0]octa-1,3,5-trien-3-yl)-adamantan-2-ol
Abstract
1. Introduction
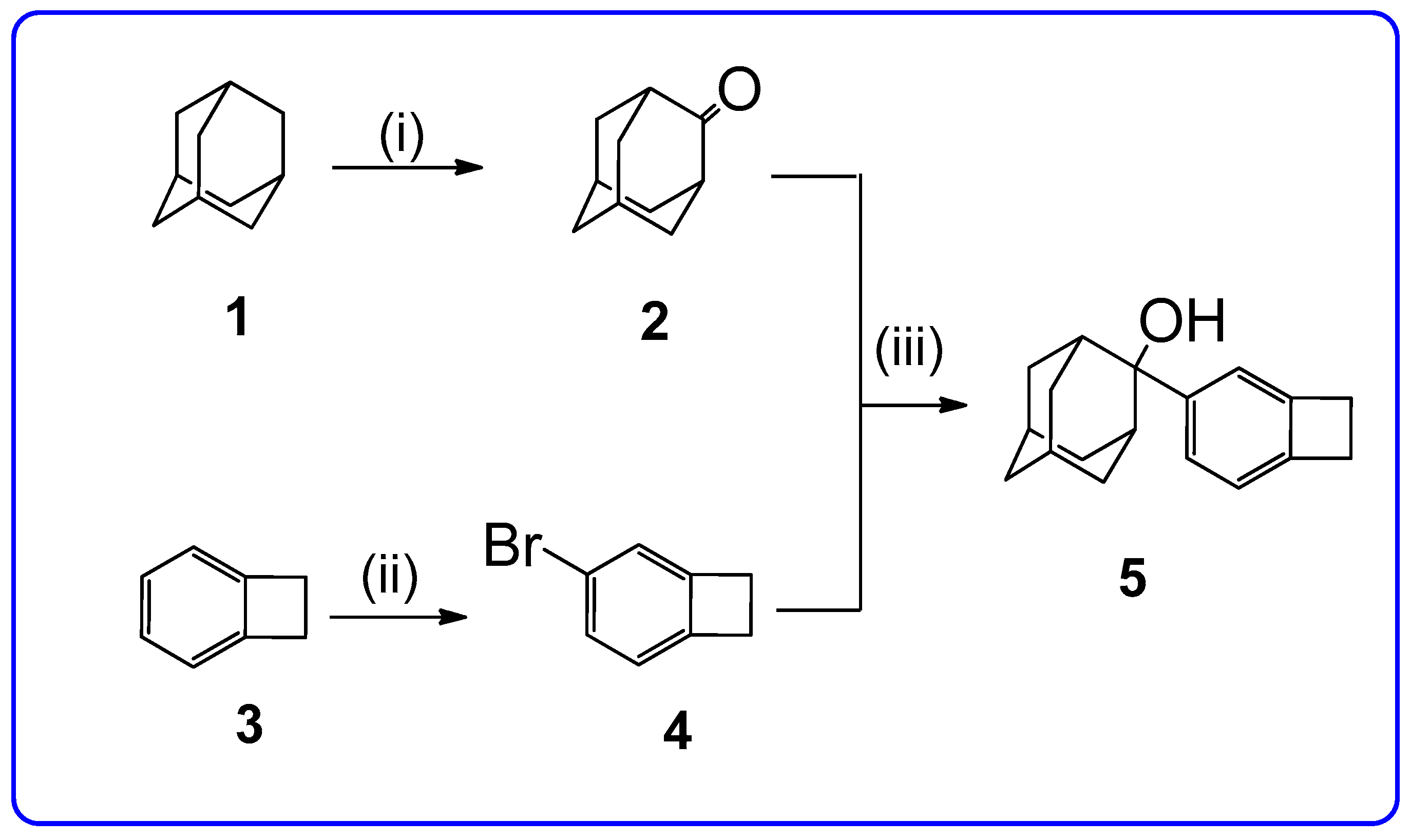
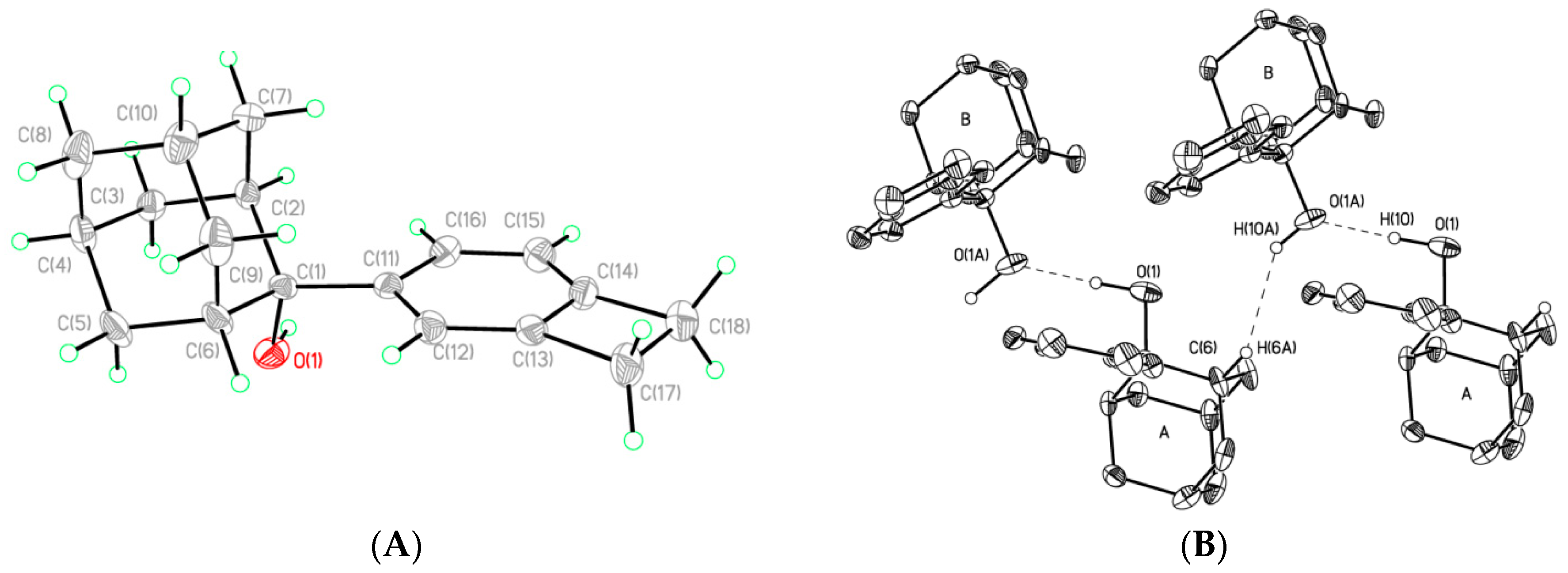
2. Results and Discussion
3. Materials and Methods
2-(Bicyclo[4.2.0]octa-1,3,5-trien-3-yl)-adamantan-2-ol (5)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References and Notes
- Robello, D.R. Moderately high refractive index, low optical dispersion polymers with pendant diamondoids. J. Appl. Polym. Sci. 2012, 127, 96–103. [Google Scholar] [CrossRef]
- Wang, K.; Cui, R.; Gu, J.; Yu, Q.; Ma, G.; Nie, J. Photopolymerization of 1-adamantyl acrylate pohotoinitiated by free radical photoinitiators. J. Appl. Polym. Sci. 2012, 123, 26–31. [Google Scholar] [CrossRef]
- Hattori, Y.; Miyajima, T.; Sakai, M.; Nagase, Y.; Nemoto, N. Synthesis and thermal characterization of novel adamantane-based polysiloxane. Polymer 2008, 49, 2825–2831. [Google Scholar] [CrossRef]
- Cheng, Y.; Chen, W.; Li, Z.; Zhu, T.; Zhang, Z.; Jin, Y. Hydrolysis and condensation of a benzocyclobutene-functionalized precursor for the synthesis of high performance low-K polymers. RSC Adv. 2017, 7, 14406–14412. [Google Scholar] [CrossRef]
- Levchenko, K.S.; Chudov, K.A.; Adamov, G.E.; Poroshin, N.O.; Shmelin, P.S.; Grebennikov, E.P.; Parshikov, Y.G. Photocurable and Thermosetting Polymer Materials on the Basis of Benzocyclobutene and Its Derivatives for Electronics. Russ. J. Gen. Chem. 2018, 88, 2793–2812. [Google Scholar] [CrossRef]
- Levchenko, К.S.; Chudov, К.А.; Demin, D.Y.; Аdamov, G.Е.; Poroshin, N.О.; Shmelin, P.S.; Grebennikov, Е.P.; Chvalun, S.N.; Zubov, V.P. Synthesis of photo and thermosetting monomers and polymers based on benzocyclobutene. Russ. Chem. Bull. 2019, 68, 1321–1342. [Google Scholar] [CrossRef]
- Kirchhoff, R.A.; Bruza, K.J. Polymers from benzocyclobutenes. In High Performance Polymers; Hergenrother, P.M., Ed.; Advances in Polymer Science; Springer: Berlin/Heidelberg, Germany, 1994; Volume 117, pp. 1–66. [Google Scholar] [CrossRef]
- Geluk, H.W.; Keizer, V.G. Adamantanone. Org. Synth. 1973, 53, 8. [Google Scholar] [CrossRef]
- Tang, P.C.; LÜ, H.; Zheng, H.; Chen, Y.; Fei, H.; Wang, S.; Wang, L. Bicyclo-Substituted Pyrazolon Azo Derivatives, Preparation Process and Pharmaceutical Use Thereof. E.P. Patent 2,236,500B1, June 2010. [Google Scholar]
- 4-Bromobenzocyclobutene (4). Benzocyclobutene (23.7 g, 22.8 mmol) was dispersed in 240 mL of water at room temperature. After cooling with ice water, (−10–5 °C), bromine (11.7 mL) was added dropwise. After complete addition, ice-water bath was removed and the reaction mixture was warmed to room temperature and stirred overnight. The reaction was monitored by TLC until the starting benzocyclobutene disappeared. The mixture was diluted with 50 mL of n-hexane and sodium sulfite (3.0 g, 23.8 mmol) was added. Upon completion of the addition, the mixture was stirred at room temperature for 30 min and becomes clear. Then, the separated organic layer was dried over anhydrous sodium sulfate, filtered to remove a drying agent, and concentrated under reduced pressure to give 4. After distillation at 110–114 °C (15 mm of Hg), compound 4 (28–30 g, 67–71%) was obtained as a colorless liquid. 1H NMR (300 MHz, CDCl3) δ 7.40 (d, J = 7.8 Hz, 1H), 7.26 (s, J = 7.0 Hz, 1H), 6.99 (d, J = 7.8 Hz, 1H), 3.31–3.10 (m, 4H).
- Roth, M.; Ahles, M.; Gawrisch, C.; Schwalm, T.; Schmechel, R.; Melzer, C.; Seggern, H.; Rehahn, M. Rodlike tetracene derivatives. Chem. Eur. J. 2017, 23, 13445–13454. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.-Q.; Fang, W.; Wei, Y.; Tang, X.-Y.; Shi, M. Gold(i)-catalyzed dehydrogenative cycloisomerization of 1,5-enynes. Chem. Commun. 2016, 52, 10799–10802. [Google Scholar] [CrossRef] [PubMed]
- Lyssenko, K.A.; Lenev, D.A.; Kostyanovsky, R.G. Self-assembly of cage structures. Paper 12: The synthesis and crystal structures of 2,5-diazabicyclo[2.2.2]octane-3,6-dione-1,4dicarboxylic acids and their diesters. Tetrahedron 2002, 58, 8525–8537. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levchenko, K.S.; Chudov, K.A.; Demin, D.Y.; Lyssenko, K.A.; Shmelin, P.S. 2-(Bicyclo[4.2.0]octa-1,3,5-trien-3-yl)-adamantan-2-ol. Molbank 2020, 2020, M1106. https://doi.org/10.3390/M1106
Levchenko KS, Chudov KA, Demin DY, Lyssenko KA, Shmelin PS. 2-(Bicyclo[4.2.0]octa-1,3,5-trien-3-yl)-adamantan-2-ol. Molbank. 2020; 2020(1):M1106. https://doi.org/10.3390/M1106
Chicago/Turabian StyleLevchenko, Konstantin S., Konstantin A. Chudov, Dmitri Yu. Demin, Konstantin A. Lyssenko, and Pavel S. Shmelin. 2020. "2-(Bicyclo[4.2.0]octa-1,3,5-trien-3-yl)-adamantan-2-ol" Molbank 2020, no. 1: M1106. https://doi.org/10.3390/M1106
APA StyleLevchenko, K. S., Chudov, K. A., Demin, D. Y., Lyssenko, K. A., & Shmelin, P. S. (2020). 2-(Bicyclo[4.2.0]octa-1,3,5-trien-3-yl)-adamantan-2-ol. Molbank, 2020(1), M1106. https://doi.org/10.3390/M1106




