8-(Pyrimidin-2-yl)-1,2,3,4,5,6,7,8-heptathiazocane
Abstract
:1. Introduction
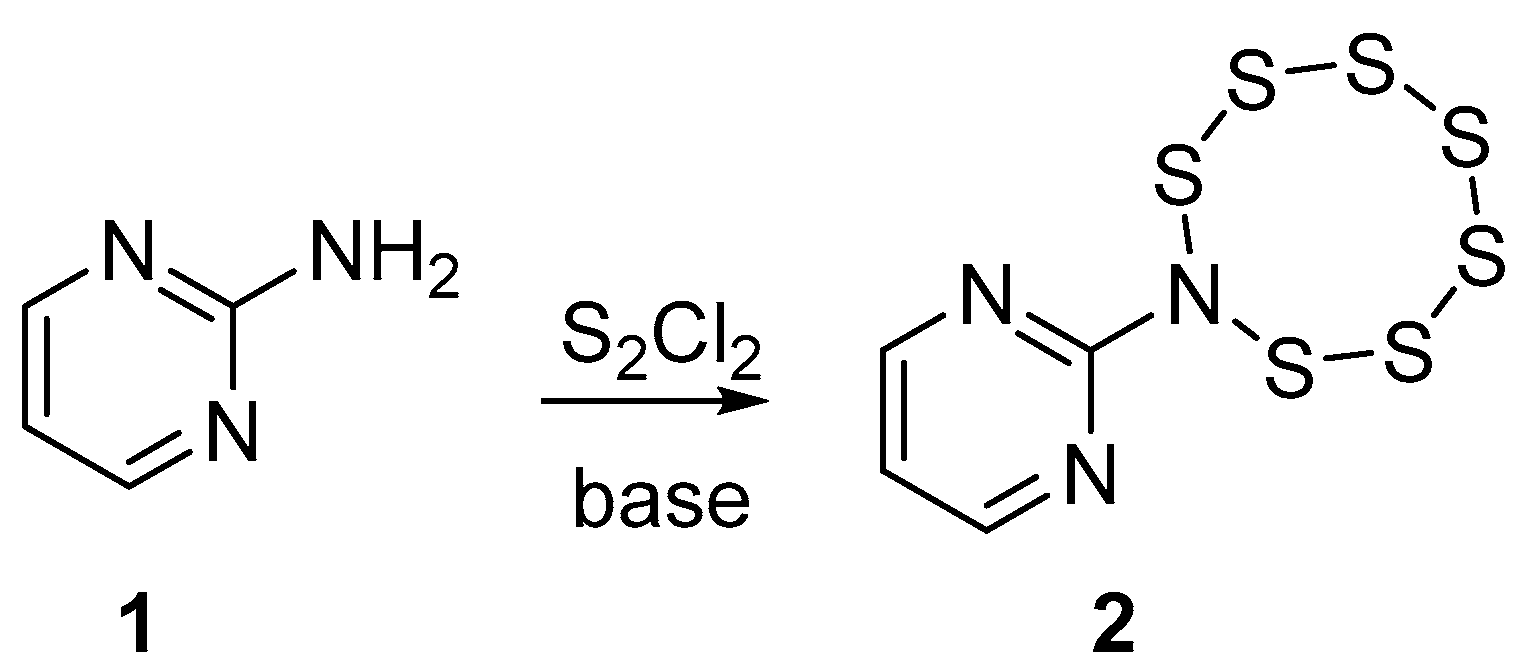
2. Results and Discussion
3. Experimental Section
3.1. General Information
3.2. Synthesis of 8-(pyrimidin-2-yl)-1,2,3,4,5,6,7,8-heptathiazocane 2
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chivers, T. Cyclic chalcogen imides. In A Guide to Chalcogen-Nitrogen Chemistry; World Scientific: Singapore, 2005; pp. 111–121. [Google Scholar] [CrossRef]
- Olsen, B.A.; Olsen, F.P. Sulfurimide anions. II. Alkylation of heptasulfurimide. Inorg. Chem. 1969, 8, 1736–1741. [Google Scholar] [CrossRef]
- Colchester, J.E.; Tavs, P.; Schulze-Steinen, H.J. Heptasulphur N-Acylimides. J. Chem. Soc. 1963, 4918–4920. [Google Scholar] [CrossRef]
- Ramsay, R.J.; Heal, H.G.; Garcia-Fernandez, H. Mercury and organomercury derivatives of cyclodiazahexathiane and cycloazaheptathiane: preparation and decomposition. J. Chem. Soc. Dalton Trans. 1976, 234–237. [Google Scholar] [CrossRef]
- Westphal, O.; Brauchle, H.H.; Hurni, H. Compounds rich in sulfur. Water-soluble derivatives of heptasulfurimides and cyclotetrathioimine. Pharm. Acta Helv. 1958, 33, 429–436. [Google Scholar] [PubMed]
- Saito, T.; Hiraoka, T. Reactions of α-amino acid derivatives with thionyl chloride. An application to a synthesis of 7α-methoxycephalosporins. Chem. Pharm. Bull. 1977, 25, 784–791. [Google Scholar] [CrossRef]
- Konstantinova, L.S.; Rakitin, O.A. Sulfur monochloride in the synthesis of heterocyclic compounds. Adv. Heterocycl. Chem. 2008, 96, 175–229. [Google Scholar] [CrossRef]
- Konstantinova, L.S.; Rakitin, O.A. Design of sulfur heterocycles with sulfur monochloride: retrosynthetic analysis and prospects. Mendeleev Commun. 2009, 19, 55–61. [Google Scholar] [CrossRef]
- Konstantinova, L.S.; Rakitin, O.A. Sulfur monochloride in organic synthesis. Russ. Chem. Rev. 2014, 83, 225–250. [Google Scholar] [CrossRef]
| Entry | S2Cl2 (equiv.) | Base (equiv.) | Solvent | Temperature, °C | Time, h | Yield of 2, % |
|---|---|---|---|---|---|---|
| 1 | 4 | DABCO (4) | CH2Cl2 | 5 | 12 | 78 |
| 2 | 4 | DABCO (4) | CHCl3 | 5 | 12 | 66 |
| 3 | 4 | Et3N (4) | CH2Cl2 | 5 | 12 | 48 |
| 4 | 4 | Pyridine (4) | CH2Cl2 | 5 | 12 | 42 |
| 5 | 4 | DABCO (4) | CH2Cl2 | Rt | 12 | 43 |
| 6 | 4 | DABCO (4) | CH2Cl2 | 5 | 6 | 65 |
| 7 | 4 | DABCO (4) | CH2Cl2 | 5 | 24 | 76 |
| 8 | 4 | DABCO (4) | CH2Cl2 | -18 | 12 | 12 |
| 9 | 2 | DABCO (2) | CH2Cl2 | 5 | 12 | 26 |
| 10 | 6 | DABCO (6) | CH2Cl2 | 5 | 12 | 77 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogurtsov, V.A.; Rakitin, O.A. 8-(Pyrimidin-2-yl)-1,2,3,4,5,6,7,8-heptathiazocane. Molbank 2019, 2019, M1091. https://doi.org/10.3390/M1091
Ogurtsov VA, Rakitin OA. 8-(Pyrimidin-2-yl)-1,2,3,4,5,6,7,8-heptathiazocane. Molbank. 2019; 2019(4):M1091. https://doi.org/10.3390/M1091
Chicago/Turabian StyleOgurtsov, Vladimir A., and Oleg A. Rakitin. 2019. "8-(Pyrimidin-2-yl)-1,2,3,4,5,6,7,8-heptathiazocane" Molbank 2019, no. 4: M1091. https://doi.org/10.3390/M1091
APA StyleOgurtsov, V. A., & Rakitin, O. A. (2019). 8-(Pyrimidin-2-yl)-1,2,3,4,5,6,7,8-heptathiazocane. Molbank, 2019(4), M1091. https://doi.org/10.3390/M1091







