2-(3,4-Dimethoxyphenyl)-N-(4-methoxyphenyl)-1-propyl-1H-benzo[d]imidazole-5-carboxamide
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Instruments
3.2. Synthetic Procedures
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Spasov, A.A.; Yozhitsa, I.N.; Bugaeva, L.I.; Anisimova, V.A. Benzimidazole derivatives: Spectrum of pharmacological activity and toxicological properties (a review). Pharm. Chem. J. 1999, 33, 6–17. [Google Scholar] [CrossRef]
- Shin, J.M.; Kim, N. Pharmacokinetics and pharmacodynamics of the proton pump inhibitors. J. Neurogastroenterol. Motil. 2013, 19, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Gurvinder, S.; Maninderjit, K.; Mohan, C. Benzimidazoles: The latest information on biological activities. Int. Res. J. Pharm. 2013, 4, 82–87. [Google Scholar]
- Ansari, N.H.; Söderberg, B.C.G. Synthesis of N-alkoxy-substituted 2H-benzimidazoles. Tetrahedron Lett. 2017, 58, 4717–4720. [Google Scholar] [CrossRef] [PubMed]
- Salahuddin; Mohammad, S.; Mazumder, A. Benzimidazoles: Biologically active compounds (A Review). Arab. J. Chem. 2017, 10, S157–S173. [Google Scholar] [CrossRef]
- Velik, J.; Baliharová, V.; Fink-Gremmels, J.; Bull, S.; Lamka, J.; Skálová, L. Benzimidazole drugs and modulation of biotransformation enzymes. Res. Vet. Sci. 2004, 76, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Desai, N.C.; Shihory, N.R.; Kotadiya, G.M. Facile synthesis of Benzimidazole bearing 2-pyridone derivatives as potential antimicrobial agents. Chin. Chem. Lett. 2014, 25, 305–307. [Google Scholar] [CrossRef]
- Shingalapur, R.V.; Hosamani, K.M.; Keri, R.S. Synthesis and evaluation of in vitro anti-microbial and anti-tubercular activity of 2-styryl benzimidazoles. Eur. J. Med. Chem. 2009, 44, 4244–4248. [Google Scholar] [CrossRef]
- Alasmar, F.A.; Snelling, A.M.; Zain, M.E.; Alafeefy, A.M.; Awaad, A.S.; Karodia, N. Synthesis and evaluation of selected benzimidazole derivatives as potential antimicrobial agents. Molecules 2015, 20, 15206–15223. [Google Scholar] [CrossRef]
- Gaba, M.; Singh, S.; Mohan, C. Benzimidazole: An emerging scaffold for analgesic and anti-inflammatory agents. Eur. J. Med. Chem. 2014, 76, 494–505. [Google Scholar] [CrossRef]
- Kalalbandi, V.K.; Seetharamappa, J.; Katrahalli, U.; Bhat, K.G. Synthesis, crystal studies, anti-tuberculosis and cytotoxic studies of 1-[(2E)-3-phenylprop-2-enoyl]-1H-benzimidazole derivatives. Eur. J. Med. Chem. 2014, 79, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Usta, A.; Yilmaz, F.; Kapucu, G.; Baltas, N.; Mentese, E. Synthesis of Some New Benzimidazole Derivatives with their Antioxidant Activities. Lett. Org. Chem. 2015, 12, 227–232. [Google Scholar] [CrossRef]
- Si, W.; Zhang, T.; Li, Y.; She, D.; Pan, W.; Gao, Z.; Ning, J.; Mei, X. Synthesis and biological activity of novel benzimidazole derivatives as potential antifungal agents. J. Pestic. Sci. 2016, 41, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Shingalapur, R.V.; Hosamani, K.M.; Keri, R.S.; Hugar, M.H. Derivatives of benzimidazole pharmacophore: Synthesis, Anticonvulsant, antidiabetic and DNA cleavage studies. Eur. J. Med. Chem. 2010, 45, 1753–1759. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, M.; Paglietti, G.; Boido, V.; Sparatore, F.; Marongiu, F.; Marongiu, E.; La Colla, P.; Loddo, R. Antiviral activity of benzimidazole derivatives. I. Antiviral activity of 1-substituted-2-[(benzotriazol-1/2-yl)methyl]benzimidazoles. Chem. Biodivers. 2008, 5, 2386–2401. [Google Scholar] [CrossRef] [PubMed]
- Nofal, Z.M.; Soliman, E.A.; Abd El-Karim, S.S.; EI Zahar, M.I.; Srour, A.M.; Sethumadhavan, S.; Maher, T.J. Novel benzimidazole derivatives as expected anticancer agents. Acta Pol. Pharm. 2011, 68, 519–534. [Google Scholar] [PubMed]
- Farmanzadeh, D.; Najafi, M. Benzimidazole derivatives as anticancer drugs: A theoretical investigation. J. Theor. Comput. Chem. 2015, 14, 1550018. [Google Scholar] [CrossRef]
- Bui, H.T.B.; Ha, Q.T.K.; Keun Oh, W.; Vo, D.D.; Chau, Y.N.T.; Thi Kim Tu, C.; Canh Pham, E.; Thao Tran, P.; Thi Tran, L.; Mai, H.V. Microwave assisted synthesis and cytotoxic activity evaluations of new benzimidazole derivatives. Tetrahedron Lett. 2016, 57, 887–891. [Google Scholar] [CrossRef]
- Ibrahim, H.S.; Albakri, M.E.; Mahmoud, W.R.; Allam, H.A.; Reda, A.M.; Abdel-Aziz, H.A. Synthesis and biological evaluation of some novel thiobenzimidazole derivatives as anti-renal cancer agents through inhibition of c-MET kinase. Bioorg. Chem. 2019, 85, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Bhambra, A.S.; Edgar, M.; Elsegood, M.R.J.; Horsburgh, L.; Krystof, V.; Lucas, P.D.; Mojally, M.; Teat, S.J.; Warwick, T.G.; Weaver, G.W.; et al. Novel fluorinated benzimidazole-based scaffolds and their anticancer activity in vitro. J. Fluorine Chem. 2016, 18, 99–109. [Google Scholar] [CrossRef]
- Baumann, M.; Baxendale, I.R. An overview of the synthetic routes to the best selling drugs containing 6-membered heterocycles. Beilstein J. Org. Chem. 2013, 9, 2265–2319. [Google Scholar] [CrossRef] [PubMed]
- Yule, I.A.; Czaplewski, L.G.; Pommier, S.; Davies, D.T.; Narramore, S.K.; Fishwick, C.W.G. Pyridine-3-carboxamide-6-yl-ureas as novel inhibitors of bacterial DNA gyrase: Structure based design, synthesis, SAR and antimicrobial activity. Eur. J. Med. Chem. 2014, 86, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Narramore, S.; Stevenson, C.E.M.; Maxwell, A.; Lawson, D.M.; Fishwick, C.W.G. New insights into the binding mode of pyridine-3-carboxamide inhibitors of E. coli DNA gyrase. Bioorg. Med. Chem. 2019, 27, 3546–3550. [Google Scholar] [CrossRef] [PubMed]
- Nuhrich, A.; Varache-Lembège, M.; Vercauteren, J.; Dokhan, R.; Renard, P.; Devaux, G. Synthesis and binding affinities of a series of 1,2-benzisoxazole-3-carboxamides to dopamine and serotonin receptors. Eur. J. Med. Chem. 1996, 31, 957–964. [Google Scholar] [CrossRef]
- Cai, W.; Liu, A.; Li, Z.; Dong, W.; Liu, X.; Sun, N. Synthesis and anticancer activity of novel Thiazole-5-carboxamide derivatives. Appl. Sci. 2016, 6, 8. [Google Scholar] [CrossRef]
- Kalpana, K.; Kumar, K.R.; Babu, A.V.; Vanjivaka, S.; Vantikommu, J.; Palle, S. Synthesis and biological evaluation of pyrazole amides fused combretastatin derivatives as anticancer agents. Curr. Bioact. Compd. 2018, 14, 357–363. [Google Scholar] [CrossRef]
- Moreau, S.; Coudert, P.; Rubat, C.; Vallee-Goyet, D.; Gardette, D.; Gramain, J.C.; Couquelet, J. Synthesis and anticonvulsant properties of triazolo- and imidazopyridazinyl carboxamides and carboxylic acids. Bioorg. Med. Chem. 1998, 6, 983–991. [Google Scholar] [CrossRef]
- Lu, X.; Tang, J.; Cui, S.; Wan, B.; Franzblauc, S.G.; Zhang, T.; Zhang, X.; Ding, K. Pyrazolo[1,5-a]pyridine-3-carboxamide hybrids: Design, synthesis and evaluation of anti-tubercular activity. Eur. J. Med. Chem. 2017, 125, 41–48. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, Y.H.; Jung, S.Y.; Kim, H.J.; Jin, C.; Lee, Y.S. Synthesis of chromone carboxamide derivatives with antioxidative and calpain inhibitory properties. Eur. J. Med. Chem. 2011, 46, 1721–1728. [Google Scholar] [CrossRef]
- Bylov, I.E.; Vasylyev, M.V.; Bilokin, Y.V. Synthesis and anti-inflammatory activity of N-substituted 2-oxo-2H-1-benzopyran-3-carboxamides and their 2-iminoanalogues. Eur. J. Med. Chem. 1999, 34, 997–1001. [Google Scholar] [CrossRef]
- Venkateswarlu, Y.; Ramesh Kumar, S.; Leelavathi, P. Facile and efficient one-pot synthesis of benzimidazoles using lanthanum chloride. Org. Med. Chem. Lett. 2013, 3, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Foals, D.; Li, J.; Yu, L.; Baldino, C.M. A versatile method for the synthesis of benzimidazoles from o-Nitroanilines and aldehydes in one step via a reductive cyclization. Synthesis 2005, 36, 47–56. [Google Scholar]
- Kumar, V.; Poojary, B.; Prathibha, A.; Shruthi, N. A Synthesis of some novel 1,2-disubstituted benzimidazole-5-carboxylates via One-pot method using Sodium Dithionite and its effect on N-Debenzylation. Synth. Commun. 2014, 44, 3414–3425. [Google Scholar] [CrossRef]
- Roy, P.; Pramanik, A. One-pot sequential synthesis of 1,2-disubstituted benzimidazoles under metal-free conditions. Tetrahedron Lett. 2013, 54, 5243–5245. [Google Scholar] [CrossRef]
- Oda, S.; Shimizu, H.; Aoyama, Y.; Ueki, T.; Shimizu, S.; Osato, H.; Takeuchi, Y. Development of safe one-pot synthesis of N-1- and C-2-Substituted benzimidazole via reductive cyclization of o-Nitroaryl amine using Na2S2O4. Org. Process Res. Dev. 2012, 16, 96–101. [Google Scholar] [CrossRef]
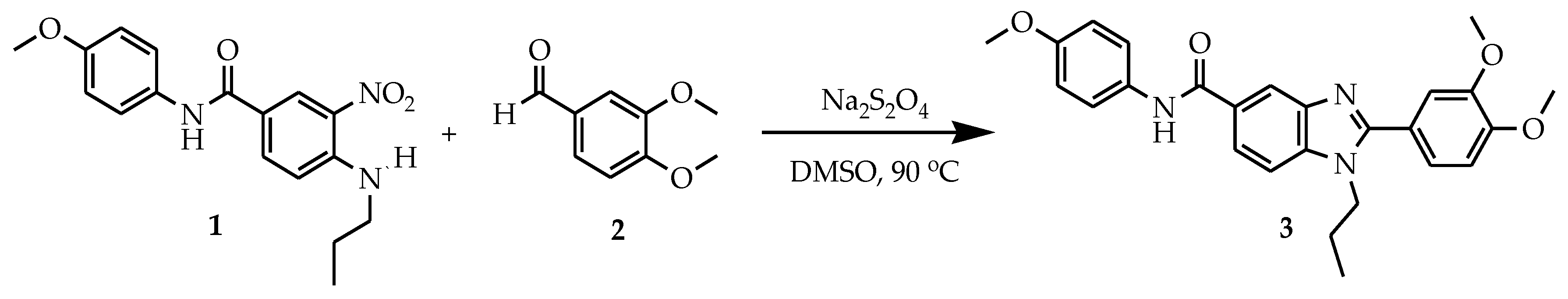
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhaskar, P.; Kumar, V.; Tholappanavara Hanumanthappa, S.K.; Haliwana Banakara Vijaykumar, S. 2-(3,4-Dimethoxyphenyl)-N-(4-methoxyphenyl)-1-propyl-1H-benzo[d]imidazole-5-carboxamide. Molbank 2019, 2019, M1079. https://doi.org/10.3390/M1079
Bhaskar P, Kumar V, Tholappanavara Hanumanthappa SK, Haliwana Banakara Vijaykumar S. 2-(3,4-Dimethoxyphenyl)-N-(4-methoxyphenyl)-1-propyl-1H-benzo[d]imidazole-5-carboxamide. Molbank. 2019; 2019(3):M1079. https://doi.org/10.3390/M1079
Chicago/Turabian StyleBhaskar, Prakash, Vasantha Kumar, Suresha Kumara Tholappanavara Hanumanthappa, and Sowmya Haliwana Banakara Vijaykumar. 2019. "2-(3,4-Dimethoxyphenyl)-N-(4-methoxyphenyl)-1-propyl-1H-benzo[d]imidazole-5-carboxamide" Molbank 2019, no. 3: M1079. https://doi.org/10.3390/M1079
APA StyleBhaskar, P., Kumar, V., Tholappanavara Hanumanthappa, S. K., & Haliwana Banakara Vijaykumar, S. (2019). 2-(3,4-Dimethoxyphenyl)-N-(4-methoxyphenyl)-1-propyl-1H-benzo[d]imidazole-5-carboxamide. Molbank, 2019(3), M1079. https://doi.org/10.3390/M1079




