2-(3,5-Dimethyl-1H-pyrazol-1-yl)thiazolo[4,5-b]pyridine
Abstract
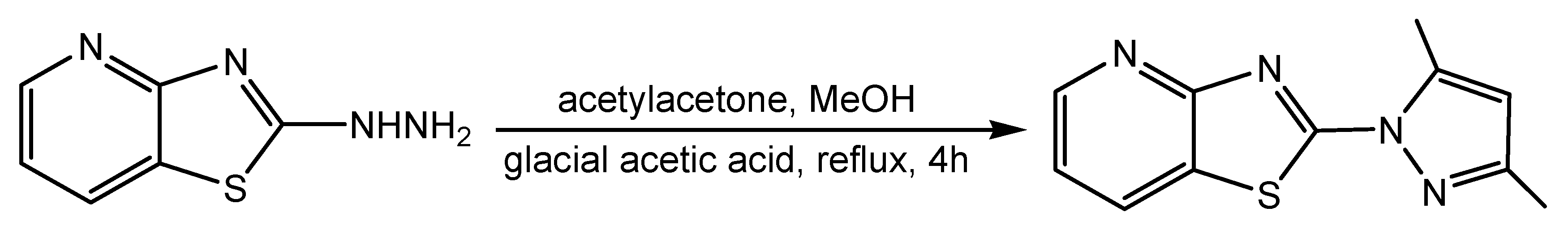
1. Introduction
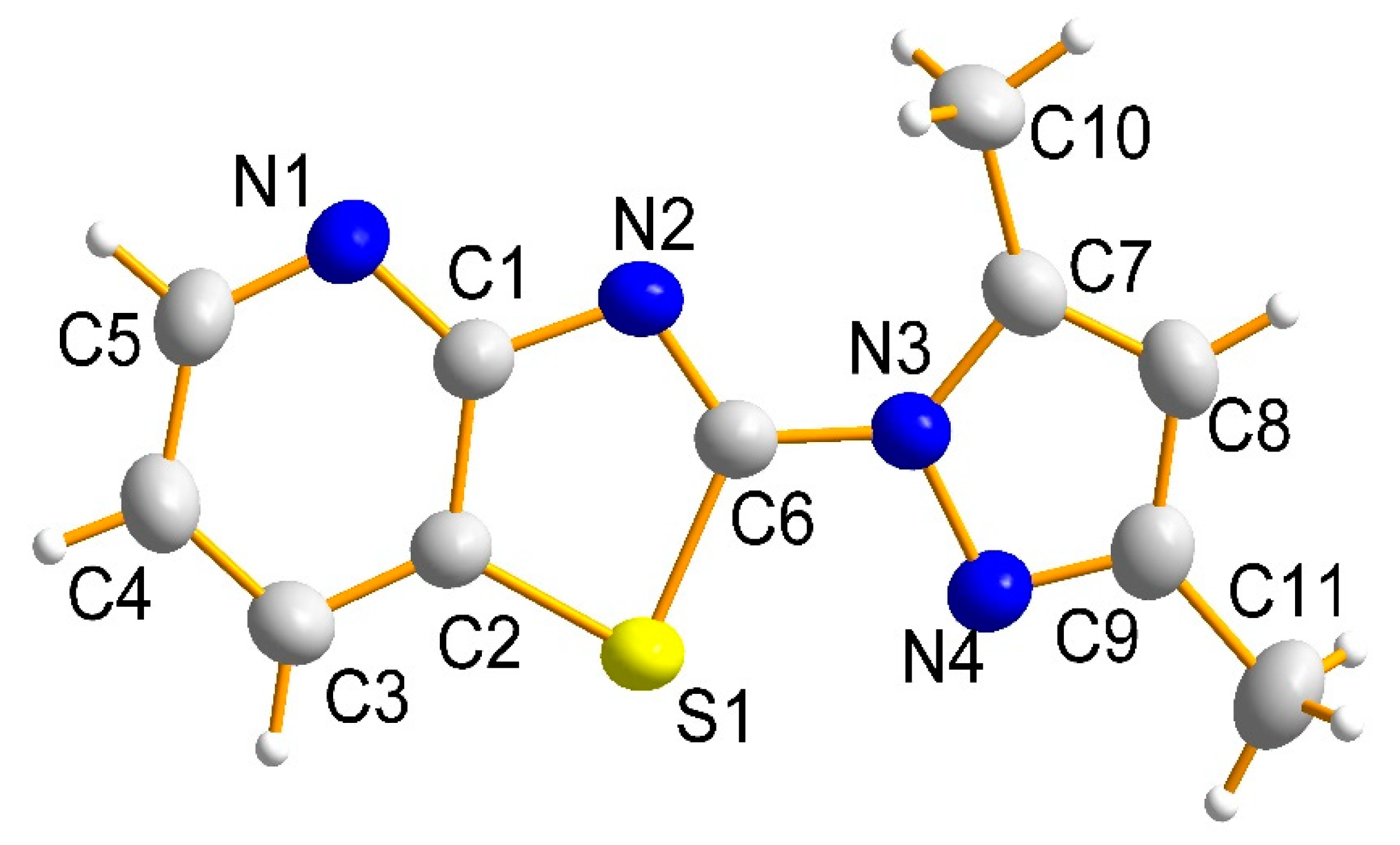
2. Results
3. Materials and Methods
3.1. General Information
3.2. Synthesis and Characterization of 1
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Samir, B.; Wesam, K.; Ahmed, A.F. Synthesis and antimicrobial evaluation of some new thiazole, thiazolidinone and thiazoline derivatives starting from 1-chloro-3,4-dihydronaphthalene-2-carboxaldehyde. Eur. J. Med. Chem. 2007, 42, 948–954. [Google Scholar]
- Holla, B.S.; Malini, K.V.; Rao, B.S.; Sarojini, B.K.; Kumari, N.S. Synthesis of some new 2,4-disubstituted thiazoles as possible antibacterial and anti-inflammatory agents. Eur. J. Med. Chem. 2003, 38, 313–318. [Google Scholar] [CrossRef]
- Bharti, S.K.; Nath, G.; Tilak, R.; Singh, S.K. Synthesis, anti-bacterial and anti-fungal activities of some novel Schiff bases containing 2,4-disubstituted thiazole ring. Eur. J. Med. Chem. 2010, 45, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Rostom, S.A.F. Synthesis and in vitro antitumor evaluation of some indeno[1,2-c]-pyrazol(in)es substituted with sulfonamide, sulfonylurea(-thiourea) pharmacophores, and some derived thiazole ring systems. Bioorg. Med. Chem. 2006, 14, 6475–6485. [Google Scholar] [CrossRef] [PubMed]
- Takate, S.J.; Shinde, A.D.; Karale, B.K.; Akolkar, H.; Nawale, L.; Sarkar, D.; Mhaske, P.C. Thiazolyl-pyrazole derivatives as potential antimycobacterial agents. Bioorg. Med. Chem. Lett. 2019, 29, 1199–1202. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.H.; Zhai, Z.W.; Sun, Z.H.; Weng, J.Q.; Tan, C.X.; Liu, X.H. Synthesis, crystal structure and biological activity of 2-chloro-5-(((5-(1-methyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)-4-phenyl-4H-1,2,4-triazol-3-yl)thio)methyl)thiazole. Chin. J. Struct. Chem. 2017, 36, 1137–1141. [Google Scholar]
- Metwally, N.H.; Badawy, M.A.; Okpy, D.S. Synthesis and anticancer activity of some new thiopyrano[2,3-d]thiazoles incorporating pyrazole moiety. Chem. Pharm. Bull. 2015, 63, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Bekhit, A.A.; Fahmy, H.T.Y.; Rostom, S.A.F.; Baraka, A.M. Design and synthesis of some substituted 1H-pyrazolyl-thiazolo[4,5-d]pyrimidines as anti-inflammatory-antimicrobial agents. Eur. J. Med. Chem. 2003, 38, 27–36. [Google Scholar] [CrossRef]
- Klenina, O.; Chaban, T.; Zimenkovsky, B.; Harkov, S.; Ogurtsov, V.; Chaban, I.; Myrko, I. QSAR modeling for antioxidant activity of novel N3 substituted 5,7-dimethyl-3H-thiazolo[4,5-b]pyridin-2-ones. Pharmacia 2017, 64, 49–71. [Google Scholar]
- Chaban, T.; Klenina, O.; Harkov, S.; Ogurtsov, V.; Chaban, I.; Nektegaev, I. Synthesis of some new N3 substituted 6-phenylazo-3H-thiazolo[4,5-b]pyridine-2-ones as possible anti-inflammatory agents. Pharmacia 2017, 64, 16–30. [Google Scholar]
- Ren, Y.L.; Wang, Q.; Wang, W.M.; Jin, C.X. Crystal structure of 2-(3,5-dimethyl-1H-pyrazol-1-yl)thiazolo[5,4-b]-pyridine, C11H10N4S. Z. Kristallogr. NCS 2014, 229, 145–146. [Google Scholar] [CrossRef][Green Version]
- Miao, S.B.; Ji, B.M.; Zhou, L. Crystal structure of 1-(diphenylmethylene)-2-(thiazolo[4,5-b]pyridine-2-yl)hydrazine. J. Struct. Chem. 2014, 55, 384–387. [Google Scholar] [CrossRef]
- Miao, S.B.; Ji, B.M.; Zhou, L. Synthesis and crystal structures of a new thiazole-hydrazone ligand and its Co(III) complex. Synth. React. Inorg. Met. Org. Nano-Met. Chem. 2013, 43, 1296–1300. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXL97. Program for the Refinement of Crystal Structure; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lan, H.; Zheng, M.; Wang, Y. 2-(3,5-Dimethyl-1H-pyrazol-1-yl)thiazolo[4,5-b]pyridine. Molbank 2019, 2019, M1077. https://doi.org/10.3390/M1077
Lan H, Zheng M, Wang Y. 2-(3,5-Dimethyl-1H-pyrazol-1-yl)thiazolo[4,5-b]pyridine. Molbank. 2019; 2019(3):M1077. https://doi.org/10.3390/M1077
Chicago/Turabian StyleLan, Honghong, Min Zheng, and Ye Wang. 2019. "2-(3,5-Dimethyl-1H-pyrazol-1-yl)thiazolo[4,5-b]pyridine" Molbank 2019, no. 3: M1077. https://doi.org/10.3390/M1077
APA StyleLan, H., Zheng, M., & Wang, Y. (2019). 2-(3,5-Dimethyl-1H-pyrazol-1-yl)thiazolo[4,5-b]pyridine. Molbank, 2019(3), M1077. https://doi.org/10.3390/M1077



