2,4-Bis[(2,6-diisopropylphenyl)imino]-3-methylpentan-3-ol
Abstract
:1. Introduction
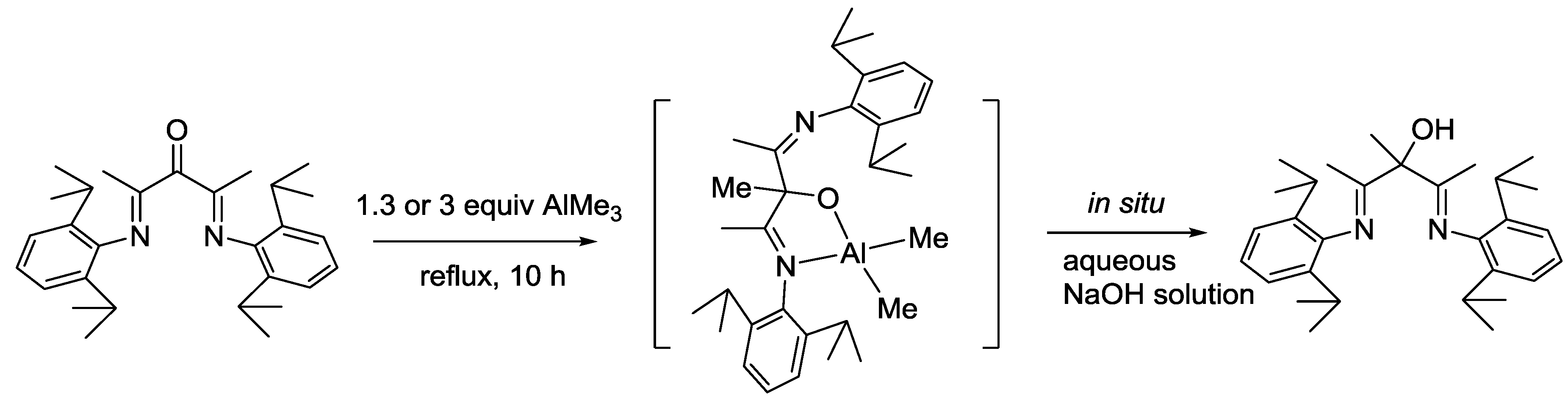
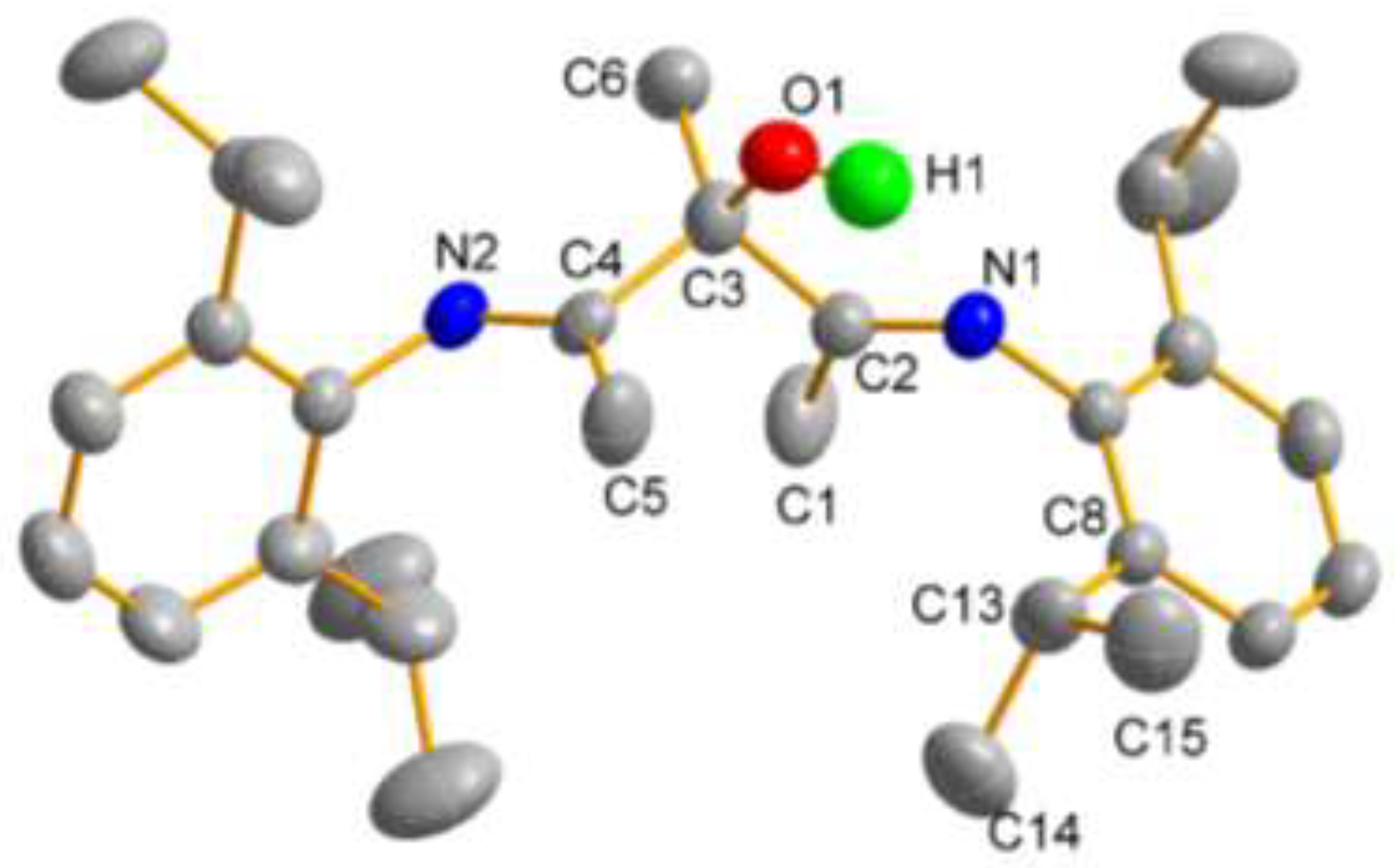
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. Synthesis of 2,4-Bis[(2,6-diisopropylphenyl)imino]-3-methylpentan-3-ol
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bourget-Merle, L.; Lappert, M.F.; Severn, J.R. The chemistry of β-diketiminatometal complexes. Chem. Rev. 2002, 102, 3031–3065. [Google Scholar] [CrossRef] [PubMed]
- Feldman, J.; Mclain, S.J.; Parthasarathy, A.; Marshall, W.J.; Calabrese, J.C.; Arthur, S.D. Electrophilic metal precursors and a β-diimine ligand for nickel(II)- and palladium(II)-catalyzed ethylene polymerization. Organometallics 1997, 16, 1514–1516. [Google Scholar] [CrossRef]
- Guo, L.H.; Dai, S.Y.; Sui, X.L.; Chen, C.L. Palladium and nickel catalyzed chain walking olefin polymerization and copolymerization. ACS Catal. 2015, 6, 428–441. [Google Scholar] [CrossRef]
- Guo, L.H.; Liu, W.J.; Chen, C.L. Late transition metal catalyzed a-olefin polymerization and copolymerization with polar monomers. Mater. Chem. Front. 2017, 1, 2487–2494. [Google Scholar] [CrossRef]
- Burford, N.; D’eon, M.; Ragogna, P.J.; McDonald, R.; Ferguson, M.J. Synthesis and structures of complexes demonstrating the coordinative versatility of the 2,4-diimino-3-phosphinopentene anion (γ-phosphino-ß-diketiminate). Inorg. Chem. 2004, 43, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Allen, S.D.; Moore, D.R.; Lobkovsky, E.B.; Coates, G.W. High-activity, single-site catalysts for the alternating copolymerization of CO2 and propylene oxide. J. Am. Chem. Soc. 2002, 124, 14284–14285. [Google Scholar] [CrossRef] [PubMed]
- Vidovic, D.; Lu, Z.; Reeske, G.; Moore, J.A.; Cowley, A.H. An N,N′-chelated phosphenium cation supported by a β-diketiminate ligand. Chem. Commun. 2006, 3501–3503. [Google Scholar] [CrossRef] [PubMed]
- Azoulay, J.D.; Rojas, R.S.; Serrano, A.V.; Ohtaki, H.; Galland, G.B.; Wu, G.; Bazan, G.C. Nickel α-keto-β-diimine initiators for olefin polymerization. Angew. Chem. Int. Ed. 2009, 48, 1089–1092. [Google Scholar] [CrossRef] [PubMed]
- Azoulay, J.D.; Koretz, Z.A.; Wu, G.; Bazan, G.C. Well-defined cationic methallyl α-keto-β-diimine. complexes of nickel. Angew. Chem. Int. Ed. 2010, 49, 7890–7894. [Google Scholar] [CrossRef] [PubMed]
- Carey, D.T.; Mair, F.S.; Pritchard, R.G.; Warren, J.E.; Woods, R.J. Borane and alane reductions of bulky N,N′-diaryl-1,3-diimines: Structural characterization of products and intermediates in the diastereoselective synthesis of 1,3-diamines. Dalton Trans. 2003, 3792–3798. [Google Scholar] [CrossRef]
- Van Vilet, M.R.P.; van Koten, G.; Rotteveel, M.A.; Schrap, M.; Vrieze, K. Reactivity of l-Aza-4-oxo-l, 3-butadienes (α-imino ketones) toward triorganoaluminum reagents. 1. X-ray crystal structures of [(AlMe3)2{(σ,σ-N,O-(MeN=C(Ph)C(Ph)=O)}], with a N,O-bridge-bonded α-imino ketone, and of Me2Al(t-Bu)N = CHC(Me),OAIMe3, a carbonyl alkylated product which is o-coordinated to AIMe3. Organometallics 1986, 5, 1389–1394. [Google Scholar]
- Van Vilet, M.R.P.; van Koten, G.; de Keijser, M.S.; Vrieze, K. Reactivity of 1-Aza-4-oxa-1,3-butadienes (α-imino ketones) toward triorganoalumlnum reagents. 2. syntheses of triorganoaluminum α-imho ketone coordination complexes, diorganoaluminum (α-imino) alkoxides, and diorganoaluminum (α-imino)enolates and study of their dynamic behavior insolution (1H and 13C-NMR). Organometallics 1987, 6, 1652–1664. [Google Scholar]
- Tang, X.; Huang, Y.T.; Liu, H.; Liu, R.Z.; Shen, D.S.; Liu, N.; Liu, F.S. α-Hydroxyimine palladium complexes: Synthesis, molecular structure, and their activities towards the suzukie-miyaura cross-coupling reaction. J. Organomet. Chem. 2013, 729, 95–102. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. C. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Mcardle, P. Oscail, a program package for small-molecule single-crystal crystallography with crystal morphology prediction and molecular modelling. J. Appl. Crystallogr. 2017, 50, 320–326. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, W.; Kong, W.; Du, Q.; Zhang, S.; Guo, L.; Liu, Z. 2,4-Bis[(2,6-diisopropylphenyl)imino]-3-methylpentan-3-ol. Molbank 2018, 2018, M995. https://doi.org/10.3390/M995
Sun W, Kong W, Du Q, Zhang S, Guo L, Liu Z. 2,4-Bis[(2,6-diisopropylphenyl)imino]-3-methylpentan-3-ol. Molbank. 2018; 2018(2):M995. https://doi.org/10.3390/M995
Chicago/Turabian StyleSun, Wenting, Wenyu Kong, Qing Du, Shumiao Zhang, Lihua Guo, and Zhe Liu. 2018. "2,4-Bis[(2,6-diisopropylphenyl)imino]-3-methylpentan-3-ol" Molbank 2018, no. 2: M995. https://doi.org/10.3390/M995
APA StyleSun, W., Kong, W., Du, Q., Zhang, S., Guo, L., & Liu, Z. (2018). 2,4-Bis[(2,6-diisopropylphenyl)imino]-3-methylpentan-3-ol. Molbank, 2018(2), M995. https://doi.org/10.3390/M995




