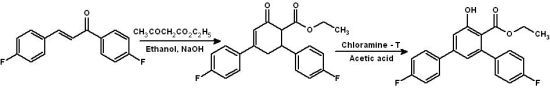
Ethyl 4,4''-Difluoro-5'-hydroxy-1,1':3',1''-terphenyl-4'-carboxylate
Abstract
: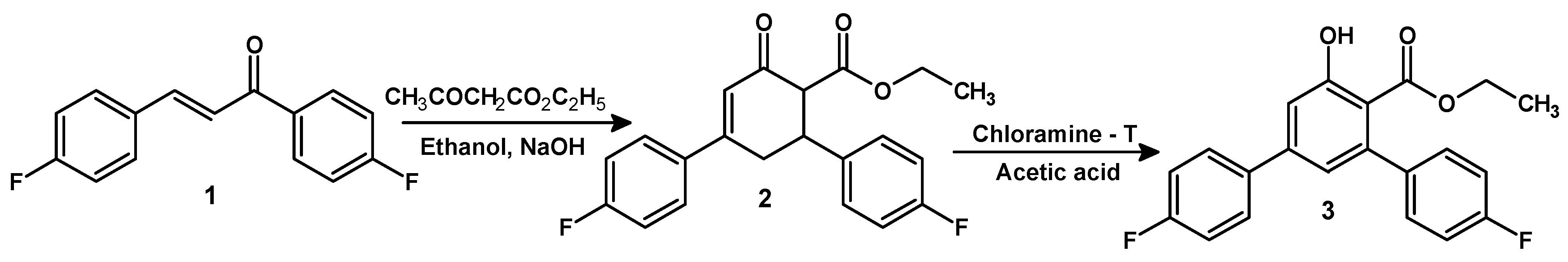
Experimental
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgments
References
- Liu, J.K. Natural Terphenyls: Developments since 1877. Chem. Rev. 2006, 106, 2209–2223. [Google Scholar] [CrossRef] [PubMed]
- Astrue, D. Modern Arene Chemistry; Wiley-VCH: Weinheim, Germany, 2002. [Google Scholar]
- Pearson, D.E.; Buehler, C.A. Friedel-Crafts Acylations with Little or No Catalyst. Synthesis 1972, 533–542. [Google Scholar] [CrossRef]
- Buncel, E.; Dust, J.M.; Terrier, F. Rationalizing the Regioselectivity in Polynitroarene Anionic σ-Adduct Formation. Relevance to Nucleophilic Aromatic Substitution. Chem. Rev. 1995, 95, 2261–2280. [Google Scholar] [CrossRef]
- Hassan, J.; Se´vignon, M.; Gozzi, C.; Schulz, E.; Lemaire, M. Aryl−Aryl Bond Formation One Century after the Discovery of the Ullmann Reaction. Chem. Rev. 2002, 102, 1359–1470. [Google Scholar] [CrossRef] [PubMed]
- Snieckus, V. Directed ortho metalation. Tertiary amide and O-carbamate directors in synthetic strategies for polysubstituted aromatics. Chem. Rev. 1990, 90, 879–933. [Google Scholar] [CrossRef]
- Trost, B.M. Atom Economy. A Challenge for Organic Synthesis. Angew. Chem. Int. Ed. Engl. 1995, 34, 259–281. [Google Scholar] [CrossRef]
- Saito, S.; Yamamoto, Y. Recent Advances in the Transition-Metal-Catalyzed Regioselective Approaches to Polysubstituted Benzene Derivatives. Chem. Rev. 2000, 100, 2901–2916. [Google Scholar] [CrossRef] [PubMed]
- Kotnis, A.S. A rapid entry to highly functionalized p-methoxy benzoates. Tetrahedron Lett. 1990, 31, 481–484. [Google Scholar] [CrossRef]
- Tamura, Y.; Yoshimoto, Y. An improved method for the conversion of cyclohexenones into anisoles. Chem. Ind. 1980, 888–889. [Google Scholar]
- Hegde, S.G.; Kassim, A.M.; Kennedy, A.I. Aromatization reactions of 2-cyclohexenones and 1,3-cyclohexadien-1-amines with iodine/sodium alkoxide. Tetrahedron 2001, 57, 1689–1698. [Google Scholar] [CrossRef]
- Yadav, J.S.; Reddy, B.V.S.; Sabitha, G.; Reddy, G.S.K.K. Aromatization of Hantzsch 1,4-Dihydropyridines with I2—MeOH. Synthesis 2000, 1532–1534. [Google Scholar] [CrossRef]
- Schoop, A.; Greiving, H.; Gohrt, A. A new analogue of rocaglamide by an oxidative dihydrofuran synthesis. Tetrahedron Lett. 2000, 41, 1913–1916. [Google Scholar] [CrossRef]
- Samshuddin, S.; Narayana, B.; Shetty, D.N.; Raghavendra, R. An efficient synthesis of 2,4,6-triaryl pyridines and their biological evaluation. Der Pharma Chemica 2011, 3, 232–240. [Google Scholar]
- Samshuddin, S.; Butcher, R.J.; Akkurt, M.; Narayana, B.; Yathirajan, H.S.; Sarojini, B.K. 1,3-Bis(4-fluorophenyl)-N,N′-(propane-1,3-diylidene)dihydroxylamine. Acta Cryst. 2011, E67, o1954–o1955. [Google Scholar] [CrossRef] [PubMed]
- Jasinski, J.P.; Guild, C.J.; Samshuddin, S.; Narayana, B.; Yathirajan, H.S. 2,3-Dibromo-1,3-bis(4-fluorophenyl)propan-1-one. Acta Cryst. 2010, E66, o2018. [Google Scholar] [CrossRef] [PubMed]
- Jasinski, J.P.; Guild, C.J.; Samshuddin, S.; Narayana, B.; Yathirajan, H.S. 3,5-Bis(4-fluorophenyl)-1-phenyl-4,5-dihydro-1H-pyrazole. Acta Cryst. 2010, E66, o1948–1949. [Google Scholar] [CrossRef] [PubMed]
- Fun, H.-K.; Hemamalini, M.; Samshuddin, S.; Narayana, B.; Yathirajan, H.S. 1-[3,5-Bis(4-fluorophenyl)-4,5-dihydro-1H-pyrazol-1-yl]ethanone. Acta Cryst. 2010, E66, o582–o583. [Google Scholar] [CrossRef] [PubMed]
- Fun, H.-K.; Hemamalini, M.; Samshuddin, S.; Narayana, B.; Yathirajan, H.S. Methyl 4,6-bis(4-fluorophenyl)-2-oxocyclohex-3-ene-1-carboxylate. Acta Cryst. 2010, E66, o864–o865. [Google Scholar] [CrossRef] [PubMed]
- Baktir, Z.; Akkurt, M.; Samshuddin, S.; Narayana, B.; Yathirajan, H.S. 2,4-Bis(4-fluorophenyl)-2,3-dihydro-1H-1,5-benzodiazepine. Acta Cryst. 2011, E67, o1262–1263. [Google Scholar] [CrossRef] [PubMed]
- Baktir, Z.; Akkurt, M.; Samshuddin, S.; Narayana, B.; Yathirajan, H.S. 3,5-Bis(4-fluorophenyl)-4,5-dihydro-1H-pyrazole-1-carbaldehyde. Acta Cryst. 2011, E67, o1292–1293. [Google Scholar] [CrossRef] [PubMed]
- Dutkiewicz, G.; Narayana, B.; Veena, K.; Yathirajan, H.S.; Kubicki, M. (1RS,6SR)-Ethyl 4,6-bis(4-fluorophenyl)-2-oxocyclohex-3-ene-1-carboxylate. Acta Cryst. 2011, E67, o336. [Google Scholar] [CrossRef] [PubMed]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Samshuddin, S.; Narayana, B.; Sarojini, B.K. Ethyl 4,4''-Difluoro-5'-hydroxy-1,1':3',1''-terphenyl-4'-carboxylate. Molbank 2011, 2011, M745. https://doi.org/10.3390/M745
Samshuddin S, Narayana B, Sarojini BK. Ethyl 4,4''-Difluoro-5'-hydroxy-1,1':3',1''-terphenyl-4'-carboxylate. Molbank. 2011; 2011(4):M745. https://doi.org/10.3390/M745
Chicago/Turabian StyleSamshuddin, Seranthimata, Badiadka Narayana, and Balladka Kunhanna Sarojini. 2011. "Ethyl 4,4''-Difluoro-5'-hydroxy-1,1':3',1''-terphenyl-4'-carboxylate" Molbank 2011, no. 4: M745. https://doi.org/10.3390/M745
APA StyleSamshuddin, S., Narayana, B., & Sarojini, B. K. (2011). Ethyl 4,4''-Difluoro-5'-hydroxy-1,1':3',1''-terphenyl-4'-carboxylate. Molbank, 2011(4), M745. https://doi.org/10.3390/M745